Abstract
One of the main questions in the design of a trial is how many subjects should be assigned to each treatment condition. Previous research has shown that equal randomization is not necessarily the best choice. We study the optimal allocation for a novel trial design, the sequential multiple assignment randomized trial, where subjects receive a sequence of treatments across various stages. A subject's randomization probabilities to treatments in the next stage depend on whether he or she responded to treatment in the current stage. We consider a prototypical sequential multiple assignment randomized trial design with two stages. Within such a design, many pairwise comparisons of treatment sequences can be made, and a multiple-objective optimal design strategy is proposed to consider all such comparisons simultaneously. The optimal design is sought under either a fixed total sample size or a fixed budget. A Shiny App is made available to find the optimal allocations and to evaluate the efficiency of competing designs. As the optimal design depends on the response rates to first-stage treatments, maximin optimal design methodology is used to find robust optimal designs. The proposed methodology is illustrated using a sequential multiple assignment randomized trial example on weight loss management.
Keywords
Introduction
In many randomized controlled trials, participants are equally allocated to intervention arms. Such a design is consistent with the view of clinical equipoise that must exist before the start of the trial. 1 However, it may be preferable to allocate more participants to one arm than to another, for instance, when variances and/or costs vary across the treatment arms,1–5 or when outcomes are categorical rather than quantitative.6–10 The derivation of the optimal allocation of units to treatment conditions has not only been done for individually randomized trials, but also for more complex trial designs such as cluster-randomized trials,11–16 and trials with partially nested data.17–19 From a statistical point of view, it is more efficient to assign more subjects to the condition with the lowest costs and highest variance. Other, more practical, reasons to use unequal allocation over equal allocation include resource constraints, administrative, political or ethical concerns or when the aim is to gain experience from an intervention and to study its feasibility.5,20
The focus of these references is on trials where subjects are randomized to either one single treatment or a combination of treatments, but do not change their assigned treatments during the course of the trial. This is a drawback since in real research practice some subjects may benefit more from one treatment and others more from another. Adaptive treatment strategies (ATSs), which are also called dynamic treatment regimens or adaptive interventions, are more flexible in the sense that they allow changing treatments over time.21–24 An ATS individualizes treatments to subjects via decision rules that adjust the type, intensity, dosage or delivery of a treatment and specify when, whether and how to proceed at certain critical clinical decisions. For instance, those subjects for whom their assigned treatment turns out to be beneficial may continue the same treatment, while those others may be assigned to another treatment. The use of sequential treatments is often necessary because of: (i) heterogeneous treatment outcomes across subjects, (ii) change in treatment goals over time, (iii) the need to balance potential risks and benefits or (iv) to reduce costs when intensive treatment is not necessary.25,26 Also, the use of sequential treatments implies multiple clinical decisions to be taken throughout the course of the study. These clinical decisions are formalized through ATSs.
Based on the number of treatments and treatment switches, various competing ATSs may be developed and they may be compared to one another in a so-called sequential multiple assignment randomized trial (SMART).25,27 SMARTs are multi-stage randomized trial designs that are used to inform on the development of multiple ATSs embedded in it. The use of SMART designs allows researchers to evaluate the timing, sequencing and adaptive selection of treatments by using randomization and developing the best sequence(s) of treatments that lead to the optimal outcomes in the long term. In SMARTs, participants are allowed to switch through multiple stages, where each stage corresponds to a clinical decision, and subjects may be randomized at each stage. Sequenced randomization ensures that at each decision point the groups of participants assigned to the intervention options are balanced in terms of patient characteristics. This adds flexibility, allowing participants to remain on those treatments that are having an effect and giving the possibility to switch away to patients being treated with less effective options. This has made SMART designs appealing in a broad variety of health care, behavioural and psychological settings.
Multiple ATSs are embedded in a SMART and the main question in the design phase of a SMART is how many subjects should be assigned to each ATS, and whether an unequal allocation is better than an equal allocation. Some recent papers studied the relation between sample size and power for SMART designs,25,28–34 but did not study the optimal allocation of units to treatment sequences and the loss of efficiency of using equal rather than unequal allocation.
The aim of this paper is to derive optimal allocations of units for a prototypical SMART design. This is a two-stage design where all units are randomized to two treatment conditions in the first stage. Those who respond to their assigned treatment are not re-randomized in the second stage, while those who do not respond are re-randomized to two second-stage treatments. This design was considered earlier by NeCamp et al. 32 in the setting of a cluster-randomized trial. In our contribution, we focus on individual randomization. We focus on sample sizes to be used when comparing two ATSs that start with different first-stage treatments. Four of such pairwise comparisons can be made in their prototypical SMART design, and one comparison may be of more importance than another. We therefore use multiple-objective optimal design methodology to consider all comparisons simultaneously, while taking into account their relative importance. 35 Multiple-objective optimal designs are useful when the study has multiple and conflicting objectives, such multiple pairwise comparisons of marginal means of ATSs in a SMART. It combines these objectives in one optimality criterion and tries to seek a design that is highly efficient for each of these criteria. We provide a Shiny App to calculate the optimal allocation of units and to evaluate the efficiency of the design with equal allocation. We demonstrate our optimal design methodology on the basis of a SMART example that compares two different treatments, nutrition (NUT) and physical activity (PHY), for weight loss management. Our focus is on SMARTs with a quantitative outcome with individual randomization. In other words, we do not focus on cluster-randomized SMARTs or other complex SMART designs with clustered data.
The remainder of our contribution is organized as follows. Section ‘Prototypical SMART design’ further discusses the prototypical SMART design and its embedded ATSs. Furthermore, this section introduces the example of weight loss management. Section ‘Derivation of the optimal design’ derives the optimal allocation of units for studies in which either the total sample size or the budget is fixed. In the latter case, we consider the realistic situation where costs may vary across treatment conditions. The optimal allocation turns out to depend on the subjects’ probabilities to respond to their first-stage treatment. We therefore also focus on maximin optimal designs that are robust to incorrect prior estimates of these probabilities. Furthermore, Section ‘Derivation of the optimal design’ introduces the Shiny App that we developed for finding the optimal design. Section ‘A SMART example’ demonstrates our optimal design methodology on the basis of the weight loss example. It shows how the optimal design is influenced by the costs per treatment, proportion of responders to first-stage treatments and the relative importance of the four pairwise comparisons. Section ‘Discussion’ summarizes our findings, discusses limitations of this contribution and gives directions for future research.
Prototypical SMART design
Before we focus on the prototypical SMART, we rehearse some general ingredients for arbitrary SMART (see for instance Ertefaie et al.,
36
but using different notation). The observed covariates and treatment assignment at stage k are denoted
The prototypical SMART design is visualized in Figure 1. This design has been used in various research fields; published examples of its use in the treatment and long-term management of many chronic conditions include weight loss,26,37,38 substance abuse,39,40 cancer research,41,42 adolescent depression, 43 adolescent conduct problems, 44 suicide, 45 and attention-deficit/hyperactivity disorder. 46
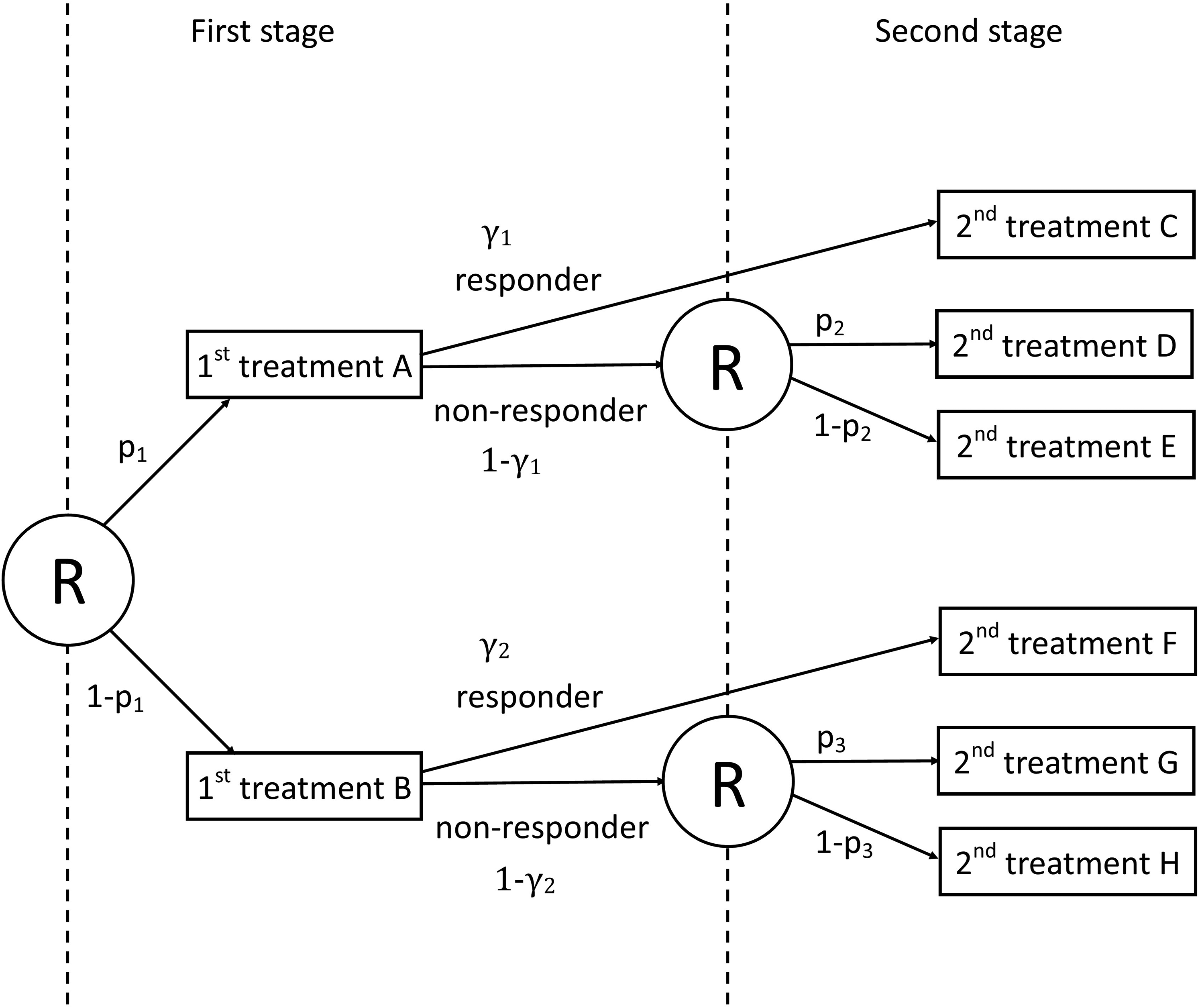
A scheme of the prototypical sequential multiple assignment randomized trial (SMART) design from NeCamp et al. 32 Circled ‘R’ denotes randomization at each stage. p1 and (1 − p1) are, respectively, the proportions of subjects receiving first-stage treatments A and B. p2 and (1 − p2) are, respectively, the proportions of subjects receiving second-stage treatments D and E for non-responders starting with first-stage treatment A. p3 and (1 − p3) are, respectively, the proportions of subjects receiving second-stage treatments G and H for non-responders starting with first-stage treatment B. γ1 and γ2 indicate, respectively, response rates for the first-stage treatments A and B.
The prototypical SMART is a two-stage design with two first-stage treatments A and B; the proportions randomized to these treatments are denoted
Four ATSs are embedded in the prototypical SMART design, see Table 1. For instance, the first ATS, denoted
The four ATSs embedded in the prototypical SMART design.

ATS: adaptive treatment strategy; SMART: sequential multiple assignment randomized trial.
The primary analysis goal of a SMART design is usually one of the following: (i) comparing first-stage intervention options; (ii) comparing second-stage intervention options; (iii) comparing two or more embedded ATSs in the study starting with the same first-stage intervention option or (iv) comparing two or more embedded ATSs in the study starting with different first-stage intervention options. 31 In the derivation of our optimal design, we focus on embedded ATSs that start with different first-stage treatments, which is a common primary aim in SMARTs. 32
Example: weight loss management
Bariatric surgery is an effective treatment for obese patients to lose weight. Given its costs, potentially harmful side effects and the risk of death, patients in the Netherlands are only considered eligible if they can demonstrate they have previously attempted other means to lose weight. Two treatments are an increase in PHY and a change in NUT.
Figure 2 visualises the example SMART design. All patients are first randomized to either PHY or NUT. Then, at the end of the first stage, subjects are categorized as responders or non-responders, according to some predefined definition of response, for example, a threshold for weight loss after a given period of time. Non-responders are then re-randomized to second-stage treatments, regardless of their treatment in the first-stage. They either switch to the other treatment or pursue with a combination of both treatments (NUT + PHY) in the second stage. Responders are not re-randomized and pursue with their first-stage treatment. This example is visualized in Figure 2. Four different ATSs are embedded within this prototypical SMART design: (i)
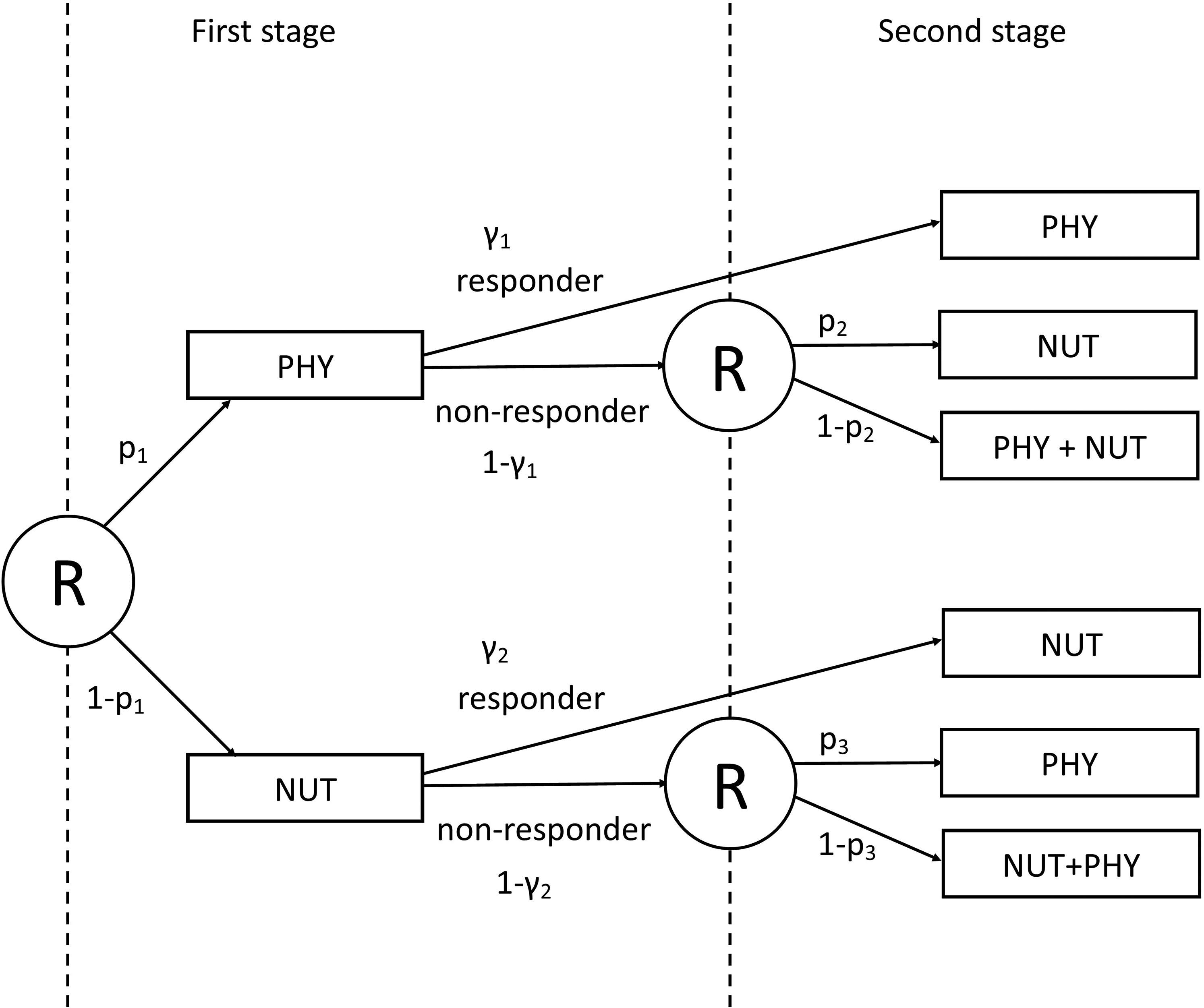
A scheme of the example SMART design on weight loss. Circled ‘R’ denotes randomization at each stage. p1 and (1 − p1) are, respectively, the proportions of subjects receiving the two first-stage treatments: PHY and NUT. p2 and (1 − p2) are, respectively, the proportions of subjects receiving second-stage treatments NUT and NUT + PHY for subjects starting with PHY as first-stage treatment. p3 and (1 − p3) are, respectively, the proportions of subjects receiving second-stage treatments PHY and NUT + PHY for subjects starting with NUT as first-stage treatment. γ1 and γ2 indicate, respectively, response rates for the first-stage treatments PHY and NUT.
The SMART design of this example is a simplification of the prototypical SMART design in the sense that just two treatments are involved. Responders continue with their first-stage treatment, while non-responders are randomized to the other treatment or a combination of both treatments. This specific SMART design was previously used for, among others, the treatment of anxiety disorder, 25 obsessive–compulsive disorder 47 and chronic pain. 48
Derivation of the optimal design
Introduction
For a given ATS
The weights follow from the fact that there is a structural imbalance between responders and non-responders: the non-responders are re-randomized but the responders are not. For instance, for ATS
The weighted mean for the continuous primary outcome of interest for ATS 



Following from (4), we obtain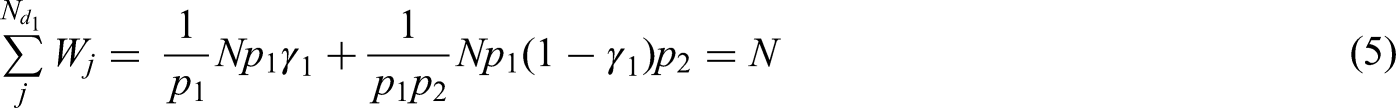
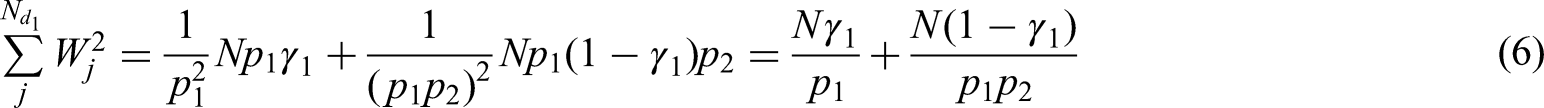

Using their respective subject-specific weights, formulae for the variance of the weighted mean
Variance for the weighted mean

We consider pairwise comparisons of ATSs that start with different first-stage treatments. The expected difference in weighted means of two such ATSs 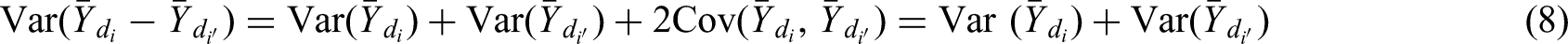
Considering the ATSs embedded in our example, four possible pairwise comparisons exist, with corresponding variances: (i)
The optimal design 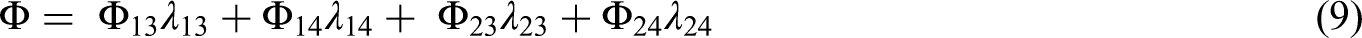
Optimal design under a fixed total sample size
In this scenario, the optimal design is sought under a fixed total sample size N. This is a realistic scenario when studying treatments for a rare disease or condition, but it can also be used when resource constraints allow recruiting a fixed number of subjects. It is assumed that a priori estimates of the response rates
The optimal design minimizes the objective in (9); it is found by taking the gradient of (9) with respect to 

The optimal randomization probability for the first-stage treatment A takes on a more complicated form:
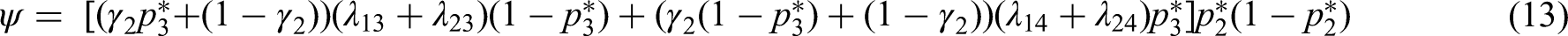
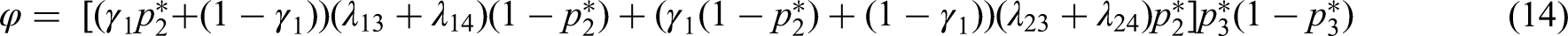
Optimal design under a fixed budget
In this scenario, we consider a budgetary constraint: the total costs C for treating subjects should not exceed the budget B. The costs are calculated as
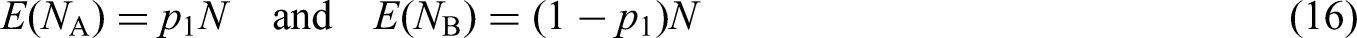
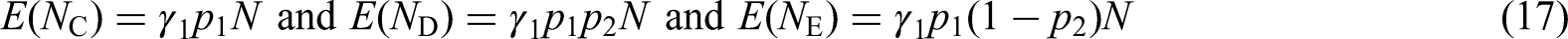
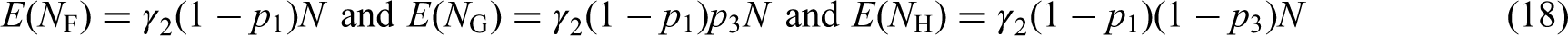
Robust optimal design
The optimal design depends on the response rates
The maximin optimal design Define the parameter space for the response rates and the design space For each possible combination of the two response rates in the parameter space, compute the locally optimal design For each design in
This procedure yields the design which is most robust to a misspecification of the response rates and it can be used when working under a fixed budget or under a fixed total sample size.
Statistical power for the optimal design
Once the optimal allocation to treatments has been derived, it makes sense to determine how much power the study has for each of the four pairwise comparisons of ATSs
51
. The following steps should be taken in such a power analysis:
Calculate the variance For each of the four pairwise comparisons of ATSs: calculate For each of the four pairwise comparisons of ATSs, get a prior estimate of the expected difference in marginal means For each of the four pairwise comparisons of ATSs, select the type I error rate For each of the four pairwise comparisons of ATSs, calculate the power. For a one-sided alternative use the following equation:
where 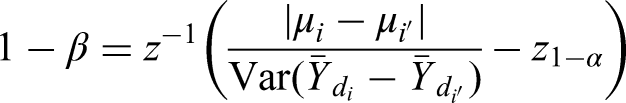
Shiny app
We developed a Shiny app 52 to facilitate finding the optimal design; it is available from https://andreamorciano.shinyapps.io/OptimalSMART/. It calculates locally optimal designs for a fixed total sample size as well as a fixed budget. In the first case, the user should specify the total sample size, in the latter case, he or she should specify the costs per treatment along with the budget. Furthermore, an a priori estimate of the two response rates should be specified to find the locally optimal design. The numerical algorithm that finds the optimal design for the budgetary constraint has a precision of 0.00002 for the optimal proportions.
The Shiny app can also be used to find the maximin optimal design. It that case intervals
A SMART example
Introduction
We apply the optimal design methodology to the example of the weight loss management study of Figure 2. Participants are randomized to two first-stage treatments: PHY and NUT. A response is defined as a (absolute or relative) loss in body weight that exceeds a user-selected threshold value. We use three sets of a priori guesses for the two response rates of the two first-stage treatments:
We consider three sets of weights for the multiple-objective optimal design (9). The first considers each comparison to be of equal importance, which implies that equal weights are used:
For this specific example, we developed another version of our Shiny app; this is available at https://andreamorciano.shinyapps.io/OptimalSMART2/.
Locally optimal design under a fixed total sample size
For each combination of
Locally optimal design: optimal proportions for first-stage
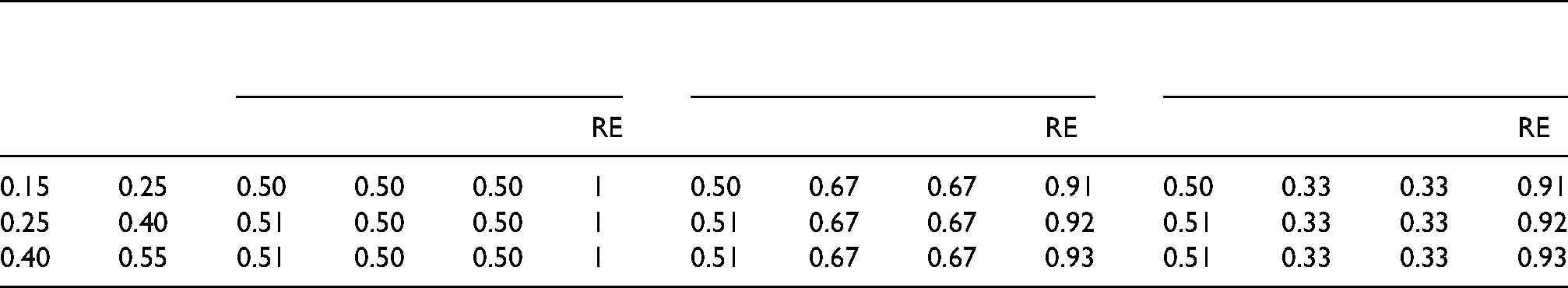
The results do not necessarily apply to other combinations of weights and response rates, so a researcher who is planning a SMART is advised to use our Shiny app to derive the optimal design for the trial at hand, and to do a sensitivity analysis to study how the optimal design is influenced using by various realistic combinations of weights and response rates.
Locally optimal design under a fixed budget
To find the optimal design under a budgetary constraint, the costs for both treatments and the budget need to be defined. We assume both stages are of equal length, so the costs do not vary across stages. The costs for combined treatment are the sum of the costs for both single treatments. We consider two sets of costs for NUT (
For
The optimal proportions
The optimal total sample size
The RE of the balanced design slightly depends on the response rates. It is also related to the weights. The RE is highest for the first set of weights, since the optimal proportions are nearest to those of the balanced design. Slightly lower relative efficiencies are found for the third set of weights, but these relative efficiencies are still above 0.9. The lowest relative efficiencies are observed for the second set of weights as the optimal proportions deviate most from those of the balanced design. The lowest RE is
Robust optimal design
The optimal designs that were presented in subsections ‘Locally optimal design under a fixed total sample size’ and ‘Locally optimal design under a fixed budget’ are locally optimal since they depend on the response rates
Tables 5 and 6 in the online supplement show maximin optimal designs using the same sets of weights and combinations of costs as in Tables 3 and 4. The ranges used for the response rates are
Locally optimal design: optimal proportions for first-stage
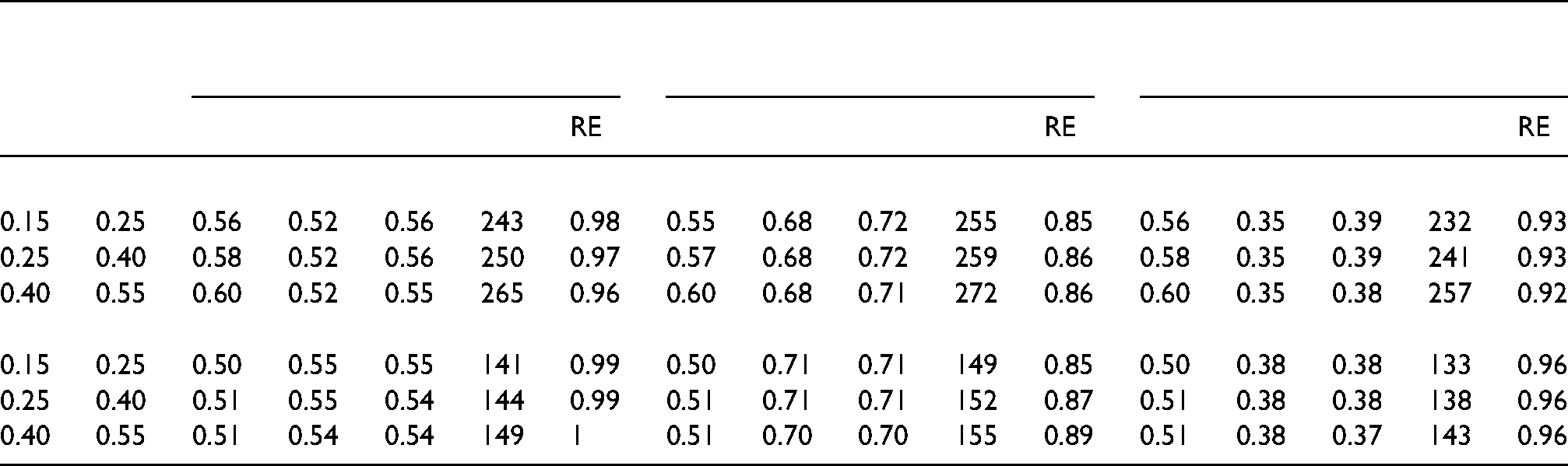
A comparison of Table 3 and Table 5 of Supplemental material, and Table 4 and Table 6 of Supplemental material shows the locally optimal designs and maximin optimal designs are (almost) identical for the chosen sets of weights, response rates and costs. As a result, the minimal RE of the balanced design as given in Tables 5 and 6 of Supplemental material is almost equal to that of the RE of the balanced design in Tables 3 and 4. This result is not surprising since in Sections ‘Locally optimal design under a fixed total sample size’ and ‘Locally optimal design under a fixed budget’, it was shown that the optimal design hardly depends on the response rates. Of course, this finding does not necessarily hold for all combinations of responses rates, weights and costs. The user is therefore encouraged to apply maximin optimal design methodology in the case the response rates are likely to be misspecified.
Discussion
Considering our example of a prototypical SMART design, we derived the optimal design
Under a fixed budget, the optimal proportions are influenced also by the cost of treatments, besides the aforementioned weights and response rates. When including cost of treatments into account, the performance in terms of RE of the optimal design
It is especially advised to use the optimal design rather than the balanced design when the second set of weights,
It should be mentioned that the optimal designs are locally optimal, as they depend on the two unknown response rates
We derived our optimal design under the assumption that outcomes of subjects in ATSs that start with different first-stage treatments are independent of each other, resulting in a zero correlation between weighted mean outcomes of ATSs starting with different first-stage treatments. There are situations in which this assumption may be violated. Consider for instance the situation in our weight loss example where just a limited number of personal trainers is available. It may then occur, a personal trainer trains subjects from ATSs starting with different first-stage treatments. In such a case, the outcomes of subjects who have been trained by the same personal trainer become dependent because of the trainer's skills, enthusiasm, experience, etc. In such a case, the assumption of independence is violated and hence our optimal design is not applicable. Such a problem can be easily solved by letting each personal trainer only train subjects from ATSs that start with the same first-stage treatment.
One limitation of this study is that it does not take clustered data structures into account, while such data may also occur in SMARTs.54,55 Clustered data occur, among others, in cluster-randomized trials and multicentre trials. In such studies not only the total number of subjects in each treatment sequence needs to be determined, but also the number of clusters and cluster size. 56 The optimal design will depend on the intraclass correlation coefficient, which measures the degree of dependence of outcomes within the same cluster.
Another limitation of this study is that formulae and methodology only apply to the prototypical SMART designs in Figures 1 and 2. Based on the number of treatments, stages and randomizations, different SMART designs can be developed, of which many examples exist in the literature57,58 and online. 59 It would be necessary to study optimal designs for such other types of SMART designs.
To our knowledge, this is the first paper that studies optimal allocation to treatments in SMARTs. Our Shiny App allows researchers in the fields of biomedical, health and social sciences to derive the optimal design for their SMART and to calculate the efficiency of a balanced design. We hope that this paper will further contribute to the development and implementation of SMARTs.
Supplemental Material
sj-docx-1-smm-10.1177_09622802211037066 - Supplemental material for Optimal allocation to treatments in a sequential multiple assignment randomized trial
Supplemental material, sj-docx-1-smm-10.1177_09622802211037066 for Optimal allocation to treatments in a sequential multiple assignment randomized trial by Andrea Morciano and Mirjam Moerbeek in Statistical Methods in Medical Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
