Abstract
The recent advent of endoscopy has enabled the endoscopic submucosal dissection (ESD) of superficial nonampullary duodenal epithelial tumors. However, the substantially thin wall and presence of bile and pancreatic juice make it technically difficult to perform duodenal ESD without perforation, which leads to lethal complications. The present study evaluated the efficacy of autologous myoblast sheet transplantation for the prevention of late perforation after duodenal ESD in a porcine model. Two weeks before ESD, skeletal muscle was surgically excised from the femur of pigs, and myoblasts were isolated and seeded in temperature-responsive culture dishes to prepare sheets. Immediately after ESD, the autologous myoblast sheets were attached to the serosal surface at the ESD site with omentopexy. The pigs were divided into two groups: the autologous myoblast sheet group (n = 5), where the myoblast cell sheet was attached to the ESD ulcer part from the duodenal serous side, and the Omentum group (n = 5), where only the omentum was used. The pigs were sacrificed and analyzed macroscopically and histologically on postoperative day 3. The macroscopic examination of the abdominal cavity revealed perforation in the ESD ulcer area and leakage of bile in the Omentum group but no perforation in the Sheet group. A histopathological examination revealed that continuity of the duodenal wall at the ESD site was maintained with dense connective tissue in the Sheet group. In conclusion, autologous myoblast sheets were useful for preventing perforation after duodenal ESD.
Introduction
Nonampullary duodenal epithelial neoplasms are rare, with an incidence among patients who undergo screening or diagnostic endoscopic surveillance of 0.098% 1 . Although the natural history of superficial nonampullary duodenal epithelial tumors (SNADETs) remains unclear, duodenal adenomas are known to occur sporadically or with genetic syndromes, such as familial adenomatous polyposis 2 –4 . They are thought to have the potential for malignant transformation into duodenal cancer via the adenoma–carcinoma sequence, and indeed, Hoteya et al. showed that SNADETs larger than 20 mm in diameter might have malignant potential 5 . Kakushima et al. reported that 36% of endoscopically treated SNADETs were upgraded after post-treatment pathology, with most moving from high-grade dysplasia to adenocarcinoma 6 . These results underscore the difficulty of making a precise diagnosis before resection, so removal is recommended for these tumors.
In other areas of the gastrointestinal tract, such as the stomach and colon, endoscopic resection, such as endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD), for early cancer has become the standard therapy. For small SNADETs, EMR is feasible, and various modified methods have been reported thus far 7,8 . However, SNADETs with a large diameter must be removed in a piecemeal fashion through repeated and overlapping snare resections. Piecemeal resection is a permissible procedure for superficial colorectal lesions 9 , whether or not this strategy is suitable for neoplasms in other organs is unclear. For example, in early gastric cancer, the local recurrence rate after piecemeal resection was reported to range from 20% to 30% 10,11 in contrast to the rate of <1% after R0 resection with ESD 12 . Yahagi et al. recommended ESD over EMR for SNADETs >21 mm from a perspective of completeness of en bloc resection 13 .
However, this procedure carries a substantial risk of bleeding and perforation due to the anatomical characteristics of the duodenum compared with other areas of the digestive tract, such as its narrow lumen, abundant vascularity in the submucosal layer, and thin muscle layer.
Fujihara et al. reported the overall rate of perforation in duodenal ESD to be 13%–50%, while that of delayed perforation is 0%–14.3% 14 . The main cause of delayed perforation is exposure of the ulceration after duodenal ESD to pancreatic juice and bile. Inoue et al. reported that the incidence of delayed perforation was significantly associated with a tumor location at the anal side of the ampulla 15,16 , where the ESD ulcer has a greater risk of being exposed to bile and pancreatic juice; consequent perforation forces the patient to undergo emergency surgery and suffer a life-threatening condition due to severe peritonitis and retroperitonitis.
We previously reported the efficacy of myoblast sheet transplantation for preventing gastric perforation and pancreatic fistula in a rat model 17,18 . These results prompted us to explore whether or not autologous myoblast sheets would tolerate chemical irritation from digestive fluids and exert a preventive effect against delayed perforation after duodenal ESD.
In the present study, we established a novel approach for treating SNADETs with endoscopic and cell sheet technology in a porcine model.
Materials and Methods
Autologous Myoblast Cell Cultures and Preparation of Myoblast Sheets
Two weeks before the duodenal ESD experiment, approximately 5 g of skeletal muscle was obtained from the quadriceps muscle of each pig (from Miyashita hog farm, Nagasaki, Japan) under general anesthesia (Fig. 1). The connective tissue was carefully removed from the retrieved specimen, and the remaining muscle tissue was minced into small pieces. The muscles were digested at 37°C in a shaker bath with TrypLE Express (Gibco-Thermo Fisher Scientific, Waltham, MA, USA) containing collagenase type I (Gibco), gentamicin sulfate (Fuji Pharma Co., Tokyo, Japan), and amphotericin B solution (Gibco) for 60 min. The fluid was discarded, and the same volume of culture medium (MCDB131; Gibco) supplemented with 15% fetal bovine serum (Invitrogen-Thermo Fisher Scientific, Waltham, MA, USA) was added to halt the enzymatic digestion process. Isolated cells were collected by centrifugation (5 min at 800 g), and cells were seeded onto two 175-cm2 gelatin-coated flasks (Invitrogen) with 30 ml of MCDB131 medium (Gibco) supplemented with 15% fetal bovine serum (Invitrogen),

Experimental protocol. This shows the day of harvesting myoblast, duodenal ESD, transplantation, and sacrifice. ESD: endoscopic submucosal dissection.
When the cells become approximately 70% confluent after 12–14 days’ cultivation, they were dissociated from the flasks with TrypLE Express and reincubated on 60-mm temperature-responsive culture dishes (Cellseed, Tokyo, Japan) at 37°C with the cell numbers adjusted to 2.2 × 107 per dish. After 12 h, the culture dishes were transferred to another incubator set at 20°C for 20 min to detach the myoblast sheets. After reducing the culture temperature, the myoblast sheet could then be detached without any need to perform enzymatic treatment; as a result, the important membrane proteins and extracellular matrix could be preserved, thereby allowing the cell sheet to successfully integrate with the tissue at the implanted site. The diameter of each detached cell sheet was approximately 2.5 cm. To increase the strength during handling, fibrin was sprayed onto the surface of the cell sheet using a Beriplast P Combi kit (CSL Behring, Tokyo, Japan).
The cell purity was measured by flow cytometry (Beckman Coulter, Miami, FL, USA) after staining with anticluster of differentiation 56 (CD56; BD Biosciences, San Diego, CA, USA).
Gene Expression Analyses
Total RNA was extracted from established myoblast cells and fibroblast cells obtained from the quadriceps muscle of pigs with an RNeasy mini kit (Qiagen, Hilden, Germany) and reverse transcribed into cDNA with the cDNA Reverse Transcription kit with RNase Inhibitor (Applied Biosystems-Thermo Fisher Scientific, Waltham, MA, USA). mRNA expression was quantified by real-time polymerase chain reaction (PCR) using TaqMan fast Universal PCR Master mix (Applied Biosystems) with primer pairs specific for DES, MyoD, PAX7, INTGα7, FGF2, TGFβ-1, IGF2, EGF, and VEGF (TaqMan Gene Expression Assays; Applied Biosystems). The mean fold-changes in the gene expression relative to beta-actin were calculated using the ΔΔCT method at each time point. The primers employed are as follows—DES: Ss03378045_u1, MyoD: Ss03378464_u1, PAX7: Ss03376692_u1, FGF2: Ss03375809_u1, TGFβ1: Ss03382325_u1, IGF2: Ss03388131_u1, EGF: Ss03391285_m1, and VEGF: Ss03393993_m1. The primers for INTGα7 were—forward: AGAGCTGGCTGCTGGTG and reverse: TGGTCGATGTCCACTCTGT.
Animal Model
Pigs (12–20 kg) from the same farm were used for the experiments. The in vivo porcine delayed perforation model after duodenal ESD is previously described 19 . This model is known to show a 100% delayed perforation rate within several days after successful duodenal ESD. In brief, we made a small incision in the upper abdomen under general anesthesia and fixed the duodenum by clamping the horizontal portion with intestinal forceps due to no adhesion between the duodenal descending part and the retroperitoneum. ESD was performed using a GIF-Q180 gastroscope (Olympus Optical Co., Ltd., Tokyo, Japan), a Videoscope System Exera II (Olympus Optical Co., Ltd.), and an Electrosurgery unit ERBE ICC 350 (ERBE Technology, Tuebingen, Germany).
The endoscope was inserted orally, and ESD was performed on the free abdominal side of the duodenal descending portion, peripheral to the papilla of Vater. Separation of the mucosal layer from the muscular layer was achieved via submucosal injection of a hyaluronic acid (MucoUp; Boston Scientific, Marlborough, MA, USA) containing indigocarmine dye. Submucosal layer dissection was performed using a Flush Knife (Fujifilm, Tokyo, Japan) and Hook Knife (Olympus Optical Co., Ltd.), and an ulcer with a surface 10–15 mm in diameter was produced (Fig. 2A). All resected pieces were retrieved for histological analyses. The pigs were divided into an autologous myoblast sheet group (Sheet group, n = 5), in which the myoblast sheet was attached to the ESD ulcer part from the duodenal serous membrane side (Fig. 2C) and coated with the omentum; and a Omentum group (n = 5), in which only the omentum was used to cover the serosal membrane side of the ESD site.

Development duodenal ESD model. (A) Make a 15-mm submucosal defect in the descending portion of the duodenum by the ESD method. (B) Macroscopic appearance of duodenum after ESD. The base of ulceration after ESD got very thin to be seen through outside. (C) In the Sheet group, the myoblast sheet was attached to the ESD ulcer part from the duodenal serous membrane side. ESD: endoscopic submucosal dissection.
The Postoperative Evaluation
All pigs began drinking liquids after ESD and started eating solids in the days following the surgery. Animals were observed for 3 days and then sacrificed. An adhesion-severity scoring system was established to measure the degree of intra-abdominal inflammation due to peritonitis (Table 1), in reference to a previous study 18 .
Adhesion Score.

Biochemical Assays of Blood Samples
To evaluate the inflammatory parameters, blood samples were collected from the cervical vein on the first postoperative day and at the beginning of the operation on the third day (Fig. 1). The plasma was separated by centrifuging blood samples at 3,000 rpm for 5 min and then stored at −80°C. Analyses of blood samples were performed using a pig C-reactive protein (CRP) enzyme-linked immunosorbent assay (ELISA) kit (Uscn Life Science Inc., Wuhan, China) and pig tumor necrosis factor-alpha (TNF-α) ELISA kit (Invitrogen).
Histopathology
After the animals were sacrificed, the duodenum was resected, and the ESD areas were harvested for the evaluation and measurement. The specimens were fixed in 10% formalin and embedded in serial-section paraffin blocks, after which they were carefully cut from the paraffin-embedded blocks and stained with hematoxylin and eosin (H&E).
To confirm the attachment of the myoblast sheets to the surface of the duodenum at the ESD serosal site, immunostaining examinations were performed. The sections were deparaffinized with xylene and rehydrated in 99% ethanol. After treatment with preheated a pH 6.0 target retrieval solution (Agilent Technologies, Santa Clara, CA, USA), the slides were made to be with the water for 20 min. The sections were rinsed in phosphate-buffered saline and soaked for 10 min at room temperature in peroxidase-blocking solution (Agilent Technologies). Monoclonal mouse anti-desmin antibody (Agilent Technologies) was used as the primary antibody. The sections were incubated at 4°C overnight. After washing, the sections were treated with horseradish peroxidase–labeled polymer conjugated to goat anti-mouse (Agilent Technologies) at room temperature for 30 min. Sections were then stained with 3,3-diaminobenzidine tetrahydrochloride containing hydrogen peroxide (Agilent Technologies).
Statistical Analyses
All numerical values are expressed as the means with the range. The data were compared using the Mann–Whitney U test, and P values of less than 0.05 were considered to be statistically significant. All statistical analyses were performed with the EZR software program (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). More precisely, it is a modified version of R commander designed to add statistical functions frequently used in biostatistics.
Results
Characteristics of the Myoblast Sheets
We successfully isolated and cultured myoblast cells and confirmed that the cultured cells were stained with desmin by immunostaining (Fig. 3A).
Cultured myoblast sheets were able to be harvested from the temperature-responsive culture dishes. The mean size was approximately 2.5 cm in diameter (Fig. 3B). H&E staining and immunostaining of desmin demonstrated that the fabricated sheets were indeed composed of myoblasts that had retained their cell-to-cell interactive connections, with a sheet thickness of approximately 80 µm (Fig. 3C, D). A flowcytometric analysis showed that the percentage of cells positive for the myoblast cell surface antigen CD56 was 67.1 ± 5.8% (Fig. 3E). These results confirmed that the fabricated cell sheets were composed of myoblasts.
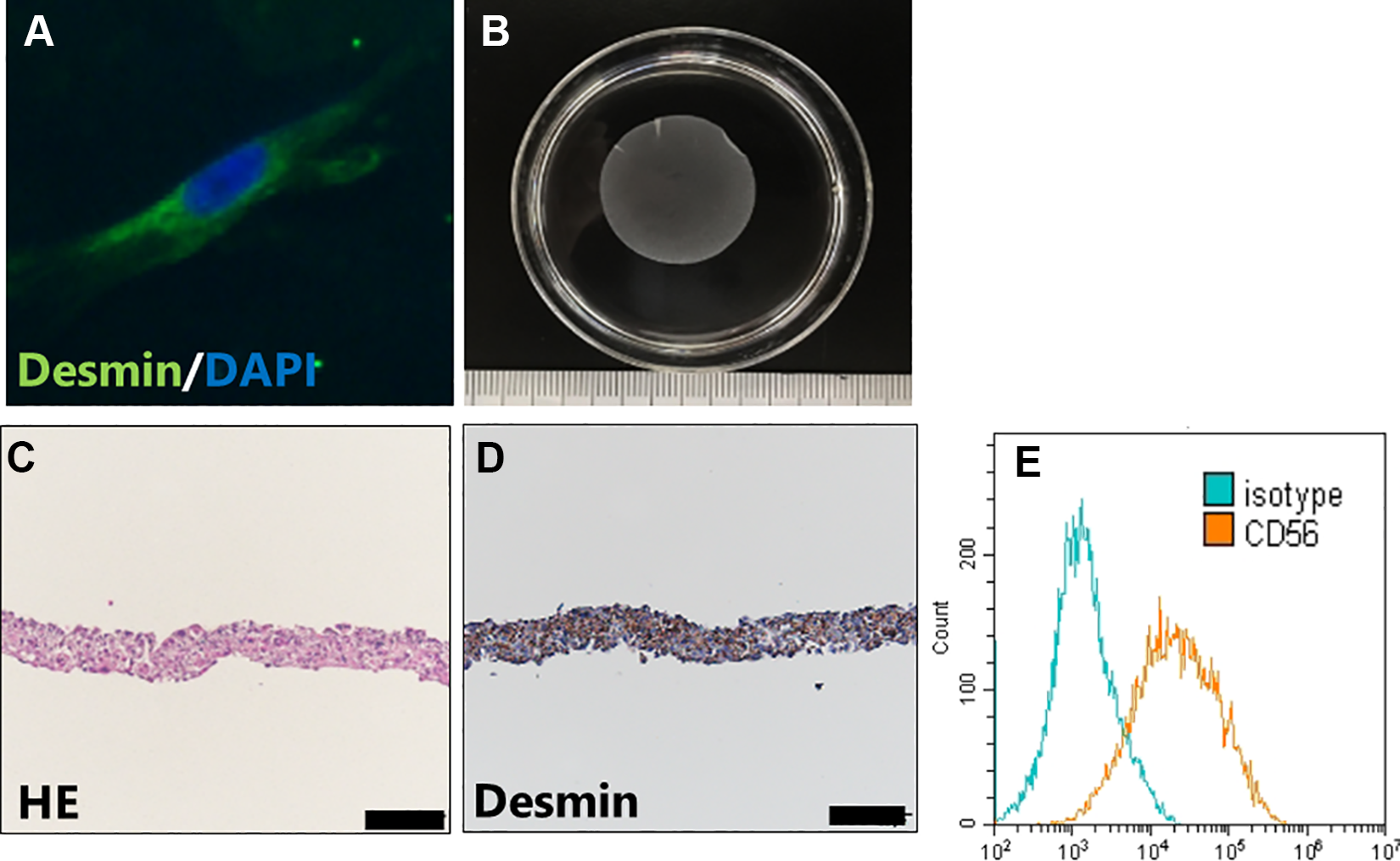
Characteristics of myoblast sheet. (A) Immunofluorescence stained images of myoblast cell special maker desmin. (B) Cultured myoblast sheets were detached from the temperature-responsive culture dishes. Its size is 2.5 cm in diameter. (C) Hematoxylin–eosin stain of myoblast cell sheet. (D) Immunostaining of desmin of myoblast cell sheet (scale bar = 100 μm). The myoblast sheet was composed of myoblasts that had retained their cell-to-cell interactive connections. (E) Flowcytometric analysis of myoblast cell surface antigens of CD56 (orange line) and isotype control (blue line). The percentages of CD56+ cells were 67.1 ± 5.8.
Gene Expression Profile Analyses of Myoblast Sheets
We confirmed that the cultured cells expressed genes specific to myoblasts, such as DES, MyoD, PAX7, and INTGα7 (Fig. 4A). In addition, it was confirmed that the myoblast sheets highly expressed several growth factor-related genes involved in wound healing, including FGF2, TGFβ-1, IGF2, EGF, and VEGF. The expression of these genes was higher than those values noted in fibroblast sheets (Fig. 4B).

Gene expression profile analysis of myoblast and fibroblast sheets. (A) qRT-PCR confirmed the expression of the specific gene of myoblast cell DES, MyoD, INTGα7, and PAX7. (B) mRNAs of growth factors TGFβ1, FGF2, and VEGF of myoblasts showed increased expression as compared to fibroblasts. The data are shown as mean ± SD. *P < 0.05. qRT-PCR: quantitative real time polymerase chain reaction; SD: standard deviation.
Effects of Myoblast Sheets on Perforation
On day three after surgery, laparotomy revealed perforations in the ESD ulcer area in the control group and leakage of bile (Fig. 5A). Perforation was observed on day three after surgery in all cases of the Omentum group, while in the Sheet group, postoperative perforation was prevented in all cases (Fig. 5C). According to the adhesive score analysis, the Sheet group had significantly lower rates of postoperative adhesion than the Omentum group (Fig. 5D).

Macroscopic findings in the abdomen 3 days after the surgery. (A) In Omentum group, perforations in the ESD area and bile leakage are observed. (B) No gross perforation of the ESD part was observed in Sheet group. (C) Percentage of perforation 3 days after the ESD. (D) Score of adhesion. The data are shown as mean ± SD. *P < 0.05, **P < 0.01. ESD: endoscopic submucosal dissection; SD: standard deviation.
Biochemical Assays of Blood Samples
The levels of CRP and TNF-α in blood increased over time after surgery in both the control and Sheet groups compared to before the operation. The CRP level in the Sheet group was significantly lower than that in the Omentum group on day three after surgery (Fig. 6A). The TNF-α level in the Sheet group tended to be lower than that in the Omentum group (Fig. 6B).

Biochemical assay of blood samples. (A) Levels of CRP in blood sample. CRP of the sheet group was significantly lower than the Omentum group on the third day after surgery. (B) Levels of TNF-α in blood sample. TNF-α of the sheet group tended to be lower than that of the Omentum group. The data are shown as mean ± SD. *P < 0.05. CRP: C-reactive protein; SD: standard deviation; TNF-α: tumor necrosis factor-alpha.
Histopathological Findings
In the Omentum group, necrosis and perforation were observed throughout the duodenal wall (Fig. 7A), whereas in the Sheet group, thickening of the intestinal wall of the ESD ulcer was observed, and no perforation was observed (Fig. 7B). Furthermore, immunostaining with desmin confirmed that the myoblast sheet remained in the ESD ulcer portion in the Sheet group (Fig. 7C). Moreover, in the Sheet group, the expression of PAX7, MyoD, and ITGα7, which are genes specific to myoblasts, was observed in the ESD ulcer tissue (Fig. 7D), confirming that the myoblast cells were alive.

Histopathological findings. (A) In the Omentum group, large defect and necrosis are observed throughout the duodenal wall (H&E; scale bar = 2 mm). (B) In the Sheet group, thickening of the intestinal wall of the ESD ulcer was observed, and no perforation was observed (H&E; scale bar = 2 mm). (C) Immunostaining of desmin of the Sheet group. Myoblast sheet was observed in the ESD ulcer area (scale bar = 400 μm). Histopathological findings. (D) Gene expression profile analysis of area of ESD. In the Sheet group, expression of myoblast-specific gene was observed. ΔΔCT method. ESD: endoscopic submucosal dissection; H&E: hematoxylin and eosin.
Discussion
Recent advent of endoscopy has made endoscopic resection such as EMR and ESD curable and safe standard therapy especially for superficial gastric and colorectal cancer. Although endoscopic resection may also be useful as a standard therapy for SNADETs, duodenal ESD carries a substantial risk of delayed perforation due to anatomical characteristics, such as a narrow lumen, abundant vascularity in the submucosal layer, and thin muscle layer. As delayed perforation is the main obstacle for prevalence of endoscopic resection as the standard treatment for SNADETs, we established a novel regenerative approach using myoblast sheets for preventing delayed perforation after ESD for SNADETs in a porcine model.
Several procedures are reported to prevent perforation after endoscopic removal of SNADETs, including cap-assisted EMR 20 , cold snare polypectomy 21 , and underwater EMR 22 . Although these procedures enable the safe and effective resection of small duodenal lesion 1,7,8,23,24 , for larger tumors, ESD is the ideal resection procedure in order to ensure an appropriate margin and removal in an en bloc manner 23 .
In addition to the technical difficulty associated with duodenal ESD, exposure of ulceration to bile and pancreatic juice is a major cause of delayed perforation. To prevent the ulcerated base from suffering chemical injury, Kato et al. suggested the importance of closing the mucosal defect after ESD 16 . They reported in a retrospective study that complete closure of the mucosal defect after duodenal ESD significantly decreased the incidence of delayed perforation in contrast to incomplete and no closure. However, they also described the difficulty of applying clips to approximate mucosal defects due to the maneuverability and location of the defect, especially in the distal or flexural portion of the duodenum. Inoue et al. described cases with delayed perforation despite prophylactic clipping to close mucosal defects due to the clips falling away 15 . Matsuda et al. also reported delayed perforation despite applying clips 25 . Recently, to perform closure for large ulcers after ESD, OTSC system (Century Medical Inc., Tokyo, Japan) have been applied to overcome the issues associated with the low grasping power of conventional clip 26 . However, prophylactic closure using OTSC system is also difficult in the distal portion of the duodenum 14 . Thus far, the effective management of delayed perforation after duodenal ESD has not been fully established.
Hiki et al. reported the efficacy of laparoscopic and endoscopic cooperative surgery (LECS) for gastric submucosal tumor 27 , and many authors have extended the indication of LECS to SNADETs as an alternative to ESD 28,29 . Irino et al. performed endoscopic total-layer resection of a duodenal tumor followed by closure of the defect using a hand-sewn technique in a laparoscopic manner 30 . However, with this procedure, surgeons must close duodenal defects with sophisticated and often painful hand-sewing techniques. In addition, there is a risk of stenosis or involvement of the ampulla of Vater with hand-sewing, along with a risk of dissemination of the tumor itself and the duodenal contents into the abdominal cavity. Although this procedure may also make it possible to securely reinforce the thin wall and prevent uncontrolled perforation after ESD, the establishment of simpler and more effective procedures than LECS is awaited.
The novel “cell sheet” technology from the field of regenerative medicine has recently been applied to the clinical practice. One approach that has shown successful outcomes involves the use of mucosal epithelial cell sheets to cover mucosal defects after ESD for superficial esophageal cancer 31,32 . Ohki et al. demonstrated the preventive effect of esophageal stricture after ESD using autologous buccal cell sheet. They performed a clinical study of 10 patients with superficial esophageal cancer. The transplanted cell sheets composed of the patients’ oral mucosa using a temperature-responsive culture dish were applied to the patients’ post-ESD esophageal ulcers. The feasibility of myoblast sheet implantation for treating cardiomyopathy has also been reported 33 –35 .
In the present study, the myoblast sheets prevented delayed perforation and severe adhesion in the abdomen 3 days after implantation, in contrast to the control group in which the ESD site was covered with only omentum. We also detected massive fibroblasts and collagen fibers among the implanted myoblasts on an immunohistochemical examination, implying the induction of regeneration at the ESD site. In both the Sheet and control groups, omentum was placed at the serosal site of ESD in order to induce a blood supply; however, the blood supply from the omentum was probably sufficient to promote local healing only in the Sheet group, since our in vitro result showed growth factor secretion from the myoblast sheet in an mRNA expression analysis. These soluble factors are also expected to be secreted in vivo and help reinforce the thinned duodenal wall after ESD.
Memon et al. revealed in their in vivo model that secretion of growth factors from implanted myoblast sheets plays a critical role in the recovery of the cardiac function through the induction of regeneration of native cardiomyocytes 36 . They confirmed the increased expression of HGF, VEGF, and SDF-1 in their myoblast sheet transplantation study. Regarding the role of omentum, Shudo et al. reported that transplantation of the cell sheet with omentopexy accelerated the secretion of angiogenesis-related cytokines, such as VEGF, compared to omentopexy only 37 . Kainuma et al. also reported that the combination of cell sheet transplantation and an omental flap acts synergistically on vessel maturation and coronary microcirculation in a rat chronic myocardial infarction model, whereas omentopexy alone failed to stabilize the long-term effect 38 . They speculated that the growth factors and cytokines secreted by the cell sheet stimulate the migration of endothelial cells derived from both the host myocardium and the omentum toward the sheet, resulting in the establishment of a robust vessel connection between the native arteries and the omentum.
In addition to the paracrine effect mentioned above, the barrier effect of the myoblast sheet might also play a key role in the prevention of delayed perforation in our model. We previously reported that myoblast sheets were effective in preventing pancreatic fistula in a rat model 17 . We also demonstrated the efficacy of myoblast sheets for preventing leakage of enteral contents in a gastric perforation rat model 18 . These results showed that myoblast sheets could be tolerated well enough to contain bile and pancreatic juice. In addition, an autologous myoblast sheet would not induce a severe immune response or rejection, which provoke abdominal adhesion and duodenal stenosis. Therefore, an autologous myoblast sheet appears to be an ideal material due to both its barrier function and delivery of growth factor.
Whether or not a myoblast sheet would also effectively prevent delayed perforation when transplanted at the “luminal side” is an important issue to be clarified. Several hurdles hamper verification of this issue at the moment: the establishment of an endoscopic transportation system of the myoblast sheet into the duodenum through a narrow lumen, such as the lower esophageal sphincter and pylorus, without the breakdown of the sheet; and methods for the subsequent successful pasting and engraftment of the myoblast sheet in a harsh environment, such as in the presence of digestive enzymes and under active peristalsis. Feasible endoscopic devices and techniques will need to be developed, and a large animal study should be conducted to obtain proof-of-concept in the future.
Several limitations associated with the present study warrant mention. First, we evaluated only a small number of animals. Second, the precise mechanisms underlying our findings remain to be elucidated. Third, preparing the autologous cell sheet is quite costly, so we must explore ways to fabricate allograft cell sheets or induced pluripotent stem cells (iPS)-derived sheets more cost effectively in the future.
In conclusion, autologous myoblast sheets were useful for preventing perforation after duodenal ESD. A clinical trial to verify the efficacy of autologous myoblast sheets in duodenal ESD is awaited.
Footnotes
Ethical Approval
This study was approved by our local ethics committee and performed according to the guidelines of the Nagasaki University on animal use.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the guidelines of the Nagasaki University on animal use, the National Society for Medical Research, and the National Institute of Health (NIH publication 86-23, revised 1985).
Statement of Informed Consent
Written informed consent was not needed for the animal experiments.
Author Contributions
SE conceived the idea of the study, designed the experiments, and supervised the project. KK performed data quality check and reviewed the manuscript. RM, YM, and SY experimented and wrote the manuscript. SK and DM assisted the experiment. KO, KH, and KN designed and experimented animal experiment. YS fabricated cell sheet devise.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RM, SY, SK, DM, KO, YS, KH, KN, and SE have no conflicts of interest or financial ties to disclose. KK and YM are members of the joint research group between Nagasaki University and TERUMO CORPORATION.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a J-CASE (Japan Consortium for Advanced Surgical Endoscopy) Research Grant in 2018 (SK), The Japanese Foundation for Research and Promotion of Endoscopy Grant in 2018 (SK), and a JSPS Grant-in-Aid for Early-Career Scientists, Grant Number 18K16368 (YM).
