Abstract
Rapamycin (RAPA) and cisplatin (CDDP) are used as clinical drugs in the treatment of various tumors, but there are few studies on the combination of RAPA and CDDP. Tumor necrosis factor α (TNF-α) plays an important role in tumorigenesis and development. This study is to explore the effects of RAPA combined with CDDP on the expression of TNF-α in osteosarcoma MG-63 cells. MG-63 cells were routinely cultured and divided into a control group, a RAPA group (20 μM), a CDDP group (20 μM), and a RAPA + CDDP group (20 μM + 20 μM). The four groups were treated with drugs for 24 and 48 h, respectively. Real-time polymerase chain reaction (PCR), Western blot (WB), and immunocytochemistry (ICC) were adopted to detect the expression of TNF-α gene and protein. The results of PCR showed that both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α gene (*P < 0.5), but the combination was more effective (*P < 0.5); the expression quantity of TNF-α gene in the RAPA + CDDP group at 48 h was much lower than that at 24 h (***P < 0.001). The results of WB showed that both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α protein, and the combination was more effective than separate drug use (*P < 0.05) and more effective at 48 h (***P < 0.001); the expression quantity of TNF-α protein in the same group at 48 h was much lower than that at 24 h (*P < 0.05). The results of ICC showed that both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α protein, and the combination was more effective than separate drug use (**P < 0.01) and more effective at 48 h (***P < 0.001); the expression quantity of TNF-α protein in the same group at 48 h was much lower than that at 24 h (**P < 0.01). RAPA combined with CDDP can significantly reduce the expression of TNF-α in MG-63 cells, which is time dependent.
Introduction
Osteosarcoma is a primary bone tumor with a high degree of malignancy in adolescents. Its incidence accounts for about 3% of adolescent malignant tumors. Osteosarcoma is mainly characterized by fast growth, strong invasion ability, high metastasis rate, and so on 1,2 . Although operation, chemotherapy, and other treatment means have been more and more effective in recent years, patients’ prognosis is still poor and is complicated with high fatality rate and disability rate 3,4 . It is reported that the 5-year survival rate of patients with localized osteosarcoma is about 60% to 70% and that of patients with metastatic osteosarcoma only 20%. Moreover, about 80% of patients often clinically have lung or other metastases when they are diagnosed with osteosarcoma 5 . Therefore, it is necessary to further learn about the molecular mechanism of its tumorigenesis and development on the basis of original therapy, so as to find effective treatment strategies. Key factors regulating invasion and metastasis of osteosarcoma were identified at the gene level, and the process was clarified to provide the corresponding theoretical basis and make contributions to the early cure of osteosarcoma 6,7 .
In 1975, Carswell et al. found a special protein in the serum of mice infected by BCG injection or bacterial endotoxin. Therefore, this substance was named tumor necrosis factor (TNF) 8 . TNF is a glycoprotein and exists in the form of TNF-α or TNF-β. Afterwards, with the deepening of research, researchers found that TNF has more and more prominent antitumor effects. In particular, TNF-α has significant effects 9 –12 . TNF-α induces apoptosis of tumor cells by combining with the TNF receptor (TNFR) 13,14 on the cytomembrane. It can also promote the proliferation and differentiation of immune cells and impose antitumor effects. In addition to antitumor effect, as one of the important inflammatory factors, TNF-α also plays an important role in the pathogenesis of insulin resistance and type 2 diabetes. Its abnormal expression in patients with nasal polyp also has a certain effect. In terms of its high expression in patients with thyroiditis, its promoting effect on the apoptosis of thyroid cells has been verified by in vivo experiment 15 –17 .
In recent years, the therapies for malignant tumors are excision and chemoradiotherapy. The use of antitumor drugs is beneficial to postoperative recovery of patients. More and more antitumor drugs have been clinically used. However, the frequent use of chemotherapy drugs leads to the increased drug resistance in patients, thus the effect is not ideal 18 . Rapamycin (RAPA) and cisplatin (CDDP) are ideal chemotherapy drugs clinically used for tumor treatment 19,20 . They are significantly effective on ovarian cancer, lung cancer, liver cancer, endometrial cancer, prostate cancer, and other tumors 21 . In this study, osteosarcoma MG-63 cells were divided into four groups, i.e., a control group, a RAPA group, a CDDP group, and a RAPA + CDDP group to detect the expression quantity of TNF. The results of statistical analysis clarified the killing effect of the combination of the two drugs on tumor cells 22 .
Materials and Methods
Cell Culture
Human osteosarcoma MG-63 cells were purchased from Shanghai Cell Bank (Shanghai, China), Chinese Academy of Sciences. Dulbecco’s Modified Eagle Medium (DMEM) culture medium was used; 10% fetal calf serum (FBS), 1% nonessential amino acid, and 2 mmol/l glutamine were added. The cells were routinely cultured at 37°C and in an environment with 5% CO2. The cell state was adjusted. Cells in logarithmic phase were selected. MG-63 cells were randomly divided into four groups and intervened with 20 μM CDDP, 20 μM RAPA (R8140; Solarbio, Beijing, China), and 20 μM CDDP + 20 μM RAPA, respectively. The normal control group was intervened with drug solvents. The cells in the four groups were collected 24 and 48 h later for subsequent experiments. CDDP was purchased from Qilu Pharmaceutical Co., Ltd (Jinan, Shandong, China). The batch number was H37021362.
Real-Time Polymerase Chain Reaction
The treated cells were collected by groups. TRIpure extraction kit (RP1001; BioTek, Beijing, China) was used as instructed to extract the total RNA in each group. The instrument NanoDrop-2000 (Thermo Fisher Scientific, MA, USA) was used to determine the RNA concentration in each group. Real-time polymerase chain reaction (PCR) (Exicycler 96, BIONEER, Daejeon, Republic of Korea) was used, and super M-MLV reverse transcriptase (PR6502; BioTek) was used for reverse transcription. 2× Power Taq PCR MasterMix (PR1702; BioTek) and SYBR Green I (SY1020; Solarbio) were used for PCR. β-actin was used as a reference. The formula 2−ΔΔCt was used to calculate the relative expression quantity of mRNA of TNF-α gene. See Table 1 for primer sequence. It is synthesized by Sangon Biotech (Shanghai) Co., Ltd. (Shanghai, China).
Primer Sequence.

TNF-α: tumor necrosis factor α.
Western Blot
The treated cells in each group were collected and split with the RIPA lysis buffer (P0100; Solarbio) containing 1 mM phenyl methane on the icebox for 5 min, and then the cells were centrifuged at 12,000 rpm and 4°C for 10 min. The supernatant was separated to obtain protein extract. Bicinchoninic acid (BCA) protein assay kit (PC0020, Solarbio) was used to quantify the total protein extracted. According to the procedures of Western blot (WB), 8% sodium dodecyl sulfate-polyacrylamide gel was used for electrophoresis. The protein bands from separation were transferred to the PVDF membrane (IPVH00010; Millipore, MA, USA). The polyvinylidene fluoride (PVDF) membrane was sealed with 5% skim milk, and then TNF-α primary antibody (diluted by 1:1,000; GB13188 -1; Servicebio, Wuhan, China) was added. The PVDF membrane was incubated at 4°C overnight. Upon the completion of incubation, the PVDF membrane was washed with Tris Buffered saline Tween (TBST). The horseradish peroxidase (HRP)-labeled sheep anti-rabbit IgG (diluted by 1:3,000; SE134; Solarbio) was added for incubation. β-actin was incubated by the same procedures. Finally, electrochemiluminescence (ECL) luminous fluid (PE0010; Solarbio) was added for exposure in a darkroom. The film was scanned. The gel image processing system (the software Gel-Pro-Analyzer, Bio-Rad, State of California, USA) was used to analyze the optical density of the target band.
Immunocytochemistry
The cells treated in each groups were made into cell slides. Paraformaldehyde solution (4%) was used for fixation for 15 min. The formaldehyde was discarded. Triton X-100 (0.1%) was added for incubation at ambient temperature for 20 min. Phosphate buffer saline (PBS) (0.01 M) was added for immersion for 5 min for three times. PBS was discarded. H2O2 (3%) was added for incubation for 15 min, and then 0.01 M PBS was added for immersion for 5 min for three times. PBS was removed. The serum was added for sealing for 15 min. The serum was discarded, and the TNF-α primary antibody diluted with PBS by 1:2,000 (GB13188-2; Servicebio) was added. The mixture was placed at 4°C overnight. The primary antibody was discarded. PBS (0.01 M) was added for immersion for 5 min for three times. PBS was discarded. PBS was used to dilute the HRP-labeled goat anti-rabbit secondary antibody by 100 times (diluted by 1:1,000; G1213; Servicebio). The mixture was added to completely cover the cells. The cells were incubated at 37°C for 1 h. The secondary antibody was removed. PBS (0.01 M) was added for immersion for 5 min for three times. PBS was discarded. DAB was added for developing. Hematoxylin was used for redyeing (G1004-100; Servicebio). Sealing was performed. The dyeing effects were observed and photographed under the microscope. The software Image-Pro Plus was used for optical density analysis.
Results
The Results of Real-Time PCR Showed that the Expression Quantity of TNF-α Gene Decreased Significantly After Drug Treatment
PCR results showed that, for the four groups at the same time point, both separate drug use and drug combination can significantly lower the expression quantity of TNF-α gene, drug combination was more effective than separate drug use, and there was significant difference; for the same group at different time points, although separate drug use can decrease the expression of TNF-α gene, the significant difference level could not be reached; the expression quantity after 48 h of drug combination was significantly lower than that after 24 h, and the extremely significant difference level could be reached (Fig. 1).
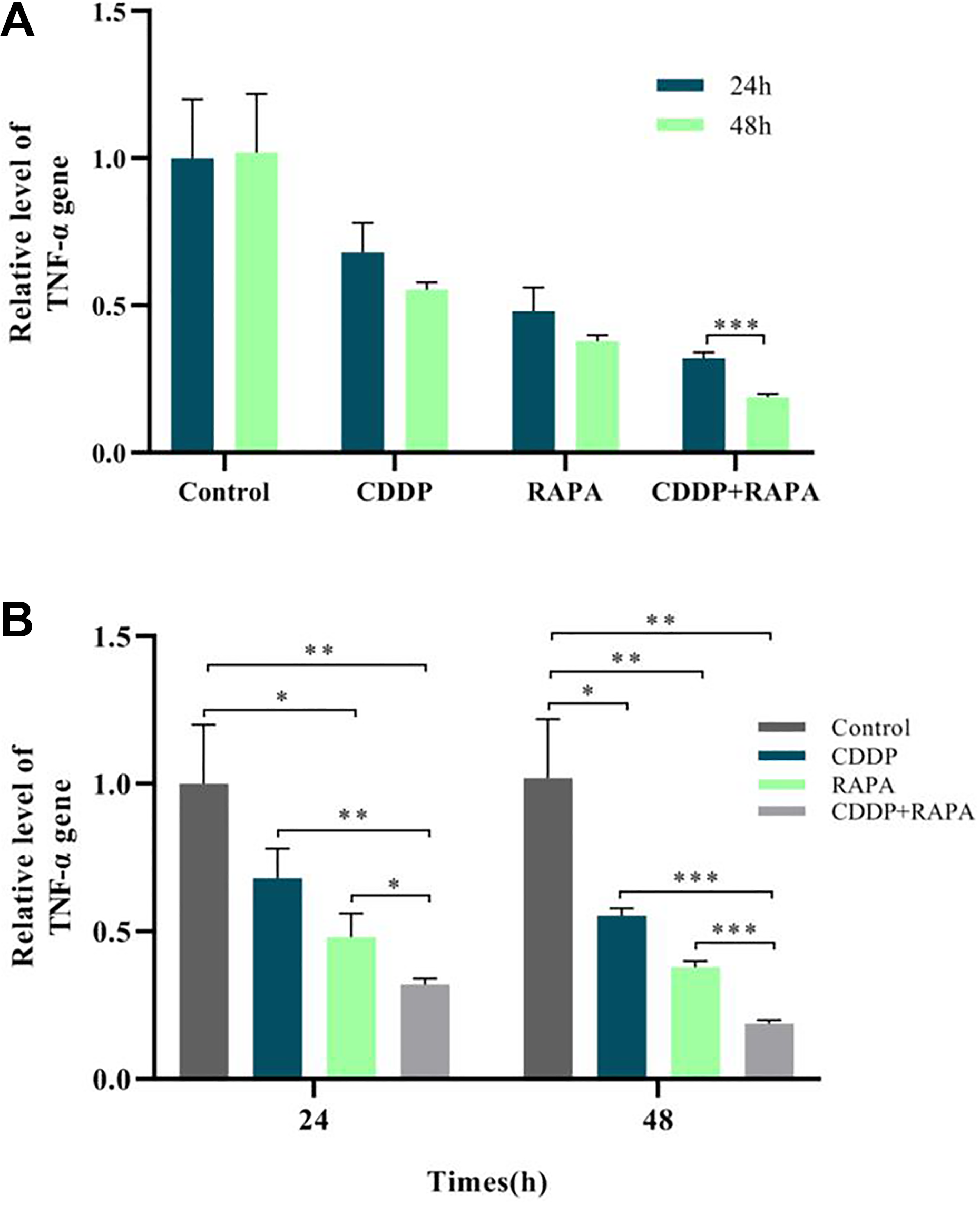
(A) The analysis of gene expression at different times in the same group. (B) The analysis of gene expression at different times in the same group. Both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α gene (*P < 0.5), but the combination was more effective (*P < 0.5); the expression quantity of TNF-α gene in the RAPA + CDDP group at 48 h was much lower than that at 24 h (***P < 0.001). CDDP: cisplatin; RAPA: rapamycin; TNF-α: tumor necrosis factor α.
WB Results Showed that the Expression Quantity of TNF-α Protein Decreased Significantly After Drug Use
Expression quantity of TNF-α protein in each group in WB quantitative analysis is shown in the Fig. 2. The expression in different groups at the same time point showed that both separate drug use and drug combination could significantly lower the expression quantity of TNF-α protein, and an extremely significant difference level could be reached; the expression quantity after 24 h of drug combination was significantly lower than that after separate drug use, and a significant difference level could also be reached; the expression quantity after 48 h of drug combination was significantly lower than that after separate drug use, reaching an extremely significant difference. In terms of the comparison of expression quantity in the same group at different time points, the expression quantity of TNF-α protein after 48 h of separate drug use and drug combination was significantly lower than that after 48 h of drug use and was time dependent to some extent (Fig. 2A, B).

(A) The experimental result of WB. (B) The statistical analysis result based on the gray value of picture A. Both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α protein, and the combination was more effective than separate drug use (*P < 0.05) and more effective at 48 h (***P < 0.001); the expression quantity of TNF-α protein in the same group at 48 h was much lower than that at 24 h (*P < 0.05). TNF-α: tumor necrosis factor α; WB: Western blot.
Immunocytochemistry Results Showed that the Expression Quantity of TNF-α Protein Decreased Significantly After Treatment with Drug(s)
The cells in the four groups were made into cell slides for immunocytochemistry dyeing. The results showed that (1) 24 h of separate drug use and drug combination could significantly lower the expression quantity of TNF-α protein and reach an extremely significant difference level; the expression quantity after drug combination was significantly lower than that after separate drug use. Forty-eight hours of separate drug use and drug combination could significantly lower the expression quantity of TNF-α protein and reach an extremely significant difference level; the expression quantity after drug combination was lower than that after separate drug use. (2) The expression quantity after 48 h of drug use was lower than that after 24 h of drug use in the same group, showing significant difference (Fig. 3A, B).
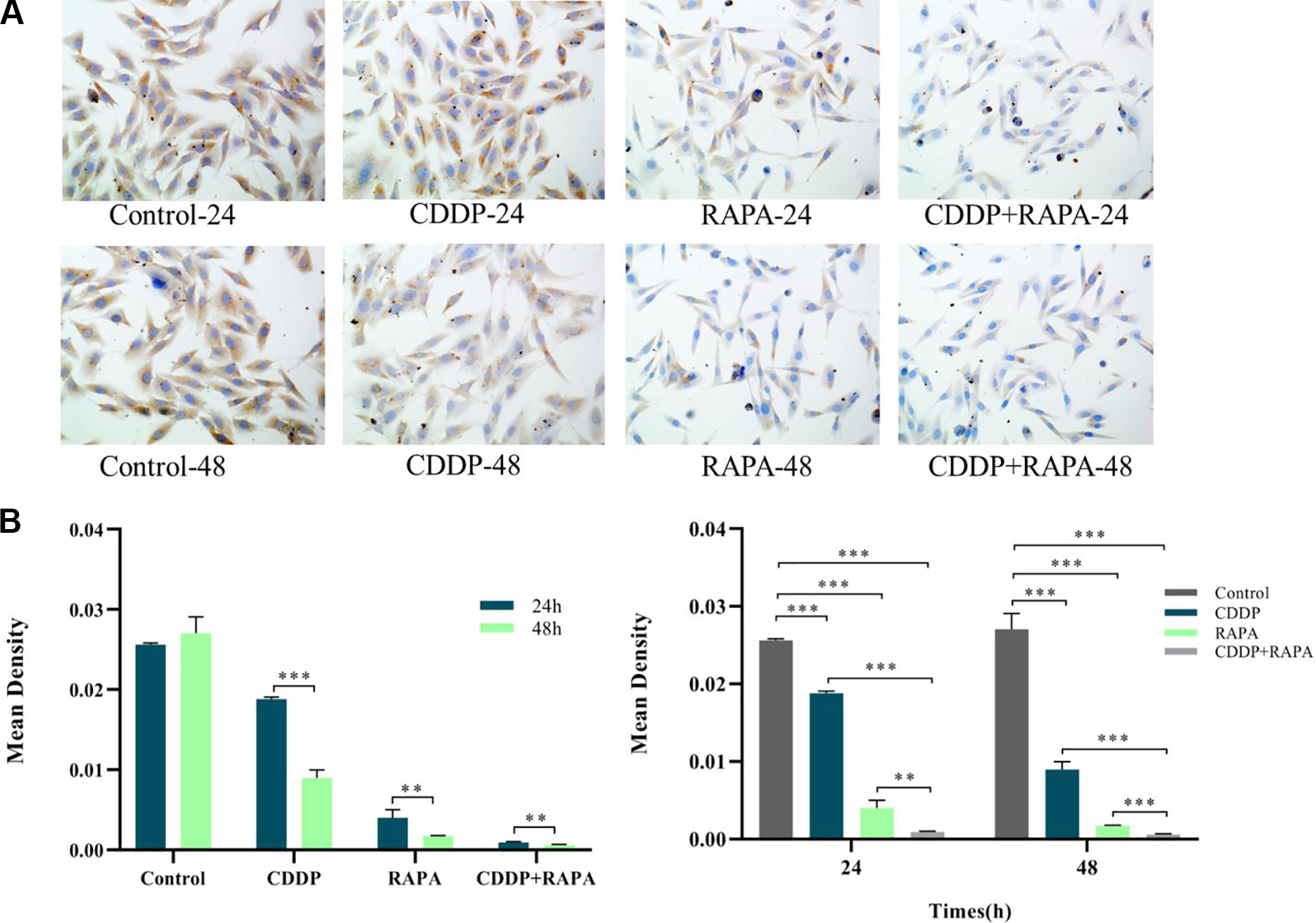
(A) The result of ICC experiment. (B) The statistical analysis based on the result of picture A. Both the separate drug use and drug combination could significantly lower the relative expression quantity of TNF-α protein, and the combination was more effective than separate drug use (**P < 0.01) and more effective at 48 h (***P < 0.001); the expression quantity of TNF-α protein in the same group at 48 h was much lower than that at 24 h (**P < 0.01). ICC: immunocytochemistry; TNF-α: tumor necrosis factor α.
Discussion
As one of the most common primary bone cancers in adolescents, osteosarcoma has seriously threatened the lives and health of children and caused a burden on families 23 . In recent years, researchers have paid more and more attention to osteosarcoma and have begun to study osteosarcoma in depth, in order to explore its mechanism, develop different chemotherapy drugs, and identify its pathogenic factors at the molecular level. They hope to primarily cure osteosarcoma, realize early detection, early diagnosis and early treatment, and improve the quality of life and health level of human beings 24 .
As an important member of TNF family, TNF-α has remarkable antitumor effect and a wide range of biological activities and now is the factor with the most powerful antitumor effect. Therefore, it is of vital importance to explore its antitumor mechanism and enhance its antitumor effect 25 . In this study, the expression of TNF-α in different groups by treating osteosarcoma with chemotherapy drugs, so as to prove its changes in the tumorigenesis and development. A great number of studies have shown that except the direct inhibition of proliferation and promotion of necrocytosis of tumor cells, TNF-α is also correlated to other diseases to some extent. The abnormal expression of TNF-α may cause apoptosis of myocardial cells, lead to heart failure and eventually cause death 26 . Besides, TNF-α is involved in several autoimmune diseases, such as rheumatoid arthritis and psoriatic arthritis 27 . Some other studies have shown that it is also associated with asthma, sepsis, chronic obstructive pulmonary disease, and other diseases 28 . As the most active cytokine, TNF-α is also involved in the development of many diseases. Therefore, it is of great importance to study its acting mechanism in depth.
CDDP has been widely applied and has developed rapidly since its discovery. It has good antitumor effect and high antitumor activity, and has been used as a preferred drug for cancers, such as breast cancer, melanoma, liver cancer, esophageal cancer, and cervical cancer 29,30 . Nevertheless, CDDP also has some side effects, mainly including nephrotoxicity, nausea, and vomiting. Its toxicity profiles differ from those of other antitumor drugs. Moreover, it does not have cross tolerance, which is beneficial to clinical drug combination. RAPA was first found to have immunosuppressive effects and might be used to deal with organ transplant rejection. Afterward, it was found to be able to specifically inhibit Mammalian target of rapamycin (MTOR) signal transduction pathway. Proteins associated with this pathway are normally expressed in multiple tumors and affect tumorigenesis by regulating this pathway. Therefore, RAPA can induce the apoptosis of tumor cells and exert its anticancer effect 31 . The effects of drug combination were proved by comparing the combination of CDDP and RAPA with separate drug use and detecting the expression of TNF-α and through experimental results. The findings also proved that both of the two drugs can lower the high expression of TNF-α, but the drug combination has better effects. This study preliminarily proved the effects of the two drugs. However, the complex internal mechanism should be further studied in depth. The subsequent topics will discuss the signal pathway from multiple perspectives and levels. Meanwhile, researchers also hope that the finders can provide theoretical basis and practical significance for clinical treatment. This study preliminarily proved the efficacy of these two drugs, and TNF-α may become a potential target for curing osteosarcoma and play a greater role in future research. The follow-up topic will explore the signal pathway from multiple perspectives and levels, and make further in-depth research on the complex internal mechanism. At the same time, I hope the research results can provide theoretical basis and practical significance for clinical treatment.
Footnotes
Ethical Approval
The study was approved by the Institutional Ethics Committee of General Hospital of Northern Theater Command, Shenyang, Liaoning, China.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
