Abstract
Personalized medicine has generated massive investments in data integration initiatives and stimulated new flows of health data among multiple actors. Such flows raise questions as to who should be able to access data, for which purposes, and how this access and use should be regulated. We suggest thinking of these questions as matters of ‘data authority’: who can legitimately do what with health data? In this article, we analyze a public debate developing in written media about personalized medicine to understand negotiations of data authority. We demonstrate how the debate creates no consensus and yet seems to stimulate selective regulatory changes. The changes are selective in the sense that they focus on the protection of autonomy but fail to address concerns about, for example, commercial interests. We argue that data authority rests on enduring conflict and that this conflict can be seen as constitutive for personalized medicine as a sociotechnical phenomenon.
Keywords
1. Introduction
Personalized medicine aims to tailor prevention, diagnostics, and treatment to the individual (Prainsack, 2017). To learn what works on an individual level, researchers and clinicians want to make data comparisons with similar patients. Personalized medicine, therefore, presupposes the integration of health data from multiple sources and at a population scale (Prosperi et al., 2018). Accordingly, investments into personalized medicine to a large extent revolve around the integration of data infrastructures. Data integration raises important questions about who should be able to access which data, for which purposes, and how this access and use should be regulated. We are interested in how humans or institutions who interact with data acquire authority to handle data. We suggest the term data authority to describe a collection or use of data, which is deemed reasonable, right, and just by the populace affected, and we believe that a good starting point for understanding the negotiation of data authority is to study how it is publicly debated. In this article, we, therefore, explore public debate about personalized medicine, and the data integration it involves, as the debate has evolved in Danish print media.
We explore what people articulate as “valuable” in relation to data authority by drawing upon what Luc Boltanski and Laurent Thevénot term “orders of worth” (Boltanski and Thévenot, 2006). We relate the debate to regulatory changes suggested by the government in the course of implementing personalized medicine in Denmark. Based on this, we reflect on the role of public debate in relation to data authority. Data authority, we contend, is not a stable state of the agreement but something that can exist to a greater or lesser degree for different people. It is a form of enduring conflict that does not reflect consensus on values or even about the practical implications of given values. Debate, however, should not be seen as simply a threat to personalized medicine: We suggest it is an element of the formation of personalized medicine as a socio-technical and legal phenomenon (Jasanoff, 2011). Data authority shapes the type of medicine a society can offer patients. In this way, critique not only undermines the introduction of new technology; it can also enable new technology (Franklin, 2003).
The ambition of individualized prevention, diagnostics, and treatment is associated with different names, including personalized medicine, precision medicine, and P4 medicine (predictive, preventive, personalized, and participatory) (Hood and Flores, 2012). Irrespective of the nomenclature, the ambition is to achieve greater precision in individual prevention, diagnostics, and treatment. Whereas personalized medicine has previously focused narrowly on genetic components, personalized medicine today has moved into a new era where genetic one of the multiple forms of health data (Prainsack, 2015). Although the discrepancies between the political expectations of what personalized medicine aspires to and the clinical ability to be in line with these aspirations have been pointed out (Dickenson, 2013), health authorities around the world continue to invest in digitizing health records and building infrastructures to enable the necessary data integration. In England, a part of the action plan for personalized medicine is to enable the integration of already existing health data (NHS England, 2016), and in the United States, a part of the Precision Medicine Initiative involves the establishment of a database where the goal is to gather extensive health information on 1 million US citizens (National Institutes of Health, 2020). In Denmark, many of these infrastructures are already in place for routine health data, but not for genomic data. Since Denmark has already invested so heavily in data integration, it constitutes an interesting case for exploring how personalized medicine affects data authority.
Denmark is a welfare state with a publicly funded healthcare system. According to international rankings, the Danish healthcare sector is among the most digitized in the world with pervasive data exchange options (Frost and Sullivan, 2017). Each citizen is provided with a 10-digit identification number (known as the “CPR-number”) assigned by birth or point of immigration. This number serves to identify individuals in interactions with both public and private health services and other sectors (Bauer, 2014). Danish registries contain data on health, housing, education, and financial data on the whole population (Terkildsen et al., 2020), which has led some to describe Denmark as a “Data Heaven” (Holm and Ploug, 2017: 516). The high level of digitization and the comprehensive registries offer a solid ground for the introduction of personalized medicine in Denmark. Previously, comprehensive digitization and data integration have been relatively uncontested in Denmark. However, with the action plans describing the national dedication to realize personalized medicine, and the legislative work necessary to enable the National Genome Center, several actors began voicing concerns about personalized medicine and the data integration it entails. Others mobilized to defend the new initiatives.
A key element in the strategy for personalized medicine in Denmark was the establishment of a National Genome Center in 2019. The purpose of the center is to centralize the storage of genetic data and to facilitate increased use of genetic data by researchers and health professionals in the clinic (Ministry of Health and Elderly Affairs, 2016). However, the centralization of genetic material was not possible under existing legislation, and as a part of establishing the new center, a legislative process was set in motion.
The Danish Health Care Act declares that consent must be obtained prior to any treatment in the healthcare system. Consent can be both implied (when the patients’ actions indisputably signal acceptance of further treatment), verbal, or written depending on the severity of the treatment (Ministry of Health and Elderly Affairs, 2018a §2). Genetic testing in relation to treatment is not explicitly mentioned in the law, but is in practice interpreted in such a way that simple genetic tests require implied consent whereas more extensive genetic testing, with the risk of secondary findings, requires written consent (The Danish Institute for Human Rights, 2019). Consent has to be obtained prior to health data being used for research, but researchers can be granted exemption through an ethics committee in the case that the use of samples and data “does not entail health related risks” and it “would be impossible or disproportionately difficult to obtain informed consent” (Ministry of Health and Elderly Affairs, 2017a §10). The use of health data from registries for research is always exempt from consent and does not require approval from an ethics committee (National Committee on Health Research, 2020). Should citizens not want their biological samples used for research, it has since 2004 been possible to register an opt-out in “The Tissue Application Register” [Vævsanvendelsesregistret]. An opt-out is not possible with other forms of health data (Ministry of Health and Elderly Affairs §29).
In September 2017, the first draft of the law proposed to promote personalized medicine in Denmark was announced. The legislative draft mentions a right to opt out from having genetic test results used for purposes other than treatment (Ministry of Health and Elderly Affairs, 2017b). As with existing samples, this formulation places the responsibility for knowing about the right to opt out with the affected patients. The draft legislation would thereby have extended the lenient legal framework. However, as we will show, it became apparent from the public debate that some participants in the debate found this alternative to consent insufficient. In the revised law following the debate, presented to parliament in February 2018 and later passed, a section called “Information about the possibility to decide over genetic information” was added (Ministry of Health and Elderly Affairs, 2018b §29a). This section states that patients have to be informed about the possibility of opting out through “The Tissue Application Register,” something that is not customary in relation to other forms of treatments in the healthcare sector. Now the responsibility for informing about the possibility of opting out rests with the health services. Still, there is no demand for explicit consent for specific uses. The legislative changes install “genetic exceptionalism” in the law (Ó Cathaoir, 2019), but the debate questioned data integration in a much wider sense. The change in the legislative draft which occurred in the course of the debate could be seen, we suggest, as a response to the debate, but a limited one. Difficult issues relating to privacy of routine data and concerns relating to economic profit were not addressed in the regulatory changes.
Before we turn to describing orders of worth in the public debate on personalized medicine in Denmark, we briefly describe our analytical approach and the empirical material. We then present four orders of worth (Integrity, The Constitutional State, Health, and The Market), which we argue inform the reasoning on data authority in Danish written media. There is only limited agreement among the people debating. Even without agreement, however, we end by suggesting that the public debate might have influenced the implementation of, and legislative framework for, personalized medicine in Denmark. We therefore conclude by suggesting to consider data authority as a state of enduring conflict.
2. Data authority and public debate
Although data infrastructures and lenient legislation are in place in Denmark, they are not the only factors affecting successful data integration. Data use presupposes authority to execute the desired tasks and to delegate this right to designated groups of people (Zambrano, 2001). Delegation is anything but trivial. Max Weber has argued that the basis of authority is that it is perceived as legitimate (Weber, 1959), that is, that the execution of power is perceived as reasonable, right, and just in the populace it affects (Troyer, 2007; Tyler, 2006). Weber argued that authority is constructed differently in different societies and during different periods: sometimes through charisma, sometimes through tradition, and sometimes through legislation based on the rationality in a society (Weber, 1959). How do people in a digitally advanced and economically wealthy country like Denmark justify access to and use of data? How does data authority become contested or confirmed?
Contestation in relation to new technology has been a focal topic in the field of Public Understanding of Science (PUS). Martin Bauer et al. (2007) have, in their extensive examination of PUS research, described how policymakers and researchers often assume that citizens’ criticism of new technology is as associated with lack of scientific knowledge. In her 2015 publication Claire Marris argues that when skepticism is viewed in this way, as based on a knowledge deficit, policymakers construct publics and the criticism they pose as potentially dangerous for the development of new technology. Maja Horst (2008) has pointed out that when people assume that skepticism is caused by insufficient knowledge, they often suggest science education as a solution to contestation around science and technology. PUS researchers have worked to move beyond this “deficit model” of public opinion (Bauer, 2016). All knowledge, including scientific, is situated (Haraway, 1988). How people understand science is affected by several social factors, and skepticism is often related to considerations other than level of scientific knowledge (Sturgis and Allum, 2004; Wynne, 1992). In what has been called the constructivist model (Kerr et al., 2016) or critical PUS (Horst, 2008), publics are viewed as people with legitimate stakes and important inputs when it comes to the development of science and technology. With this movement, publics are increasingly valued as lay experts (Kerr et al., 2016). Although scholars argue that the “deficit model” is still present in science communication (Simis et al., 2016) and public engagements (Marris, 2015), PUS has brought into the light how publics can hold valuable insights when it comes to the development of new science and technology, albeit different insights than those that scientists’ can provide (Wynne, 2008).
The institutional engagement of the publics in the formation of new technology has also become an ideal in relation to personalized medicine (Budin-Ljøsne and Harris, 2015). However, citizens need not necessarily be engaged at a policy level; since the publics are closely bound up with new technology, they are naturally also involved in its development (Amelung and Machado, 2019). In this study, we focus on the people who themselves choose to express their opinion through a public debate and ask: What affects whether people perceive administrations,’ clinicians’, and researchers’ collection of, and access to, health data as reasonable, right, and just—that is, as legitimate?
To answer this question, we draw on the work of French sociologist Luc Boltanski and economist Laurent Thévanot. In their book On Justification: The Economies of Worth (Boltanski and Thévenot, 2006), they define orders of worth as ways of justifying action based on different “modes of reasoning” (Boltanski and Thévenot, 2006: 17). An order of worth involves criteria for evaluating what is reasonable, right, and just. It is a way of looking for expressions of what is perceived as the common good. Accordingly, orders of worth can be used to understand how people evaluate situations and what people see as valuable. Whereas Boltanski and Thévanot identify six specific orders of worth, we take inspiration from David Stark (2011) who suggests identifying different orders of worth from various different empirical settings. We are interested in the values, attitudes, and convictions that inform how people reason on the key issue: who can and should access and process data. Therefore, we look at how the investments in the data integration needed to facilitate personalized medicine are justified in a public debate in Denmark. We explore when data access for different parties appears reasonable, right, and just, that it is how people or institutions gain what we term data authority.
Who participates in a public debate? In 1925, Walter Lippman challenged the idea of the public as one group (Lippmann, 1925), and John Dewey shortly after argued that multiple publics exist (Dewey, 1927). Already here, they questioned the division between a “general public” as a group of ignorant people with no opinion toward an issue and “stakeholders” with strong opinions toward a specific topic. In her re-actualization of the work of Lippman and Dewey Noortje Marres argues that publics emerge around issues. She argues that publics consist of people who are affected by a given issue (Marres, 2005). In line with this, Horst has argued that “the public” should be seen as changing groups of people emerging and dissolving (Horst, 2008). One single issue can also create multiple publics over time (Amelung and Machado, 2019). Databases and population cohorts that establish particular groups of people through data gathering come to construe publics of their own (Hinterberger, 2012). However, different constellations also emerge through different technological mediations: television, newspapers, and social media platforms create different publics (Koed Madsen and Munk, 2019). Accordingly, not only issues but also technological platforms generate publics.
A public is thereby not a predefined group whose convictions can be captured and analyzed. Publics are analytical constructions emerging in reaction to, and in reflection of, issues and technological mediation (Birkbak, 2016). When we study a public debate about personalized medicine, we therefore need to choose not only the specific issue for our focus, but also the technological platform through which we wish to study the issue, and we should not expect, or wish, to find a representative sample of a “total Danish public.” For this article, we decided to explore how data authority is negotiated by those who react to the issue of personalized medicine by articulating their opinion in national written media. The opinions expressed through the opinion pieces and news articles included in this study thus constitute a particular public, which is not representative of all citizens, but informative about opinions curated by the media in which they appear.
3. Method and materials
The debate about personalized medicine in Denmark has unfolded in several forums, including televised and written media, radio programs, social media, the parliament, and professional circles. In this study, we focus our analysis on what we term “the curated written public debate.” It is the unsolicited opinions that people themselves decide to submit to written media to express a particular concern or perspective. We conceive of a debate as attempts by different people to promote opinions about a given topic, in this case through opinion pieces and newspaper articles. We wish to explore debate among people who seek to address the broader population and therefore concentrate on institutionally mediated communication with a national reach. This consists of news articles and opinion pieces written in the national Danish media, including daily papers, weekly magazines, and online media which have been through a form of curation of relevance by editors. Although different magazines and newspapers appeal to specific political stances, we try to take account of this by including all the national newspapers. We excluded regionally specific publications and magazines circulated only within specific professional circles or interest groups. In the following, we will refer to this curated written debate simply as “the debate.”
Literature search
Data were collected from June 2015, when the first policy paper on personalized medicine was published, through April 2020. We used the database Infomedia which is specialized in media surveillance and has digitalized Danish media dating back to 1990. We used the following search words: “personlig medicin” (personalized medicine) and “genomcenter” (genome center) (in the various Danish spellings and deviations). The material included consists of 133 articles and opinion pieces from 11 different news sources (Berlingske, BT, Den Offentlige, Danmarks Radio, Ekstrabladet, Information, Jyllandsposten, Politiken, Sundhedspolitisk Tidsskrift, Videnskab.dk and Weekendavisen). Since many of the documents are from web sources, page numbers do not follow quotations.
Coding
The coding had two stages. The first stage was inspired by what Adele Clarke calls situational mapping focusing on the interests of the speakers (Clarke, 2015) and was carried out as brainstorms on posters. After we used the situational map to stimulate critical thinking and challenge our preexisting assumptions, we applied what Attride-Stirling terms Thematic Network Analysis (Attride-Stirling, 2001). At this stage, we used the data analysis software NVivo 12 to structure the coding. We gathered subcategories into overall themes and only later organized the material into the four orders of worth (see below), which we argue are important for data authority. We returned to the situational map several times during the coding process to revisit previous ideas, to challenge our thematic network, and to secure an overview of the material.
Our first and primary finding in this coding process was that the debate about personalized medicine revolved around the legitimacy of data integration and was thereby basically a debate about data authority. In the thematic coding, we then identified four orders of worth used to justify or challenge particular forms of access or use. One order of worth, which we call Integrity, was especially prevalent in the debate and is presented in its own section. The other three—Constitutional State, Health, and The Market—are described afterward. Before we unfold the different orders of worth, we will describe the material included in the analysis.
4. The contours and participants in the curated written debate
The curated written debate included in this study consists of 83 opinion pieces and 63 news articles. As apparent from Figure 1, the contributions did not occur evenly within the timeframe of the study. Rather, it seems that the contributions are centered around political initiatives to realize personalized medicine, and in particular in time periods where these initiatives are still open for change. There was a clear rise in contributions to the debate in the time after the draft legislation and before the modified legislation was passed.
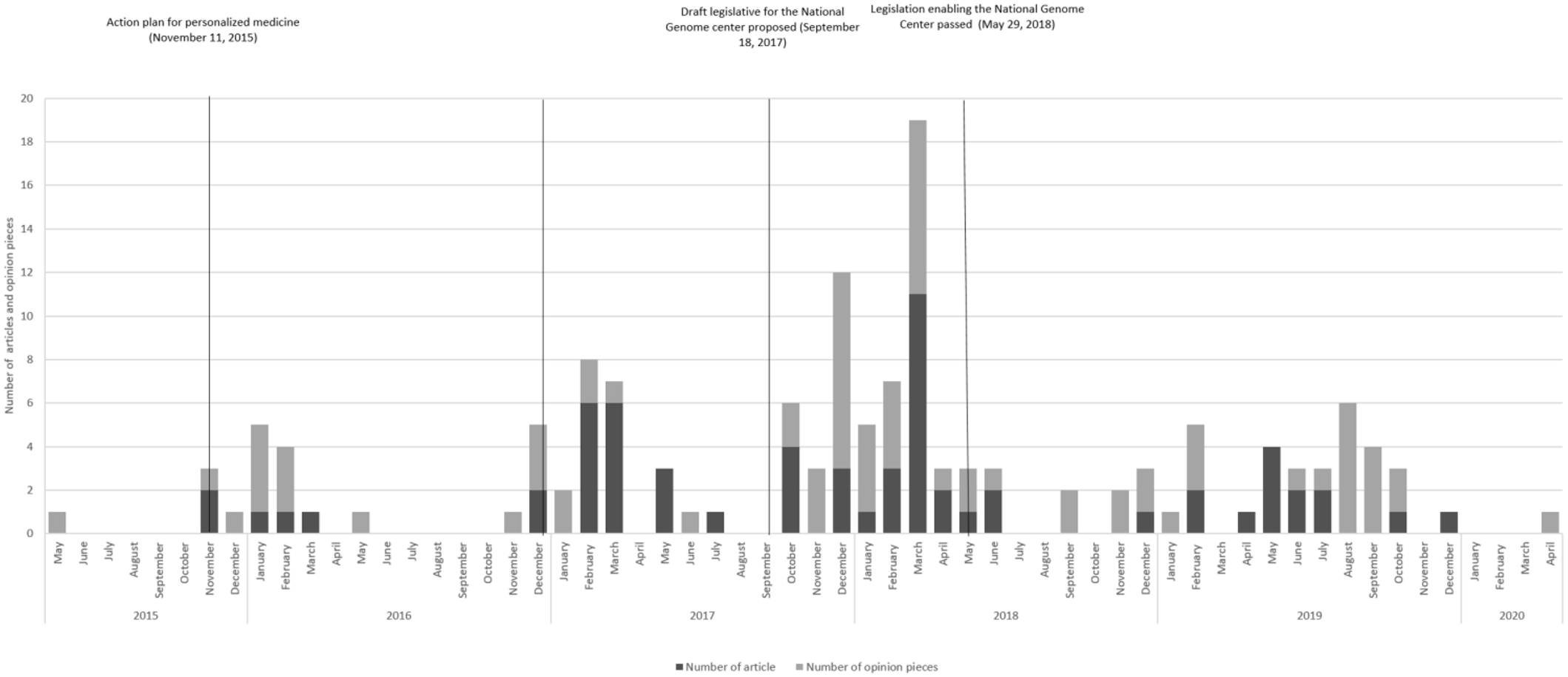
Number of articles over time with political initiatives regarding personalized medicine.
It was not all kinds of citizens who took part in the curated written debate. The opinion pieces and articles we have studied come from a relatively small group of people, a group with certain positions relevant in relation to personalized medicine. As apparent from the quotations below, many names appear several times. Accordingly, the debate contains the views of a small group of clinicians, policymakers, patient organizations, and university professors—all people or institutions with a professional interest in personalized medicine. People who do not hold such professional positions—the patients or potential patients—did not participate in this debate. Accordingly, the opinions presented in the following, are the opinions of people who have an professional interest in personalized medicine. What, then, affects perceptions of data authority in this particular public? Who do they think should access data, for which purposes and under which regulatory framework, and why?
5. Integrity
The key questions about who gains access to which information and how this should be regulated were often debated in a language of respect for the Integrity of the individual. It was the order of worth mentioned by most participants in our material. Integrity as expressed in the debate consists of privacy and the right to act autonomously. Everybody seemed to agree on the inviolability of the individual and that efforts to realize personalized medicine should respect this. However, what this should entail in practice caused considerable dispute. Some saw increased state collection of data as problematic per se, while others argued that the new initiatives taken to implement personalized medicine will help secure privacy thanks to enhanced technological protective measures. The order of worth which we term “integrity” is also about procedures regulating who should decide who can access data on an individual. Here people debate which procedures best secure respect for the autonomy of individuals, primarily focusing on consent and opt-out measures. In relation to the use of genetic tests in treatment and the storage of genetic data for potential reuse, there was no question as to whether consent should be sought. However, what a satisfactory consent entails is not straightforward. In the following, we describe the opinions about privacy and autonomy separately.
Privacy
For many contributors to the discussion, it was crucial that storage and use of health and genetic data do not come at the expense of privacy. However, whose access should be prevented to secure individual privacy was contested. Genetic data were seen as the “main key to the life and existence of every single citizen” (Beich and Birk Kristiansen, 2018), and the right to privacy is argued to be a “human right” (Beich and Birk Kristiansen, 2018). To some, the mere collection of health data threatens privacy because it was seen as a part of the “surveillance happening drop by drop” in Denmark (Lyhne, 2018). The measures taken to realize personalized medicine are described in line with other public initiatives for data pooling, and some argued that “Denmark is opening the way for a surveillance society” (Politiken, 2018). To the people who were worried about surveillance, it was seen as problematic that the state has access to health and genetic information about citizens. In an opinion piece, two general practitioners (GPs) wrote, Prevention in recent years is increasingly aimed at the individual. The state cannot regulate at a citizen level, that is illegal. However, through research you can nevertheless be allowed to make individual profiling about single citizens. In that way it is often the research aim that opens the door to the Danish register data, making it possible to pool and withdraw data without consent. (Birk Kristiansen and Jeppesen, 2018)
Others feared that groups other than the state, namely, private companies or IT (Information Technology) criminals, can access data. The head of the IT Political Association of Denmark was cited to say, My great fear is that information is just going to be registered in big databases where all of it can be pooled. That would be a major catastrophe because we will have a ticking bomb in our hands—and it will simply be a matter of time before the information is leaked. (Jesper Lund cited in Jensen, 2017)
For the group of people who used this line of argument, protecting privacy was seen largely as a question of trustworthy data security. However, many questioned whether data can ever really be secured and hence saw the collection of data in itself as problematic. A third group of people did not see the collection of, or access to, data in relation to personalized medicine as challenging the privacy of individuals. On the contrary, this group argued that the National Genome Center, the institution responsible for centralized storage of genetic data, is a part of the solution to secure privacy, rather than an initiative that will make storage insecure. A politician was quoted explaining that “Today (. . .) the security level is uneven” (Kirsten Norman cited in Stryhn Kjeldtoft, 2018). She argued that the centralization of data in a National Genome Center would secure “a higher, uniform security level” and thereby help secure the privacy of individuals (Kirsten Norman cited in Stryhn Kjeldtoft, 2018).
In short, people who participated in this debate agreed that respect for privacy is decisive for data authority, but disagreed on which types of access by whom could be seen as a breach of privacy. While some feared surveillance from the state, others problematized potential access from stakeholders with commercial interests—and yet others saw investments in personalized medicine as steps toward securing privacy through technological means.
Autonomy
The sentiment that personalized medicine must respect autonomy was manifested through debates about consent and the right not to know. There seemed to be a general agreement that consent should be sought, but there were different notions about what counts as sufficient consent. The debate about consent was messy and people with disparate opinions accused each other of spreading myths and misinformation. This might partly be due to the change in the law regulating information given in relation to genetic testing described in the introduction so that it is now mandatory for health professionals to inform about the possibility to opt out. But, as will become apparent in the following, the messiness is also likely to have arisen from people’s strong emotions about how the regulation of collection, storage, and use of genetic data should look to secure individual autonomy. Some argued that autonomy is respected when patients consent to treatment that involves the sequencing of genetic material. Two representatives from a Patient Organization wrote, “Patients always have to consent to receive treatment. That is a constituted right in Denmark which is called informed consent in the Health Care Act” (Hersom and Freil, 2017). They continued the opinion piece with the argument that a healthcare sector, to maintain full functionality, requires data to be stored. In addition, they argued that access to data is necessary if we are to ensure high-quality care and develop new medicine. Furthermore, some argued that the possibility to opt out of one’s genetic data being used for research, something which in Denmark is only possible for genetic information, tissue and blood samples, is a measure which additionally secures that autonomy is being respected. A former health minister wrote, “The patients can choose themselves that genetic information is only allowed to be used for the purpose of the person’s own treatment. Consequently, you can say no to the information being used for research and statistics” (Nørby, 2019). Others argued that opt-out is not sufficient to count as consent for the storage of genetic information: First of all it is not true consent but only a possibility to opt out, and this is only possible if you know that your data are being transferred to the center at all. Secondly, this opt-out is only possible after the data is gathered in the center (Beich and Birk Kristiansen, 2017).
The two GPs who wrote this opinion piece, along with multiple others in the debate, were not convinced that opt-out respects the autonomy of the individual. They were already discontent with the existing system and used this new initiative to problematize the existing modus operandi where patients themselves had to find out about the opt-out register. Besides, opt-out was seen as problematic by the two GPs because patients could not, and still cannot, prevent genetic information gathered in relation to treatment from being transferred to the center. The fact that genetic data are going to be stored for possible use in quality assessment, for future treatment, or in case of complaints—even if the individual opts out of data being used for other purposes than treatment—caused them to argue that the consent procedure is a “pseudo blocking” (Beich and Birk Kristiansen, 2018). They see it as a pseudo-blocking because patients cannot prevent genetic information from being used for all purposes other than treatment, despite opting out. This concern was echoed by several patient organizations who in a joint letter wrote that it “leaves patients no choice” (Huniche et al., 2018) but to accept that the genetic data are stored. This is because the only way to prevent the storage of genetic information is by declining treatment that involves genetic sequencing. However, the two GPs cited above did not only find it problematic that genetic data were collected without consent. They also took the debate on personalized medicine as a chance to voice their dissatisfaction with the fact that multiple forms of routine data about patients can be correlated without consent (Beich and Birk Kristiansen, 2018).
Another theme discussed in relation to autonomy is the right not to know, and this revolved around protecting individuals from unwanted information about the risk of disease. Specifically, it was about the individual’s right not to receive information on secondary findings in relation to genetic sequencing, although researchers or clinicians might have access to this information. Three geneticists wrote, “Patients’ autonomy is also secured through their possibility to choose whether they want knowledge about secondary findings” (Østergaard et al., 2018). In the debate there seemed to be broad agreement that it is important to respect the right not to know and it was often, as in the quotation above, tied to the autonomy of the individual. However, secondary findings potentially have implications beyond the individual since they can result in information about hereditary genetic disease. While the right not to know was relatively uncontested in the debate, multiple notions of what it takes to respect the autonomy of the individual were expressed.
In sum, for collection, storing, access, use, and distribution of data to be perceived as legitimate by this select group of stakeholders, the integrity of individuals needs to be respected. While people agree on this, they disagree about what this implies in terms of practical solutions just as they disagree on the meaning of the words “privacy” and “autonomy.”
6. Legislation and the importance of purpose
Three other orders of worth also inform perceptions of data authority: The first of these relates to the importance of The Constitutional State to manage data integration. The other two relate to the purposes for which data are used. Where one purpose—the one related to Health—can help legitimize the data authority purposes that are related to commercialization and economic profit challenges it, as will be described below.
The constitutional state
To some, data authority was a question of adequate legislation. However, some challenged this with reference to the flexibility inherent in the law. To those who saw the legislation as a way of securing legitimate data collection and use, it became a question of whether the constitutional state takes charge of setting a framework for the new technology. A policymaker and a clinical doctor wrote, The solution is to make rules and some secure framework for the development of personalized medicine and besides this to include patients in a whole new way from what is being done today. As we see it, this has to happen through a combination legislation which clearly states the rules for sharing of health data, including rules for consent and protection of privacy, and some agreements on information and ethics. (Hersom and Ullum, 2016)
In this opinion piece, we see how a policymaker and a clinical doctor argued that proper legislation can solve the problems around the elements of data authority relating to sharing, consent, access, and protection of privacy, as well as solving the ethical challenges personalized medicine gives rise to. This rationale could not justify to all participants in the debate the implementation of personalized medicine: There is no future guarantee that all purposes are equally good, and after the data are collected in the first place, the law can always be changed. There is no guarantee that some future Folketing [the Danish Parliament] is not going to liberalize the rules for blocking [of data] in for instance ten or 50 years. (Beich and Birk Kristiansen, 2017)
The possibility to change the law in the future led these two GPs to argue that the legislation adopted today will not protect from future harm because the law changes according to the existing normativity in society. By criticizing the law’s ability to regulate personalized medicine in the long term, they also seem to argue that although values and norms in society might change, their own perception of what it takes to respect integrity will not. That legislation can mitigate the risks of personalized medicine therefore did not justify, for all people in the debate, the comprehensive collection and sharing of data.
Health
The purposes for which data are going to be used affected whether the collection, storage, and use of data were perceived as legitimate. In the debate, there was a general sentiment that data integration should be used for purposes that benefit the health of the population, yet there was no agreement on how to do this in practice. Many expected personalized medicine to underpin the health of the population and praised implementation based on expectations of better healthcare: We dare promise that we have the potential to create a medical revolution where we can design treatments that in a new way help the individual and avoid a massive overconsumption of ineffective treatments and the side effects they might have. (Flyvberg et al., 2015)
What counts as population health here is ambiguous: It can be collective health thanks to better health services, while for others it is the health of the individual which matters, and population benefits only arise as a form of accumulation of individual benefits. The manager of the Danish Medicines Agency wrote, The medicine of the future is “tailored to the individual”—also called personalized medicine or more precisely precision medicine. That is, medicine which is developed to be able to be adjusted to the single individual’s genes, physiology and lifestyle. We have to leave the one-size-fits-all mindset where all get a standardized treatment regardless of gender, genetics, age, lifestyle etc. (Senderovitz, 2018)
While no one openly contested the value of promoting better health, some challenged the claim that personalized medicine can really deliver that. People who doubted the efficacy of personalized medicine often pointed to expenditure and argued that the returns to the health of the population are uncertain, such as in this opinion piece from two professors: “Though it sounds appealing, there is limited knowledge on whether personalized medicine will lead to better treatment or only make it more expensive” (Linnberg and Jørgensen, 2016). The two professors continue to argue that Denmark should instead invest in preventive measures such as those targeting smoking and obesity.
The very reasoning behind collective health benefits and more effective service delivery is in this way disputed in the body of opinion we surveyed: There is agreement on the order of worth, but disagreement about the ability of personalized medicine to deliver. Although benefiting the health of the general population seemed like a purpose which secures data authority, not everyone in the debate shared the conviction that personalized medicine is able to do that. In fact, to some, the purposes that legitimize access to and use of health data are the very same as others invoke when contesting access to and use of health data.
The market
While securing a healthy population was almost universally agreed to be a legitimate use of data, economic growth was not—in this debate—considered to be a legitimate purpose. Arguments with roots in utility maximization and the economic value of things were problematized. Despite this, some did argue that the healthcare sector can save money and increase economic growth through investments in personalized medicine. Two policymakers wrote, It will benefit patients through more effective medicine and earlier preventive efforts based on research. But it will also prepare the ground for [economic] growth and help Danish biotech and pharmaceutical companies to develop those new treatment which can be beneficial for patients (Løhde and Hansen, 2016)
However, people who used this rationale normally had a twofold argument: For instance, we can save lives and earn money, or we can enhance research and save money. The Market rationale could not alone justify personalized medicine and was subordinated to other rationales. Many viewed profit motives a key argument against personalized medicine, such as in an opinion piece by a politician: Commercial dreams about [economic] growth and the prospect of economic benefits are the pivotal points of the state’s plan to exploit Danes’ health data and the mapping of the Danes’ DNA profiles in the National Genome Center (. . .). As patient, you might imagine that the state invests money for the sake of the patients with purpose of achieving better treatment. (. . .) Here [in a governmental report] the focus is on creating jobs, and on secondary economic effects and growth in GDP. (Kjær, 2016)
This politician challenged the legitimacy of using health and genetic data for the purpose of economic growth. That potential profit could not justify the implementation of personalized medicine was also obvious when health researchers and policymakers in a joint opinion piece, as a reaction to the accusation of the profit motive behind the introduction of personalized medicine, wrote, “They [health information data] are not going to be used as a commodity (. . .). They are going to be used to develop new treatments for the Danes” (Ullum et al., 2018). Here we see how accusations of motivation based on market values were met with assurances that personalized medicine would offer new treatments to patients. Actions taken based on marketplace rationale alone challenged data authority understood as legitimate access, use, storage and distribution of health data. It is the only order of worth where there seemed to be a general agreement, and yet some sought to suggest that economic profit also could arise along with beneficial outcomes, but without serving as a motivation.
7. Discussion
In the course of studying the debate on personalized medicine, integrity, autonomy, and privacy became key topics, around which strong support aligned. Yet ambiguity remained as to what the support of these orders of worth should imply at a practical level. The inherent disagreement about how to respect Integrity, how and whether The Constitutional State can mitigate potential harm caused by the data integration needed for personalized medicine, and how best to secure the Health of the population showed that conditions for what might be considered legitimate access, use, storage, and distribution of health data were not stable. Data authority seems to be a matter of degree and not something which is there or absent.
The specific public we have studied through an analysis of the written curated debate mainly featured people who are professionally involved with personalized medicine. The orders of worth affecting data authority found in this public might therefore not be applicable to publics made up by patients, potential patients, or their relatives and who may have other considerations when deciding on when they think access to, and use of, health and genetic data is reasonable, right, and just. However, even within this selective group, participants do not view data authority in a homogeneous way. Although people share a professional interest in personalized medicine, they do not necessarily share ideas about what counts as legitimate data collection and use. Furthermore, similar orders of worth can give rise to different positions on the practical implications to be derived from these rationalities. Despite lack of agreement on how to secure legitimate data collection and use, we would like to suggest that the debate in itself might have had implications for the development of personalized medicine in Denmark, and more specifically on the legislation on consent and use of genetic material.
Modification of the legal framework in the course of the debate
As described in the introduction, the first legislative proposal to promote personalized medicine in Denmark extended the lenient legal framework regarding the reuse of health data for research purposes to genomic medicine. This draft suggested that the responsibility for knowing about the right to opt out of genetic data being reused for research should rest with the patient (Ministry of Health and Elderly Affairs, 2017b). However, in the revised law, which was presented to the parliament in February 2018 and passed in May 2018, a new section was added that patients have to be informed about the possibility of opting out through “The Tissue Application Register” (Ministry of Health and Elderly Affairs, 2018b §29a). This only applies for genetic information. Thereby, the new legislation installs a form of genetic exceptionalism (Ó Cathaoir, 2019). In the debate, however, we saw concerns about existing practices around all types of data reuse, including other types of tissue-based research and routine health data. The political reaction nevertheless narrowed down to what was needed to pass the law needed for establishing the National Genome Center. The changes in the final law can be interpreted as a reaction to the debate, although we cannot claim a causal link. However, it seems that a call for autonomy and privacy has translated into a changed emphasis on helping individuals to make their own choices, but only in relation to genomic medicine. Whether these are truly meaningful choices in practice is another issue.
We have seen that orders of worth relating to the promotion of population health are strong and that many clearly feel unease about the potential for economic profit based on health data. However, the law was implemented without stipulating tighter regulation of the profit motive, and to the best of our knowledge, the plans of actions have seen no changes in relation to the promotion of industry access to data. On the contrary, in 2021, the government issued a new life science strategy with the ambition of increasing “fast and easy access” for industrial stakeholders to health data in Denmark (Regeringen, 2021). It appears that public debate cannot affect everything. It would not be the first time that individual autonomy has captured the agenda in relation to ethical and political issues in medicine, and marginalized other social, economic, and political disagreements (O’Neill, 2002).
Although personalized medicine increasingly blurs the boundaries between research and clinic, policies regulating the necessary data access seek to retain the distinction. As of 15 June 2020, the use of genetic information gathered in relation to treatment for research requires ethical committee approval, since it is considered sensitive bioinformatic data (National Committee on Health Research, 2020). At the same time, we see efforts to ease access to genetic information for health professionals in the clinic (Committee of Health and Elderly Affairs, 2020). However, the debate analyzed in this article does not indicate that concerns are specifically related to access for researchers. On the contrary, there is a general sentiment that data should be used to benefit the health of the general population—something that research can advance. Paradoxically, the policy response now potentially impedes public research aimed at furthering population health by making procedures for access to data difficult for them, as people are now actively invited to opt-out of research. Opt-out has become a generic choice, which does not allow patients to distinguish between different motives, researchers, and public and private stakeholders.
8. Conclusion
In this article, we have offered insights into the making of data authority in Denmark, understood as the authority to decide who should access data, for which purposes, and under which regulatory framework. We have done this by looking for orders of worth in a public debate about personalized medicine. We have described four orders of worth (Integrity, The Constitutional State, Health, and The Market) which in different ways affect perceptions of the legitimacy of collection of, access to, and use of health data. The debate indicates a general sentiment about the need to respect Integrity, understood as respect for privacy and autonomy, yet people hold disparate notions of what these terms imply at a practical level. In the order of worth we call The Constitutional State we described how some think that proper legislation can secure data authority, while the possibility of changing the law later made others question whether legislation can mitigate potential harms of personalized medicine. The purposes for which data are used also matter. While using data for the purpose of benefiting the health of the general population seems to sustain data authority, the use of data for the purpose of economic growth challenges this authority. Accordingly, the public debate in Denmark, as presented within news media, was not a forum where an agreement on what counts as legitimate data collection and use could be reached.
The legislative changes that came about in the course of the debate brought about a changed emphasis on helping individuals to make their own choices in relation to genetic testing. It has changed the conditions under which personalized medicine will develop in Denmark—both as clinical practice (clinicians have to talk about the opt-out framework) and as research opportunity (which data will be available). This suggests that debates about data authority are also a part of the development of personalized medicine. However, we have also pointed to the limitations of impact from a debate. Concerns in relation to the potential economic benefits from health data and requests for opt-in solutions are articulated without affecting the implementation of personalized medicine. One could see this as a challenge to data authority, but perhaps we should instead see the clear articulation of many different views as part of establishing authority. It is obvious that there cannot be agreement among all and thereby choices and priorities become necessary. This leads us to suggest that data authority rests on enduring tension between conflicting values. Tensions need not undermine new technology but might contribute to the type of data authority that emerges in response to the new technology.
The concerns in relation to commercial involvement that are expressed in this study are a reiteration of concerns expressed in surveys and qualitative studies of public opinion on the use of health data for purposes other than treatment (Skovgaard et al., 2019). Therefore, the selective Danish legislative response to the public concerns articulated, where the focus is on issues relating to integrity and autonomy—while concerns relating to economics, profit, and who should have access are disregarded—might pose a threat to the long-term sustainability of data integration. There is reason to go deeper into the justification of commercial involvement and address—rather than neglect—these controversial issues. An acceptance of enduring tensions and unresolvable conflict as constitutive of data authority might make open engagement with protests less threatening for authorities. It might even facilitate a more open debate about commercial stakeholders and profit motives when it comes to future uses of health data.
Footnotes
Acknowledgements
We would like to thank Henriette Langstrup, Ivana Bogicevic and Zainab Afshan Sheikh who commented on an earlier version of this article with highly constructive feedback.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from the Carlsberg Foundation (CF17-0016) and from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement number 682110).
