Abstract
Parkinson’s disease (PD) is the second most common degenerative disorder of the central nervous system in the elderly. It is characterized by progressive loss of dopaminergic neurons in the substantia nigra pars compacta, as well as by motor dysfunction. Although the causes of PD are not well understood, aggregation of α-synuclein (α-syn) in neurons contributes to this disease. Current therapeutics for PD provides satisfactory symptom relief but not a cure. Treatment strategies include attempts to identify new drugs that will prevent or arrest the progressive course of PD by correcting disease-specific pathogenic process. Betulin is derived from the bark of birch trees and possesses anticancer, antimicrobial, and anti-inflammatory properties. The aim of the present study was to evaluate the potential for betulin to ameliorate PD features in Caenorhabditis elegans (C. elegans) models. We demonstrated that betulin diminished α-syn accumulation in the transgenic C. elegans model. Betulin also reduced 6-hydroxydopamine-induced dopaminergic neuron degeneration, reduced food-sensing behavioral abnormalities, and reversed life-span decreases in a pharmacological C. elegans model. Moreover, we found that the enhancement of proteasomes activity by promoting rpn1 expression and downregulation of the apoptosis pathway gene, egl-1, may be the molecular mechanism for betulin-mediated protection against PD pathology. Together, these findings support betulin as a possible treatment for PD and encourage further investigations of betulin as an antineurodegenerative agent.
Introduction
Parkinson’s disease (PD), which is the most common neurodegenerative disorders other than Alzheimer’s disease, results in selective death of dopaminergic (DA) neurons in the substantia nigra of the midbrain, dopamine deficiency in the basal ganglia, as well as impaired the motor control and recognition of the body, 1 including slow movement and tremor. PD is becoming increasingly prevalent in elderly populations and is both a care-intensive major health problem and an economic challenge. 2
PD is characterized by formation of Lewy bodies in neurons of the brain, which occur via α-synuclein (α-syn) aggregation. 3 Causes of sporadic PD (>90% of cases) remain unclear and likely result from complex interactions of environmental and genetic factors. 4 Familial PD (5% to 10% of cases) is associated with several specific genes, including α-syn (SNCA), leucine-rich repeat kinase 2 (LRRK2), parkin (PRKN), DJ-1(PARK7), phosphatase and tensin homolog (PTEN)-induced putative kinase 1 (PINK1), ATP13A2, eukaryotic translation initiation factor 4 gamma 1 (EIF4G1), vacuolar protein sorting-associated protein 35 (VSP35), and Ras-related protein Rab-39B (RAB39B). 5 The small acidic protein, α-syn, is found mostly at the presynaptic terminals of neurons in the brain and may supply synaptic vesicles at neuronal terminals, as well as control dopamine release to regulate voluntary and involuntary movements. 6 Insoluble fibrils characterized by Lewy bodies are formed when α-syn aggregates abnormally and are the pathological hallmark of PD. These fibrils are detected in both sporadic and familial forms of PD. 7 Furthermore, overexpression of α-syn or 3 major mutations (A53T, A30P, and E46) in the α-helical domain of this gene induce oligomer/fibril formation and are associated with sporadic and familial PD. 8,9 Increased levels of human α-syn have also been observed in Caenorhabditis elegans (C. elegans) models, 10 –12 and are associated with neurodegeneration.
Many studies have found relationships between toxin exposure and increased risk of PD. 13,14 The compound 6-hydroxydopamine (6-OHDA) is a specific neurotoxin that interferes with catecholamine neurons via the dopamine active transporter . When 6-OHDA is injected into the median forebrain bundle or neostriatum of animal brains, it causes irreversible damage to DA neurons in the ventral midbrain. This consistent loss of dopamine innervation is associated with a range of long-term, behavioral deficits. Thus, 6-OHDA-induced lesions are the most widely used animal models of PD. 15 –17
There is currently no cure for PD. Medication (levodopa, dopamine agonists, and monoamine oxidase B inhibitors) and surgery (deep brain stimulation) only relieve and delay the symptoms. 18 Stem cell transplants are a recent research target 17,19 ; however, stem cell therapies for PD are not yet ready for use in patients, and considerable work is necessary before clinical trials can occur. Therefore, the most practical therapeutic direction for PD treatment requires discovery of small compounds that can prevent PD and reduce symptoms. A number of phytocompounds have been established as effective neuroprotective agents. 20 –22 Betulin, a naturally active compound, is a pentacyclic lupine-type triterpene 23 (Fig. 1). It is found in the outer bark of birch trees and has various biological activities on animal and human health, including the following properties: antibacterial, 24 –27 antiparasitic, 28 –30 antiviral, 31,32 anti-inflammatory, 33 –37 anticancer, 38 –44 antivenom, 45 liver protective , 46 –48 kidney protective, 49 lung protective, 24 anticonvulsant, 50 and a cognitive enhancer in patients with Alzheimer’s disease (AD). 51 Betulin also improves diet-induced obesity, ameliorates the stability of atherosclerotic plaques, and can be used to treat type II diabetes. 52

Chemical structure of betulin.
C. elegans is an idea model for neurobiology studies. The nematode has a conserved DA pathway, a simple nervous network, a short life cycle, an easy culture method, and a fully transparent body. 53 –56 Here, we consider that betulin may be a prophylactic as well as an adjuvant agent for its advantageous effects on PD using pharmacological and transgenic C. elegans model system. We also clarified the possible mechanism of betulin action.
Materials and Methods
C. elegans Strains, Maintenance, and Synchronization
Wild-type Bristol N2 C. elegans, transgenic BZ555 (P dat-1 :: gfp; green fluorescent protein [GFP] visible in DA neurons), transgenic OW13 (P unc-54 ::α-synuclein:: yfp+unc-119; human α-syn protein with yellow fluorescent protein [YFP] observable in the muscles), as well as their food source, Escherichia coli (E. coli) strain OP50, were obtained from the Caenorhabditis Genetics Center (CGC, University of Minnesota, USA). According to standard procedures, 57 we maintained the worms on nematode growth medium (NGM) plates seeded with live OP50 as a food source, at 22 °C. For the synchronization, fertilized eggs were acquired by sodium hypochlorite/sodium hydroxide treatment of gravid adults. After 20 h of incubation at 22 °C in M9 buffer to collect synchronized L1 larvae, the worms were spread on OP50/NGM plates and then incubated for 24 hours at 22 °C, in order to obtain L3 larvae.
Food Clearance Assay
Synthesized betulin (mol wt. 442.72, 98% purity) was purchased from Sigma-Aldrich (St Louis, MO, USA) and dissolved in dimethyl sulfoxide (DMSO) to 1 M as a master stock solution. A food clearance assay was used to assess the effects of betulin on C. elegans physiology and to determine the concentration of betulin treatment. 58 E. coli grew overnight and were then resuspended at a final optical density (OD) of 6.6 in nematode S-medium. Betulin was diluted into the E. coli suspension, in order to achieve the desired concentrations. Each well of a 96-well plate received 50 µL of the E. coli suspension. Approximately 20 to 30 synchronized L1 worms in 10 µL of S-medium were added to an E. coli suspension containing a series of betulin concentrations and were then incubated in a 96-well microtiter plate at 25 °C. The absorbance (OD 595 nm) of the culture was measured once/day (d) for 6 d, using a SpectraMax M2 Microplate Reader (Molecular Devices, Silicon Valley, CA, USA).
Exposure to 6-OHDA and Treatment with Betulin
We used a previous method of 6-OHDA-induced DA neuron degeneration in C. elegans, with slight modifications. 59 In brief, 50 mM 6-OHDA and 10 mM ascorbic acid were added to OP50/S-medium mix with or without the indicated concentration of betulin. Synchronized L3 larvae were then transferred onto the treated cultures, incubated for 1 h at 22 °C, and agitated gently every 10 min. After 1 h of treatment, worms were washed 3 times with M9 buffer, transferred to fresh OP50/NGM plates with or without betulin, and then cultured at 22 °C. After 24 h, worms were transferred to OP50/NGM plates containing betulin and 0.04 mg/mL fluoro-2′-deoxyuridine,2′-deoxy-5-fluorouridine (FUDR, Sigma-Aldrich) to reduce progeny production. Worms were used for various assays 1 or 3 d following treatment.
Quantitative Analysis of Dopaminergic Neurodegeneration
Analysis of DA neuron degeneration was performed in worms treated with 6-OHDA or betulin/6-OHDA, as described above. After 3 d of treatment at 22 °C, BZ555 worms were washed 3 times with M9 buffer and then mounted onto a 2% agar pad on a glass slide, using 100 mM sodium azide (Sigma-Aldrich), before being enclosed with a coverslip. Imaging of the head region of the immobilized worms was conducted with an Axio Observer inverted fluorescence microscope (Carl Zeiss MicroImaging GmbH, Göttingen, Germany). Fluorescence intensity was determined using ImageJ software (Carl Zeiss, Göttingen, Germany). Loss of the GFP signal from DA neurons indicated DA neuron degeneration.
Quantitative Analysis of α-Syn Accumulation
Accumulation of α-syn protein was measured in control and betulin-treated OW13 worms. Synchronized OW13 L3 larvae were cultured on OP50/NGM plates containing 0.04 mg/mL FUDR, with or without the indicated concentrations of betulin, for 3 d at 22 °C, then washed 3 times with M9 buffer. Larvae were then transferred to 2% agarose pads on glass slides, mounted with 100 mM sodium azide, and enclosed with a coverslip. Immobilized worms were observed and imaged using an Axio Observer inverted fluorescence microscope (Carl Zeiss) at 200×, in order to monitor the YFP signal (the accumulation of α-syn protein) for the body region of each worm. The signal was quantified by measuring fluorescence intensity using ImageJ software.
Quantitative Analysis of Protein Expression
Protein extracts were obtained from whole worm frozen pellets in phosphate buffered saline containing proteinase inhibitors (Sigma-Aldrich) by using Fastprep24 (MP, Solon, OH, USA). Samples were boiled 10 min with sodium dodecyl sulfate sample buffer, and separated on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Then proteins were transferred to polyvinylidene difluoride (PVDF) membranes. Antibody binding was visualized by binding of horse-radish peroxidase (HRP)-coupled secondary antibody and Amersham-enhanced chemiluminescence system (Piscataway, NJ, USA). Signals were detected by using a UVP BioSpectrum Imaging System (Upland, CA, USA). Antibodies used were antihuman α-synuclein, anti β-actin (GeneTex, Irvine, CA, USA), anti RPN-1, and anti EGL-1 (Allbio Science, Taichung, Taiwan). Goat–antimouse and goat–antirabbit immunoglobulin G (IgG)-HRP (PerkinElmer Life Sciences, Boston, MA, USA) were used as secondary antibodies.
Analysis of Food-Sensing Behavior
Food-sensing behavior analysis was conducted in order to measure the function of DA neurons in C. elegans, using a previously described method with slight modifications. 60 Briefly, assay plates were prepared by spreading E. coli in a ring with an inner diameter of 1 cm and an outer diameter of 8 cm, and incubated overnight at 37 °C on 9-cm diameter NGM agar plates to prevent the worms from reaching the edge of the plate during the assay. Well-fed 6-OHDA-treated or betulin/6-OHDA-treated adult worms were washed with M9 buffer and then transferred in a drop of M9 buffer to the center of an assay plate with or without a bacterial lawn. Five minutes after transfer, the locomotor rate of each worm was counted at 20-s intervals. The slowing rate was calculated as the percentage of the locomotor rate in the bacteria lawn compared to that in the absence of a bacteria lawn. The average slowing rate of 10 worms was calculated for each analysis. In all analyses, plates were numbered so that the experimenter was blind to worm treatment.
Life-span Analysis
Life-span analysis of C. elegans was conducted using a previously described method with slight modifications. 61 The test plates were prepared by adding betulin stock solution, at various concentrations, to NGM plates just before use. The NGM plates were then seeded with OP50. Life-span analyses were performed by transferring control, 6-OHDA-treated, and betulin/6-OHDA-treated L3 stage worms to a new control or betulin-containing plate every 3 d, until all worms were dead. A total of 0.04 mg/mL of FUDR was added to each plate to reduce progeny production. Survival was calculated daily, and the worms were counted as dead if they failed to respond to mild, repeated touches with a platinum pick. Age 1 d was defined as the first day of adulthood. Worms that moved off the walls of the plates and died from dehydration were excluded from the analyses. Three different analyses were conducted. Survival curves were plotted using the product-limit method of Kaplan and Meier, using SPSS software (IBM, Armonk, NY, USA).
RNA Isolation and Real-Time Quantitative PCR (qPCR)
Total RNA was extracted from synchronized control or experimental adult animals using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and an RNeasy Mini kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions. 62 The quantity and integrity of the RNA were assessed with a UV-Vis spectrophotometer Q5000 (Quawell Technology, San Jose, CA, USA) and agarose gel electrophoresis.
The RNA samples were stored at −80 °C until use. For quantitative gene expression analyses, high capacity complementary DNA (cDNA) was generated from 2 µg of RNA, using the SuperScript one-step Reverse transcription-polymerase chain reaction (RT-PCR) kit (Invitrogen). qPCR analyses were performed with a 1:20 dilution of cDNA using SYBR Green I Master kit (Roche Diagnostics, Indianapolis, IN, USA) and an ABI StepOnePlus system (Applied Biosystems, Inc, Foster City, CA, USA). Primer sets for this study were based on our previous study. 57
Data were calculated by the comparative 2ΔΔC t method, using the geometric means of cdc-42 and pmp-3, as well as Y45F10D.4 as an endogenous control. 63
Proteasome Activity Assays
Our proteasome activity assays in C. elegans used a previously described method, with slight modifications. 64 Briefly, using a Precellys 24 homogenizer (Bertin Technologies, Montigny-le-Bretonneux, France), worms were lysed in a proteasome activity assay buffer containing 50 mM Tris-HCl (pH 7.5), 250 mM sucrose, 2 mM adenosine triphosphate (ATP), 5 mM MgCl2, 1 mM dithiothreitol, and 0.5 mM ethylenediaminetetraacetic acid (EDTA). The lysate was centrifuged at 10,000g for 15 min at 4 °C. For each test, 25 μg of total lysate was loaded into each well of a 96-well microtiter plate, after which fluorogenic substrate was added. Z-Gly-Gly-Leu-AMC (Enzo Life Sciences, Farmingdale, NY, USA) was used as a substrate for testing the chymotrypsin-like activity of the proteasome. After incubation for 1 h at 25 °C, fluorescence (excitation wavelength = 380 nm, emission wavelength = 460 nm) was measured with a SpectraMax M2 Microplate Reader (Molecular Devices, Silicon Valley, CA, USA).
RNA-Mediated Interference Approaches
We initiated RNA-mediated interference (RNAi) by feeding worms with RNAse III-resistant E. coli expressing the gene-specific double-stranded RNA (dsRNA) by Isopropyl β-D-1-thiogalactopyranoside (IPTG)-inducible T7 polymerase vector and resulted in rpn-1 or egl-1 gene inactivation through the specific degradation of the targeted endogenous mRNA. 65 In brief, the gene-specific dsRNA-expressing E coli were streaked onto LB/tetracycline plates. A positive colony was inoculated into LB/ampicillin liquid medium and incubated at 37 °C overnight. Then the culture was transferred to center of NGM/IPTG/ampicillin plate (RNAi plate). The plate was dried overnight. Starved L1 larvas were transferred to RNAi plates and were fed. Worms were transferred every 24 h to new feeding plates until desired stage for further experiments.
Statistical Analyses
Statistical analyses were performed using SPSS software. Data are expressed as means ± standard deviations from independent tests. Each test was replicated 3 times. The differences between 2 means were assessed by independent Student’s t tests. Statistical significance was indicated when *P < 0.05.
Results
Determining the Betulin Concentration Range for the Food Clearance Assay
In order to evaluate the effects of betulin on DA neuron degeneration and α-syn accumulation, we first used the food clearance assay to determine the optimal concentrations of betulin for use in our C. elegans PD models. Given the short life cycle and the capability of C. elegans to grow in a liquid culture of E. coli, betulin was examined by assessing the rate of E. coli suspension consumption. Each adult worm can produce hundreds of progeny, which rapidly consume the restricted E. coli supply. Therefore, the OD of the wells without betulin was significantly reduced after 3 d for N2, BZ555, and OW13 strains (Fig. 2). The addition of 0.02 mM, 0.1 mM, or 0.5 mM betulin to the culture containing N2, BZ555, or OW13 strains did not significantly impact food clearance compared to control worms. However, worms exposed to 2.5 mM betulin had significantly postponed food clearance (Fig. 2A). Furthermore, worms exposed to the higher concentration of 2.5 mM betulin had lower numbers of offspring, with reduced body sizes throughout the experiment (Fig. 2B), which is related to the lack of E. coli source clearance. Thus, betulin, at concentrations of 0.02, 0.1, or 0.5 mM, did not affect general worm health. Consequently, worms were treated with betulin at concentrations of up to 0.5 mM in subsequent assays.

The concentrations of betulin for subsequent experiments were determined with a food clearance assay in Caenorhabditis elegans. Between 20 and 30 newly hatched L1 synchronized worms of N2, BZ555, or OW13 strains were incubated at 25 °C in Escherichia coli (E. coli; OD A595 = 0.6), in a 96-well plate containing 0.02, 0.1, 0.5, or 2.5 mM of betulin, to a total volume of 60 µL. The optical density (OD) of the plate was measured daily for 6 d. (A) The OD of E. coli was recorded daily for each concentration of betulin. Data are presented as means ± standard deviations from 3 independent experiments. (B) Concentrations >2.5 mM caused death. Scale bar = 200 μm.
Betulin Protects C. elegans from 6-OHDA-Induced DA Neuron Degeneration
C. elegans has 8 DA neurons, including 1 pair of anterior deirid (ADE) neurons and 2 pairs of cephalic (CEP) neurons in the head, and 1 pair of posterior deirid (PDE) neurons in the posterior lateral region. Selective degeneration of these DA neurons was detected following exposure to 6-OHDA. The fate of DA neurons following treatment with a neurotoxic agent can be monitored in the transgenic strain BZ555, which expresses GFP in DA neurons. In order to test the efficacy of betulin, we determined neuronal viability by measuring reduced GFP expression in DA neurons of 6-OHDA-treated BZ555 worms. At 3 d postexposure of the synchronous L3 worms to 6-OHDA, we found that ADE and CEP neurons had partially reduced GFP expression and that there was also a slight reduction in GFP expression in PDE neurons (Fig. 3A). Worms treated with betulin displayed the highest percentage of intact DA neurons, with ADE and CEP neurons expressing increased GFP (Fig 3A).

Betulin protects Caenorhabditis elegans from 6-hydroxydopamine (6-OHDA)-induced dopaminergic (DA) neuron degeneration. L3 stage worms of the transgenic strain BZ555, with green fluorescence protein (GFP) expression in DA neurons, were exposed to 6-OHDA with or without betulin, prior to further culture for 3 d in the presence or absence of betulin. (A) GFP expression pattern in DA neurons of the transgenic C. elegans strain BZ555. Fluorescence images are shown. Scale bar = 50 µm. (B) Graphical representation of the GFP fluorescence intensity pattern in DA neurons of the BZ555 strain, as quantified using ImageJ software. Data are presented as the means ± standard deviations (n = 10). #Indicates significant differences between 6-OHDA-treated and untreated worms (***P < 0.001); *Indicates significant differences between the 6-OHDA-treated control and betulin/6-OHDA-treated groups (*P < 0.05. **P < 0.01).
We further measured the fluorescence intensity in DA neurons using ImageJ software. In 6-OHDA-treated worms, the mean fluorescence (GFP) intensity diminished by 62% (P < 0.001) compared to untreated worms (Fig. 3B). Furthermore, betulin dose-dependently augmented GFP expression. At 0.5 mM betulin, the fluorescence intensity of GFP expression in DA neurons of 6-OHDA-treated worms increased 1.9-fold (**P < 0.01) compared to worms treated only with 6-OHDA (Fig. 3B).
Betulin Diminishes α-Syn Protein Accumulation
C. elegans lacks the orthologous SNCA (α-syn) gene. However, its genetic flexibility allows transgenic expression of the human SNCA gene. Constitutive expression of YFP-fused human α-syn in the body wall enabled visual assessment of α-syn accumulation by fluorescence microscopy in the C. elegans OW13 strain. Synchronized L3 worms were treated with various concentrations of betulin until the third day of adulthood. Untreated or betulin-treated OW13 worms were observed for α-syn protein deposition range in the head region. Worms treated with betulin displayed notably reduced fluorescence intensity accumulation compared to untreated animals (Fig. 4A). Additionally, betulin dose-dependently decreased YFP expression in OW13 worms. In OW13 worms treated with 0.5 mM betulin, the fluorescence intensity of YFP expression linked to α-syn protein accumulation declined by 49% (***P < 0.001) compared to untreated worms (Fig. 4B). We used the Western blot approaches to verify the improvement of α-syn-YFP chimeric protein is recognized by an antibody specific for human α-syn. Worms treated with betulin showed decreased α-syn protein level compared to untreated worms (Fig. 4C). Moreover, betulin dose-dependently reduced α-syn expression in OW13 worms. In OW13 worms treated with 0.5 mM betulin, the α-syn expression linked to α-syn protein accumulation declined by 55% (***P < 0.001) compared to untreated worms (Fig. 4C).Thus, the accumulation of α-syn protein decreased with betulin treatment in C. elegans.
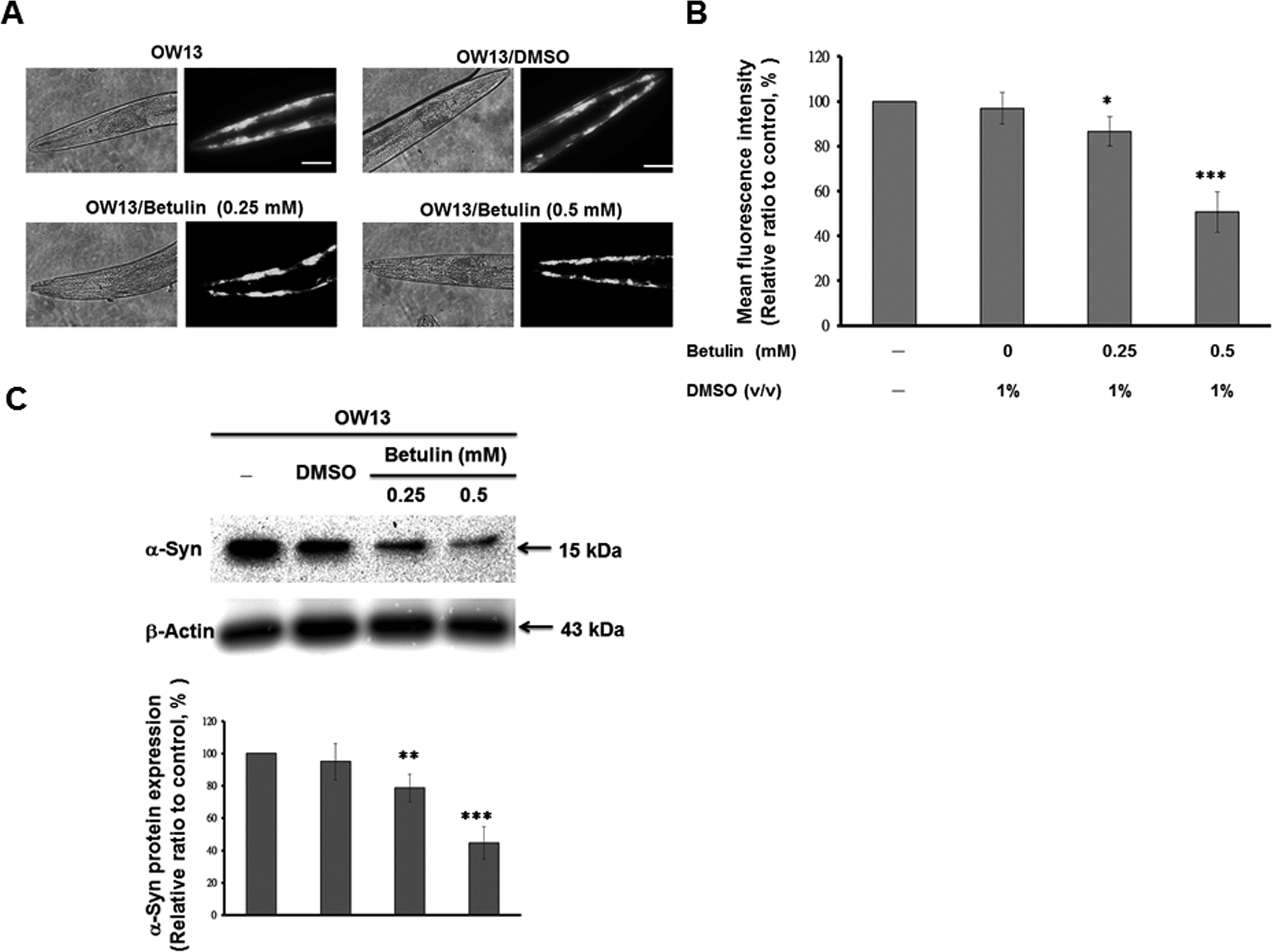
Treatment with betulin decreased α-synuclein (α-syn) accumulation in the OW13 strain of Caenorhabditis elegans. Synchronized L3 stage OW13 transgenic worms were treated with betulin (0, 0.25, or 0.5 mM), cultured until the third day of adulthood. (A) Yellow fluorescent protein (YFP) expression pattern in muscles of the transgenic C. elegans OW13 strain. The figures show representative fluorescence images of α-syn accumulation in the head region. Scale bar = 50 µm. (B) Quantification of α-syn from the fluorescence intensity of YFP expression patterns in muscles of the transgenic C. elegans OW13 strain, using ImageJ software. Comparisons are between the control and betulin-treated groups. Data represent 3 independent experiments and are presented as the means ± standard deviations (n = 10). *Indicates significant differences between the control and the betulin-treated groups (*P < 0.05. ***P < 0.001). (C) Worms were lysed, and the protein level of α-syn was analyzed by immunoblot analysis. One representative result from 3 independent experiments is shown. The expression of β-actin was used as an internal control. The relative fold in protein level was represented as the level in betulin-treated groups relative to that in betulin-untreated controls. The data represent the mean ± SD (n = 3). *Indicates significant differences between the control and the betulin-treated groups (**P < 0.01. ***P < 0.001).
Betulin Improves Food-Sensing Behavior in C. elegans Treated with 6-OHDA
Previous findings indicate that 6-OHDA-treated worms display DA neuron degeneration, which is predicted to disturb food-sensing behavior. 59,66 C. elegans bend their bodies to accomplish migration, and the rate of movement is related to the bending frequency. When worms encounter food, they diminish the bending frequency in order to feed themselves more effectively. 6-OHDA-treated worms, however, fail to decrease their bending frequency in response to food. Hence, the function of DA neurotransmission in C. elegans is related to this food-sensing behavior. We tested whether 6-OHDA treatment in C. elegans induces a defect in this food-sensing response in 3 d worms that were synchronized for age. At 3 d post-6-OHDA, wild-type N2 worms displayed a 41% reduction in bending frequency upon contact with bacteria (Fig. 5). In contrast, 6-OHDA-treated worms displayed a significant reduction in this decremental response compared to wild-type N2 worms (18%, #P < 0.05). Betulin dose-dependently reversed the decremental response of 6-OHDA-treated worms. At 0.5 mM betulin, 6-OHDA-treated worms displayed 1.9-fold reduced bending movements upon contact with bacteria (*P < 0.05), compared to untreated worms (Fig. 5). Therefore, in the 6-OHDA-induced C. elegans model of DA neuron degeneration, treatment with betulin improved food-sensing behavior in C. elegans at 3 d following 6-OHDA exposure.
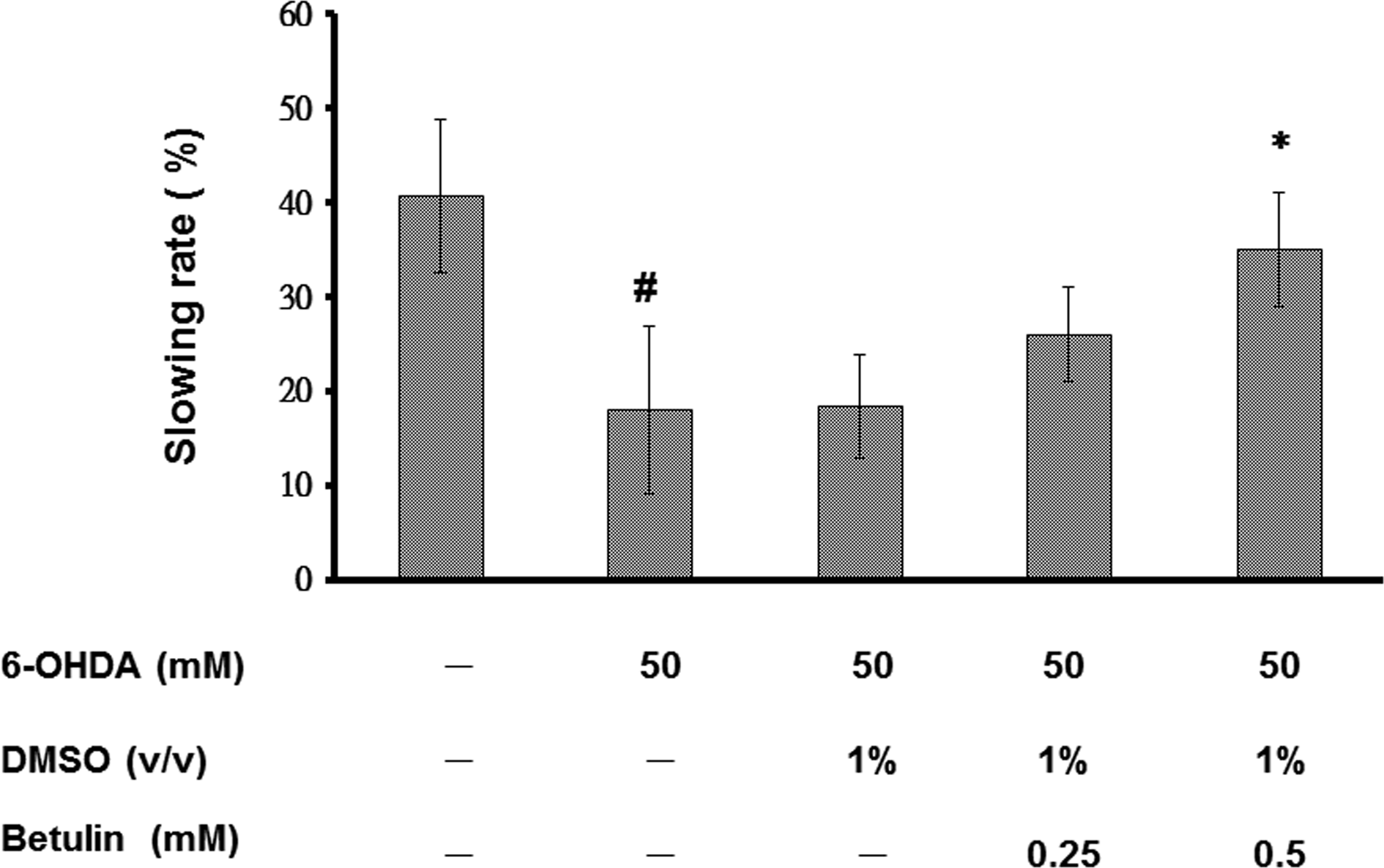
Betulin ameliorated food-sensing behavior in 6-hydroxydopamine. (6-OHDA)-treated N2 Caenorhabditis elegans. L3 stage worms of the wild-type strain, N2, were exposed to 6-OHDA with or without betulin, prior to further culture for 3 d in the presence or absence of betulin. The locomotor rate (frequency of bending) of 6-OHDA-untreated worms, 6-OHDA-treated worms, or betulin/6-OHDA-treated worms with or without bacteria lawns was examined by counting the number of body bends per 20 s. Slowing rates were calculated as the percentage decrease in locomotor rate in the bacteria lawn compared to without a bacteria lawn. Data are presented as the means ± standard deviations (n = 20). #Indicates significant differences between 6-OHDA-treated and untreated worms (P < 0.05); *Indicates significant differences between the 6-OHDA-treated control and betulin/6-OHDA-treated groups (*P < 0.05).
Betulin Augments the Life Span of 6-OHDA-Treated C. elegans
We next investigated the influence of betulin on the longevity of 6-OHDA-treated worms. 6-OHDA-treated worms have a shorter life span compared to wild-type N2 worms (Fig. 6). We found that the DMSO solvent had no effect on the longevity of 6-OHDA-treated worms, and betulin dose-dependently extended the life span. Furthermore, 0.5 mM betulin significantly recovered the life span of 6-OHDA-treated worms. Figure 6 indicates the cumulative survival patterns, as calculated by Kaplan–Meier survival analyses for each group. The mean survival for the betulin/6-OHDA (0.5 mM) group was 20.4 ± 2.13 d versus 14.6 ± 2.44 d for the 6-OHDA group (P < 0.001).

Betulin increases longevity in 6-hydroxydopamine (6-OHDA)-treated N2 C. elegans. Cumulative survival curves of wild-type N2 worms grown on OP50, 6-OHDA-treated worms grown on OP50, and 6-OHDA-treated worms grown on OP50/betulin.
Decreased α-Syn Accumulation by Betulin Treatment Is Associated with Raised Somatic Proteasome Activity via Enhancing Proteasome Regulatory Subunit RPN-1 Expression
Multiple factors, including proteasome dysfunction, cellular apoptosis, and autophagy deficiencies, may contribute to the etiology of PD. Here, we investigated the effects of betulin on the ubiquitin proteasome system in OW13 α-syn transgenic worms. Synchronized L3 stage worms were cultured in the presence or absence of betulin for 3 d. In order to detect whether reductions in total α-syn in the muscle of OW13 worms was the result of increased proteasomal activity, we assayed 26 S proteasome activity upon treatment with betulin, using a proteasome activity test with a fluorescent substrate. As shown in Fig. 7A, the basal level of chymotrypsin-like proteasome activity was 37% lower in OW13 worms compared to N2 worms (#P < 0.001). Betulin treatment significantly and dose-dependently amplified the chymotrypsin-like proteasome activity in OW13 worms, with a 1.7-fold increase following 0.5 mM betulin treatment (*P < 0.01; Fig. 7A). These data indicate that enhanced proteasome activity results in reduced α-syn and that betulin treatment can improve proteasome activity in this C. elegans model of PD.

Betulin enhances proteasome activity by increasing rpn-1 expression in the OW13 strain of C. elegans. OW13 transgenic worms expressing human α-synuclein (α-syn) were cultured from the synchronized L3 stage, in the presence or absence of betulin. (A) Chymotrypsin-like activity of the proteasome was monitored by Z-Gly-Gly-Leu-AMC digestion in a 3 d of adulthood extract of worms containing equal amounts of total protein. Data are presented as means ± standard deviations (n = 3). #Indicates significant differences between N2 and OW13 worms (P < 0.001); *Indicates significant differences between the OW13 control samples and the betulin-treated OW13 samples (*P < 0.05, **P < 0.01). (B) Betulin increases the expression of rpn-1 of the regulatory subunit of proteasome in 3 d adult OW13 C. elegans. Quantitative real-time RT-PCR (qPCR) experiments quantified the expression of the RPN subunit of the 26 S proteasome, using complementary DNA (cDNAs) isolated from OW13 control or betulin-treated worms. Data are presented as means ± standard deviations
In order to investigate the molecular mechanism for the protective effect of betulin, we used qPCR to assess whether the proteasome activity of betulin-treated OW13 worms is associated with increased expression of the regulatory particles of the 19 S proteasome or with the catalytically active subunits of the 20 S proteasome. The basal levels of the subunits were not different in OW13 worms compared to N2 worms, with the exception of proteasome regulatory particle, non-ATPase-like family member (rpn-1), which was slightly decreased. Additionally, betulin increased expression of the rpn-1 regulatory subunit. Expression of rpn-1 following 0.5 mM betulin treatment was amplified 2.1-fold in the OW13 worms (***P < 0.001; Fig. 7B). We also used the Western blot approaches to analyze protein levels of RPN-1 following treatment of betulin in OW13 worms. OW13 worms treated with betulin showed increased RPN-1 protein level compared to untreated worms (Fig. 7C). At 0.5 mM betulin, the RPN-1 expression raised 5.6-fold (***P < 0.001) compared to untreated worms (Fig. 7C).Moreover, we examined the effects of betulin on the mRNA level of rpn-1 at different time points (0, 1, 2, 3, 4, 5, 6, 8, 10, 15, and 20 d after L3 stage) by using qPCR in OW13 worms. Data showed maximum increase in rpn-1 expression at 5 d time point and subsequent lessening of the betulin effect on rpn-1 expression with longer incubation times (Fig. 7D).
Diminished 6-OHDA-Induced DA Neuron Degeneration Following Betulin Treatment Is Associated with Downregulation of Apoptosis Modulator EGL-1
We also aimed to evaluate whether the reduction in DA neuron degeneration in 6-OHDA-treated C. elegans was the result of decreased apoptotic activity induced by betulin treatment. We used qPCR to analyze the mRNA levels of egl-1, ced-3, ced-4, and ced-9, which are associated with apoptosis in C. elegans. As shown in Fig. 8, expression of ced-3, ced-4, and ced-9 did not differ in 6-OHDA-treated worms compared to untreated worms, with the exception of egl-1, which was slightly increased (#P < 0.5). At 0.5 mM betulin, the expression level of egl-1 in 6 OHDA-treated worms was reduced by 55% (**P < 0.01) compared to worms treated with only 6-OHDA (Fig. 8A). We also used the Western blot approaches to analyze protein levels of EGL-1 following treatment of betulin in 6-OHDA-pretreated BZ555 worms. 6-OHDA-pretreated BZ555 worms treated with betulin showed decreased EGL-1 protein level compared to untreated worms (Fig. 8B). At 0.5 mM betulin, the EGL-1 expression diminished by 63 % (**P < 0.01) compared to untreated worms (Fig. 8B). Moreover, we examined the effects of betulin on the mRNA level of egl-1 at different time points (0, 1, 2, 3, 4, 5, 6, 8, 10, 15 and 20 d after L3 stage) by using qPCR in 6-OHDA-pretreated BZ555 worms. Data showed maximum reduction in egl-1 expression at 4 d time point and subsequent lessening of the botulin effect on egl-1 expression with longer incubation times (Fig. 8C).

Betulin diminishes egl-1 expression during modulation of apoptosis in 6-hydroxydopamine (6-OHDA)-treated N2 C. elegans. 6-OHDA-treated L3 stage N2 worms were cultured in the presence or absence of betulin for 3 d. (A) Quantitative real-time RT-PCR experiments quantified expression of egl-1, ced-3, ced-4, and ced-9 using cDNAs isolated from N2, 6-OHDA-pretreated, or 6-OHDA-pretreated betulin-treated worms. Data are presented as means ± standard deviations (n = 3). *Indicates significant differences between the 6-OHDA-pretreated control and 6-OHDA-pretreated betulin-treated groups (**P < 0.01). (B) Worms were lysed, and the protein level of EGL-1 was analyzed by immunoblot analysis. One representative result from 3 independent experiments is shown. The expression of β-actin was used as an internal control. The relative fold in protein level was represented as the level in betulin-treated groups relative to that in betulin-untreated controls. The data represent the mean ± standard deviation (n = 3). *Indicates significant differences between the control and the betulin-treated groups (**P < 0.01). (C) The effects of betulin on the messenger RNA (mRNA) level of egl-1 in 6-OHDA-pretreated N2 worm at different time points. qPCR experiments quantified the expression of egl-1. Data are presented as means ± standard deviations (n = 3). *Indicates significant differences between the 6-OHDA-pretreated control and 6-OHDA-pretreated betulin-treated groups (***P < 0.001).
Downregulation of Rpn-1 and Egl-1 Abolished the Capacity of Betulin to Ameliorate PD Pathology
We used RNA-mediated interference (RNAi) approaches to verify the role of rpn-1 and egl-1 in betulin-treated OW13 worms and 6-OHDA pretreated, betulin-treated BZ555 worms, respectively. In OW13 worms, RNAi decreased expression of RPN-1 by 86% (No. 2, ***P < 0.001) compared to uninterfered worms (Fig. 9A). Rpn-1-downregulated, betulin-treated OW13 worms didn’t display reduction of YFP fluorescence intensity (α-syn accumulation) compared to rpn-1-downregulated betulin-untreated controls (Fig. 9B and C). In BZ555 worms, RNAi decreased the expression of EGL-1 by 84% (No. 1, ***P < 0.001) compared to uninterfered worms (Fig. 9D). Egl-1-downregulated, 6-OHDA-pretreated, betulin-treated BZ555 worm didn’t reveal augment of the GFP fluorescence intensity (intact DA neurons) compared to egl-1-downregulated, 6-OHDA-pretreated, betulin-untreated controls (Fig. 9E and F). Consequently, downregulation of rpn-1 and egl-1 abolished the capacity of betulin to ameliorate PD. Betulin modulated the activation of proteasome system and apoptosis signal transduction pathways that involved in degradation of DA neurons by changing rpn-1 and egl-1 activity.
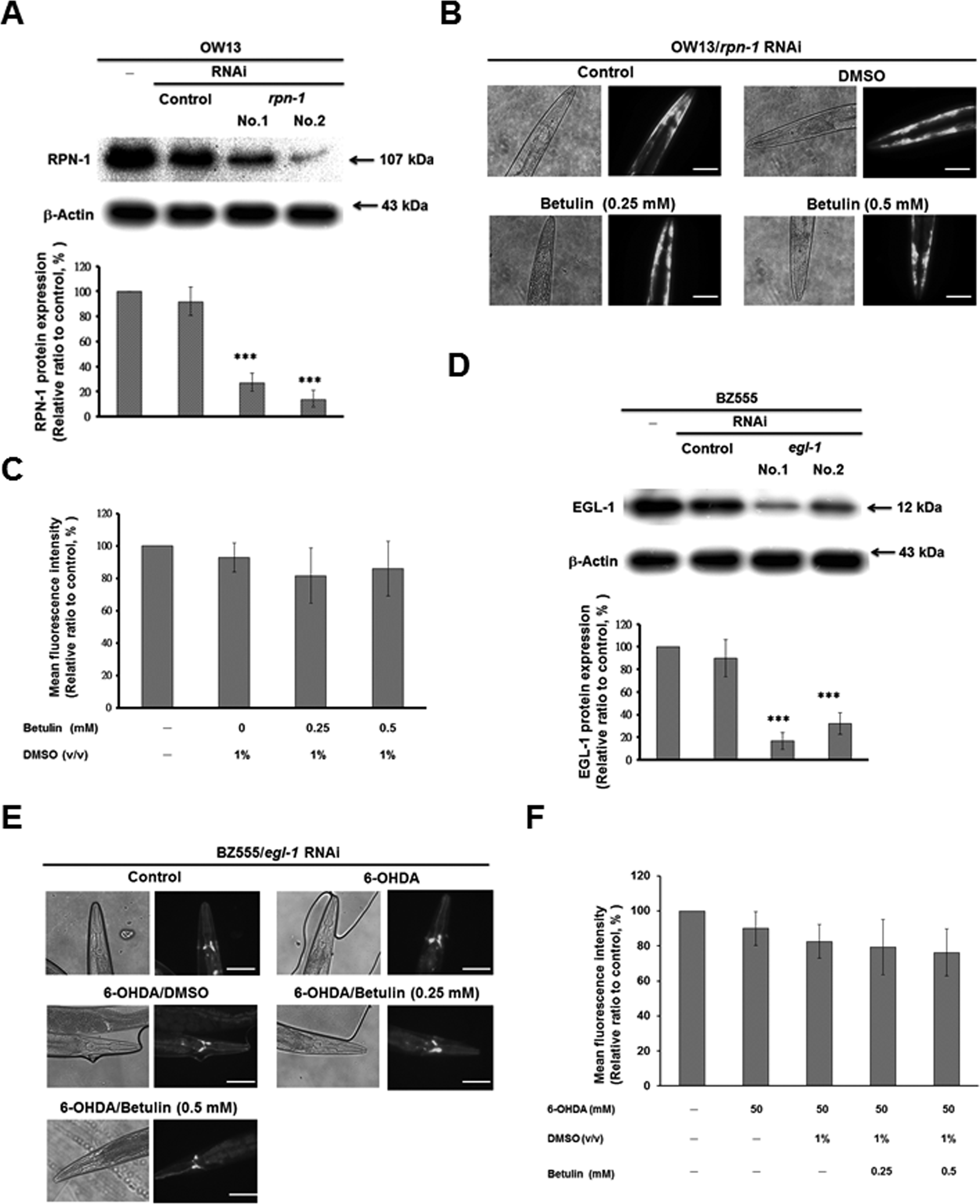
Downregulation of rpn-1 and egl-1 by RNA-mediated interference (RNAi) approaches abolished the capacity of betulin to ameliorate Parkinson’s disease. (A) Rpn-1 RNAi-treated OW13 worms were lysed, and the protein level of RPN-1 was analyzed by immunoblot analysis. The expression of β-actin was used as an internal control. The data represent the mean ± standard deviation (n = 3). *Indicates significant differences between the control RNAi and rpn-1 RNAi-treated groups (***P < 0.001). (B) Yellow fluorescent
Discussion
In the present study, we confirmed that betulin attenuates DA neuron degeneration, reduces human α
DA neurons are more vulnerable to oxidative stress in the 6-OHDA model because this compound generates reactive oxygen species, which are the primary regulators of neuronal apoptosis and death. In particular, 6-OHDA generates the superoxide radical. 69,70 The neuroprotective roles of betulin in 6-OHDA-induced DA neuron degeneration, food-sensing behavior defects, and life span may be related to its antioxidant and antiapoptotic activity. 71 Additionally, mitochondrial dysfunction induced by 6-OHDA results in the release of cytochrome c and activation of caspase-3. 72 Caspase-3 is a key effector in the apoptosis pathway, and is activated through different molecular mechanisms in various mammalian cell types, particularly during cytochrome c–mediated apoptosis. 73 In C. elegans, the BH3-only domain protein EGL-1 antagonizes Apoptosis regulator ced-9, resulting in Cell death protein 4 (CED-4) oligomerization and Cell death protein 3 subunit 2 (CED-3) caspase activation-induced apoptosis. 74 In the present study, we found that betulin reduced egl-1 expression and abolished 6-OHDA-induced apoptosis of DA neurons.
The protective properties of betulin on DA neurons may be associated with these actions. However, the specific mechanisms underlying these results require further investigation.
In various PD models, α-syn accumulation is toxic, particularly because it aggregates in inclusions and aggresomes, blocks the cellular functions that support degradation of these aggregates, and thereby results in neuronal degeneration. 75
These effects may be associated with dysregulation of PD-related chaperones, downregulation of the degradation system, and rapid loss of whole cell homeostasis. 76,77 Betulin reduced α-syn accumulation, thereby blocking its toxic effect in cells. Previous research has demonstrated that an ubiquitin-proteasome mechanism repairs cells from damage under stressful conditions. 78 Therefore, we suggest that the effect of betulin may be associated with amplified expression of rpn1, a subunit of the 19 S proteasome. RPN-1 is exclusively appropriate to coordinate substrate recruitment, deubiquitination, and movement toward the 20 S catalytic core. 79 –81 Enhancing RPN-1 protein levels may increase ubiquitin-proteasome system efficiency and increase α-syn degradation, but the mechanisms of this effect must be studied in more detail.
Autophagy is a lysosomal-mediated pathway causing the degradation of protein aggregates. 82 The steps of autophagy include the formation of a phagophore, elongation, development of autophagosome; fusion of lysosome (autophagolysosome), degradation of the contents, and recycling of degraded material. 83 In mammals, LC3 has been the marker of choice to detect autophagosomes. 84 During autophagosome maturation, LC3 on the outer membrane is cleaved and recycled, whereas LC3 located to the inner membrane remains, as the autophagosome fuses with the lysosome. In C. elegans, the orthologs of LC3 exist as Protein lgg-1(LGG-1) and LGG-2. 85 Monitoring changes in the expression of LGG-1/2 has become a widely accepted method to detect autophagosomes of C. elegans. 86 We used fluorescent reporters of LGG-1 coupled with GFP to monitor autophagosomes of C. elegns in vivo. Results showed that no significant change of level of autophagosomes was observed in the betulin-treated OW13 strain of C. elegans (data not shown).
Evidence suggests that chronic neuroinflammation contributes to the pathophysiology of PD. 87 Activation of microglia and increased levels of pro-inflammatory mediators such as interleukin 1β (IL-1β), tumor necrosis factor-α, and IL-6 have been reported in the substantia nigra of patients with PD, as well as in animal models of PD. 88 These mediators may result in neuroinflammation and neurodegeneration. Moreover, DA neurons are more susceptible to proinflammatory mediators than other cell types. In a previous report, betulin derivatives suppressed LPS-induced inflammatory responses in J774 macrophages. 33 Betulin also markedly decreases the transcriptional and translational levels of inducible nitric oxide synthase (iNOS), as well as the production of nitric oxide, and downregulates the expression of IL-6, monocyte chemotactic protein-1 , and prostaglandin synthase-2 .
Hence, betulin may also improve inflammation in the brains of patients with PD and reduce damage to DA neurons.
Several pentacyclic triterpenes have neuroprotective effects. For example, maslinic acid from the Olea europaea plant promotes synaptogenesis and axonal regeneration by regulating the Akt/GSK-3β signaling pathway. 89 Asiatic acid from Centella asiatica decreases glutamate-induced cognitive deficits, blood-brain barrier permeability, and mitochondrial injury during cerebral ischemia. 90,91 Our results suggest that betulin is a new pentacyclic triterpene with antiparkinsonian properties. This highly accessible compound is a convenient, low-cost, and highly effective means of regulating the survival and function of DA neurons. In the future, we plan to clarify the precise mechanism by which betulin maintains DA neuron activity in SH-SY5Y cells and mouse models of PD, as well as evaluating the suitability of betulin for disease control in clinical trials.
Footnotes
Ethical Approval
Ethical Approval is not applicable.
Statement of Human and Animal Rights
Statement of Human and Animal Rights is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Ministry of Science and Technology (Taiwan) (MOST 104-2314-B-039 -026), the Taiwan Ministry of Health and Welfare Clinical Trial Center (MOHW106-TDU-B-212-113004), and China Medical University (DMR105-058).
