Abstract
Currently, islet isolation is performed using harsh collagenases that cause nonspecific injury to both islets and exocrine tissue, negatively affecting the outcome of cell transplantation. We evaluated a novel islet isolation protocol utilizing high concentrations of glucose to cause selective osmotic shock (SOS). Islets have a membrane glucose transporter that allows adaptation to changes in glucose concentrations while exocrine tissue can be selectively destroyed by these osmolar shifts. Canine pancreata were obtained within 15 min after euthanasia from animals (n = 6) euthanized for reasons unrelated to this study. Each pancreas was divided into 4 segments that were randomized to receive 300 mOsm glucose for 20 min (group 1), 600 mOsm for 20 min (group 2), 300 mOsm for 40 min (group 3), or 600 mOsm for 40 min (group 4). Islet yield, purity, and viability were compared between groups. Mean ± standard error of the mean islet yield for groups 1 to 4 was 428 ± 159, 560 ± 257, 878 ± 443, and 990 ± 394 islet equivalents per gram, respectively. Purity ranged from 37% to 45% without the use of density gradient centrifugation and was not significantly different between groups. Islet cell viability was excellent overall (89%) and did not differ between treatment protocol. Islet function was best in groups treated with 300 mOsm of glucose (stimulation index [SI] = 3.3), suggesting that the lower concentration of glucose may be preferred for use in canine islet isolation. SOS provides a widely available means for researchers to isolate canine islets for use in islet transplantation or in studies of canine islet physiology.
Introduction
Islet endocrine tissue comprises only 1% to 2% of total pancreatic cell mass; therefore, isolation of islets from the exocrine pancreatic tissue can allow focused transplantation of only the necessary islet tissue, minimizing the host immune response and avoiding an invasive surgical procedure needed for pancreas transplantation. Traditional isolation of pancreatic islets involves enzymatic digestion of pancreatic connective tissue followed by cell separation using density gradient centrifugation 1,2 . The limited islet yield and high rate of cell death resulting from current isolation protocols have led to many human diabetic patients receiving at least 2 islet transplants before achieving an adequate effect 3 . Progression from a crude collagenase to a purified blend has been shown to improve the efficiency of islet isolation with significantly higher postpurification recovery and function 4 –6 . However, there remains considerable variation in the number, purity, viability, and secretory capacity of isolated islets.
Collagenase enzyme preparations used in standard islet isolation techniques are intended to disrupt the connective tissue structure of the pancreas at an organ level, producing a cell suspension. An inherent weakness of the current isolation techniques is that the enzymes also cause injury to the islet cells, resulting in a loss of extracellular matrix and basement membrane, apoptosis, necrosis, and release of immunogenic proteases producing pro-inflammatory cytokines and free radicals 7 –10 . Damage caused during islet isolation is suspected to explain the large number of islets required to obtain treatment effects, the high rate of primary graft failure, and the short-term euglycemia experienced by many human islet transplant recipients 11 .
Given the limitations of the current enzyme-based procedures for islet isolation, an alternative approach based on selective osmotic shock (SOS) of pancreatic acinar cells has been reported for pig islet isolation 12 . This procedure is purely a nonenzymatic approach to minimize islet cell injury in porcine islet isolation. The working hypothesis is that islet cells would be resistant to rapid changes in extracellular glucose concentrations when compared to surrounding acinar cells due to the presence of a specialized membrane glucose transporter (GLUT2) protein in pancreatic β cells of islets 13 . Because the GLUT2 cell membrane transporters are not saturated by high concentrations of glucose, the glucose molecule is essentially osmotically inactive in these cells and they are resistant to rapid changes in glucose concentrations 12 . In acinar cells that lack GLUT2, an increased concentration of extracellular glucose would lead to instantaneous cell shrinkage due to water efflux. Exocrine cells recover almost immediately via a process called regulated volume increase, leaving their cytoplasm hypertonic. Subsequent immersion of these cells in media without glucose would cause rapid water influx, selectively rupturing the acinar cells. In the porcine pancreas, Atwater et al. found that SOS with glucose was simple and inexpensive and avoided the inherent enzymatic damage of collagenase-based isolation methods. Isolated islets were found to have an intact pericapsular membrane with higher islet yield per gram of tissue (13,423 islets/g) than historical reports from Liberase-treated porcine pancreata (4210 islets/g) 14 or human cadaveric donors (2,279 islets/g) 1 . While SOS has been documented to be effective in the pig pancreas, variation of GLUT2 expression in β cells and differences in connective tissue density may affect the utility of this technique in other species, requiring optimization of the procedure 15 –17 .
Our work focuses on the use of the dog as a naturally occurring large animal model of human diabetes 6,18,19 . Diabetes is a common endocrinopathy in dogs, and this species is considered to be a spontaneous large animal model for human diabetes mellitus with at-risk breeds sharing several major histocompatibility complex II haplotypes and other genetic polymorphisms with human diabetics 20,21 . Given their extreme β cell deficiency at the time of diagnosis 22 , dogs are completely insulin-dependent and fit many of the criteria for islet transplantation in human diabetic patients. Our goals in the current study were to use the dog model (1) to evaluate the efficacy of SOS for islet isolation in canine pancreata and (2) to optimize osmolality and exposure time to maximize islet yield and viability. We hypothesized that increasing glucose concentrations and duration of exposure would lead to significant increases in islet yield; however, we expected that prolonged exposure to hyperglycemic solutions would eventually cause islet cell injury, contributing to islet cell death and diminished function.
Materials and Methods
Biochemicals/Chemicals
Glucose (
Animals
Six medium to large breed, skeletally mature dogs (2–3 yr of age) were used in this study. Body weight ranged from 25 to 45 kg, and the following breeds were identified: Staffordshire Terrier (n = 4), Hound (n = 1), and German Shepherd (n = 1). Two dogs were intact males, 1 was a neutered male, and 3 were intact females. All animals were euthanized under approved Institutional Animal Care and Use Committe (IACUC) protocols for reasons unrelated to the current study. Procurement of tissues after euthanasia does not require separate IACUC approval at our institution.
Pancreatectomy
Pancreata were procured immediately after confirmation of death in canine cadavers (n = 6) euthanized for reasons unrelated to this study. All animals were euthanized by administration of pentobarbital-based euthanasia solution administered intravenously. After confirmation of death, dogs were placed in dorsal recumbency, and the ventral abdomen was aseptically prepared using iodine solution and alcohol. A ventral midline incision was made from the xyphoid process and extending 20 cm caudally to expose the cranial abdomen. The duodenum was exteriorized, and the resection of the pancreas was initiated at the distal right limb of the pancreas, proceeding to the body and left limb. The pancreas was separated from the duodenum, portal vein, and omentum using a combination of blunt and sharp dissection. No attempt was made to perfuse the organ before or after procurement. Pancreata were placed in cold phosphate-buffered saline and transported on ice to the lab for immediate processing. Time from euthanasia to completed organ procurement was recorded.
SOS-based Method of Islet Isolation
Once in the lab, the pancreas was divided into 4 equal sized pieces and labeled as segments A (distal right limb), segment B (central right limb), segment C (body of the pancreas), and segment D (distal left limb; Fig. 1). Segments were then block randomized into 1 of the 4 treatment groups for each dog using a computer-generated program (https://www.randomizer.org/) to prevent bias due to differences in islet density in each segment. Pancreatic segments were then exposed to hyperosmolar glucose treatment with the following protocols: group 1 (300 mOsm glucose for 20 min exposure time), group 2 (600 mOsm for 20 min exposure time), group 3 (300 mOsm for 40 min exposure time), and group 4 (600 mOsm glucose for 40 min exposure time). Each sample was weighed, so that yield could be standardized and reported per gram of tissue. Using aseptic technique under a laminar flow hood, pancreatic tissue was minced into <3 mm fragments with scalpel blades and divided into sterile 50 mL centrifuge tubes leaving room in each tube for the sample to be submerged in media.

Illustration of the division of the canine pancreas into sections including the distal right limb (A), the central right limb (B), the body (C), and the distal left limb (D). Pancreatic segments were block randomized for each dog and assigned to 1 of the 4 treatment groups to avoid bias due to asymmetrical distribution of islets in the canine pancreas.
Zero glucose RPMI 1640 media was supplemented with glucose to achieve either 300 or 600 mmol/L glucose solution. Tissue fragments were then suspended in the 300 or 600 mmol/L glucose + RPMI 1640 solutions and periodically agitated during incubation at room temperature. After the prescribed incubation time for each of the 4 treatment groups, the tubes were centrifuged for 5 min at 180 relative centrifugal force (rcf) to produce a pellet, then media was decanted and replaced with zero glucose RPMI. Each sample was then gently mixed, resuspended, and centrifuged with an additional 2 rinses using zero glucose RPMI for a total of 3 wash steps. Mechanical disruption of any remaining tissue fragments was achieved by trituration (suction/expulsion) using a 6-mL sterile syringe for 10 min. Remaining connective tissue was removed using a 500 μM steel sieve followed by a 70 μM disposable cell strainer to remove single cells and small tissue fragments. No density gradient centrifugation was used in this study. The islets and any remaining acinar tissue were washed from the cell strainer into 100 mm culture plates containing standard islet culture media composed of RPMI 1640 + 10% FBS + Penicillin Streptomycin 1% and incubated at 37 °C, 5% CO2.
At 24 h after islet isolation, islets were resuspended by agitating the media, and 100 μL aliquots were obtained for quantification of islet yield, purity (% islet tissue vs. % exocrine tissue), and assessment of islet morphology. Islets were quantified by counting the number of islets in each 100 μL aliquot, and islet yield was standardized by dividing the number of islet equivalents (IEq) by the weight of pancreatic tissue used in each group.
Cell death was measured using the exclusion dye PI, a red nuclear stain used to identify dead cells, and Hoecht 33342, a blue nuclear stain used to identify all cells. Cells were imaged using 4′,6-diamidino-2-phenylindole (blue) and Texas Red (red) channels via epifluorescent microscopy, and acquired images were analyzed using a proprietary software program (NIS-Elements Basic Research, Version 4.5) to determine the percentage of cell death in each islet and in each acinar tissue fragment.
After stabilization for 24 h in culture, islet β cell function was measured with a modified version of the glucose-stimulated insulin secretion (GSIS) assay used in human islet transplant centers (Integrated Islet Distribution Program City of Hope, Standard Operating Procedure for GSIS, https://iidp.coh.org/Investigators/Policies-Standard-Operating-Procedures). Groups of 10 islets were handpicked from each of the treatment groups using a 20-μL pipette and placed into separate 1.5 mL cryovials containing 500 μL zero glucose. Islets were preincubated for 1 h at 37 °C in zero glucose RPMI 1640 with 2.8 mM glucose and 1% bovine serum albumin. After 1 h, tubes were spun at low speed (180 rcf, 2 min), and 500 μL media was removed and replaced with 500 μL zero glucose RPMI 1640 with either low (2.8 mM) or high (28 mM) glucose concentration. Assays were performed in duplicate, using 2 cryovials with 10 islets for each group at each concentration for a total of 16 samples. Thus, a total of 40 total islets were collected from each group, with two 10-islet cryovials tested at low glucose concentration and two 10-islet cryovials tested at high glucose concentration. Islets were incubated for 1 h at 37 °C. After 1 h, tubes were spun at low speed (180 rcf, 2 min), and 500 μL media was removed and stored in a freezer for insulin determination.
Statistics
A linear mixed model was created with the treatment group and exocrine versus endocrine as fixed effects and dog as a random effect using JMP Statistical Software Program (JMP Pro 12, SAS Institute, Cary, NC, USA). Significance was set at a P value < 0.05. Where significant effects were found in the model, individual comparisons of islet yield, cell death, and islet function were performed between treatment groups using one-way analyses of variance with post hoc Tukey’s tests.
Software Programs
http://www.ramdomizer.org. Copyright © 1997–2017 by Geoffrey C. Urbaniak and Scott Plous
NIH Element Analysis Software (Version 4.5), Nikon Metrology, Inc., Melville, NY, USA
JMP Statistical Software Program, ÓSAS Institute Inc., Cary, NC, USA
Results
Islet Yield, Purity, and Morphology
SOS was successful in achieving islet isolation for all dogs. Overall, the treatment group was not found to have a statistically significant effect on islet yield (Fig. 2). In evaluating the specific effects of exposure time and osmolality of the glucose solution, mean ± standard error of the mean (SEM) IEq/g for tissue exposed to 300 mmol/L glucose (613 ± 240 for groups 1 and 3) was not significantly different from yield for tissue exposed to 600 mmol/L glucose (718 ± 240 for groups 2 and 4). Islet yield for tissue exposed to 40 min of hyperosmolar solution was nearly double that of the groups exposed for the shorter 20 min period (858 ± 294 vs. 474 ± 522 IEq/g), although this difference was not statistically significant (P = 0.26).
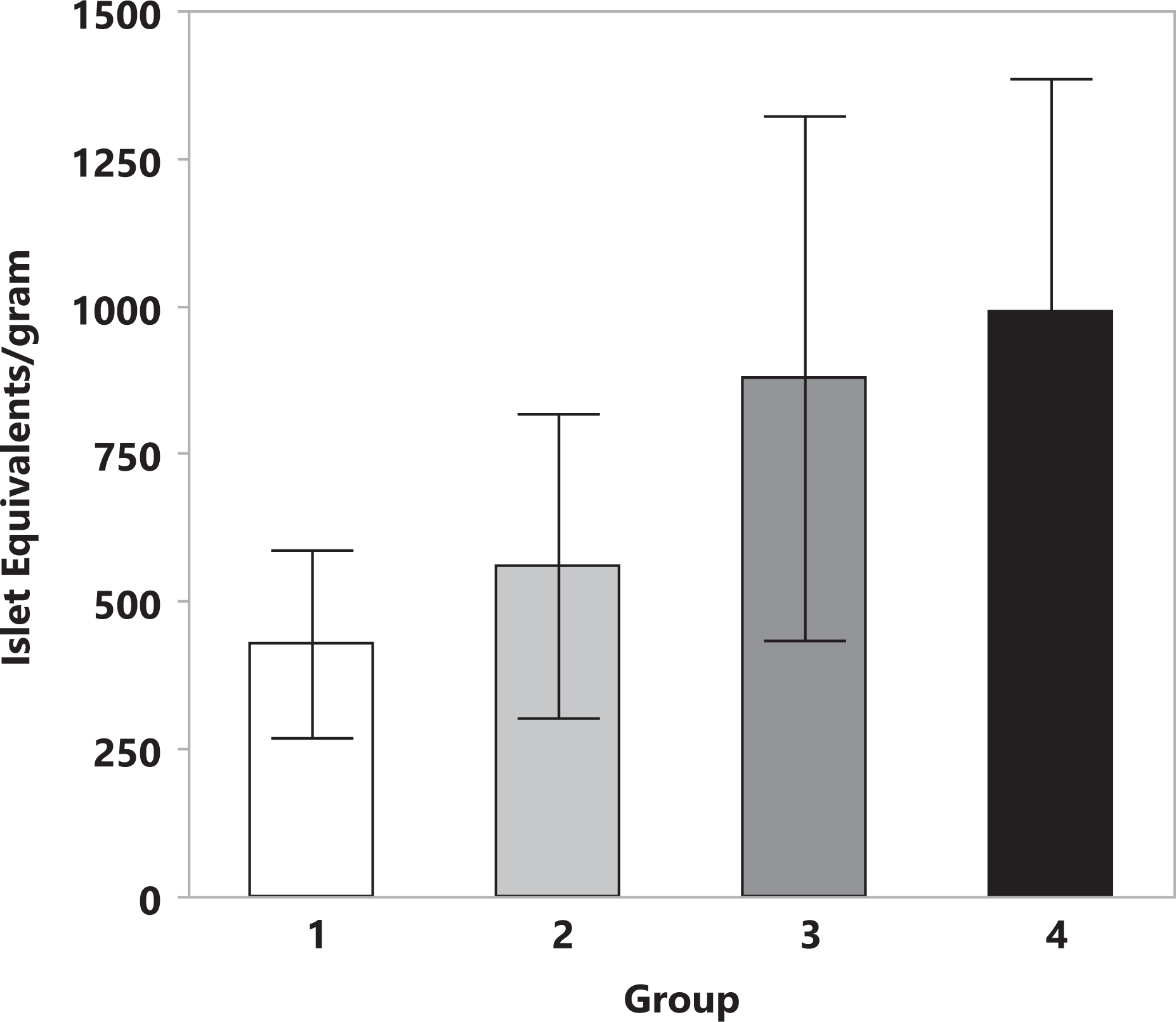
Islet yield is presented as the mean ± standard error of the mean islet equivalents/g of pancreatic tissue obtained from canine pancreata (n = 6) after selective osmotic shock. Treatment group is indicated on the x-axis for group 1 (300 mOsm glucose for 20-min exposure time), group 2 (600 mOsm for 20-min exposure time), group 3 (300 mOsm for 40-min exposure time), and group 4 (600 mOsm glucose for 40-min exposure time). While mean islet yield was nearly doubled in groups 3 and 4 (exposed to 40 min of hyperosmotic solution) when compared to groups 1 and 2 (20 min exposure), results were highly variable and this was not statistically significant (P = 0.26).
Mean ± SEM islet yield for the 4 pancreatic segments was 796 ± 463 IEq/g (right distal), 655 ± 250 IEq/g (right central), 957 ± 397 IEq/g (left central), and 448 ± 150 IEq/g (left distal). Overall islet yield was similar for left limb of the pancreas when compared to the right limb (725 ± 252 vs. 703 ± 217 IEq/g, respectively). Islet purity (percentage of islets vs. acinar tissue) was similar for all groups and ranged from 37% to 45%. Differences between individual dogs contributed to variability in this study with a small number of dogs, as there were significant variations in islet yield identified between dogs (P = 0.0004 for effect of dog on islet yield, Fig. 3).

Islet yield (mean ± standard error of the mean islet equivalents/g) is presented for each dog, # 1 to 6. Dog had a significant effect on islet yield (P = 0.0004) and contributed to the variability in islet yield seen in Fig. 1.
Islet Viability and Percent Cell Death
Overall, quantification of PI uptake as a measure of cell death showed that glucose-mediated osmotic shock caused selective destruction of acinar tissue, with significantly higher cell death in the acinar cells when compared to islet tissue (P < 0.0001, Fig. 4). Cell death in islets was low, with cell death ranging from 10% to 13% in all groups (Fig. 5A). Neither exposure time to hyperglycemic conditions nor osmolality of the solutions had a significant effect on islet viability. Interestingly, the treatment group had a significant effect on percentage of cell death in acinar tissue (P = 0.0015), with higher cell death in tissues exposed to longer (40 min) incubation times in hypertonic glucose solutions (P = 0.0021 for 20 min vs. 40 min groups, Fig. 5B).

Cell death was measured using exclusion dye (propidium iodide) to identify cells with loss of nuclear membrane integrity at 8 to 24 h after islet isolation using selective osmotic shock (SOS). Data were combined for all treatment groups to show the overall effect of SOS on pancreatic tissue (A), which produced a high rate of cell death in acinar tissue, while sparing islet viability. A representative image (B) was obtained at 200× magnification. Islets are identified by arrow and are stained blue (Hoechst) to identify cell nuclei. Minimal red propidium iodide staining seen in the islets, reflecting the low rate of cell death. In contrast, the acinar tissue (arrowheads) has nearly uniform staining with propidium iodide, showing a high rate of cell death.

Cell death data (mean ± standard error of the mean) in canine pancreatic islets (A) and acinar tissue (B) after selective osmotic shock are presented for each treatment group. Cell death in islets was low in all groups and did not appear to be affected by changes in exposure time or osmolality of glucose solutions used in the 4 treatment groups. However, acinar cell death was significantly increased in groups 3 (300 mOsm glucose) and 4 (600 mOsm glucose), which were exposed to longer (40 min) duration of exposure to hyperosmolar glucose solutions (P = 0.0021).
In concordance with the data for islet yield, significant differences in islet viability were noted between dogs (P = 0.0031 for effect of dog on percentage of islet cell death). Variations in islet viability did not appear to be dependent upon operator experience leading to improvements in latter isolation procedures but appeared to be related to differences in tissue quality between dogs (Fig. 6A). On the contrary, there was no significant difference between dogs in the acinar cell death rates, suggesting that the procedure itself was relatively specific in its effect on the tissues for effect of dog on percentage of acinar cell death (Fig. 6B).

Mean ± standard error of the mean islet cell death (A) and acinar cell death (B) are presented for each dog. Similar to the effects on islet yield in Fig. 2, each dog had a significant effect on islet viability (P = 0.0031). In contrast, acinar cell death rate was relatively constant, suggesting that differences in islet viability may be due to factors related to the dog rather than inconsistencies in isolation technique.
GSIS
In healthy islets, insulin secretion is expected to increase at least 3-fold when islets are moved from a low to high concentration glucose solution if assessed by the standard GSIS assay 23 –25 . This ratio of insulin production after stimulation is termed the stimulation index (SI). Overall, the mean SI for islets in the current study was 2.5, with highest values in the groups that were treated with 300 mOsm glucose (group 1: 3.75 ±2.49, group 3: 2.93 ± 3.51) when compared to groups treated with 600 mOsm glucose (group 2: 1.96 ± 1.67 and group 4: 1.51 ± 1.17), although this difference fell short of reaching statistical significance because of the small number of samples (P = 0.10, Fig. 7). Measured insulin secretion data for each group are presented in Table 1.
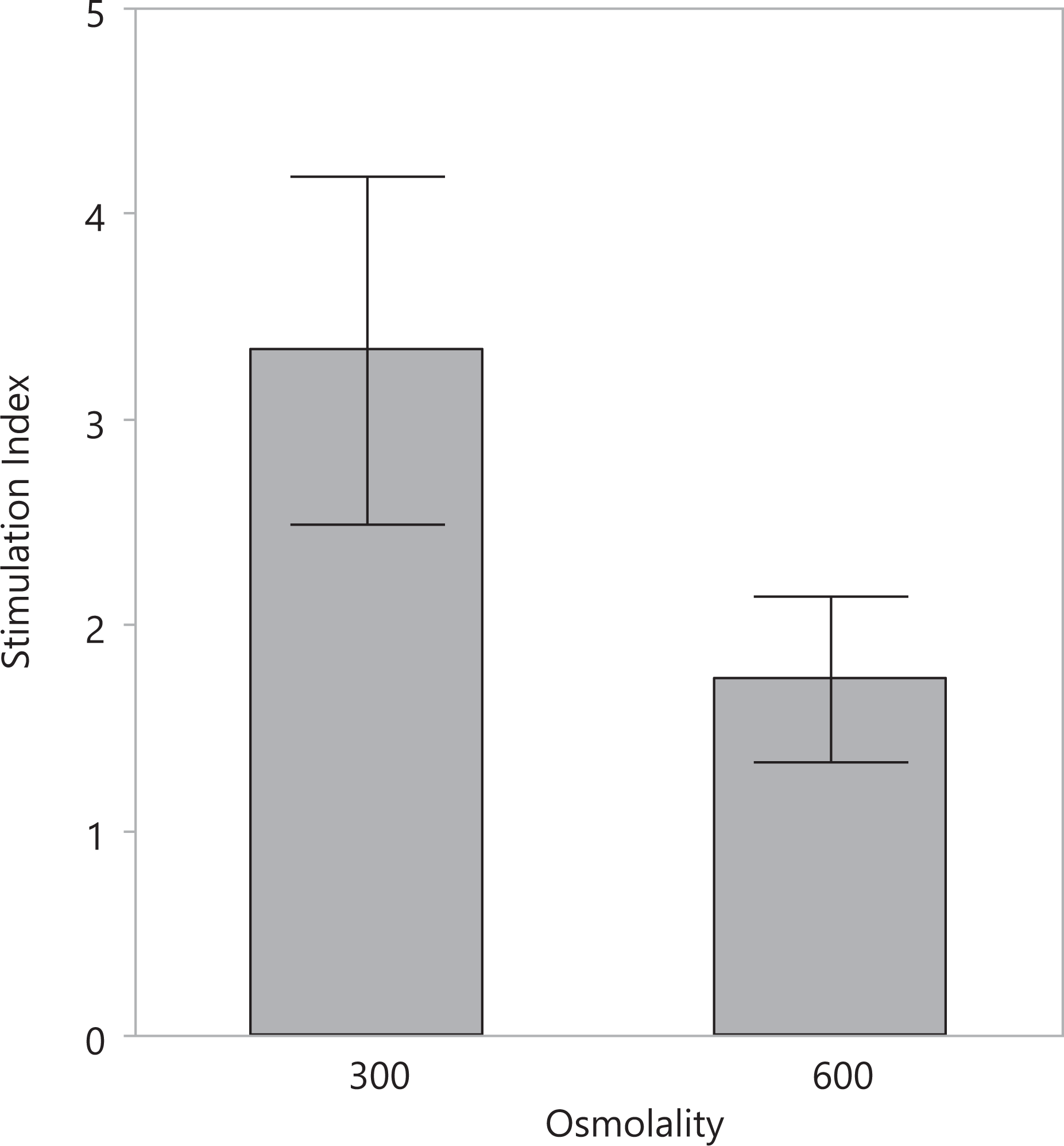
Glucose-stimulated insulin secretion was measured 24 h after islet isolation as a method of assessing islet function. Stimulation index (SI) represents a ratio of insulin produced when islets are placed in a high-glucose environment (28 mM) when compared to insulin production in a low-glucose environment (2.8 mM). SI is expected to increase 3-fold in healthy islets. While SI appeared to be increased in islets from treatment groups 1 and 3 (exposed to 300 mOsm glucose) when compared to islets from groups 2 and 4 (exposed to 600 mOsm), this failed to reach statistical significance (P = 0.10).
Insulin Production by Isolated Islets.

Note: Glucose-stimulated insulin secretion was measured as described in Fig. 6. In vitro insulin production by canine islets at 24 h after isolation is reported at low (2.8 mM) and high (28 mM) concentrations of glucose for islets treated with either 300 mOsm or 600 mOsm of hyperosmolar glucose solutions during selective osmotic shock.
Discussion
The SOS protocol using hyperosmolar solutions of glucose caused selective destruction of acinar tissue and resulted in canine islet isolation, thus confirming our primary hypothesis. Overall, islet viability and function were excellent after SOS and are consistent with accepted standards for islet viability and potency used for human islet transplant programs 26 . While this technique was successful in isolating high-quality islets from canine pancreata, further refinements to the protocol will be required to increase islet yield and purity.
Although the SOS technique was successful in producing clinical grade islet quality, the quantity of islets recovered per pancreas (approximately 28 × 10 3 IEq) was far lower than previous reports of canine islet isolation techniques using enzymatic digestion (49 to 234 × 10 3 IEq) 4 . Islet yield was calculated after 24 h of culture in our study, and previous reports suggest that islet yield decreases by 40% to 50% in the first 24 h of culture due to cell death induced by the isolation process, thus islet quantification immediately after isolation would have revealed a higher yield 27 . Another likely contributor to the markedly lower islet yields in our study is that we used cadaveric donors from an animal shelter that underwent some period of warm ischemia while the authors of the previously referenced study performed organ procurement from living anesthetized dogs 4 , essentially eliminating warm ischemia. The use of cadaveric donors is known to significantly decrease islet yields when compared to the extraction of pancreatic tissue from living anesthetized animals—a finding that is consistent with results in our lab using the enzymatic technique in pancreata that were obtained after euthanasia of dogs at an animal shelter 6,28 .
Despite using a relatively homogenous population of apparently healthy young dogs, our data showed that significant variability of islet yield between dogs contributed to the wide range of islet yield in each treatment group, affecting the ability to discern differences in efficacy of the various isolation protocols. In particular, islet yield for groups that were exposed to hyperosmolar glucose solution for 40 min was almost double that of groups that received 20 min exposure, but the inter-dog variability caused enough overlap between the groups to prevent these results from achieving statistical significance. While procurement of organs from living dogs would likely increase the islet yield and decrease variability between experiments, ethical obligations in the treatment of companion animal species preclude the procurement of organs prior to euthanasia, and our current method reflects the modern reality of organ availability when using a companion animal model.
Automation of the enzymatic digestion process by Camillo Ricordi using a perfusion circuit with a digestion chamber containing silicone spheres that produce gentle tissue disruption while the chamber is agitated with an automated shaker was an important step in achieving consistent results for human islet isolation 29,30 and a similar process may be required following SOS. In an attempt to reproduce the techniques used in the Atwater study and to minimize the need for complex equipment, we used scalpel blades and syringe aspiration to mechanically disrupt tissues and produce cell suspensions for the current study. While we standardized the duration of scalpel and syringe trituration, we noted that variable results occurred depending upon how vigorous the operator was in manipulating the tissues—particularly with the performance of fast versus slow aspiration and ejection from the syringe tip.
The 2 glucose solutions used in our studies are within the range of hypertonic solutions reported to be tolerated by canine islets 31 . We had originally hypothesized that glucose concentration and incubation time would significantly increase islet yield but that increased exposure to hyperosmolar solutions would cause increased islet cell death and diminished insulin secretion. While our relatively small sample size (n = 6 per group) affected the power of the current study, it was the high degree of variability between islet isolations that prevented making any strong conclusions comparing the various treatment groups. Thus while increased incubation time in hyperosmolar glucose (40 min) produced nearly double the islet yield per gram that was seen in the shorter (20 min) exposure groups, this difference was not statistically significant. In contrast to the results of Atwater’s work in pigs, alterations in glucose concentration to 600 mOsm had no discernible effect on islet yield 12 . Surprisingly, longer exposure time to hyperosmolar solutions did not lead to negative effects on islet viability, and cell death was extremely low in all 4 groups (10–13%). Higher concentrations of glucose in our study did lead to increased cell death in the acinar tissue, reflective of the lack of GLUT2 transport capabilities in these cells.
Islet function testing suggested that the SOS technique preserved islet function at a level that would be acceptable for islet transplantation or for use in in vitro studies. In concordance with our results for islet viability, islet function was within the desired range for islets exposed to 300 mOsm (SI of 3.3); however, islets exposed to 600 mOsm had an SI that would predict a lower level of viability (SI of 1.7). In contrast, incubation time in hypertonic solutions did not have any measurable effect on islet function. Based on our findings for this functional assay, a lower concentration of glucose (300 mOsm) for a shorter duration of time (20 min) may be preferred in canine islet isolation—a finding that differs from those in a previous study using porcine tissues 12 and emphasizing that species differences in glucose transporters or in sensitivity of β cells to glucotoxicity may be required for species-specific SOS protocols currently being explored in our laboratories 32 .
Islet purity in the current study (37–45%) was lower than typical reports for clinical islet transplant materials (78.5%) 27 and in canine enzymatic islet isolation procedures performed in our lab (87.5%) 6 . It is important to note that previous enzymatic digestion techniques require a separate step in which density gradient centrifugation is performed to separate the islets from the acinar tissue fragments using ficoll or dextrans—a step that we excluded in the current study. After completing initial steps in the SOS protocol, we noted that islet purity prior to any cell separation was remarkably high when compared to our previous experience with enzymatic techniques. While islets comprise only 1% of the total pancreatic tissue mass, we were seeing approximately 40% purity without any steps beyond SOS. This effective “islet isolation” was based solely on the selective death and subsequent filtration of acinar cells, which had become fragmented or lysed. In avoiding the use of density gradient separation, we attempted to further decrease the injury to islet cells that is known to occur during high-speed centrifugation and exposure to ficoll solutions. While the 40% purity of the islet preparations created in our current study would be considered lower than ideal in human islet transplant centers, one recent publication suggested that islet transplants having <50% purity had better outcomes when compared to higher purity preparations, attributing the improvement to paracrine and endocrine contributions for other cell types 33 . One negative to the approach that we took in this study was that continued culture of the islets in an environment containing large number of dead and dying acinar cells could lead to islet injury and loss of function or add to the innate immune response after transplantation, as dead cells release antigens, cytokines, and damage-associated molecular patterns that trigger the immune system 34,35 .
Since the current study provides evidence that the concept of SOS can be effective in canine islet isolation, future efforts will attempt to refine the process in a similar manner to the evolution of automated steps that have been adopted in enzymatic digestion for islet isolation. By avoiding the use of collagenase enzymes, the SOS technique indeed has a high requirement for mechanical disruption of the connective tissue of the pancreas in order to produce a homogenous cell suspension. Use of a tissue homogenizer or a semi-automated digestion chamber as described by Ricordi 29 would improve islet yield and would minimize operator-induced variability in the aggressiveness of tissue disruption. Further purification of the sample could also be performed via density gradient centrifugation after the filtration process in order to eliminate more exocrine debris as is typically recommended for clinical transplantation 36,37 .
Conclusion
SOS for isolation of canine islets is an inexpensive protocol requiring minimal equipment that produces viable islets but still requires significant improvements in islet yield and purity prior to clinical application. Islet viability was excellent in all 4 protocols that were evaluated, while islet function was highest in islets exposed to the lower 300 mOsm glucose solution, suggesting that lower osmolality treatment protocols may be preferred in dogs. Further refinements using an automated method of mechanical disruption and identification of an ethically acceptable protocol to obtain high-quality tissues from canine donors will be essential to successful use of this technique in islet transplantation in diabetic dogs.
Footnotes
Ethical Approval
All animals were euthanized under approved Institutional Animal Care and Use Committee (IACUC) protocols for reasons unrelated to the current study.
Statement of Human and Animal Rights
All animals were euthanized by administration of pentobarbital-based euthanasia solution administered intravenously.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
