Abstract
Successful immunosuppressive therapy is critical for liver transplantation; however, a considerable number of patients experience fatal rejection or alternatively exhibit serious infection resulting from excessive immunosuppression. The in vitro tacrolimus response of peripheral blood mononuclear cells (PBMCs) before transplantation was compared to the clinical outcome up to 4 weeks after operation in 28 living-donor liver transplant recipients treated with tacrolimus. The tacrolimus IC50 values against concanavalin A-induced PBMC blastogenesis in vitro were calculated. These recipients were classified into two groups with the mean tacrolimus IC50 (0.18 ng/ml) as the cutoff point, after which the clinical outcome between the patient groups was compared. The allograft rejection incidence in the low-sensitivity group (IC50 < 0.18 ng/ml; n = 16) was 6/12 (50.0%), which was significantly higher than the incidence of 2/16 (12.5%) in the high-sensitivity group (IC50 > 0.18 ng/ml; n = 12) (p = 0.0297). In contrast, the infection incidence in the high-sensitivity group was 6/16 (37.5%), which was significantly higher than that of the low-sensitivity group (1/12; 8.3%) (p = 0.0401). These data suggest that patients exhibiting a low PBMC sensitivity to tacrolimus have a risk of rejection, whereas highly sensitive patients have a risk of infection in living-donor liver transplantations under tacrolimus therapy.
Keywords
Introduction
Calcineurin inhibitors including tacrolimus have been widely used for maintenance immunosuppressive therapy in liver transplantations (20,22,25,27,28). However, variations in the clinical efficacy of tacrolimus in individual recipients have been observed and thus recipients with poor responses to the drug therapy have required relatively high doses and experienced fatal rejection episodes and/or serious side effects (19,24).
One of the attractive ways to predict the clinical efficacy of immunosuppressive drugs in individual patients is the cellular pharmacodynamics of drugs using the patients' peripheral blood mononuclear cells (PBMCs) (12, 14). The in vitro response of PBMCs to the suppressive effects of immunosuppressive drugs correlates with the clinical efficacy in renal transplantation (3,5,7,15), asthma (29), minimal change nephrotic syndrome (4,13), psoriasis (16,30), rheumatoid arthritis (18), and ulcerative colitis (11). In addition, there are large individual differences in the PBMC-suppressive effects of glucocorticoids, cyclosporine and tacrolimus in renal transplant recipients, nephrosis and psoriasis (14). However, the PBMC sensitivities to immunosuppressive drugs in liver transplant recipients have not been investigated.
The present study was undertaken to retrospectively examine the relationship between the in vitro response of PBMCs to tacrolimus before the operation and the clinical outcome, including allograft rejection episodes and infection under tacrolimus therapy, in living-donor liver transplantation recipients. The findings are informative for planning individualized immunosuppressive therapy with tacrolimus in living-donor liver transplantations.
Materials and Methods
Patients and Medications
The present research was conducted in accordance with the Declaration of Helsinki and was approved by the Ethical Committee of Tokyo Medical University and the Ethical Committee of Tokyo University of Pharmacy and Life Sciences. Written informed consent was obtained from all patients included in the study. The study included 28 cirrhosis patients (12 male and 16 female; 53.1 ± 8.1 years of age).
The cirrhosis patients examined in this study included 16 with hepatitis C virus (HCV) infection (11 of which had hepatocellular carcinoma), five with hepatitis B virus (HBV) infection (three of which had hepatocellular carcinoma), four with primary biliary cirrhosis, two with autoimmune hepatitis, and one unknown. Liver transplantation is applicable to all of these patients and blood sampling was carried out 1–2 weeks before transplantation. The Child-Pugh classification (8) of these patients was grade A (n = 5), Grade B (n = 9), and Grade C (n = 14), respectively. Profiles of these patients are presented in Table 1. None of the patients included in this study had a history of taking immunosuppressive drugs, including tacrolimus or glucocorticoids, before blood sampling.
Patient Profiles, Tacrolimus IC50 Data, and Clinical Outcome Until 4 Weeks After Living-Donor Liver Transplantation
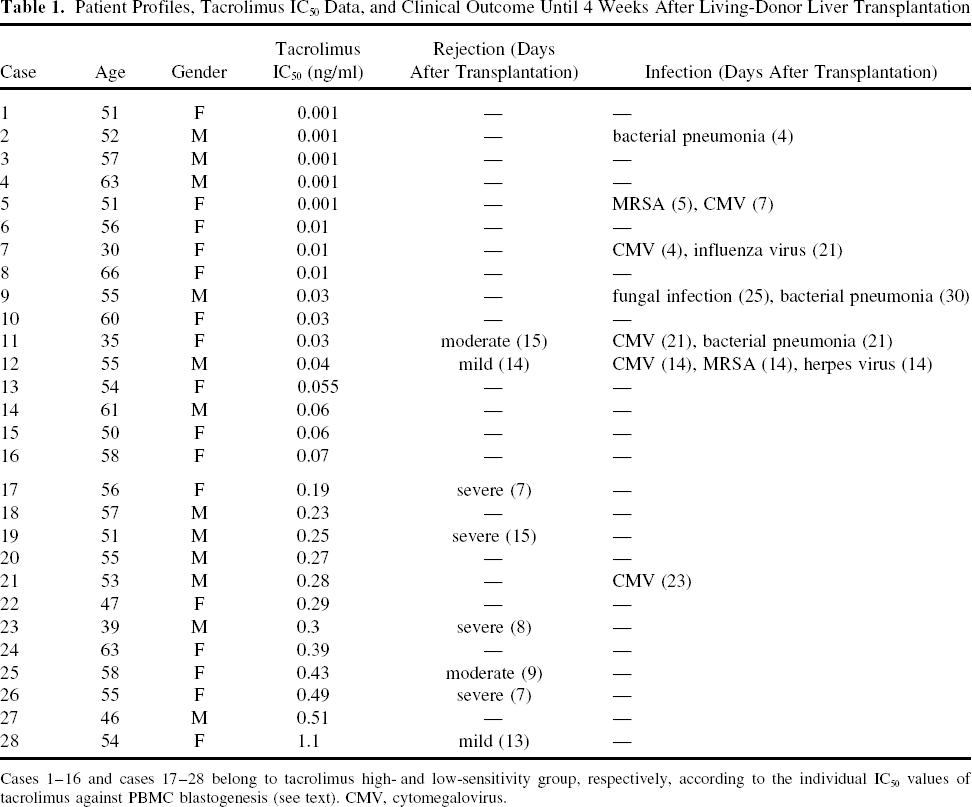
Cases 1–16 and cases 17–28 belong to tacrolimus high- and low-sensitivity group, respectively, according to the individual IC50 values of tacrolimus against PBMC blastogenesis (see text). CMV, cytomegalovirus.
All 28 patients received liver transplantations from living donors during March 2001 to November 2005. The clinical courses of these recipients were retrospectively surveyed until 4 weeks after the operation. All of these patients had received maintenance immunosuppressive therapy with tacrolimus in combination with methylprednisolone. Basically, from 1 to 10 mg/day of tacrolimus was administered to give blood trough concentrations of 8–16 ng/ml during the monitoring period. Tacrolimus blood concentrations were measured by generalized enzyme immunoassay procedures.
Acute allograft rejection episodes were diagnosed by hyperthermia and increases in serum AST, ALT, and/or bilirubin concentrations and were further confirmed by a biopsy of the graft, except in cases 12 and 28. Rejection episodes in cases 12 and 28 were mild and therefore graft biopsies were not carried out in these cases. For the treatment of acute rejection episodes, intravenous pulses of 125–1000 mg methylprednisolone hemisuccinate were given daily for 1–3 days.
Isolation of PBMCs and Evaluation of Drug Effects In Vitro
Twenty milliliters of venous blood was taken from patients and healthy subjects between 0930 and 1100 h and heparinized. This 20-ml sample size was the smallest possible to carry out the drug sensitivity test. The heparinized blood was loaded on 3 ml of Ficoll-Hypaque (Nakarai Co., Japan), centrifuged at 1,300 × g for 20 min, and PBMCs were separated as described previously (12,14,15). For the evaluation of PBMC sensitivity to immunosuppressive drugs, cells were washed and resuspended in RPMI-1640 medium containing 10% fetal calf serum, 100,000 IU/L penicillin, and 100 mg/L streptomycin to a final density of 1 × 106 cells/ml. Concanavalin A, as a mitogen, was added to each well to a final concentration of 5.0 μg/ml. Subsequently, 4 μl of an ethanol solution containing tacrolimus was added to give final agent concentrations of 0.001–10.0 ng/ml. Four microliters of ethanol was added to the control wells. The plate was incubated for 96 h in 5% CO2/air at 37°C. The cells were pulsed with 18.5 KBq/well of [3H]thymidine for the last 16 h of incubation and then collected on glass-fiber filter paper using a multiharvester device and dried. The radioactivity retained on the filter was further processed for liquid scintillation counting. The mean of the counts for a duplicate of each sample was determined. A PBMC stimulation index was calculated from the formula: [3H]thymidine incorporated in the presence of mitogen (dpm)/ [3H]thymidine incorporated in the absence of stimulant (dpm). Drug concentrations that would give 50% PBMC blastogenesis inhibition (IC50) were determined from the dose-response curve.
Materials
RPMI-1640 medium and fetal calf serum were purchased from Gibco Co. (USA). Concanavalin A was obtained from Seikagaku Kogyo Co. (Japan). Tacrolimus was kindly provided by Astellas Co. (Japan). [3H]Thymidine (5.55 × 1011 Bq/mmol) was from New England Nuclear Corporation (USA). All other reagents were of the best available grade.
Statistics
Two-tailed unpaired t-tests and a variance analysis were used for comparisons of the mean values for age, tacrolimus dose, tacrolimus blood concentration, and dose of coadministered methylprednisolone between the two recipient groups (i.e., the high tacrolimus sensitivity group and the low tacrolimus sensitivity group). Fisher's tests were used to compare the proportion of recipients with and without rejection episodes or infections between the two recipient groups. The IC50 values for immunosuppressive drugs are reported as the mean (SD) or median (range). These analyses were performed using the Statview software program (10). In each case, two-sided values of p < 0.05 were considered to be significant.
Results
Individual Variations in IC50 Values of Tacrolimus on the Blastogenesis of PBMCs in Liver Transplant Recipients
Typical dose–response curves for tacrolimus on mitogen-induced blastogenesis of PBMCs from two patients with relatively high sensitivities (IC50 values are 0.001 and 0.03 ng/ml) and two patients with low sensitivities (IC50 values are 0.3 and 0.49 ng/ml) are presented in Fig. 1. The IC50 values of tacrolimus on the blastogenesis of PBMCs from the 28 liver transplant recipients ranged from 0.001 to 1.1 ng/ml (Fig. 2). The baseline characteristics for these recipients, tacrolimus IC50 values and the clinical events including allograft rejection episodes and infections, are shown in Table 1. These recipients were classified into two groups on the basis of their PBMC sensitivity to tacrolimus in vitro with the mean tacrolimus IC50 (0.18 ng/ml) of these recipients as the cutoff point (Fig. 2), after which the clinical outcomes between the patient groups were compared retrospectively (Tables 2 and 3). The recipient group with high tacrolimus sensitivity (IC50 less than 0.18 ng/ml; n = 16) and the group with low tacrolimus sensitivity (IC50 more than 0.18 ng/ml; n = 12) were similar with respect to age, male/female ratio, and their primary liver diseases (Tables 2 and 3).

Typical dose–response curves of tacrolimus against concanavalin A-stimulated blastogenesis of PBMCs derived from four cirrhosis patients (cases 1, 9, 23, and 26; see Table 1) awaiting living-donor liver transplantation.

Distribution of the IC50 values of tacrolimus on the PBMC blastogenesis in the 28 liver transplant recipients. These recipients were classified into two groups according to their PBMC sensitivity to tacrolimus with the mean tacrolimus IC50 value of these recipients (0.18 ng/ml) as the cutoff point (dashed line).
Comparison of the Baseline Characteristics Between Cirrhosis Patients Exhibiting High and Low PBMC Sensitivity to Tacrolimus Before Living-Donor Liver Transplantation

NS, not significant between the two groups.
Number of Cases With Allograft Rejection Episode and Infection in Relation to PBMC Sensitivity to Tacrolimus in Living-Donor Liver Transplantation

Comparative Study of the Clinical Outcome After Liver Transplantation Between the High and Low Tacrolimus Sensitivity Groups
Acute Allograft Rejection and Infection. Acute rejection episodes occurred in eight recipients 7–15 days after transplantation (cases 11, 12, 17, 19, 23, 25, 26, and 28; Table 1). The number of recipients experiencing allograft rejection in the low-sensitivity group was six (cases 17, 19, 23, 25, 26, and 28) out of 12 (50.0%), whereas the number of recipients experiencing allograft rejection in the high-sensitivity group was two (cases 11 and 12) out of 16 (12.5%). Therefore, the incidence in the low-sensitivity group was significantly higher than that in the high-sensitivity group (p = 0.0297) (Table 3). Moreover, the severity of acute rejection episodes in the low tacrolimus-sensitive group was moderate to severe in five cases and was mild in one case, whereas the severity in two cases of the high tacrolimus-sensitive group was mild. In contrast, viral, fungal, and bacterial infections were observed in seven recipients 4–30 days after transplantation (Table 1). The incidence of infections in the high tacrolimus-sensitive group was six (cases 2, 5, 7, 9, 11, and 12) out of 16 cases (37.5%), which was significantly higher than the incidence of 1 (case 21) out of 12 cases (8.3%) of the low-sensitivity group (p = 0.0401) (Table 3). Four recipients of the low-sensitivity group (cases 17, 19, 23, and 26; 33.3%) died from acute allograft rejection, whereas two recipients of the high-sensitivity group (cases 11 and 12) survived instead of experiencing rejection episodes. On the other hand, one patient (case 5; 6.25%) of the high-sensitivity group died from infection after transplantation, while none of the recipients in the low-sensitivity group died from infection.
Other Clinical Indices. The total tacrolimus doses during the 4 weeks after the operation were not significantly different between the high tacrolimus-sensitive group and the low-sensitivity group (Fig. 3a). In addition, tacrolimus blood concentrations were not significantly different between the two groups during the monitoring period (Fig. 3b). The incidences of tacrolimus-related side effects, including nephrotoxicity and cardiotoxicity, other than infection, were also not significantly different between the two groups. These recipients included in the present study occasionally used mycophenolate mofetil and/or basiliximab, but the incidence of any of these drugs between the high and low tacrolimus sensitivity groups was not significantly different. The IC50 values for tacrolimus on the blastogenesis of PBMCs were not significantly different between the cirrhosis patients with hepatitis virus infection and those without hepatitis virus infection (data not shown).
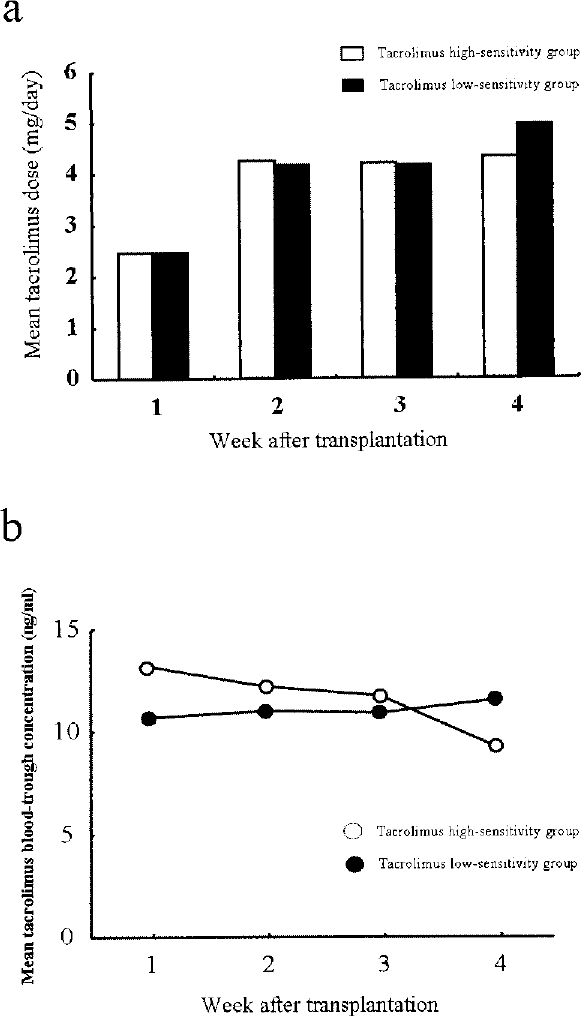
Mean tacrolimus doses (a) and blood trough concentrations (b) in liver transplant recipients with PBMCs exhibiting relatively high (open symbols) and low (closed symbols) sensitivity to tacrolimus in vitro. No significant differences in these indices were observed between the two recipient groups.
Discussion
Acute allograft rejection resulting from insufficient immunosuppression and, conversely, severe infection resulting from excess immunosuppression are both fatal in the early phases of liver transplantation. Therefore, the evaluation of individual responses to the therapeutic efficacy of the drugs before the operation might be critical for successful maintenance immunosuppressive therapy in liver transplantation. The present study showed that the recipients with PBMCs exhibiting low sensitivity to tacrolimus in vitro have a significantly high risk of acute rejection, whereas the recipients with PBMCs exhibiting relatively high sensitivity to tacrolimus have a significantly high risk of infection after living-donor liver transplantation.
Several investigations have been conducted to correlate the cellular pharmacodynamics of immunosuppressive drugs in vitro and the clinical efficacy of the drugs in renal transplantation (3,5,7,15), asthma (29), minimal change nephrotic syndrome (4,13), psoriasis (16,30), rheumatoid arthritis (18), and ulcerative colitis (11). Most of these studies have shown that PBMC sensitivity to immunosuppressive drugs including glucocorticoids in vitro before drug therapy can help in predicting the clinical outcome of the patients after immunosuppressive therapy based on the corresponding drugs (12). In liver transplantation, however, little information is available concerning this topic. Specifically, this study first raised the possibility that the cellular pharmacodynamics of tacrolimus can predict not only the occurrence of acute rejection episodes but serious infection in living-donor liver transplantation.
PBMC responses to tacrolimus in vitro in liver cirrhosis patients showed wide individual differences between subjects in comparison to those in healthy subjects (23). Specifically, the population exhibiting extremely high PBMC sensitivity to tacrolimus was large, in comparison to healthy subjects, in the liver cirrhosis patients (23). These observations suggest that PBMCs of a considerable number of liver cirrhosis patients are vulnerable to the immunosuppressive effects of tacrolimus. These patients may therefore have a high risk of infection after liver transplantation and tacrolimus therapy. The molecular basis for such increased sensitivity of cirrhosis PBMCs to tacrolimus is unclear; however, several changes in immune functions have been reported in patients with liver dysfunction and cirrhosis (2,6,26). However, the increased PBMC sensitivity to tacrolimus does not appear to be associated with HBV infection, HCV infection, hepatocellular carcinoma, primary biliary cirrhosis, or autoimmune hepatitis based on the patient data shown in Table 1 of this study.
A persistent infection with HCV has been suggested to be associated with a weak CD4+ or CD8+ T-cell response during the acute phase (17). The depressed function of peripheral blood dendritic cells is also reported to be associated with the pathogenesis of hepatocellular carcinoma with HBV or HCV infection (31). In addition, reconstitution of HCV-specific T-cell-mediated immunity has been suggested in liver transplantation (1,21). Therefore, HCV infection possibly influences T-cell function and T-cell homing to the liver in HCV-infected cirrhosis patients. However, the IC50 values for tacrolimus on the blastogenesis of PBMCs were not significantly different between cirrhosis patients with hepatitis virus infection and those without virus infection in the present study. Therefore, these observations suggest that a vulnerable change of PBMCs to immunosuppressive drugs is not mainly due to hepatitis virus infection, but is caused by hepatic failure. However, little is known about the direct implication of cirrhosis pathophysiology with immune responses to tacrolimus.
PBMCs of cirrhosis patients tended to show high sensitivities to both glucocorticoids and calcineurin inhibitors (23). Therefore, the common signal transduction pathway(s), through which glucocorticoids and calcineurin inhibitors act to exhibit immunosuppressive effects, may participate in the mechanism(s) of increased PBMC sensitivity to these drugs in the treatment of cirrhosis. A decreased response of lymphocytes to the T-cell mitogen, phytohemagglutinin, has been reported in patients with acute hepatitis and patients with fulminant hepatic failure (9). These patients may show altered PBMC sensitivity to tacrolimus in the present mitogen assay system. However, neither patients with acute hepatitis nor those with fulminant hepatitis were included in this study.
In contrast to the patients with PBMCs exhibiting high sensitivity to tacrolimus, approximately 43% of the patients showed relatively low PBMC sensitivity to tacrolimus. However, the tacrolimus IC50 values in these patients fell into the range of those of healthy subjects (23). Therefore, the data showed that few liver cirrhosis patients exhibit PBMC resistance to tacrolimus, which is different from the observations characterized in renal transplantation (12,14,15). A significantly high incidence of PBMC low sensitivity to tacrolimus is observed in patients with chronic renal failure awaiting renal transplantation, in comparison to healthy subjects (12,14,15). According to these observations, the threshold of tacrolimus IC50 value for the differentiation of the high and low tacrolimus-sensitive patients to carrying out tacrolimus individualized medicine should be determined based on the data of cirrhosis patients, not on the data of healthy subjects.
In conclusion, the present study raises the possibility that evaluating the PBMC sensitivity to tacrolimus before transplantation might be helpful for individualized tacrolimus therapy in living-donor liver transplantation. Tacrolimus doses for maintenance immunosuppressive therapy in patients exhibiting high PBMC sensitivity to tacrolimus is recommended to be reduced, whereas the dose in patients exhibiting relatively low PBMC sensitivity is recommended to be increased. Alternatively, the latter cases should be treated with tacrolimus and glucocorticoids combined with other drugs such as basiliximab and/or mycophenolate mofetil.
Footnotes
Acknowledgments
This study was supported by a Grant-in-Aid for Scientific Research (Grant number 19590162) and the Initiative for Attractive Education in Graduate Schools, from the Ministry of Education, Science, Sports and Culture, Japan.
