Abstract
A number of degenerative conditions affecting the neural retina including age-related macular degeneration have no successful treatment, resulting in partial or complete vision loss. There are a number of stem cell replacement strategies for recovery of retinal damage using cells from variable sources. However, literature is still deficit in the comparison of efficacy of types of stem cells. The purpose of the study was to compare the therapeutic efficacy of undifferentiated cells, i.e., lineage negative stem cells (Lin-ve SC) with differentiated neurosphere derived from ciliary epithelium (CE) cells on retinal markers associated with laser-induced retinal injury. Laser-induced photocoagulation was carried out to disrupt Bruch’s membrane and retinal pigmented epithelium in C57BL/6 mouse model. Lineage negative cells were isolated from human umbilical cord blood, whereas neurospheres were derived from CE of post-aborted human eyeballs. The cells were then transplanted into subretinal space to study their effect on injury. Markers of neurotropic factors, retina, apoptosis, and proliferation were analyzed after injury and transplantation. mRNA expression was also analyzed by real-time polymerase chain reaction at 1 week, and 3-month immunohistochemistry was evaluated at 1-week time point. CE cell transplantation showed enhanced differentiation of rods and retinal glial cells. However, Lin-ve cells exerted paracrine-dependent modulation of neurotrophic factors, which is possibly mediated by antiapoptotic and proliferative effects. In conclusion, CE transplantation showed superior regenerative outcome in comparison to Lin-ve SC for rescue of artificially injured rodent retinal cells. It is imperative that this source for transplantation may be extensively studied in various doses and additional retinal degeneration models for prospective clinical applications.
Keywords
Introduction
Degenerative conditions of the retina, viz. retinitis pigmentosa, age-related macular degeneration, diabetic retinopathy, glaucoma, etc., do not have any successful treatment to reverse the vision loss. Their widespread use in industrial, medical, and military fields has caused severe vision loss in a number of individuals 1 . Laser injuries are also a part of severe retinal damage, as a result of occupational eye injuries 2 .
Cell-based therapies may provide treatment avenues for injuries as well as degenerative disorders by using reproducible laser injury models. Current ongoing stem cell work findings have also raised hopes of individuals suffering from such untreatable disorders 3 –5 . Stem cells from different sources such as neural stem cells, retinal progenitors, hematopoietic, mesenchymal, embryonic, and induced pluripotent stem cells (iPSCs) have been studied for their differentiation potential into neuronal, retinal as well as glial cell lineages 3,6,7 . Stem cell therapies for targeting such chronic diseases may provide effective solutions. However, the field suffers from lack of comparative analysis of the various types of stem cells even though there is overemphasis on iPSCs 8 and embryonic stem cells 9 without any comparative studies between the two and/or other types of stem cells.
Recent studies show that stem cell transplantation at the site of neural injury or neuronal niche may facilitate differentiation into neurons, resulting in functional improvements in animal models 10,11 . Although the use of stem cells derived from umbilical cord blood (UCB) has been employed with several injury/disease models involving the central nervous system, it is limited as compared to the burgeoning number of cord banks being set up in the Asian countries 12 . The study tested the effect of UCB-derived stem cells in laser-induced injury in rabbit trabecular meshwork 13 . Similarly, the efficacy of transplantation of ciliary epithelium (CE) cells has not been adequately investigated let alone compared with other cell types despite repeated failure to regenerate the damaged or diseased retina. However, several groups have identified and continued with studies of transplantation of CE stem cells in retinal injury models 14,15 .
In our previous study, we characterized human umbilical cord blood (hUCB)-derived lineage negative stem cells (Lin-ve SC) on the basis of morphology and cell surface marker expression 16 . We identified these cells under scanning electron microscopy and reported homogenous morphology, i.e., size, shape, and structure as compared to lineage positive and mononucleated cells. Lin-ve SC showed significantly increased expression of hematopoietic stem cells expression, i.e., CD117 and CD34, which ameliorated amyloid-induced memory loss upon transplantation in mouse model. The presence of CD34+ and CD117+ in Lin-ve SC showed 99% positive expression of CD45 marker 17 . These cells exerted neurotrophic factors mediated paracrine effects causing antiapoptotic activity and amyloid clearance by the activation of astrocytes 18,19 .
As lasers have been used for creating the models of retinal degeneration in fish, rodents, and primates 20,21 , we established a modified animal model, injuring the Bruch’s membrane and the surrounding retinal pigment epithelium (without causing choroidal neovascularization). CE cell–derived neurospheres were cultured under in vitro conditions using recombinant fibroblast growth factor (rhFGF) and recombinant epidermal growth factor (rhEGF), which showed increased size and number on sixth day of culture 22 . These neurospheres were dissociated and transplanted in the subretinal space of mice after laser injury. This resulted in marked increase in neurotrophic marker expression.
This study describes the comparative efficacy of differentiated human fetal CE-derived neurospheres and undifferentiated Lin-ve hUCB-derived stem cells, in the subretinal space of laser-induced retinal injury mouse model. We wanted to understand which source provides effective outcomes. Our results showed that the transplantation of CE cells presumably differentiated it into rods and retinal ganglion cells (RGCs) after migration from subretinal space. However, Lin-ve SC rescued the retinal damage by inducing neurotrophic factors, which may exert antiapoptotic and proliferation activity. Hence, we conclude that differentiated (CE cells) cells can be a superior source for cell transplantation therapy in comparison to undifferentiated (Lin-ve SC) cells after laser injury.
Materials and Methods
Animals
C57BL/6 J syngeneic mice (N = 3 per group) of 6- to 8-week-old male were used in the study after the approval from Institutional Animal Ethics Committee (IAEC), Post Graduate Institute of Medical Education and Research, Chandigarh, India [71(69)/IAEC/423]. Animals were kept in animal house facility in a 12-h light/dark cycle (LD 12:12). Mice were fed with standard chow diet and access them freely to clean drinking water.
Laser-Induced Retinal Injury in Mouse Model
The laser-induced retinal injury was established using Argon green laser (532 nm, Iris Medical, USA). The C57 mouse was anesthetized with xylazine hydrochloride (10 mg/kg) (Sigma-Aldrich, St. Louis, MO, USA) and ketamine hydrochloride (100 mg/kg) (Nirlife, Sachana, Gujarat, India) in 1:10 ratio. Mice were kept over heating pad to prevent cold cataract. The local anesthesia, i.e., lignocaine solution, was given to the cornea and 1% tropicamide solution (Akums Drugs and Pharmaceuticals Ltd, Haridwar, Uttarakhand, India) applied for pupil dilation. The fundus shots were obtained through slit lamp by placing the anesthetized mice in front of laser photo-coagulator (IRIDEX, Mountain View, CA, USA). Eight even laser spots in circular fashion were subjected around optic disk in both the eyes. The standardized parameters used in laser photocoagulation were spot size of 100 μm, power of 200 mW, and duration of pulse of 100 ms. Sham control group was operated for subretinal injection without injecting any liquid/vehicle.
Isolation of Lin-ve Stem Cells from hUCB for Subretinal Transplantation
Human UCB samples were obtained after the approval from Institutional Committee for Stem Cell Research and Therapy (IC-SCRT: Approval No. PGI-ICSCRT-53-2014/1469), Post Graduate Institute of Medical Education and Research, Chandigarh-India. Pregnant women of age 20–35 years and ≥28 weeks of gestation period were included in the study. UCB was taken from the umbilical cords of newborns after filling proper informed consent, in the presence of an independent witness. The collected UCB was layered over histopaque solution (Sigma-Aldrich) for enrichment of MNCs using density gradient centrifugation. These MNCs were then subjected to magnetic associated cell sorter (MACS) (Miltenyi Biotech, Bergisch Gladbach, Germany) for the isolation of Lin-ve SC using human Lin-ve isolation kit (Miltenyi Biotech). The kit works on the basis of biological affinity of biotin and streptavidin. The primary antibody cocktail consisted of biotinylated monoclonal antibodies, which include CD123, CD235a (Glycophorin A), CD56, CD19, CD16, CD15, CD14, CD11b, CD3, and CD2.
MNCs were suspended in MACS-BSA buffer and 10 µl of biotin antibody cocktail was added per 107 cells. These were incubated for 15–20 min at 4–8°C and tapped/gently agitated regularly. The cells were then incubated with the second solution, i.e., streptavidin coated magnetic microbeads. Twenty microliters of these microbeads per 107 cells was added and incubated again with gentle tapping for 20–30 min at 4–8°C. The cells were then diluted with 1–2 ml of the buffer, mixed well, and washed twice at 1,500 rpm for 5 min at 4°C. Supernatant was completely pipetted out. Up to 108 cells were suspended in 500 µl of freshly made PBS/MACS-BSA buffer. The cells were then subjected to magnetic separation. The magnetic separation unit along with magnetic separation column was kept in a laminar hood and the column was washed with appropriate amount of buffer solution twice. This was shifted to a fresh collection tube, and the cell suspension was passed through the column at a very slow and steady rate, taking care that there are no air spaces/bubbles inside the column. One milliliter of the buffer was then passed through the column twice to completely elute the cells of interest. Turbid white suspension was obtained as flow through.
Ciliary Epithelial Isolation and Culture
Human fetal eye globes were obtained from abortus after legal termination of pregnancy up to 20 weeks of gestation in accordance to ethical guidelines approved by IC-SCRT (Approval no. PGI-ICSCRT-53-2014/1469). The samples were collected from the terminations at the MTP-OT, SLR, and the Emergency-OT, PGIMER, Chandigarh.
Appropriate informed consents were obtained for every donor, on a prescribed consent form, and all the detailed procedures and objectives related to sample collection were explained to them. All the donors were screened and the following conditions were noted before collection of eye samples:
Inclusion Criteria
The samples were obtained of mid-trimester and up to 20 weeks of fetal abortions. This was on suggestion from the experts at both Department of Obstetrics and Gynecology and the Institutional Committee on Stem Cell Research (IC-SCR), PGIMER, Chandigarh.
Exclusion Criteria
Hepatitis virus B and C Human immunodeficiency virus Samples bearing congenital abnormalities Any malformation of fetus effecting head Evidence of chorioamnionitis (fever, foul smell liquor) Intrauterine fetal death
The fetal eyes were carefully enucleated immediately after abortion and transported in an ice cold sterile Hanks’ Balanced Salt Solution (HBSS) (Gibco, Grand Island, NY, USA). The surgical procedure was performed under stereozoom microscope (Leica EZ4, Leica Microsystems, Wetzlar, Germany) in order to isolate CE. Eye was held with forceps and a cut at the anterior edge of pars plana was made in order to carefully get the strip of ocular tissue containing CE. Ciliary rings were isolated and washed with sterile HBSS without taking nonpigmented epithelium, retinal pigmented epithelium (RPE), iris as well as retina. CE was dissociated first mechanically using blade and trypsinized using 4–5 ml of 0.25% trypsin (Gibco, Life Technologies, Burlington, ONT, Canada) with ethylenediaminetetraacetic acid at 37°C for 20–30 min. The action of trypsin was further neutralized with advanced Dulbecco’s modified Eagle’s medium/F12 (Gibco). The unwanted debris was removed by 0.70 µm cell strainer (BD Biosciences, Discovery Labware, Durham, NC, USA) and then centrifuged at 800G for 10 min. The number of pigmented cells was estimated with hemocytometer (Hausser Scientific, Horsham, PA, USA) and also with automatic cell counter (Millipore, Darmstadt, Germany). Finally, 3,000 cells/well were cultured in 96-well plate using retinal culture medium containing advanced DMEM/F12 (Gibco), 2 mM
Transplantation of hCE-Derived Neurospheres and hUCB Lin-ve Stem Cells
We labeled our transplanted cells with fluorescent dye–carboxyfluorescein succinimidyl ester (CFDA-SE) (Sigma-Aldrich) in order to track the Lin-ve SC population and the CE-derived neurospheres after transplantation. For CFDA staining, cells were counted and 50,000 were suspended in 1 µl of PBS, then transplanted after 24 h of injury through transcorneal subretinal route. The endpoint analysis was carried out at 1 week and 3 months after transplantation. The subretinal injection in mice was performed under surgical microscope after giving anesthesia. The prick was made at the cornea–scleral junction with beveled 31G insulin needle and pressure was released without injuring the cornea. A microsyringe (EXMIRE, Fuji, Shizouka, Japan) of 33G was introduced through the pricked area and moved behind the lens and moved till it touched the retina without exerting pressure on it. One milliliter suspension of 50,000 cells was injected carefully and bleb formation was observed. This served as a marker for successful subretinal delivery.
Immunohistochemistry
The eye balls were enucleated after sacrifice and frozen at −80°C and cryosectioned (Leica Cryostat, Buffalo Grove, IL, USA). Immunohistochemistry was performed to analyze the protein expression changes in the retinal layers viz. rhodopsin (Santa Cruz Biotechnology, Dallas, TX, USA), Thy1 (Santa Cruz Biotechnology), brain-derived neurotrophic factor (BDNF; Santa Cruz Biotechnology), and ciliary neurotrophic factor (CNTF; Santa Cruz Biotechnology). The sections were fixed using HistoChoice (Sigma-Aldrich) and then incubated with primary antibody (1:100 dilution) at 4°C overnight. Next day, the sections were kept in Cy3 labeled secondary antibody solution (1:200 dilution) (Jackson Immunoresearch, West Grove, PA, USA) for half an hour and nuclei were counterstained with 4′,6-diamidino-2-phenylindole (Sigma-Aldrich) (1:1,000 dilution) (Table 1). The sections were subsequently imaged under a confocal microscope to analyze the protein expression.
List of Antibodies Used in Immunohistochemical Experiments Real-Time PCR.

BDNF: brain-derived neurotrophic factor; CNTF: ciliary neurotrophic factor; PCR: polymerase chain reaction.
Real-Time Polymerase Chain Reaction
Relative fold change in the expression of the mRNA levels was detected using real-time polymerase chain reaction (PCR). The RNA was isolated from the dissected retinae of the enucleated eye balls, and converted to cDNA libraries, using standard kit protocols (Qiagen, Velno, The Netherlands and Thermo Fisher Scientific, Waltham, MA, USA). Real-time PCR was performed using primers for rhodopsin (Eurofins, Bangalore, India), Thy1 (Eurofins), BDNF (Sigma-Aldrich, New Delhi, India), CNTF (Sigma-Aldrich, New Delhi, India), BCl2 (Eurofins), and Ki67 (Eurofins). β-Actin (Sigma-Aldrich, New Delhi, India) was used for an endogenous housekeeping control (Table 2). Expression levels were quantified and analyzed using StepOne, Applied Biosystems real-time PCR software (Thermo Fisher Scientific).
List of Genes Analyzed and Their Primer Sequences Used for Real-Time PCR.

BDNF: brain-derived neurotrophic factor; CNTF: ciliary neurotrophic factor; PCR: polymerase chain reaction.
Statistical Analysis
Data were analyzed by calculating mean ± standard error of the mean, and normality of data was analyzed using 1-KS sampling test. The data were analyzed by one-way analysis of variance (ANOVA) followed by LSD and Scheffe and Dunnett’s test for post hoc analysis. Statistical analysis of results was analyzed using 16.0 version of SPSS. *P ≤ 0.05, **P ≤ 0.01, and ***P ≤ 0.001 were considered as statistically significant.
Results
CE Cell Transplantation Enhances the Formation of Rod Cells
We used laser-induced retinal injury mouse model without causing CNV. For this, we targeted eight shots in a circular pattern around the optic disc. These shots were focused at the Bruch’s membrane, which resulted in the damage to RPE and caused the breach of blood retinal barrier. We validated this model using fundus fluorescein angiography showing leakage in the retinal blood vessels as well as electroretinogram that showed changes in the wave pattern 22 . RPE plays crucial role in providing nutrients to the retina and phagocytosis of photoreceptor’s outer segment for its renewal 23 . Therefore, we wanted to analyze the rhodopsin expression, a pigment that makes up rod photoreceptors. The immunohistochemistry for its expression showed significant reduction in rhodopsin levels in mice injured with laser, in comparison to the control mice. We then transplanted two types of cells, i.e., sixth day dissociated neurospheres differentiated from CE cells or undifferentiated Lin-ve SC derived from human UCB. The protein expression of rhodopsin at 1 week was found to be significantly higher in the retina transplanted with CE differentiated cells in comparison to the group transplanted with Lin-ve SC (Fig. 1A, B). The mRNA expression of rhodopsin was also estimated at 1 week and 3 months after transplantation. We found increased expression of rhodopsin in CE cells transplanted group as compared to Lin-ve SC transplanted group (Fig. 1C).

Differentiated CE cells transplantation enhances rod cells expression. (A) Immunohistochemistry of rhodopsin showing Cy3 (red fluorescence marked by arrows) expression bound to primary antibody at 20× visualized under confocal microscope in normal control, laser injured retina mice, laser injured transplanted with Lin-ve SC mice, and laser injured transplanted with CE cells mice. (B) Quantitative protein expression of rhodopsin at 1-week time point measured by corrected total cell fluorescence of immunohistochemistry images using Image J software. (C) mRNA expression of rhodopsin in all four groups was analyzed using real-time PCR at 1 week and 3 months after transplantation. Statistical analysis was performed using one-way ANOVA test for immunohistochemistry and real-time PCR results. This was followed by post-hoc analysis using LSD, Scheffe, and Dunnett’s test. *P ≤ 0.05 and **P ≤ 0.01 were regarded as statistically significant. ANOVA: analysis of variance; CE: ciliary epithelium; Lin-ve SC: lineage negative stem cells; PCR: polymerase chain reaction.
Differentiated CE Neurospheres Increase the Retinal Ganglion Cells (RGCs) of Retina
RGCs are a type of neurons present on the inner layer of retina. They transmit the visual stimulus at photoreceptor layer by engaging two types of neurons, i.e., bipolar and amacrine cells 24 . As rhodopsin was found to be significantly altered by laser-induced injury therefore, we wanted to analyze the effect of injury as well as transplantation of Lin-ve SC on RGCs. We found reduction of Thy1 protein expression after laser-induced retinal injury. However, transplantation of CE cells induced the upregulation of Thy1 expression (in comparison to control and laser injured mice) evident from quantitative analysis of immunohistochemical staining carried out at 1 week after transplantation (Fig. 2A, B). The mRNA expression of Thy1 showed significant increase after Lin-ve SC transplantation at 1 week, but this expression was abolished significantly when analyzed at 3 months after transplantation (Fig. 2C).

Differentiated CE cell transplantation enhances expression of retinal ganglion cells. (A) Immunohistochemistry of Thy1 showing Cy3 (red fluorescence marked by arrows) expression bound to primary antibody at 20× visualized under confocal microscope in normal control mice, laser injured retina mice, laser injured transplanted with Lin-ve SC mice, and laser injured transplanted with CE cells mice. (B) Quantitative protein expression of Thy1 at 1 week after transplantation was measured by corrected total cell fluorescence of immunohistochemistry images using Image J software (C). mRNA expression of Thy1 in all four groups was analyzed using real-time PCR at 1 week as well as 3 months after transplantation. Statistical analysis was performed using one-way ANOVA test for immunohistochemistry and real-time PCR results. This was followed by post hoc analysis using LSD, Scheffe, and Dunnett’s test. *P ≤ 0.05, **P ≤ 0.01, and ***P ≤ 0.001 were regarded as statistically significant. ANOVA: analysis of variance; CE: ciliary epithelium; Lin-ve SC: lineage negative stem cells; PCR: polymerase chain reaction.
Neurotrophic Modulation by Paracrine Effects Exerted by Undifferentiated hUCB Lin-ve SC Transplantation
The existing literature of stem cell transplantation suggests that these cells either differentiate or aid integration in the host tissues, while others argue that stem cells act by providing neuroprotection mediated by the release of paracrine factors 25 . In the light of paracrine effects, we analyzed the expression levels of BDNF as well as CNTF in all the groups. We report that BDNF levels were upregulated more by transplantation of undifferentiated Lin-ve SC as compared to differentiated CE cells, when analyzed at 1 week (Fig. 3A, B). The mRNA expression of BDNF was concomitantly found increased after Lin-ve SC transplantation (in comparison to laser injured mice at 1 week). However, this effect was again diminished when analyzed at a longer time duration, i.e., 3 months (Fig. 3C).

Neurotrophic factor modulation by the Lin-ve SC transplantation. (A) Immunohistochemistry of BDNF showing Cy3 (red fluorescence marked by arrows) expression bound to primary antibody at 20× visualized under confocal microscope in normal control, laser injured mice transplanted with Lin-ve SC, and laser injured mice transplanted with CE cells. (B) Quantitative protein expression of BDNF at 1-week time point measured by corrected total cell fluorescence of immunohistochemistry images using Image J software. (C) mRNA expression of BDNF in all four groups was analyzed using real-time PCR at 1-week as well as 3-month time point. Statistical analysis was performed using one-way ANOVA test for immunohistochemistry and real-time PCR results. This was followed by post hoc analysis using LSD, Scheffe, and Dunnett’s test. *P ≤ 0.05 was regarded as statistically significant. ANOVA: analysis of variance; BDNF: brain-derived neurotrophic factor; CE: ciliary epithelium; Lin-ve SC: lineage negative stem cells; PCR: polymerase chain reaction.
CNTF is a ciliary neurotrophic factor and regarded as a survival factor for various neuronal types 26 . We estimated CNTF by immunohistochemistry, using confocal microscopy (Fig. 4A). By quantitative analysis (by Image J software), we found significant CNTF expression in laser injured mice. CNTF protein expression was also found upregulated in mice transplanted with CE cells in comparison to Lin-ve SC transplanted mice (Fig. 4B). The mRNA expression of CNTF, analyzed by real-time PCR, showed significant increase in Lin-ve SC transplanted mice in comparison to all other groups at 1-week after transplantation. However, this expression was diminished after 3 months of transplantation (Fig. 4C).
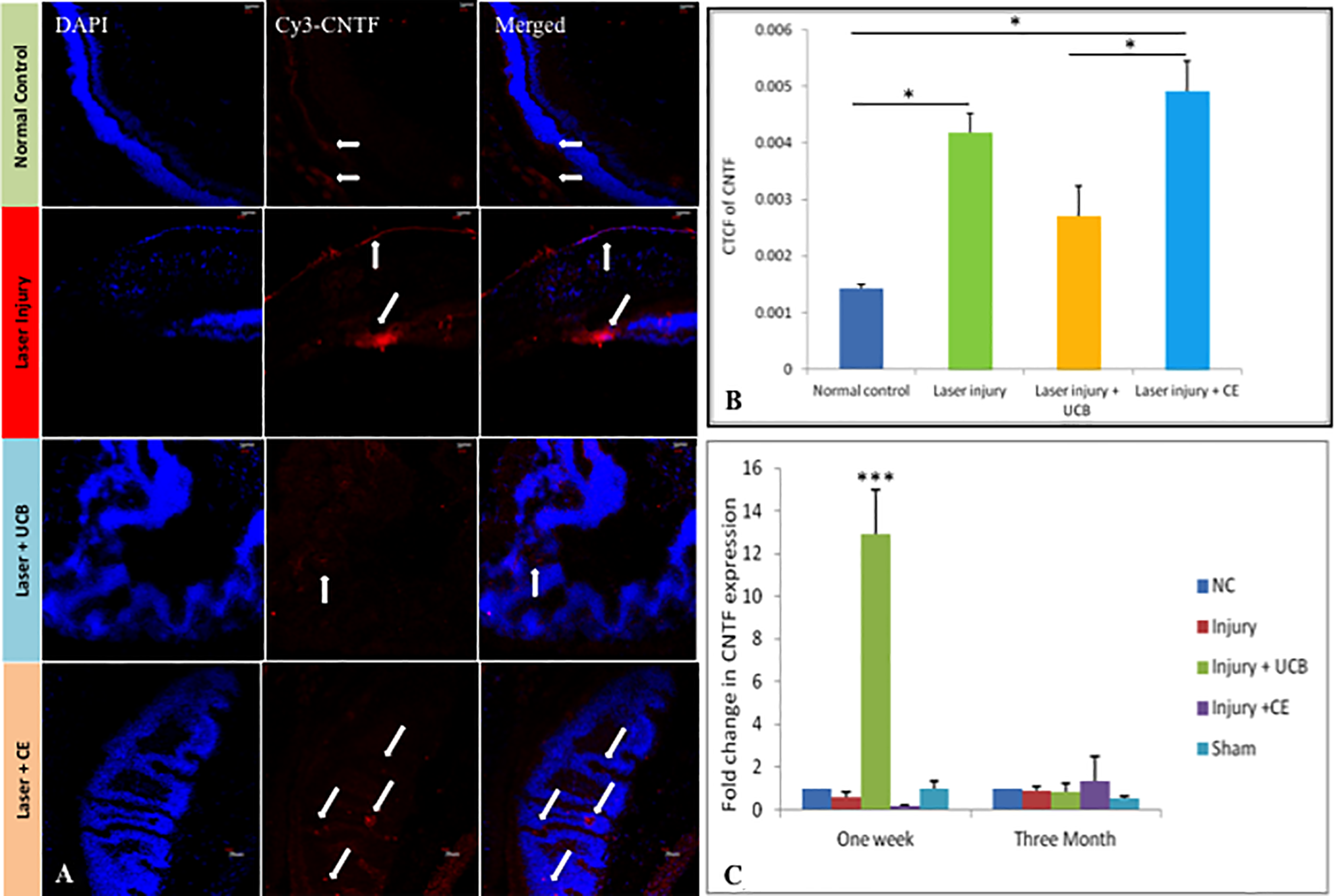
CE cell transplantation upregulated ciliary neurotrophic factor in the laser injured retina. (A) Immunohistochemistry of CNTF showing Cy3 (red fluorescence marked by arrows) expression bound to primary antibody at 20× visualized under confocal microscope in normal control, laser injured mice, laser injured mice transplanted with Lin-ve SC, and laser injured mice transplanted with CE cells. (B) Quantitative protein expression of CNTF at 1-week time after transplantation measured by corrected total cell fluorescence of immunohistochemistry images using Image J software. (C) mRNA expression of CNTF in all four groups was analyzed using real-time PCR at 1 week as well as 3 months after transplantation. Statistical analysis was performed using one-way ANOVA for immunohistochemistry and real-time PCR results. This was followed by post hoc analysis using LSD, Scheffe, and Dunnett’s test. *P ≤ 0.05, **P ≤ 0.01, and ***P ≤ 0.001 were regarded as statistically significant. ANOVA: analysis of variance; CE: ciliary epithelium; CNTF: ciliary neurotrophic factor; Lin-ve SC: lineage negative stem cells; PCR: polymerase chain reaction.
hUCB Lin-ve SC Transplantation Enhances Antiapoptotic and Proliferative Activity in Laser Injured Retina
The stem cells are reported to exert paracrine effects by releasing neurotrophic factors upon transplantation. These neurotrophic factors act as a ligand and initiate cell signaling pathways. BDNF acts as a ligand for TrkB receptor and activates CREB transcriptional factor, further promoting cell survival and maintenance of neuronal cells 27 . Likewise, CNTF is known to initiate Jak-STAT pathway and provides antiapoptotic and proliferative effect 28 . Bcl2, a well-known antiapoptotic marker, acts as a downstream molecule of CNTF initiated pathway. We found significant upregulation of Bcl2 mRNA expression after Lin-ve SC transplantation in laser injured mice in comparison to all the groups of 1 week and 3 months after transplantation. However, Bcl2 expression remained unaffected at 3 months after cells transplantation (Fig. 5A).

Antiapoptotic and proliferative activity enhanced by Lin-ve SC transplantation in laser injured retina. (A, B) mRNA expression of antiapoptotic marker Bcl2 and proliferative marker, i.e., Ki67 in all four groups was analyzed using real-time PCR at 1 week as well as 3 months after transplantation. Statistical analysis was performed using one-way ANOVA for real-time PCR results. This was followed by post hoc analysis using LSD, Scheffe, and Dunnett’s test. *P ≤ 0.05, **P ≤ 0.01, and ***P ≤ 0.001 were regarded as statistically significant. ANOVA: analysis of variance; CE: ciliary epithelium; Lin-ve SC: lineage negative stem cells; PCR: polymerase chain reaction.
As proliferation markers are routinely analyzed in stem cell therapies, Ki67, a nuclear antigen of proliferation 29 , was found highly upregulated upon transplantation of hUCB-derived Lin-ve SC (as compared to other groups at 1 week after transplantation). However, Ki67 expression was unaltered in all the groups at 3 months after transplantation (Fig. 5B).
Discussion
The series of degenerative changes in retina are due to diseased state with certain injuries resulting in irreversible damage. The currently used drugs have potential for symptomatic relief without halting the disease progression. Our data provide the comparative outcomes of transplantation of differentiated versus undifferentiated cells by utilizing a reproducible model of laser injured mouse retina. We wanted to examine the role of candidate markers of retinal repair and proliferation by transplantation of stem cells isolated from different origins. For that purpose, human eye from abortus fetuses was used to harvest the CE sourced stem cells. Second, we purified the Lin-ve SC from hUCB, a richly harvested source of undifferentiated stem cells.
Retina is an accessible and well-studied part of central nervous system. The retinal cellular structures have been studied in detail 30,31 . The existing studies provide evidence of the existence of stem cells in rodent retina. CE is one of the sources of stem cells shown as a rich source of cells with tremendous differentiation potential 15 . It has also been shown to possess the capacity to differentiate into specific retinal cell lineages for better integration into host tissue because of a similar niche. The pigmented CE has been shown to result in BrdU incorporation when provided with mitogens in vivo along with increase in cyclin D1 and Ki67 expression 32 . These cells, when stimulated with growth factors, rhEGF and bFGF, have been shown to form self-renewing colonies expressing retinal progenitor marker, i.e., Chx10 33 . Therefore, we used neurospheres that were differentiated from CE cells from human fetal eyes to examine how the stem cell markers are altered in laser-induced retinal injury in comparison to other cell types. We subretinally transplanted sixth day dissociated neurospheres into the laser-induced injury in retina of the mouse model. A significant expression of rhodopsin and Thy1 was reported in the mice retina after 1 week of transplantation (Figs. 1, 2). These data suggest that CE cells are involved in the rescue of retinal injury by activating the progenitor cells residing in the retina, which aid their differentiation into rods and RGCs. In comparison, CE cells are better positioned in the repair by activating the retinal markers and exerting neurotrophic effects superior to Lin-ve SC.
It has been shown that mesenchymal stem cells derived from hUCB exert neuroprotective effects by secreting several trophic factors including TGFbeta-1, NT-3, BDNF, and CNTF. These stem cells showed repair and regeneration of damaged neurons when transplanted in rat optic tract model 34 . Further, studies have shown transplantation of these cells intravenously in a neonatal hypoxic ischemia induced injury rat model could recover motor abilities mediated by neurotrophic factors, i.e., GDNF, BDNF, and NGF 35 . Endogenous cells secrete these neurotrophic factors mediated by paracrine effectors of transplanted cells. These trophic factors generally play an active role in neuronal development and survival by promoting proliferation, regeneration, and maturation of neurons 32 . The administration of trophic factors, e.g., CNTF, BDNF, and NT-4 has resulted in prevention of ON-injury-induced RGC death 36 . Other studies have described the effect of trophic factor administration (CNTF, BDNF, or PEDF) via intravitreal route on prevention of phototoxic-induced degeneration of photoreceptors cells 37 . Hence, such studies provide the necessary basis of studying the levels of these paracrine factors in transplantation studies. Our results that there is upregulation of BDNF after Lin-ve SC transplantation are consistent with other studies (Fig. 3).
Our results showed that there was significant enhancement in the expression of CNTF in the Lin-ve SC transplanted retina (when compared to the laser injured and CE cells transplanted group). CNTF is known to regulate via JAK/STAT pathway 28 . Bcl2 comprises the downstream effector molecule 38 and hence the upregulation of Bcl2 and CNTF mRNA (Figs. 4C, 5A). However, immunohistochemistry shows the shoot in the level of neurotropic factors in injured group, which are maintained sustainably by the CE injected group while there is not the similar sustenance in the UCB injected group. The dichotomy of CNTF mRNA and protein expression can be ascribed to post-translational differences in both the experimental and intervention groups. This can be further examined by using specific inhibitors of CNTF.
Ki67 is the universal molecule for investigating proliferation. It was found to have increased expression upon Lin-ve SC transplantation (Fig. 5B). The activity of the enhanced endogenous proliferation needs further elaboration. Therefore, our results unanimously reveal that hUCB-derived Lin-ve SC, when transplanted at the site of retinal injury, induce expression of neurotrophic factors, which may exert neuroprotective effect mediated by the antiapoptotic and proliferative mechanisms. Importantly, our study indicates the need for the multiple doses of Lin-ve SC transplantation instead of single dose as the paracrine effects noted in the study did not last for longer duration investigated.
Briefly, we explain the superior outcomes from transplantation of CE-related stem cells by ascribing it to same niche, i.e., retina providing conducive niche for rescue when compared to undifferentiated Lin-ve SC. In the context of noted paracrine effects, Lin-ve SC provide comparable outcomes but mediated by antiapoptotic and proliferative mechanisms. However, as Lin-ve SC effects lasted for a few weeks, it is suggested to examine the associated pathway at extended time frame using multiple doses of Lin-ve SC. Such studies will be helpful in utilizing the cord blood stored in the banks and for developing therapies for untreatable disorders of the retina. The main limitation of the study is that we were not able to perform immunohistochemistry of BDNF due to limited availability of eye samples.
Conclusion
The transplantation of CE cells showed superior outcomes than the undifferentiated Line-ve UCB-derived SC when tested in retinal injury mouse model. Based on the retinal marker analysis, we hypothesize that while the CE cells aid the formation of rods and ganglion cell layer, the Lin-ve SC transplantation promotes antiapoptotic and proliferative activity by neurotrophic modulation. We could not test the therapeutic outcome on the neuroprotective or paracrine effects from transplantation of multiple doses of Lin-ve SC or those at extended time points as the study was limited for a few weeks. Also, we have neither tested the per se effect of the transplantation of prominent markers on retinal repair nor evaluated the physiological changes. Hence, this study provides the rationale for more comprehensive analysis of the different sources of stem cells and their transplantation effects for extended duration. It is pertinent to note that clinical trial recently used RPE cells differentiated from autologous iPSCs and resulted in successful transplantation 39,40 . Our study thus provides preliminary data for future clinical application of the stem cell therapy for treating several retinal injuries.
Footnotes
Data Availability
All raw data are available with the first author of this manuscript.
Ethical Approval
Ethical approval for animals was obtained from Institutional Animal Ethics Committee (IAEC), Post Graduate Institute of Medical Education and Research, Chandigarh, India [71(69)/IAEC/423]. Human UCB samples were obtained after the approval from Institutional Committee for Stem Cell Research and Therapy, Post Graduate Institute of Medical Education and Research, Chandigarh, India (IC-SCRT: Approval No. PGI-ICSCRT-53-2014/1469).
Statement of Human and Animal Rights
All the experimental procedures were conducted in accordance with Committee for the Purpose of Control and Supervision of Experiments on Animals (CPSEA) guidelines.
Statement of Informed Consent
The samples were obtained after filling the proper informed consent forms by participants after explaining the complete protocol.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was obtained from Department of Science and Technology, Science and Engineering Research Board, India (EMR/2016/000190) and Department of Biotechnology, India.
