Abstract
Tripartite motif 8 (TRIM8) is a member of the TRIM protein family that has been found to be implicated in cardiovascular disease. However, the role of TRIM8 in myocardial ischemia/reperfusion (I/R) has not been investigated. We aimed to explore the effect of TRIM8 on cardiomyocyte H9c2 cells exposed to hypoxia/reoxygenation (H/R). We found that TRIM8 expression was markedly upregulated in H9c2 cells after stimulation with H/R. Gain- and loss-of-function assays proved that TRIM8 knockdown improved cell viability of H/R-stimulated H9c2 cells. In addition, TRIM8 knockdown suppressed reactive oxygen species production and elevated the levels of superoxide dismutase and glutathione peroxidase. Knockdown of TRIM8 suppressed the caspase-3 activity, as well as caused significant increase in bcl-2 expression and decrease in bax expression. Furthermore, TRIM8 overexpression exhibited apposite effects with knockdown of TRIM8. Finally, knockdown of TRIM8 enhanced the activation of PI3K/Akt signaling pathway in H/R-stimulated H9c2 cells. Inhibition of PI3K/Akt by LY294002 reversed the effects of TRIM8 knockdown on cell viability, oxidative stress, and apoptosis of H9c2 cells. These present findings defined TRIM8 as a therapeutic target for attenuating and preventing myocardial I/R injury.
Keywords
Introduction
Myocardial infarction (MI) is one of the leading causes of morbidity and mortality in patients with coronary heart diseases worldwide 1 . Myocardial ischemia/reperfusion (I/R) is a very complex pathophysiological process that has been demonstrated to be a critical mechanism of MI 2,3 . Although early reperfusion is well acknowledged to provide oxygen and nutrients to the ischemic area, it also has side effects on myocardium which is called myocardial I/R injury 4,5 . These insults significantly diminish the therapeutic benefits of reperfusion. Better understanding of the mechanisms of I/R injury may be helpful for exploring more suitable strategies to minimize myocardial damage.
Myocardial I/R injury is a very complex pathophysiological process with multiple molecular and cellular events, such as ion accumulation, mitochondrial dysfunction, reactive oxygen species (ROS) formation, activation of oxidative stress and inflammation, and apoptosis 6,7 . Among these, ROS are critical mediators in myocardial I/R injury, as evidenced by interventions that enhancement of ROS scavenging protects against reperfusion injury 8 . Enhanced ROS production induces oxidative stress, which may contribute to myocardial injury and cardiomyocyte death 9 . Therefore, it is necessary to develop new strategies for attenuating ROS production and apply them in the clinical patient care.
Tripartite motif 8 (TRIM8), a member of the TRIM protein family, was reported to be involved in various biological processes, such as cell survival, differentiation, inflammation, innate immune response, and apoptosis 10 –13 . Recently, accumulating studies demonstrate that TRIM8 is involved in multiple diseases, including cardiovascular disease. Overexpression of TRIM8 exaggerates cardiac hypertrophy both in vivo and in vitro 14 . In addition, TRIM8 has been found to be involved in hepatic I/R injury 15 . However, the role of TRIM8 in mycardial I/R injury remains unclear. The aim of this study was to investigate the role of TRIM8 in H9c2 cells exposed to hypoxia/reoxygenation (H/R).
Materials and Methods
Cell Culture
Rat cardiomyocyte H9c2 cells obtained from American Type Culture Collection (ATCC, Rockville, MD, USA) were cultured in Dulbecco’s modified Eagle’s medium/F12 medium (Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum (Hyclone, Logan, UT, USA), 100 U/ml penicillin, and 100 mg/ml streptomycin (Sigma-Aldrich, St. Louis, MO, USA) at 37°C.
H/R Model
The protocol of H9c2 cells exposed to H/R stimulation was performed as follows. Briefly, cultured H9c2 cells were subjected to hypoxia in a hypoxic chamber (1% O2, 5% CO2, and 94% N2) for 2 h, followed by 24 h of reoxygenation in a normoxic chamber (95% air and 5% CO2). Cells in control group were kept in normoxic condition. Cell viability, oxidative stress, and apoptosis were examined using the methods described afterward.
Knockdown of TRIM8
For knockdown of TRIM8, the siRNA against TRIM8 (si-TRIM8: GGCGGCCTGTTATATGGAATTTGGA) and negative control siRNA (si-NC: GGGGGGCCCTTTTTTTTGGGGAAAAA) were designed and synthesized by GenePharma (Shanghai, China). Then the siRNAs were transfected into H9c2 cells by Lipofectamine™ 2000 kit (Invitrogen) according to the manufacturer’s instructions.
Rescue Assay
To confirm that PI3K/Akt signaling pathway contributes to the regulation of the TRIM8 inhibition-mediated cardiac-protective effect, H9c2 cells were transfected with si-TRIM8 or si-NC in the presence of LY294002 (10 μM) for 48 h, followed by H/R stimulation.
Overexpression of TRIM8
For overexpression of TRIM8, the cDNA for TRIM8 gene was cloned into a commercial pcDNA3.0 vector (Invitrogen) to generate the TRIM8 overexpressing plasmid pcDNA3.0-TRIM8. The pcDNA3.0-TRIM8 or pcDNA3.0 empty vector was transfected into H9c2 cells by Lipofectamine™ 2000 kit (Invitrogen).
Quantitative Real-time Polymerase Chain Reaction
The total RNA of the H9c2 cells was extracted by RNA extraction kit (Applied Biosystems, Foster, CA, USA) according to the instructions. The reverse transcription was performed using 2 µg of total RNA with a SuperScript III First-Strand Synthesis system (Invitrogen). Quantitative assay of TRIM8 gene expression was performed using a QuantiTect SYBR Green kit (Toyobo, Osaka, Japan) on an ABI Prism 7700 Sequence Detection System (Applied Biosystems). The gene expression was calculated by the d2−ΔΔCt method. The specific primer sequences were—TRIM8 F: 5′-GAC GGA TTC ACG GAC AGT AA-3′, R: 5′-TTG ATG CTG GCC AGG C-3′; β-actin F: 5′-GGG AAA TTC AAC GGC ACA GT-3′, R: 5′-AGA TGG TGA TGG GCT TCC C-3′.
Cell Viability Assay
Cell viability was assessed by 3-(4,5-dimethylthiazol 2-yl)-2,5-(diphenyltetrazolium bromide) (MTT) assay. Briefly, H9c2 cells were seeded into 96-well plates (5 × 103 cells/well). Overnight, 20 µl of MTT (5 mg/ml) was then added to each well and incubated for additional 4 h at 37°C. After that, the medium was removed and 150 µl dimethyl sulfoxide was added into each hole. Finally, the absorbance was recorded at 490 nm by using a microplate reader (Bio-Rad Laboratories, Hercules, CA, USA).
Measurement of Intracellular ROS
The detection of intracellular ROS levels was depended on the fluorescent probe 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA), which is a cell-permeable indicator for ROS. In brief, H9c2 cells were incubated with 10 μM of DCFH-DA for 20 min at 37°C. Then, the fluorescence of formed DCF was visualized under a fluorescent microscope with excitation/emission set at 502/523 nm.
Western Blot
H9c2 cells were homogenized with ice-cold RIPA lysis buffer (Beyotime, Shanghai, China). Protein concentration in the lysates was measured according to the Bradford method using a commercial kit (Beyotime). Equal amounts of protein were proceeded to 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and then transferred electrophoretically to polyvinylidene difluoride membranes (Thermo Fisher Scientific, Waltham, MA, USA). After blocking with 5% nonfat milk for 1 h at room temperature, the membranes were incubated with the specific primary antibodies against bax, bcl-2, p-PI3 K, PI3 K, p-Akt, Akt, and β-actin (Invitrogen) diluted in blocking buffer at 4°C overnight. Following washing three times with tris-buffered saline Tween-20, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies (Invitrogen) for 1 h at 37°C. The proteins signals were visualized using an enhanced chemiluminescent detection kit (Pierce, Rockford, IL, USA). The optical density was analyzed using Bio-Image Analysis System (Bio-Rad Laboratories).
Enzyme-linked Immunosorbent Assay
The levels of superoxide dismutase (SOD), glutathione peroxidase (GPx), and caspase-3 in lysates were measured using commercial assay kits purchased from MyBioSource (San Diego, CA, USA) according to the manufacturer’s instructions.
Statistical Analysis
Quantitative results were analyzed by GraphPad Prism version 6.0 (GraphPad Software, Inc., San Diego, CA, USA) from at least three separate experiments and expressed as mean ± standard deviation. Comparisons were made by Student’s t-tests or one-way analysis of variance with Tukey’s post hoc tests. P-value less than 0.05 was considered statistically significant.
Results
The Expression of TRIM8 was Upregulated in H9c2 Cells Exposed to H/R
H9c2 cells were exposed to hypoxia condition for 2 h, followed by reoxygenation for another 24 h. The results showed that expression of TRIM8 at both mRNA and protein levels was significantly upregulated by H/R treatment compared to the control H9c2 cells (Fig. 1A, B).

TRIM8 expression in H9c2 cells with or without exposure to H/R. (A and B) The expression levels of TRIM8 in H9c2 cells were measured using qRT-PCR and western blot after incubation in hypoxia condition for 2 and 24 h of reoxygenation condition. Data are presented as mean ± SD. n = 5. *P < 0.05. H/R: hypoxia/reoxygenation; qRT-PCR: quantitative real-time polymerase chain reaction; SD: standard deviation; TRIM8: Tripartite motif 8.
Knockdown of TRIM8 Improved the Viability of H9c2 Cells Exposed to H/R
Subsequently, we explored the role of TRIM8 in H/R-treated H9c2 cells through transfection with si-TRIM8. The western blot revealed that TRIM8 protein expression was markedly decreased by si-TRIM8 in H9c2 cells, as compared with the si-NC group (Fig. 2A). Cell viability of H9c2 cells exhibited a significant reduction after H/R exposure, while TRIM8 knockdown caused a recovery of cell viability (Fig. 2B).
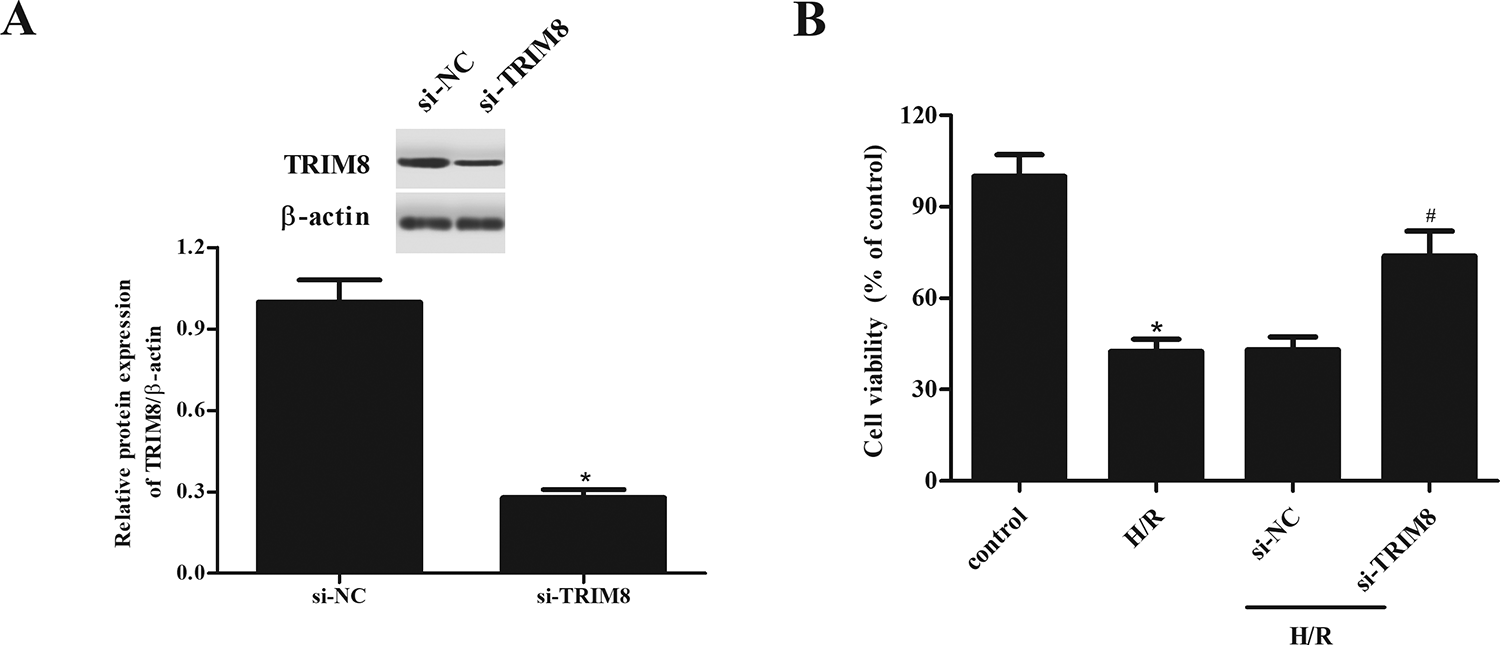
Improvement of H9c2 cells viability by TRIM8 knockdown exposed to H/R. (A) Transfection efficiency was confirmed by western blot after transfection with si-TRIM8 and si-NC. (B) Cell viability of H9c2 cells was detected by MTT assay. Data are presented as mean ± SD. n = 4. *P < 0.05 vs control group, # P < 0.05 vs H/R + si-NC group. H/R: hypoxia/reoxygenation; MTT: 3-(4,5-dimethylthiazol 2-yl)-2,5-(diphenyltetrazolium bromide); SD: standard deviation; si-NC: negative control siRNA; TRIM8: Tripartite motif 8.
Downregulation of TRIM8 Inhibited H/R-Induced Oxidative Stress in H9c2 Cells
At the end of reperfusion, as shown in Fig. 3A, ROS production was significantly increased by H/R treatment, as compared with control H9c2 cells, which was markedly attenuated by downregulation of TRIM8. In addition, downregulation of TRIM8 also induced an improvement in activities of SOD and GPx when compared with H/R group (Fig. 3B, C).
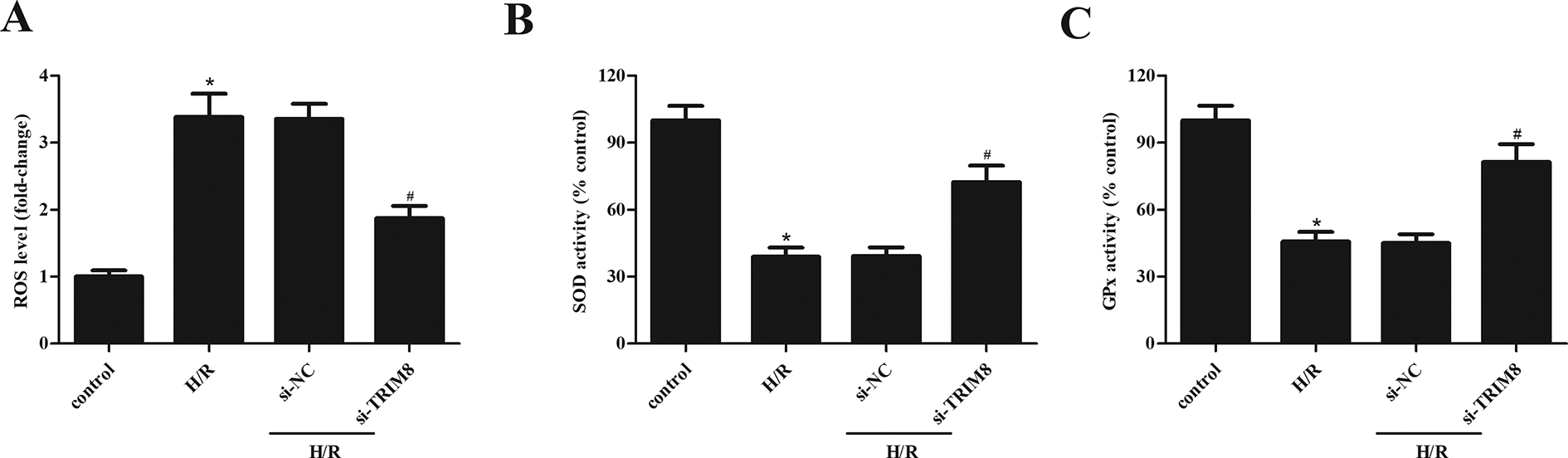
Inhibitory effect of TRIM8 downregulation on H/R-induced oxidative stress in H9c2 cells. (A) TRIM8 downregulation suppressed ROS production. (B and C) TRIM8 downregulation elevated the activities of SOD and GPx. Data are presented as mean ± SD. n = 6. *P < 0.05 vs control group, # P < 0.05 vs H/R + si-NC group. GPx: glutathione peroxidase; H/R: hypoxia/reoxygenation; ROS: reactive oxygen species; SD: standard deviation; si-NC: negative control siRNA; SOD: superoxide dismutase; TRIM8: Tripartite motif 8.
TRIM8 Knockdown Inhibited Apoptosis in H9c2 Cells After H/R
Next, we found that H/R-caused increase in caspase-3 activity was suppressed by TRIM8 knockdown in H9c2 cells (Fig. 4A). Meanwhile, the bcl-2 expression was decreased, while bax expression was increased in response to H/R, when compared with control cells. However, H/R-induced changes in bcl-2 and bax expressions were significantly reversed by knockdown of TRIM8 (Fig. 4B–D).

Inhibitory effect of TRIM8 downregulation on H/R-induced apoptosis in H9c2 cells. (A) TRIM8 downregulation decreased caspase-3 activity. (B) TRIM8 downregulation caused changes in bax and bcl-2 expressions. (C and D) Quantification analysis of bax and bcl-2. Data are presented as mean ± SD. n = 4. *P < 0.05 vs control group, # P < 0.05 vs H/R + si-NC group. H/R: hypoxia/reoxygenation; SD: standard deviation; si-NC: negative control siRNA; TRIM8: Tripartite motif 8.
TRIM8 Promoted H/R-Induced Oxidative Stress and Apoptosis in H9c2 Cells
To further study the function of TRIM8 in H/R-treated H9c2 cells, we overexpressed TRIM8 in H9c2 cells by transfection with pcDNA3.0-TRIM8, which was confirmed by western blot (Fig. 5A). MTT assay results disclosed that TRIM8 overexpression promoted the decreased cell viability upon H/R induction (Fig. 5B). The H/R-induced production of ROS and increase in caspase-3 activity were enhanced by TRIM8 overexpression (Fig. 5C, D).

TRIM8 overexpression exhibited opposite effects of TRIM8 downregulation on H/R-induced oxidative stress and apoptosis in H9c2 cells. (A) Transfection efficiency was confirmed by western blot after transfection with pcDNA3.0-TRIM8 or pcDNA3.0. (B) Cell viability of H9c2 cells was detected by MTT assay. (C and D) Effects of TRIM8 overexpression on ROS production and caspase-3 activity. Data are presented as mean ± SD. n = 4. *P < 0.05 vs control group, # P < 0.05 vs H/R + pcDNA3.0 group. H/R: hypoxia/reoxygenation; MTT: 3-(4,5-dimethylthiazol 2-yl)-2,5-(diphenyltetrazolium bromide); ROS: reactive oxygen species; SD: standard deviation; TRIM8: Tripartite motif 8.
TRIM8 Knockdown Enhanced the Activation of PI3K/Akt Signaling Pathway in H/R-Stimulated H9c2 Cells
To further explore the mechanism of TRIM8, we studied the effect of TRIM8 knockdown on PI3K/Akt signaling pathway through detecting the expressions of PI3 K, p-PI3 K, Akt, and p-Akt. As shown in Fig. 6, H/R treatment significantly decreased the expression levels of p-PI3 K and p-Akt in H9c2 cells, as compared with the control group. However, knockdown of TRIM8 greatly enhanced the activation of PI3K/Akt signaling pathway in H/R-stimulated H9c2 cells.

Enhancement of PI3K/Akt signaling pathway by TRIM8 knockdown in H/R-stimulated H9c2 cells. (A) The expressions of PI3 K, p-PI3 K, Akt, and p-Akt were measured using western blot after different treatments. (B) The ratio of p-PI3K/PI3 K. (C) The ratio of p-Akt/Akt. Data are presented as mean ± SD. n = 5. *P < 0.05 vs control group, # P < 0.05 vs H/R + si-NC group. H/R: hypoxia/reoxygenation; SD: standard deviation; si-NC: negative control siRNA; TRIM8: Tripartite motif 8.
Inhibition of PI3K/Akt Reversed the Effects of TRIM8 Knockdown on H9c2 Cells
In addition, LY294002 was used to block the activation of PI3K/Akt signaling pathway. As shown in Fig. 7A, the improvement of cell viability in H/R + si-TRIM8 group was lost in cells treated with LY294002. Inhibition of PI3K/Akt signaling pathway was also shown to prevent the antioxidative and antiapoptotic effects of si-TRIM8 in H/R-stimulated H9c2 cells, as evidenced by increased ROS production and caspase-3 activity (Fig. 7B, C).
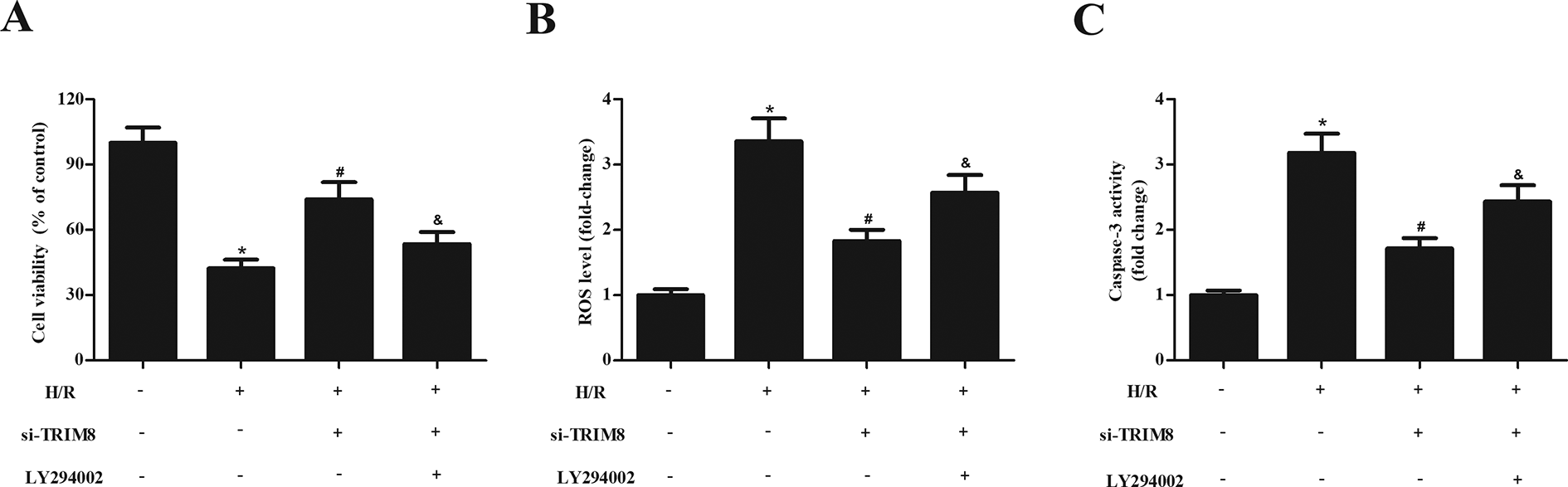
Effects of LY294002 on cell viability, oxidative, and apoptosis in si-TRIM8-transfected H9c2 cells exposed to H/R. (A) Effects of LY294002 on cell viability. (B and C) Effects of LY294002 on ROS production and caspase-3 activity. Data are presented as mean ± SD. n = 4. *P < 0.05 vs control group, # P < 0.05 vs H/R group, & P < 0.05 vs H/R + si-TRIM8 group. H/R: hypoxia/reoxygenation; ROS: reactive oxygen species; SD: standard deviation; TRIM8: Tripartite motif 8.
Discussion
I/R-induced injury has been described as one of the main factors that contribute to the observed morbidity and mortality in MI. ROS have been found to play a key role in the pathophysiology of I/R injury and mediate injury to the insulted tissues 16 . During the reperfusion stage of an ischemic tissue, a burst of ROS is produced due to the abundance of oxygen supply 17 . The excessive production of ROS induces oxidative stress, which can result in direct cytotoxic effects. Besides, the generated oxidative stress also induces the production of ROS, as well as the formation of inflammatory mediators through redox-mediated signaling pathways, leading to post I/R inflammatory injury 18 . These oxidative and inflammatory responses may cause cell apoptosis. Therefore, the methods eliminating the I/R injury include: preconditioning techniques, and minimizing the oxidative stress during reperfusion with the use of antioxidants, anti-inflammatory agents, and scavengers for ROS.
TRIM8 is a member of the TRIM family that is mostly ubiquitous in murine and human tissues 10 . TRIM8 plays an important role in response to various physiological and pathological conditions. Blocking of TRIM8 protects against lipopolysaccharide-induced acute lung injury in mice through its anti-inflammatory and antioxidative activities with decreased ROS production; increased SOD; and lessened IL-1β, IL-6, and TNF-α expression in lung tissues 19 . Additionally, TRIM8 was found to be upregulated in liver of mice subjected to hepatic I/R injury. TRIM8 deficiency relieves hepatocyte injury triggered by I/R. Silencing of Trim8 expression alleviates hepatic inflammation responses and inhibits apoptosis in vitro and in vivo 15 . TRIM8 overexpression exaggerates cardiac hypertrophy in pressure overload–induced mice and Ang II–induced cardiomyocyte hypertrophy in vitro 14 . Thus, we speculated that TRIM8 might be involved in myocardial I/R injury. Our results showed that TRIM8 expression was markedly upregulated in H9c2 cells after stimulation with H/R. Knockdown of TRIM8 improved the cell viability and inhibited oxidative stress and apoptosis of H9c2 cells, while TRIM8 overexpression exhibited apposite effects.
The PI3K/Akt signaling pathway is a conserved pathway to many aspects of cell growth and survival, in physiological as well as in pathological conditions 20,21 . Recent studies have identified that PI3K/Akt is crucial for limiting oxidative stress, proinflammatory, and apoptotic events in response to I/R stimuli 22 –24 . It has been well documented that activation of PI3K/Akt is associated with decreased myocardial ischemic injury. PI3K/Akt activation inhibits cardiomyocytes apoptosis induced by hypoxia, and it protects hearts against I/R injury 25 –27 . Troxerutin reduces myocardial infarct size, improves cardiac function, and decreases the levels of inflammatory cytokines as well as some apoptosis markers in a myocardial I/R injury model in rats via activating PI3K/Akt pathway 28 . Urolithin A reduces myocardial infarct size and cell apoptosis, and enhances antioxidant capacity in mice after I/R through PI3K/Akt pathway 29 . In the current study, we aimed to evaluate the role of PI3K/Akt pathway in the protective effects of TRIM8 on H/R-stimulated H9c2 cells. The data showed that knockdown of TRIM8 enhanced the activation of PI3K/Akt signaling pathway in H/R-stimulated H9c2 cells. While inhibition of PI3K/Akt by LY294002 reversed the effects of TRIM8 knockdown on H9c2 cells, implying that the protective effects of TRIM8 knockdown were mediated by PI3K/Akt signaling pathway.
Conclusion
In summary, the present study has demonstrated that knockdown of TRIM8 has protective effects on H/R-stimulated H9c2 cells with improvement of cell viability, decreased ROS production, increased antioxidants, and decreased cell apoptosis markers. Knockdown of TRIM8 protected H9c2 cells against H/R stimulation through the activation of PI3K/Akt signaling pathway. These present findings defined TRIM8 as a crucial mediator in myocardial I/R injury, thus providing a potentially novel therapeutic target for attenuating and preventing MI. However, to translate our present discoveries into clinical usage, future in vivo studies need to be addressed.
Footnotes
Ethical Approval
This study was approved by the Ethics Committee at The Second Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with The Second Affiliated Hospital of Xi’an Jiaotong University of Ethics Committee’s or Institutional Review Board’s (Approval Number: 00024) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Key Project of Research and Development Plan of Shaanxi Province in China (No.: 2019SF-167).
