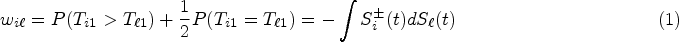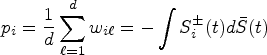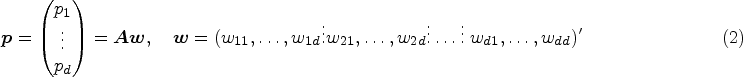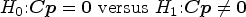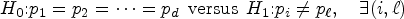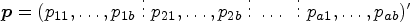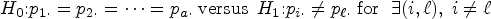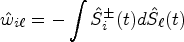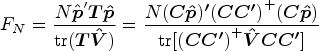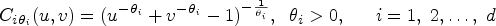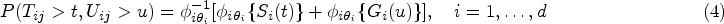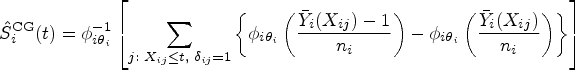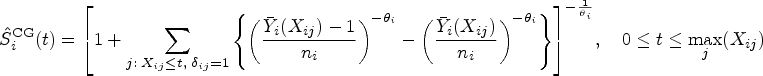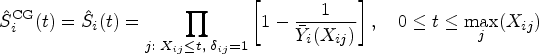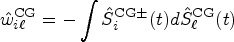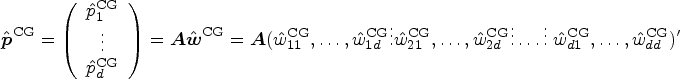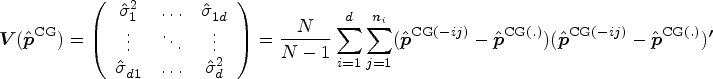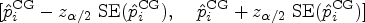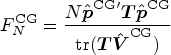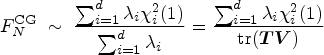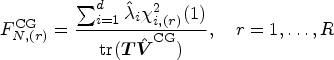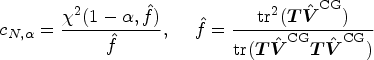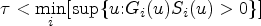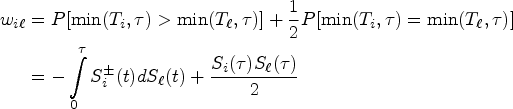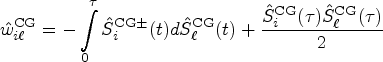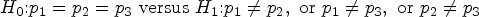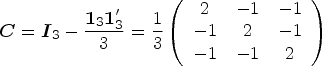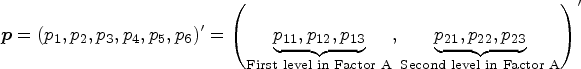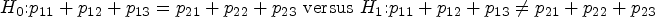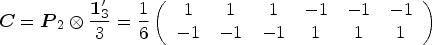Abstract
Factorial analyses offer a powerful nonparametric means to detect main or interaction effects among multiple treatments. For survival outcomes, for example, from clinical trials, such techniques can be adopted for comparing reasonable quantifications of treatment effects. The key difficulty to solve in survival analysis concerns the proper handling of censoring. So far, all existing factorial analyses for survival data have been developed under the independent censoring assumption, which is too strong for many applications. As a solution, the central aim of this article is to develop new methods for factorial survival analyses under quite general dependent censoring regimes. This will be accomplished by combining existing nonparametric methods for factorial survival analyses with techniques developed for survival copula models. As a result, we will present an appealing F-test that exhibits sound performance in our simulation study. The new methods are illustrated in a real data analysis. We implement the proposed method in an R function surv.factorial(.) in the R package compound.Cox.
Introduction
Factorial designs allow researchers to assess treatment effects in one-way, two-way, and other layouts of experimental factors. 1 Factorial analyses offer a variety of treatment contrasts involving multiple treatment factors and their levels. The classical analysis of variance (ANOVA) is a test for detecting treatment effects via the F-statistic under normally distributed outcomes. The F-test is an omnibus test2,3 for the global null hypothesis, where any significance indicates the presence of treatment effects, providing scientific evidence for further explorations of individual factors/treatments.
For survival outcomes, there is a keen interest in adopting factorial analyses for testing treatment effects in cancer clinical trials: see for instance, an ongoing phase III clinical trial for patients with metastatic prostate cancer with a 2 × 2 factorial design. 4 A 2017 survey identified 30 clinical trials with the 2 × 2 factorial design for phase III cancer treatment, 5 including a trial that examined the effect of treatments on survival for patients with advanced cervical cancer. 6
Due to the non-normality of survival data, treatment effects are formulated nonparametrically via the Mann-Whitney effects, 7 pairwise effects using sample partitions, 8 the effects on median survival, 9 or the effects on cumulative hazards. 10 While these metrics provide different survival aspects of treatment effects, they can be identified without the aid of specific distributions. These techniques offer a powerful nonparametric means to detect the main/interaction effects of treatments.
The key difficulty to solve in survival analysis concerns proper handling of censoring owing to the limited follow-up time of clinical trials. 11 However, the existing factorial analyses7–10 were developed under the assumption that the censoring mechanism is independent of survival. Unfortunately, this assumption is imposed for the sake of mathematical convenience, and its validity in real applications is largely questionable.12–18
Dependent censoring arises in the analysis of survival data, especially in data obtained from biomedical studies.14,15,19–29 Typical examples of dependent censoring are the dropout, withdrawal, or removal of patients because of worsening health conditions. For instance, Staplin et al. 15 analyzed the survival of patients registered for an elective liver transplant. Censoring occurs when patients are removed from the waiting list for liver transplants. Transplantation potentially yields dependent censoring because patients closer to death are more likely to receive transplants. Also, patients removed due to deteriorating conditions may have died on the day of removal.
The Kaplan-Meier (KM) estimator for a survival function is systematically biased when censoring is associated with survival time. 30 Andersen and Perme 19 suggested reducing the bias of the KM estimator with the aid of observed covariates. Emura and Chen 16 analyzed the bias of Cox regression and variable selection under dependent censoring. Emura and Hsu 31 studied the bias on two-sample tests based on the Mann-Whitney type treatment effects. Other statistical tools for dependent censoring are reviewed by the books of Emura and Chen 17 and Collett. 20 However, dependent censoring has not been studied in the context of factorial designs.
In this article, we propose a new method for factorial survival analyses, which can take into account dependent censoring. Our method extends the factorial analysis of Dobler and Pauly 7 to the copula-based factorial analysis under dependent censoring. We employ copula models to account for dependent censoring as in Emura and Hsu 31 who worked on a two-sample test. The proposed method leads to an omnibus test for the global null hypothesis and other tests for composite null hypotheses under factorial designs. The method also allows researchers to conduct sensitivity analyses for treatment effects under a variety of copula models for dependent censoring. We implement the method in an R function surv.factorial(.) in the R package compound.Cox. 32
This article is organized as follows. Section 2 reviews the background for factorial survival analyses. Section 3 proposes a new method for factorial survival analyses under dependent censoring. Section 4 explains the software implementation of the proposed method. Section 5 conducts simulation studies to check the performance of the proposed method. Section 6 provides a real data example. Section 7 concludes the article.
Background
We introduce the nonparametric survival analysis method of factorial designs, which was proposed by Dobler and Pauly. 7 This method defines treatment effects and tests them by using censored survival data collected from one-way, two-way, or other layouts.
Treatment effects
In clinical trials and animal experiments, the effects of treatments are often assessed by survival outcomes. The classical ANOVA for testing the equality of normal means is inappropriate since survival data are non-normally distributed. Therefore, the nonparametric formulations of treatment effects are needed.7,33
Consider patients allocated to d different treatments. Let
Following the framework of the nonparametric ANOVA,33,39 Dobler and Pauly
7
defined the relative treatment effect on the i-th treatment relative to the average treatment effect as
A variety of hypotheses can be formulated using a contrast matrix
For a two-way layout, we assume “Factor A” with a levels and “Factor B” with b levels. The treatment effects with interactions are
We review the nonparametric test for
Let
The null distribution of
In this context, our goal is to modify
Proposed method under dependent censoring
We first formulate a survival copula model for dependent censoring. Then, we propose a new estimator of treatment effects and a new F-test for treatment effects under dependent censoring.
Preliminary: Estimating survival under dependent censoring
Recall that survival time
In order to model the structure of dependent censoring, we postulate the survival copula model:
One can estimate
Under the Clayton copula, the generator function takes
We shall derive a new estimator for the relative treatment effects
We first propose to estimate the pairwise effect
Under mild conditions and model (4), the estimator
The variance of
The proposed test
We shall propose a new test for a linear hypothesis
We construct an approximately level
A technical correction is necessary when survival data are collected within a restricted follow-up time. In this situation, the pairwise effects
Sensitivity analysis
The proposed method needs to specify a copula (including its parameter
In this context, we propose a sensitivity analysis under a given copula function with different values of
Software implementation
We implemented the proposed method in an R function surv.factorial(.) available in the R package compound.Cox.
32
The function surv.factorial(.) can compute the estimates of treatment effects
For implementation, we assume that the copula is identical across treatments, namely,
Simulation
Simulation studies were conducted to investigate the performance of the proposed methods. In particular, we evaluate the estimators
Simulation designs
The sample size was
Based on the data, we calculated the estimator
The rate parameters
Parameter settings for the data-generating process are defined by nine scenarios. True treatment effects are beneficial (Bold ) or harmful (Italic) relative to the overall effect (Normal).
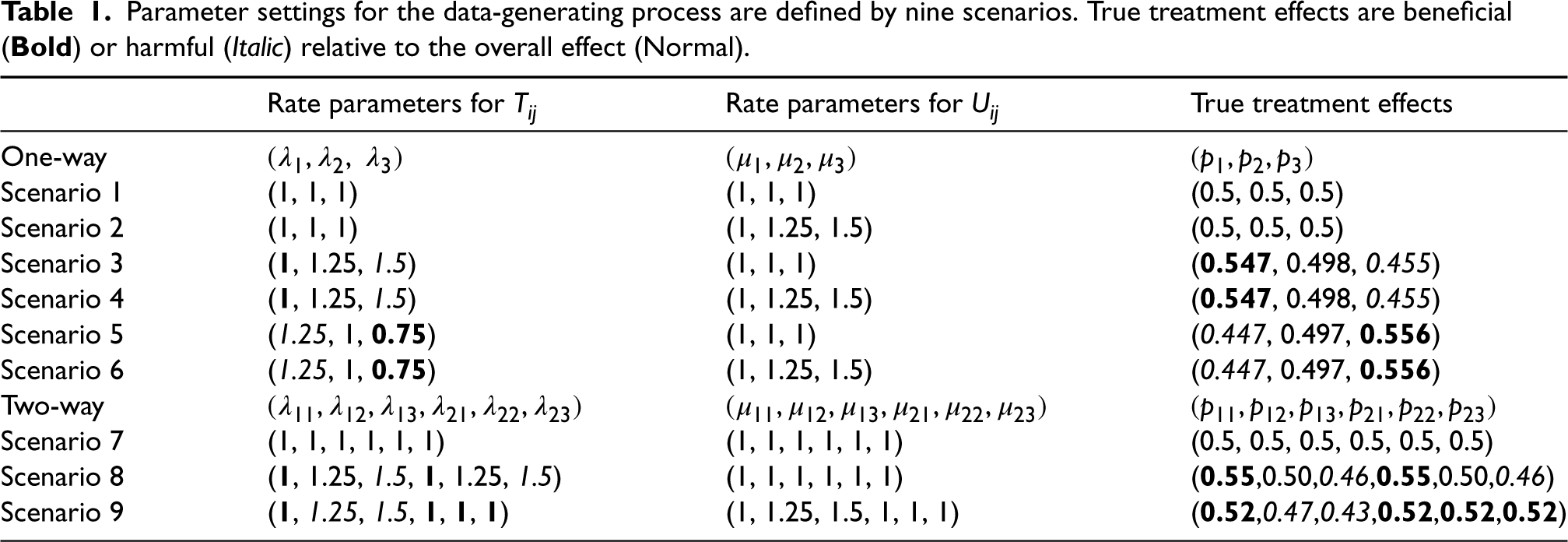
Parameter settings for the data-generating process are defined by nine scenarios. True treatment effects are beneficial (

Based on 1000 replications of simulated data, we assessed the accuracy of the proposed estimators for treatment effects
We first show the results of the estimators and tests when the true value of
When the true value
Table 2 shows the results for the proposed F-test when the true value of
Rejection rates for the proposed test based on 1000 simulation runs under level
.
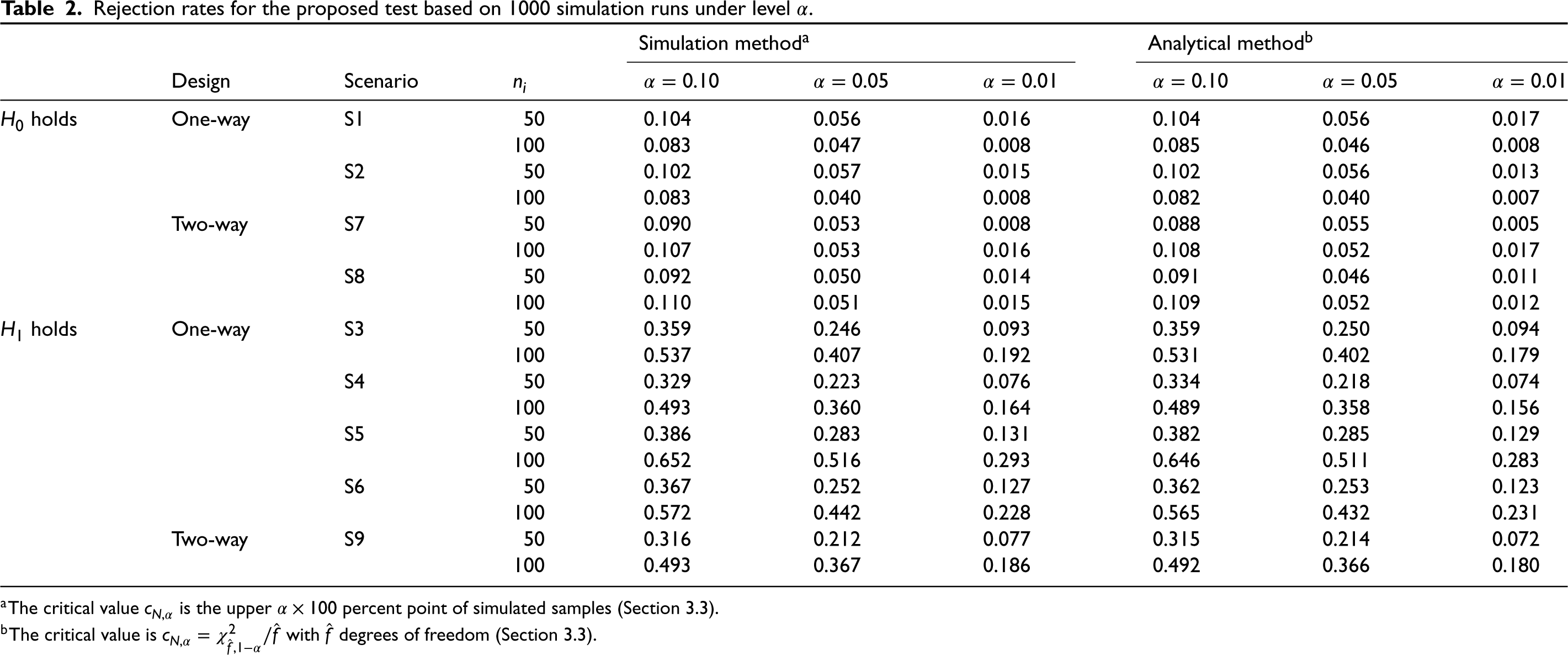
Rejection rates for the proposed test based on 1000 simulation runs under level

The critical value
The critical value is
Figure 1 (the box plots) shows the performance of the estimator
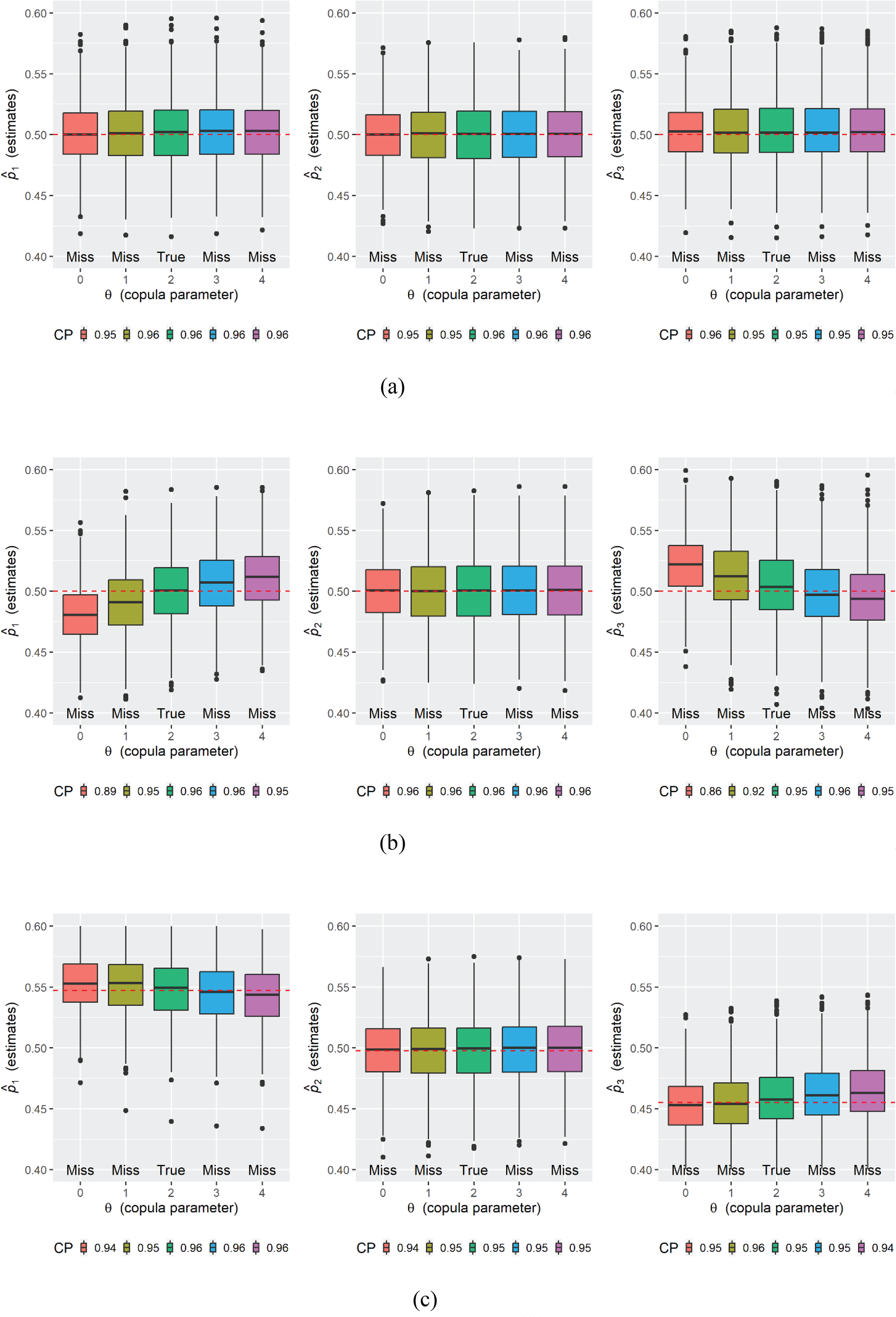
Simulation results for
Therefore, if the independent censoring assumption is mistakenly imposed, the estimator
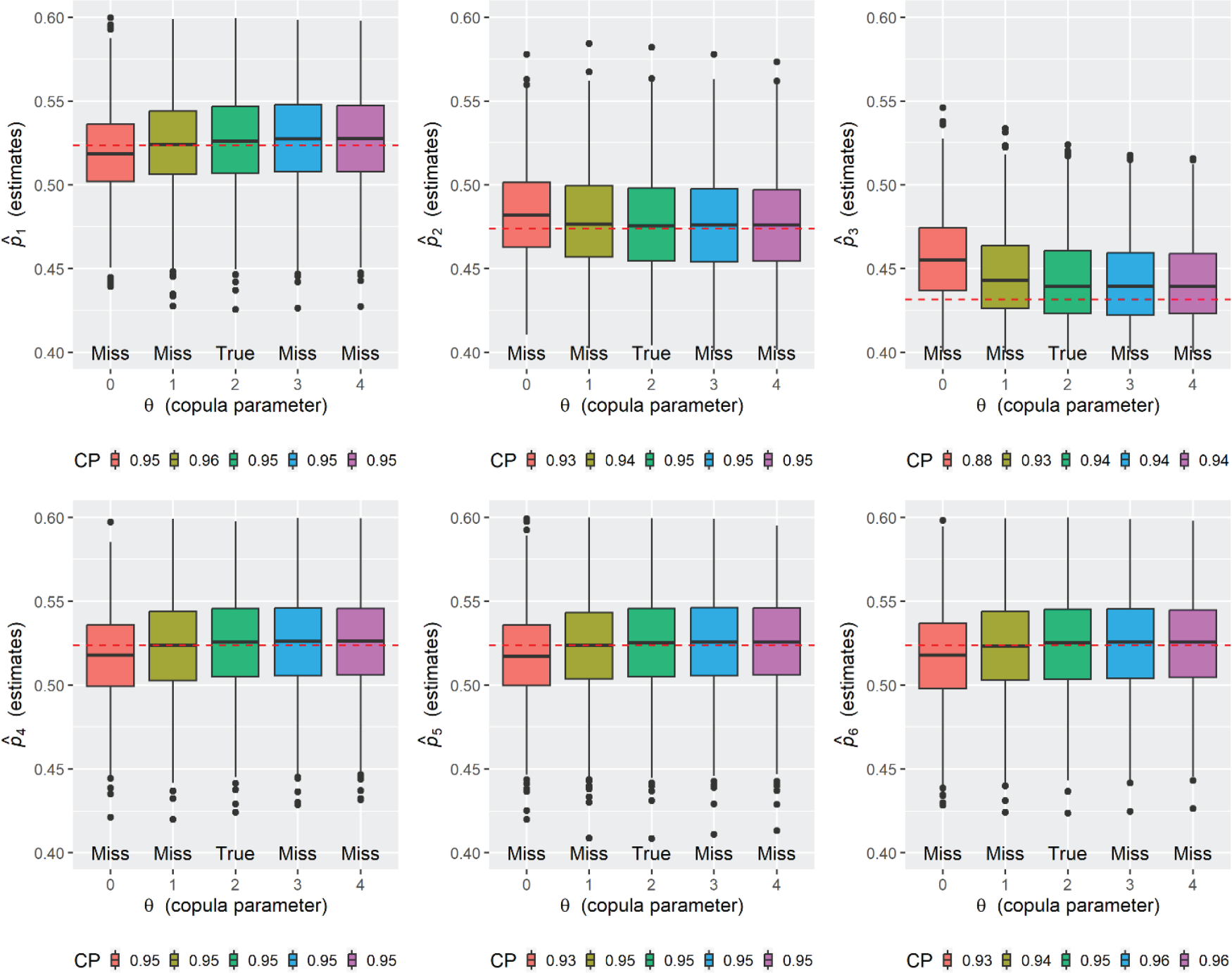
Simulation results for
We analyze breast cancer data available from an R Bioconductor package, curatedBreastData.
53
The data contain survival outcomes and other clinical covariates on 2719 breast cancer patients with advanced breast cancer. In the following analysis, we chose a subset of the data having complete information on both disease-free survival (DFS) outcomes and treatment types. There are three treatment types: adjuvant, neoadjuvant, and mixed (both neoadjuvant and adjuvant). Excluding patients with missing DFS or treatment, we shall analyze a subset of 635 patients. We analyze this subset to compare three treatments in the one-way layout (
Figure 3 displays the estimated survival probabilities for DFS for the three treatments (adjuvant, neoadjuvant, or mixed treatment) by using the CG estimators with the Clayton copula. The adjuvant treatment yields higher survival probabilities than the mixed and neoadjuvant treatments, for all the copula parameters,
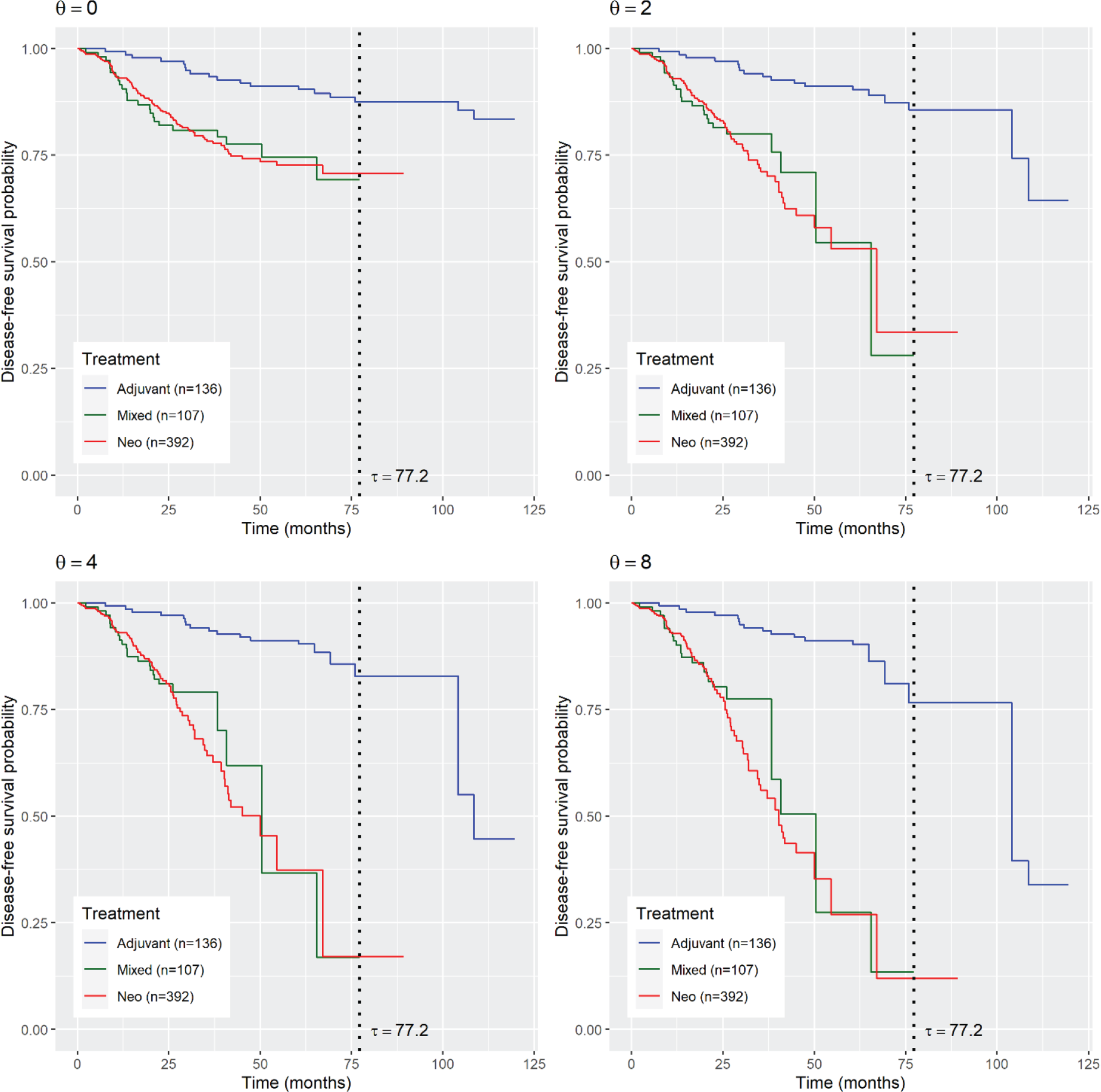
Copula-graphic estimates of disease-free survival (DFS) probabilities for the breast cancer data based on three treatments: adjuvant, neoadjuvant, and mixed (both neoadjuvant and adjuvant). The four panels correspond to the fitted Clayton copula with the parameters
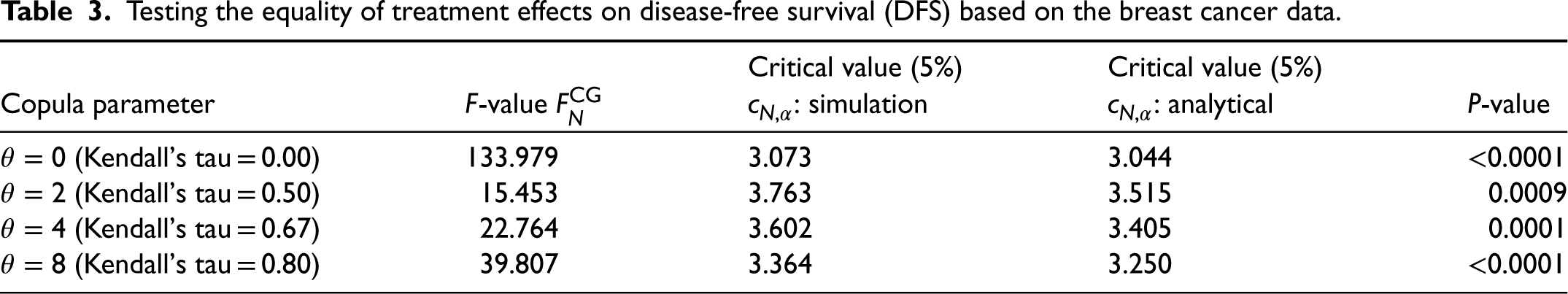
To check the significance of the difference in treatments, we shall perform a hypothesis test. The log-rank test rejected the global hypothesis
In order to see how the conclusion might change under dependent censoring, we applied the proposed F-test. To this end, we perform the test under the parameters
Testing the equality of treatment effects on disease-free survival (DFS) based on the breast cancer data.
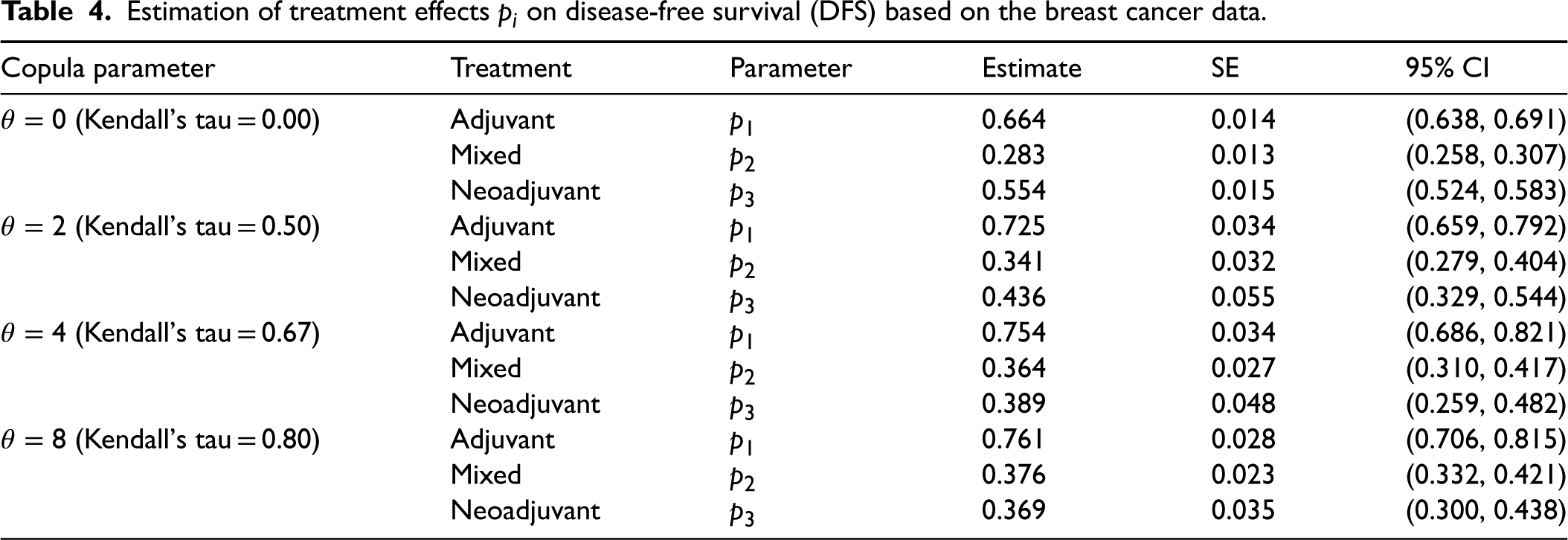
As we detected significant differences in treatment effects, we estimated them by the proposed estimators. Table 4 shows the estimates of
Estimation of treatment effects
We develop a novel method for factorial survival analysis under a copula-based dependent censoring model. While the majority of the traditional survival analyses were formulated under the independent censoring model, we try to challenge this assumption in a situation of factorial designs. This article is the first attempt to examine dependent censoring in factorial designs. Even though copula-based dependent censoring models have already been considered in a variety of settings and applications,12,14,16,17,21,22,27–31 we extend these works toward a new setting: factorial designs.
We derive an F-test that can be applicable to factorial designs with survival outcomes. For the one-way and two-way layouts, the type I error and power properties of the proposed test are well-behaved according to our simulation studies. While we offer two methods to calibrate a critical value of the F-test (the simulation method vs. the analytical method), the practical difference is negligible. The simulation method may have a theoretical advantage due to its more direct approximation to the null distribution, while the analytical method forces a chi-squared distribution to the null distribution. On the other hand, the minor drawback of the simulation method is the need for generating random numbers (i.e. of size
While factorial designs refer to experimentally controlled designs (e.g. randomized clinical trials), the proposed method can also be applicable for observationally collected data with treatment groups as covariates. Indeed, we analyze survival data of 635 breast cancer patients treated by one of three treatments (adjuvant, neoadjuvant, or mixed). The proposed method offers a nonparametric means of estimating treatment effects without assuming any model, such as the proportional hazards model.
Significant survival advantage of the adjuvant treatment is found over the neoadjuvant and mixed treatments. This conclusion is shown to be robust for a variety of dependent censoring scenarios. Nonetheless, the estimated advantage of the adjuvant treatment may be biased since the treatments were not experimentally controlled (i.e. not randomized) and the data were collected observationally. For instance, if the neoadjuvant treatment were administered mainly for patients with poor survival prognosis, their survival would be inherently poor, irrespective of the effect of treatments. Therefore, we cannot exclude the possible bias of the estimated treatment effects due to unbalanced patient characteristics across treatments. It is of interest to develop a method to utilize covariates to adjust for potential biases.
The proposed method relies on the continuity assumption for both survival time and censoring time distributions. Rivest and Wells
30
imposed this assumption to derive the CG estimator and its asymptotic properties. For the CG estimator in the proposed method, this assumption is partially relaxed by the restricted follow-up time (Section 3.4). That is, we only need to assume the continuity for
In our numerical analyses, the Clayton copula was fitted. Nonetheless, other copulas could be tried, such as the Frank copula, especially when negative dependence is suspected for dependent censoring. 12 Our R package (Section 4) allows users to choose the Gumbel and Frank copulas, though we reported our results only for the Clayton copula due to the space limitation. Indeed, it is difficult to justify a suitable copula function by survival data.
The metric of treatment effects is formulated on the basis of pairwise comparison, which is free from model assumptions, such as the proportional hazards model. Recently, pairwise comparison has been adopted for many clinical trials with multiple endpoints 54 with the aid of Buyse's generalized pairwise comparison. 55 This approach was extended to survival outcomes with independent censoring by Péron et al. 56 along with the asymptotic theory. 57 Extension of the generalized pairwise comparison under dependent censoring is of great interest; see a relevant work for the extension to competing risks outcomes. 58
Supplemental Material
sj-r-1-smm-10.1177_09622802231215805 - Supplemental material for Factorial survival analysis for treatment effects under dependent censoring
Supplemental material, sj-r-1-smm-10.1177_09622802231215805 for Factorial survival analysis for treatment effects under dependent censoring by Takeshi Emura, Marc Ditzhaus, Dennis Dobler and Kenta Murotani in Statistical Methods in Medical Research
Supplemental Material
sj-r-2-smm-10.1177_09622802231215805 - Supplemental material for Factorial survival analysis for treatment effects under dependent censoring
Supplemental material, sj-r-2-smm-10.1177_09622802231215805 for Factorial survival analysis for treatment effects under dependent censoring by Takeshi Emura, Marc Ditzhaus, Dennis Dobler and Kenta Murotani in Statistical Methods in Medical Research
Supplemental Material
sj-pdf-3-smm-10.1177_09622802231215805 - Supplemental material for Factorial survival analysis for treatment effects under dependent censoring
Supplemental material, sj-pdf-3-smm-10.1177_09622802231215805 for Factorial survival analysis for treatment effects under dependent censoring by Takeshi Emura, Marc Ditzhaus, Dennis Dobler and Kenta Murotani in Statistical Methods in Medical Research
Footnotes
Acknowledgements
The authors thank the Editor and two referees for their valuable suggestions that improved the paper. Emura T was supported financially by JSPS KAKENHI (22K11948; 20H04147). Ditzhaus M was funded by the Deutsche Forschungsgemeinschaft (grant no. DI 2906/1-2). Dobler D would like to thank his new affiliations where a small part of the work has been done: Department of Statistics, TU Dortmund University, and Research Center Trustworthy Data Science and Security, University Alliance Ruhr, Germany. A draft of this manuscript was presented by Emura T in an organized session (EO350: Beyond proportional hazards and standard survival), CM Statistics 2022, London. Comments from the audience helped improve the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deutsche Forschungsgemeinschaft, Japan Society for the Promotion of Science (grant number DI 2906/1-2, 20H04147, 22K11948).
Supplementary data
Online supplemental materials related to this article are additional simulation results, the R code for the simulation studies, and the R code for the data analysis.
Appendices
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.