Abstract
Background
Lupus nephritis (LN) is a major cause of morbidity and mortality among patients with systemic lupus erythematosus. However, there is a paucity of literature on the clinical characteristics and standard of care of patients in the Arabian Gulf. The LUNELORD study aimed to describe the demographics, clinical characteristics, treatment patterns and health-related quality of life (HRQoL) of patients with LN in the region.
Methods
Eligible adult patients with LN were enrolled from rheumatology centres in the UAE, Qatar, Oman and Kuwait and followed up for 12 months of routine care. Data were abstracted retrospectively from available medical charts up to 12 months prior to study enrolment, and prospectively from patients’ medical charts after each follow-up visit. Patients completed the 36-Item Short Form Health Survey (SF-36) at the baseline and Month 12 visits to assess HRQoL.
Results
Included patients (N = 193) were predominantly female (83.9%) and of Arab ethnicity (85.0%), with a mean (standard deviation [SD]) age of 37.2 (10.4) years. Among patients with available data, renal biopsy was performed in 123 (64.4%) patients, and most had ISN/RPS class V (23.4%), IV (22.7%) or III (20.6%) LN. At baseline, the most frequently used medications were hydroxychloroquine (96.4%), prednisolone (78.8%) and mycophenolate mofetil (69.3%). Renal remission was achieved by 123 (63.7%) patients during the follow-up period; end-stage renal disease occurred in 12/177 (6.8%) patients. A significant association was found between reduced anti-double-stranded DNA levels and occurrence of renal remission at Months 3, 6 and 9. Significant improvements from baseline to Month 12 were observed in the mean (SD) general health SF-36 domain score (+9.5 [42.5]; p = 0.002); overall SF-36 and other domain scores remained constant.
Conclusions
LUNELORD demonstrated a substantial burden of LN disease among patients and highlighted variable adherence to international management and treatment guidelines among rheumatologists in the Arabian Gulf. These findings emphasise the need for early diagnosis, careful monitoring and effective management to optimise renal outcomes and patient HRQoL, which may be achieved by increasing awareness of, and compliance with, international guidelines.

Introduction
Systemic lupus erythematosus (SLE) is a complex, chronic autoimmune condition that can affect multiple organ systems, including the kidneys. 1 Lupus nephritis (LN) is a common and potentially serious renal manifestation of SLE that most frequently occurs within 5 years of disease onset, although some patients develop this complication later.2,3 Globally, 40–75% of patients with SLE develop LN. 2 Prevalence ranges from 18 to 54% in the Arabian Gulf region, 4 with rates of 52% reported in Oman and 41% in the UAE.5,6 LN prevalence has also been reported to vary from 29 to 82% across races/ethnicities. 7 In the Middle East, LN was present in 37–69% of patients within Arab-dominated SLE patient populations, particularly among those from Saudi Arabia, and in 50% of an exclusively Arab population from the UAE. 2
The clinical manifestations of LN range from ‘silent’ presentation and subtle urinary abnormalities to nephrotic syndromes, rapid decline in renal function and chronic renal failure.4,8,9 As many patients do not present with overt signs in the early disease course, it is recommended that patients with SLE undergo screening for LN at the initial SLE diagnosis, and at least yearly thereafter, or more frequently in high-risk patients.10–13 Histological classification of LN by renal biopsy is also recommended to guide treatment approach based on disease severity.11,14
Although early diagnosis and treatment are associated with improved prognosis, treatment effectiveness is not uniform; ∼35% of patients who initially respond to treatment experience relapses. 15 Even with immunosuppressive treatment, 5–20% of patients with LN will develop end-stage renal disease (ESRD) within 10 years of an initial SLE diagnosis. 15
LN is therefore recognised as a major risk factor for morbidity and mortality among patients with SLE; patients with LN have a higher standardised mortality rate compared to patients with SLE but without LN (6–6.8 vs 2.4 across cohort studies). 7 Despite improvements in care, the long-term morbidity experienced by patients often impairs health-related quality of life (HRQoL) and contributes to high healthcare resource utilisation (HCRU).16,17 Factors attributed to poor HRQoL in patients with LN include frequent visits to healthcare facilities, side effects of medication and limited treatment efficacy.15,17
To aid physicians in LN management, several guidelines are available, including those published by the joint European League Against Rheumatism and European Renal Association–European Dialysis and Transplant Association (EULAR/ERA-EDTA), American College of Rheumatology (ACR), Kidney Disease: Improving Global Outcomes (KDIGO) and Asia-Pacific League of Associations for Rheumatology (APLAR).11–13,18 Nevertheless, a recent literature review highlighted that while Arabian Gulf countries generally follow a mixture of these recommendations for treating LN, only 24–56% of patients achieve complete remission and 16–37% achieve partial remission. 4
The diverse manifestations of LN, unpredictability of disease course and variability in treatment effectiveness result in a high unmet need, particularly for patients from the Arabian Gulf, where LN appears to manifest more severely compared with other regions. 4 However, there is limited knowledge of the clinical characteristics and current standard of care of patients in the region. The LUpus NEphritis and Long-term ORgan Damage (LUNELORD; NCT04971590) study aimed to describe the demographics, clinical characteristics, treatment patterns and HRQoL of patients with LN in the Arabian Gulf. The results would help improve understanding of the characteristics and treatment patterns among this population and may inform development of targeted management guidelines to improve patient outcomes in this region. A graphical abstract summarising the study is available (Supplemental Figure S1).
Methods
Study design and objectives
LUNELORD is an observational, multicentre, prospective study (collecting data for a 1-year period following enrolment) with a patient-completed survey and retrospective medical chart review component (collecting data covering the 1-year period prior to enrolment; Figure 1). The primary objective was to describe the demographics, clinical characteristics, disease activity, treatment patterns and HRQoL of patients with LN over a prospective 1-year observation period, across four Gulf countries. LUNELORD study design. LN: lupus nephritis.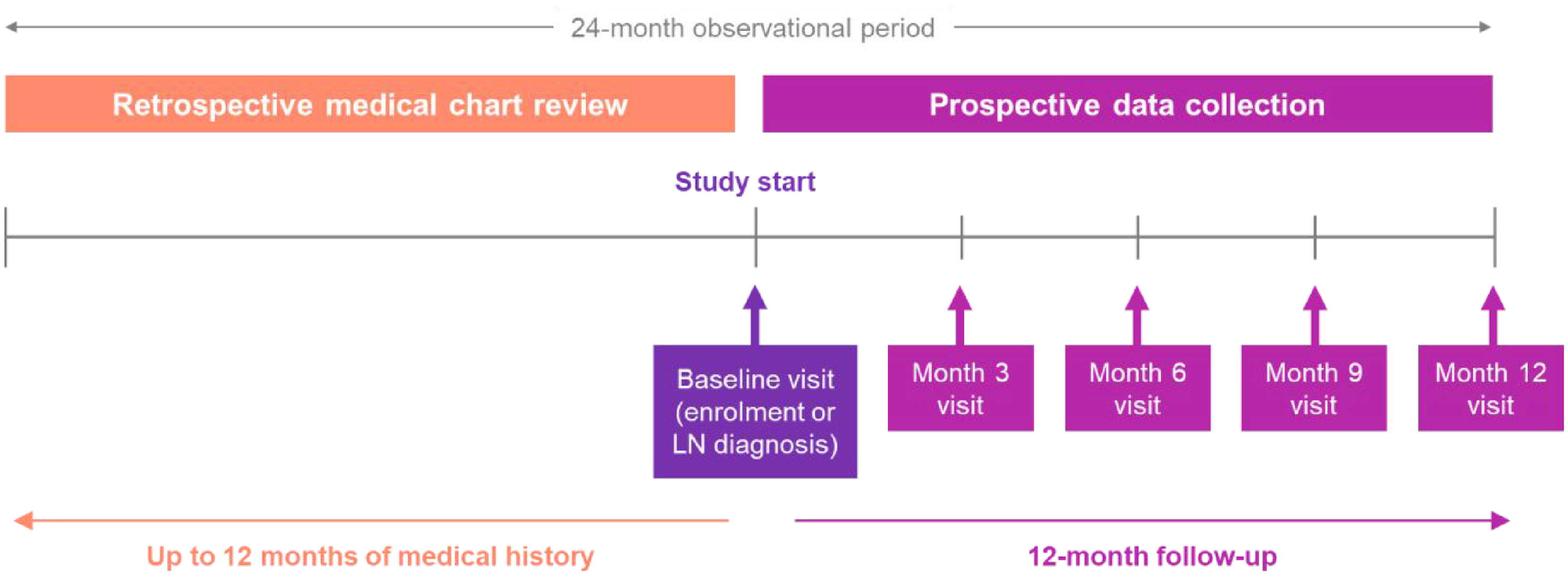
Secondary objectives were to: (1) Describe refractory LN cases and assess changes in serological markers of disease activity over the treatment period. (2) Assess the association between clinical factors, biomarkers, and treatment patterns with achievement of renal remission. (3) Assess the impact of LN on HRQoL to 1 year after diagnosis or enrolment. (4) Explore differences in treatment patterns by physician profile (public vs private sector). (5) Describe HCRU associated with LN management to 1 year following enrolment.
Study population
Patients were enrolled over a 6-month period by rheumatologists in private and public sector centres in Kuwait, Oman, Qatar and the UAE. Staff at participating sites reviewed the medical charts of patients with LN to identify patients who met the study eligibility criteria. Potentially eligible patients were contacted by trained site staff via phone or on-site at the patient’s routine care visit to obtain informed consent in English or Arabic.
Included patients were aged ≥18 years and had clinician-diagnosed LN prior to/at the enrolment visit, with ≥1 documented visit to the healthcare centre within the 12 months preceding enrolment. The study excluded patients with incomplete medical records that precluded assessment of disease severity, or medical records lacking any of the following renal laboratory results within the last 12 months: (1) urine protein-creatinine ratio or 24-hour proteinuria or urine sediment; or (2) serum creatinine or estimated glomerular filtration rate (eGFR; or measured GFR if eGFR was not available). Patients with current or medical history of congenital or acquired immunodeficiency, malignancy in active treatment phase and acute viral infection requiring hospitalisation were also excluded.
Data sources and collection
The prospective phase of LUNELORD consisted of 5 patient visits over 12 months as per routine care, including the enrolment (baseline) visit and subsequent follow-up visits at Months 3, 6, 9 and 12. Data were abstracted from patients’ medical charts after each visit and entered directly into the study’s electronic case report forms (eCRFs). Post-baseline data collection visits were intended to occur near the Month 3, 6, 9 and 12 timepoints, but exact timings varied due to the real-world nature of this study; thus, the closest available visit to these timepoints were selected. If a patient missed a visit, data analyses were performed based on available data and it was assumed that clinical status did not change from the previous visit. This approach minimised the influence of study procedures on routine patient assessments and associated HCRU.
To assess HRQoL, patients completed 36-Item Short Form Health Survey (SF-36) forms in Arabic or English at the baseline and final Month 12 visits. Site staff entered completed questionnaire data into the eCRFs.
For the retrospective phase, site staff performed one-time data abstraction from the medical charts of enrolled patients. Available data in the charts, collected as part of usual care from diagnosis or up to 12 months prior to enrolment, were abstracted and recorded directly in eCRFs. Prospectively and retrospectively collected data variables are summarised in Supplemental Table 1.
Sample size and statistical analysis
A sample size of 200 patients was selected to provide significant power to interpret study results. The margin of error (0.067 significance level) indicates that 200 patients with LN are required to achieve a 93% confidence level that the data will accurately reflect the entire LN population in the Gulf countries. The sample size (N = 200), of which 35% are in renal remission and 65% are not, would provide 80% power at a 0.05 significance level to detect a change in the probability of achieving renal remission from the baseline value of 0.200 to 0.385 to assess secondary objective 2. This corresponds to an odds ratio of 2.508. Sample size calculations were performed using PASS® version 2019.
The primary data analysis was performed in all patients who met the eligibility criteria (‘all enrolled patients [AEP]’ population). For each variable, summary statistics at each data collection timepoint were calculated using the number of patients without missing data and reported descriptively; missing data were not imputed. Statistical analyses for the secondary outcomes are described in the Supplemental Methods. No adjustments were made either for multiple comparisons when testing associations, or for potential confounders of the odds ratios for anti-dsDNA. All statistical analyses were performed using GNU PSPP version 2.0.1.
Ethical approval and informed consent
This study was performed according to the legal requirements of each participating country and principles outlined in the Good Pharmacoepidemiological Practice guidelines and Declaration of Helsinki. The study protocol and any amendments were approved by the relevant Ethics Committee/Institutional Review Board/Regulatory Authorities for each site prior to study commencement (Supplemental Methods). All patients, or their legally acceptable representative, provided written informed consent before participating in the study. This study complied with all applicable laws regarding participant privacy.
Results
Patient disposition and characteristics
The medical records of 200 patients were reviewed between 8 September 2021 and 1 August 2023. LUNELORD enrolled 193 patients attending rheumatology centres across the UAE (n = 137; 71.0%), Qatar (n = 30, 15.5%), Oman (n = 15, 7.8%) and Kuwait (n = 11, 5.7%).
Included patients were predominantly female (83.9%) and of Arab ethnicity (85.0%), with a mean (standard deviation [SD]) age of 37.2 (10.4) years at baseline (Supplemental Table S2). Approximately two-thirds of patients (69.4%) had comorbidities diagnosed since their first SLE/LN diagnosis; endocrine/metabolic disorders were diagnosed in one-third of patients (32.1%).
Clinical characteristics of SLE
Clinical characteristics of SLE.
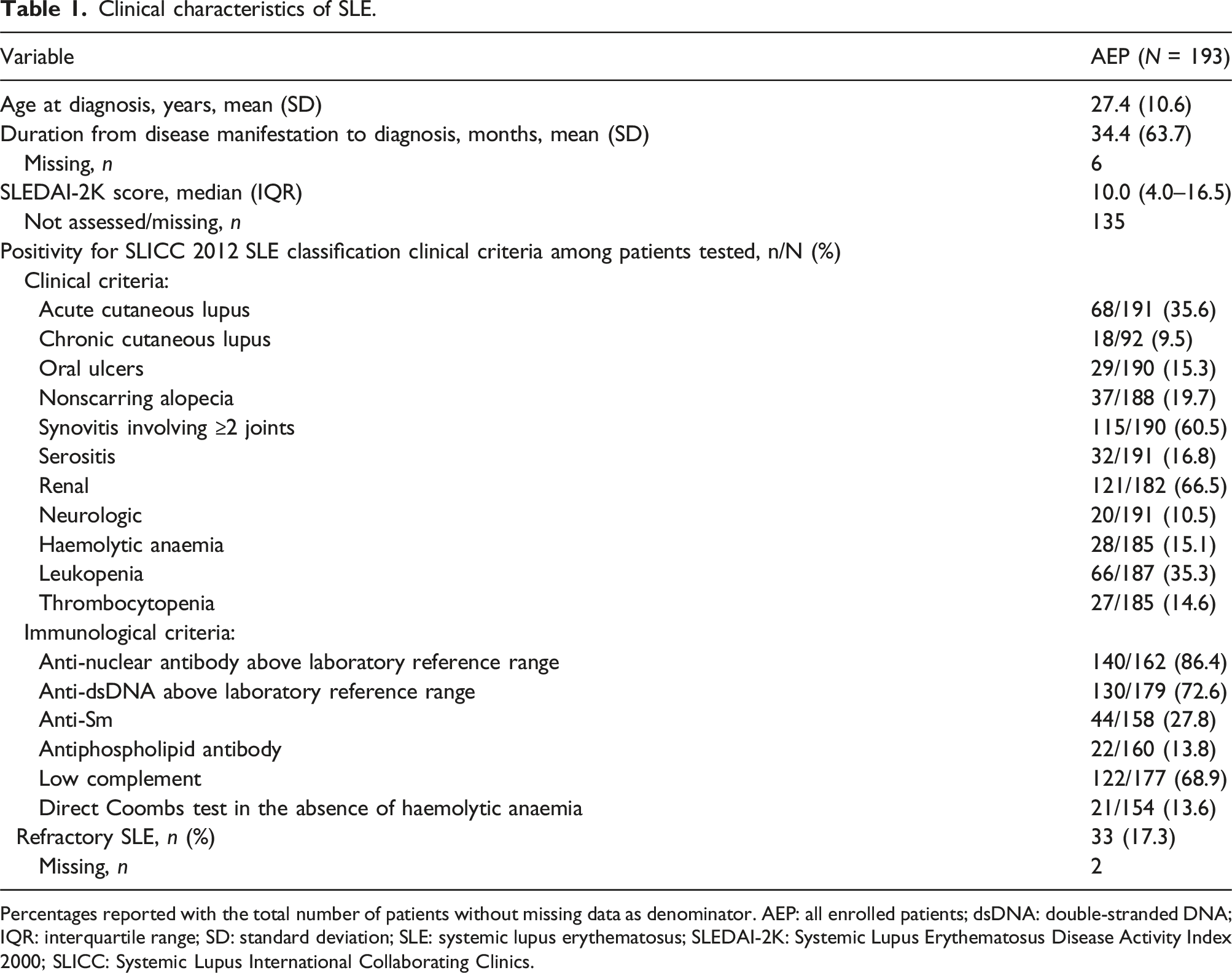
Percentages reported with the total number of patients without missing data as denominator. AEP: all enrolled patients; dsDNA: double-stranded DNA; IQR: interquartile range; SD: standard deviation; SLE: systemic lupus erythematosus; SLEDAI-2K: Systemic Lupus Erythematosus Disease Activity Index 2000; SLICC: Systemic Lupus International Collaborating Clinics.
Among the SLICC 2022 clinical criteria for SLE classification, most patients tested positive for the renal criterion (121/182 [66.5%]) and synovitis involving two or more joints (115/190 [60.5%]) prior to LN diagnosis and study inclusion. Among the immunological criteria, the most frequently occurring were antinuclear antibody above the laboratory reference range (140/162 [86.4%]), anti-double-stranded DNA (dsDNA) above the laboratory reference range (130/179 [72.6%]) and low complement levels (122/177 [68.9%]).
Clinical characteristics of LN
Clinical characteristics of LN.

Percentages reported with the total number of patients without missing data as denominator. ACR: American College of Rheumatology; AEP: all enrolled patients; dsDNA: double-stranded DNA; eGFR: estimated glomerular filtration rate; IQR: interquartile range; ISN/RPS: International Society of Nephrology/Renal Pathology Society; LN: lupus nephritis; NIH: National Institute of Health; SD: standard deviation; SLICC: Systemic Lupus International Collaborating Clinics.
Patients presented with diverse active clinical LN manifestations. Renal manifestations were prevalent in the cohort, including proteinuria (71.5%), haematuria (52.8%), hypertension (33.2%) and renal failure (11.4%; based on local site definitions). Extrarenal manifestations, such as joint pain (48.7%), skin rashes (8.9–16.6%) and haematological abnormalities (12.5–42.2%), also occurred frequently. Among the 42 (26.4%) patients with organ damage assessed per the SLICC/ACR Damage Index, mean (SD) organ damage score was 1.9 (2.2).
In total, 123 (63.7%) patients achieved renal remission during the 1-year follow-up period. ESRD occurred in 12/177 (6.8%) patients, with a median (IQR) duration from LN diagnosis to ESRD of 3.7 (1.8–11.3) years.
Changes in serological biomarker levels over 12 months in the AEP population (N = 193).
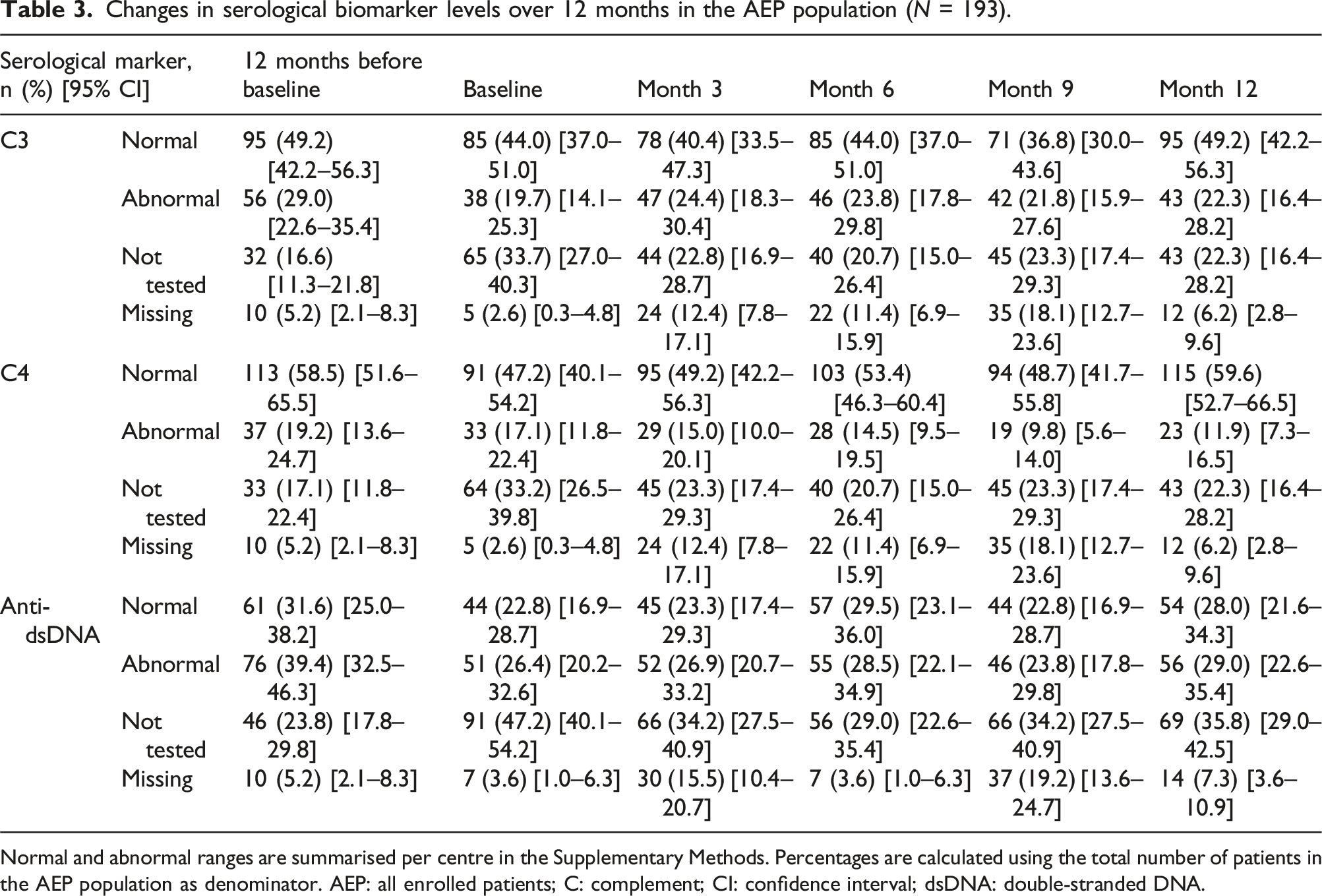
Normal and abnormal ranges are summarised per centre in the Supplementary Methods. Percentages are calculated using the total number of patients in the AEP population as denominator. AEP: all enrolled patients; C: complement; CI: confidence interval; dsDNA: double-stranded DNA.
Association between clinical factors, treatment, and biomarkers, and renal remission in the AEP (N = 193).
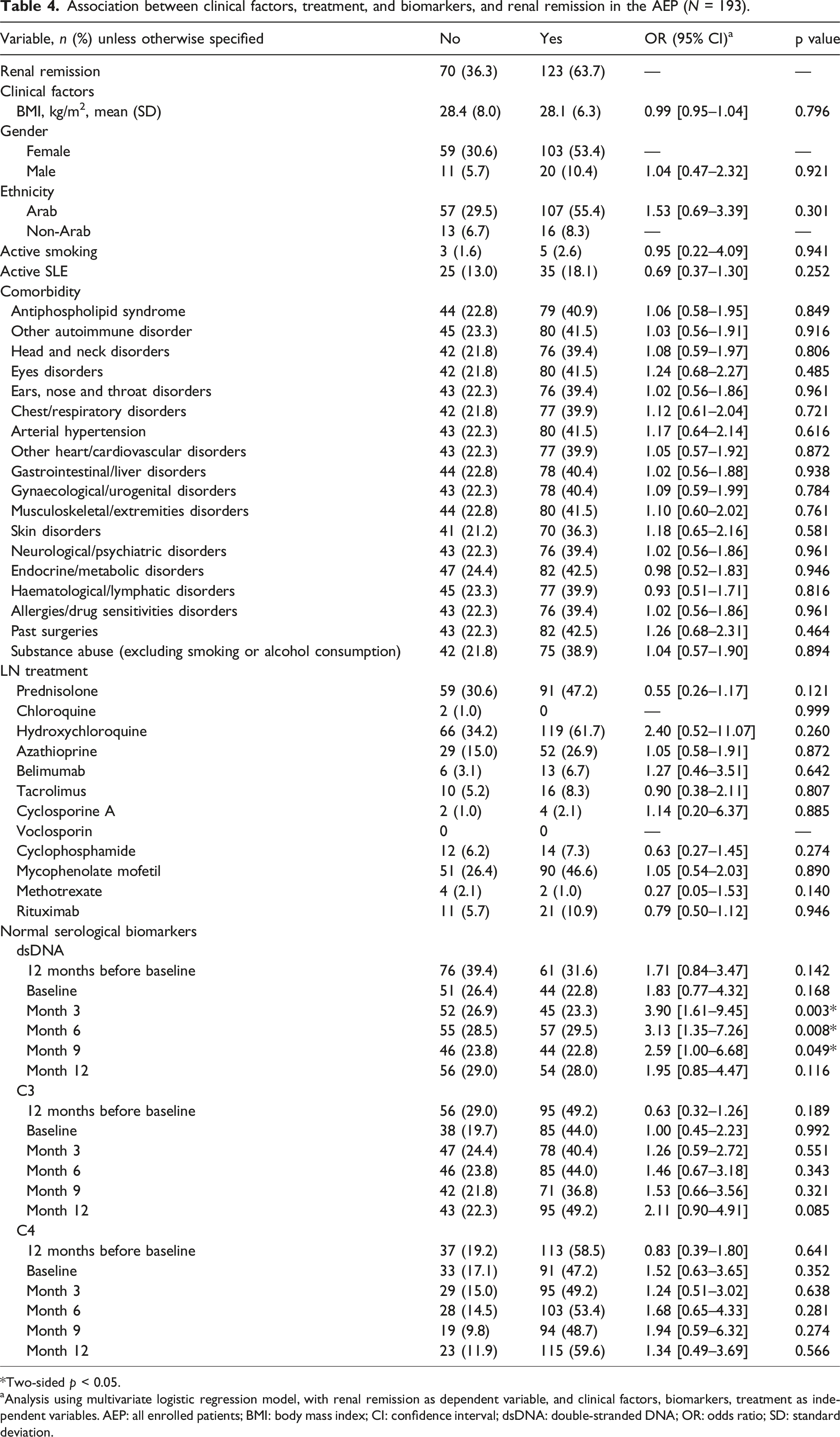
*Two-sided p < 0.05.
aAnalysis using multivariate logistic regression model, with renal remission as dependent variable, and clinical factors, biomarkers, treatment as independent variables. AEP: all enrolled patients; BMI: body mass index; CI: confidence interval; dsDNA: double-stranded DNA; OR: odds ratio; SD: standard deviation.
Changes in serological markers over 12 months in patients with refractory disease (n = 33) are reported in Supplemental Table S3; statistical comparisons were not performed due to the small sample size and lack of serological data. Due to the low number of patients with refractory LN, haematology and biological laboratory values were not analysed.
Treatment history and patterns
At baseline, patients most frequently used hydroxychloroquine (96.4%), prednisolone (78.8%) and mycophenolate mofetil (69.3%). Median (IQR) number of prior treatment failures, defined as LN relapse following treatment, was 1.0 (0–2.0) in 151 patients. LN treatment history data are summarised in Supplemental Table S4.
More patients were treated by physicians in the public sector (n = 126) than the private sector (n = 67). The most commonly prescribed medications for patients with LN by public versus private physicians were hydroxychloroquine (93.7% vs 100%), prednisolone (81.0% vs 71.6%) and mycophenolate mofetil (68.3% vs 82.1%; Supplemental Table S5).
HRQoL
SF-36 scores at baseline and month 12 in the AEP population (N = 193).
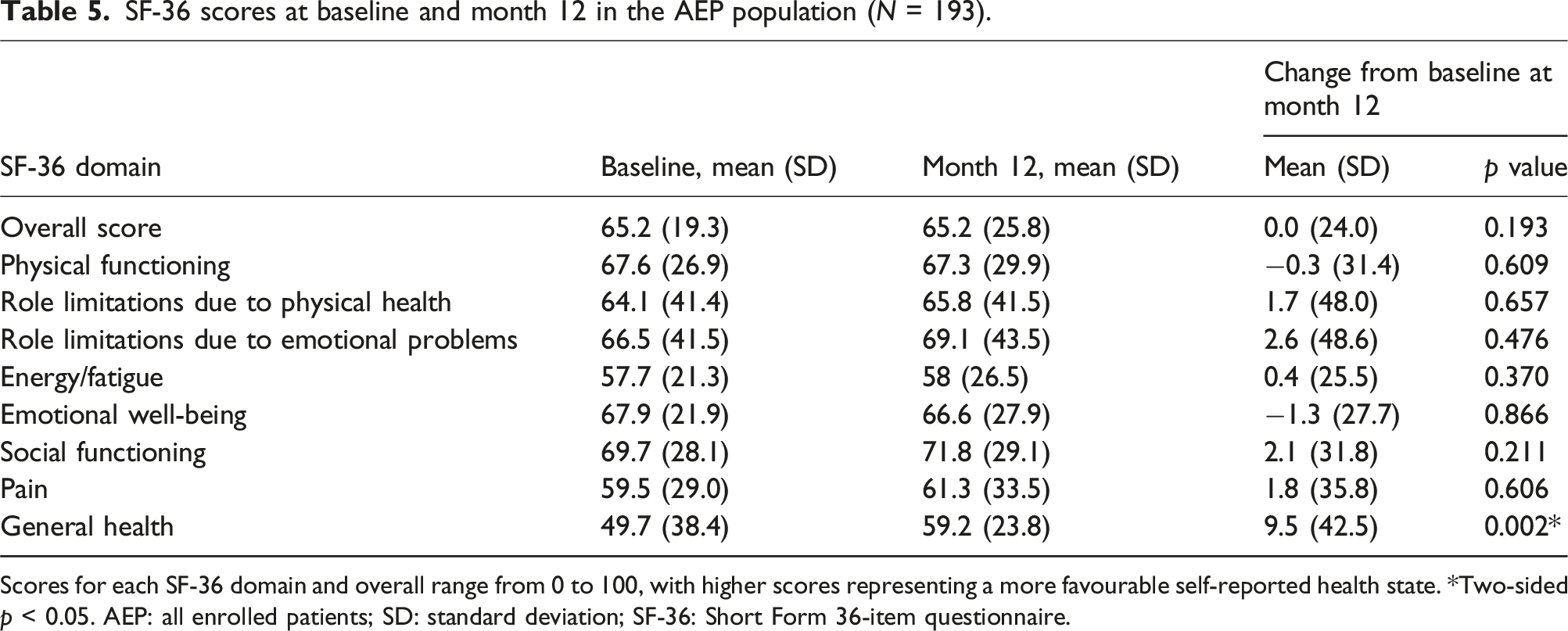
Scores for each SF-36 domain and overall range from 0 to 100, with higher scores representing a more favourable self-reported health state. *Two-sided p < 0.05. AEP: all enrolled patients; SD: standard deviation; SF-36: Short Form 36-item questionnaire.
At Month 12, mean (SD) overall SF-36 score was 65.2 (25.8), with no significant changes from baseline. However, mean general health domain score increased significantly from 49.7 (38.4) at baseline to 59.2 (23.8) at 12 months (p = 0.002). Mean scores of the other SF-36 domains did not exhibit significant changes after 1 year (p > 0.05).
HCRU associated with LN management
Estimated HCRU for patients over 12 months is summarised in Supplemental Table S6. Mean (SD) number of office and outpatient visits was 7.05 (9.31) and 12.90 (14.58) visits per patient per year, respectively, while the number of hospitalisations and intensive care unit hospitalisations was 0.90 (1.72) and 0.06 (0.29), respectively. There was one death, following cardiac arrest, in the UAE.
Discussion
LUNELORD described the demographics, clinical manifestations, treatment patterns and HRQoL of patients with LN over 1 year across rheumatology centres in Kuwait, Oman, Qatar and the UAE, improving understanding of LN in the Arabian Gulf.
The cohort was comparable with other LN patient populations. Patients were predominantly female (female-to-male ratio: 5.2:1), consistent with studies conducted in individual Gulf countries and worldwide which reported female-to-male ratios of 3–8.3:1.19–23 Mean age at SLE diagnosis in the LUNELORD cohort (27.4 years) was younger than that reported in Western cohorts,24,25 whilst mean age at LN diagnosis in LUNELORD (30.5 years) is at the upper end of previously reported ranges (23.4–32 years),19–23 suggesting earlier SLE disease onset but delayed recognition of renal involvement. This may be because non-renal SLE manifestations are more clinically prominent and prioritised in Arab populations, or due to lower screening rates and delayed recognition of renal involvement in Arab populations. Mean duration from LN manifestation to diagnosis in LUNELORD (27.7 months) was consistent with other LN cohorts (25.5–31.2 months).20,26
LN may manifest more severely in patients from the Arabian Gulf compared to other regions. 4 SLEDAI-2K in the LUNELORD cohort was indicative of high disease activity (median [IQR] score: 10.0 [4.0–16.5]), higher than previously reported in Korean and Spanish patients.27–29 Additionally, the most common LN pathological type in LUNELORD was ISN/RPS Class V (23.4%), whereas in previous cohorts from Lebanon, Jordan, Oman, Iran and Hong Kong, Class IV was most frequently reported (44.1–49.1%).5,20,22,30,31 Despite the high burden of advanced disease, patient-reported outcomes revealed preserved psychosocial wellbeing, with social functioning and emotional health scores reflecting resilience and strong social support networks.
A variety of renal and extrarenal manifestations were reported in the LUNELORD cohort, with a substantial proportion presenting with severe renal involvement. 71.5% and 52.8% of patients demonstrated significant proteinuria and haematuria, respectively, underscoring the renal disease burden in this population. Stratified analysis of proteinuria levels may further refine risk prediction and therapeutic response. Furthermore, 11.4% of patients presented with renal failure and 6.8% of patients had ESRD, with progression to ESRD occurring at a median of 3.7 years post-LN diagnosis. This rapid progression highlights the aggressive course of LN in this population, consistent with the higher prevalence of proliferative pathology. Therefore, findings from LUNELORD emphasise the need for early diagnosis, close monitoring and intensive treatment to manage systemic symptoms and prevent further renal damage and ESRD progression among patients with LN.
Variable renal remission rates have been reported across previous studies,32–34 which may depend on factors such as criteria used, histological class at presentation, time to LN recognition, treatment type and time to treatment. In LUNELORD, renal remission was achieved in 63.7% of patients. Normal anti-dsDNA test results at 3, 6 and 9 months after LN diagnosis were significantly associated with the occurrence of renal remission, with lower detectable anti-dsDNA levels during renal remission than active disease. Normal anti-dsDNA levels at these timepoints may therefore be a positive indicator of renal improvement and a predictor of renal relapse in patients with LN. Other clinical factors or treatments were not found to be correlated with renal remission, potentially owing to the relatively small sample size and low statistical power, although an association between absence of anti-C1q autoantibodies and protection against LN has previously been reported. 7
As clinical presentations often do not reflect the extent of renal involvement, EULAR, KDIGO and APLAR guidelines recommend a diagnostic renal biopsy be considered for patients showing any sign of renal involvement at initial presentation to inform treatment approach, and repeat renal biopsies for patients with persistently active serological markers to guide further treatment.11,14,18 Despite this, only 64.4% of patients in LUNELORD underwent renal biopsy in the year before enrolment, which is lower than reported in international cohorts. 35 This highlights both practice variability and potential limitations in access to/acceptance of invasive diagnostics, and suggests variable compliance with international LN diagnosis and management guidelines in the Arabian Gulf, as per previous findings in the region. 4 These results underscore the need to improve awareness of international guidelines and emphasise the importance of adherence among rheumatologists in the Arabian Gulf. Targeted training on local and international guidelines could be considered, as well as development of local policies and multidisciplinary protocols. For example, recently developed consensus-based recommendations for SLE management in the region emphasise the role of repeat renal biopsy, including in pregnant women.36,37
Studies in international cohorts have demonstrated remarkably diminished HRQoL among patients with SLE and LN, especially among those with severe disease.38–40 To the authors’ knowledge, this is the first study assessing the impact of LN on HRQoL in the Arabian Gulf. Baseline SF-36 scores ranged between 49.7 and 69.7, which is slightly higher than the range reported for patients with LN (40–60) but lower than that for healthy controls/general population (70–80). 16 Although overall SF-36 score did not improve after 1 year in LUNELORD, a 9.5-point gain was observed in the general health domain. This suggests that patients perceived their health to be better overall, which may reflect improved disease control or psychological adaptation to the challenges of living with LN.
Prescribing practices were consistent across the LUNELORD cohort, which may partially be because most patients (65.3%) were treated in government facilities. There was remarkably high hydroxychloroquine usage at enrolment (96.4%), reflecting strong adherence to international guidelines.11,12 This is a therapeutic strength of the cohort, as antimalarials such as hydroxychloroquine increase renal remission and survival rates, reduce frequency of flares and risk of ESRD progression and are associated with improved HRQoL.39,41,42 However, prednisolone use reported at enrolment remained high (78.8%), raising concerns regarding long-term toxicity, and few patients received biologic treatments such as rituximab (14.1%) and belimumab (9.9%) despite international guidelines recommending the use of biologics in combination with standard therapy for refractory, active or severe LN.11–13 The incorporation of biologic therapies into standard treatment regimens has been demonstrated to reduce disease activity, fatigue and overall symptom burden, and may help achieve sustained remission and greater improvements in HRQoL.43–45 It should be noted that the study period occurred prior to the latest guidelines emphasising early use of biologics in LN.11–13,36
Although several studies have shown that renal involvement and flares increase the costs associated with SLE management, there is a paucity of literature on HCRU and costs among the subgroup of patients with LN.17,46,47 A previous systematic literature review reported annual outpatient visits and inpatient hospitalisations ranging from 6.6 to 7.4 and from 0.6 to 1.0 per patient with LN, respectively, in the US. 17 In LUNELORD, mean number of outpatient visits and hospitalisations was estimated to be 12.9 and 0.9 per patient, respectively, through a year of follow-up. This may reflect greater disease severity and higher burden of LN in the Arabian Gulf, or differences in healthcare delivery models between geographies. Less frequent scheduled follow-up visits in the Arabian Gulf may lead patients to seek the opinion of other physicians, accessing specialists in private non-governmental facilities. Future studies should quantify the economic burden associated with LN management in the Arabian Gulf region and ascertain how disease severity impacts this burden.
Strengths and limitations
A major strength is that LUNELORD is the first study of its kind to comprehensively assess demographics, disease characteristics, treatment patterns and HRQoL of patients with LN in the Arabian Gulf, addressing a large knowledge gap in the region. The pool of 193 patients represents one of the largest dedicated LN cohorts in the Gulf region, enhancing the study’s external validity for Arab populations. Owing to the prospective, observational cohort design with a retrospective chart review component, the study was able to collect comprehensive data, including clinical, laboratory, renal biopsy, treatment and HRQoL data. This approach enabled the assessment of renal remission, progression to ESRD, trends in serological markers and changes in HRQoL over time. Furthermore, inclusion of both public and private sector sites across multiple Gulf countries ensures that the findings have broad real-world applicability.
Nevertheless, this study was subject to some limitations. By its nature, the study sample was geographically restricted, limiting extrapolation. Results for several variables were missing, likely due to the retrospective study component. For instance, some laboratory tests were not performed, or data in medical records were incomplete. In addition, SF-36 results may not reflect the sole impact of LN on HRQoL since most patients presented with other comorbidities. However, these results may be representative of the wider LN population as comorbidities are common in these patients.
Although LN diagnosis and management are performed by both rheumatologists and nephrologists, only rheumatology centres were included. Patients with more advanced renal dysfunction or isolated renal flares, or patients requiring dialysis, are often managed by nephrology centres; excluding nephrology-led cases may have biased the sample towards less severe patients and resulted in an underestimation of clinical outcomes. Thus, the LUNELORD cohort may not be fully representative of patients with LN and their management in the region.
This study used a broad definition for renal remission, which included treating physicians’ discretion as a criterion. This definition was selected to enable remission status to be determined for all patients, including those with missing laboratory data. Nevertheless, the lack of a standardised definition for renal remission may have introduced heterogeneity and information bias, thereby limiting internal validity of these results. In addition, the sample size is relatively small, especially considering the inclusion of four countries, resulting in limited statistical power to assess the secondary outcomes. Although a large proportion of patients achieved renal remission, the lack of association found between clinical factors, biomarkers and treatments, and renal remission suggests that further studies with a larger sample size are required to better understand the factors influencing remission in patients with LN.
Conclusions
LUNELORD addressed important knowledge gaps and provided a greater understanding of the demographics, clinical manifestations, outcomes and treatment patterns of patients with LN in four Arabian Gulf countries. A substantial disease burden was found in terms of disease activity and organ involvement. The results also suggest that reduced anti-dsDNA levels may be a positive indicator of renal improvement. Overall HRQoL did not improve, although the gains seen in general health suggest that patients’ perception of their health may have improved due to adaptation to living with chronic illness. Altogether, these findings underscore the need for early diagnosis, close monitoring and adherence to international guidelines to reduce ESRD progression and improve HRQoL in patients with LN in the Arabian Gulf.
Supplemental material
Supplemental Material - LUNELORD: A descriptive, prospective study on the demographics, disease characteristics and health-related quality of life of patients with LUpus NEphritis and long-term ORgan damage in rheumatology clinics in the Arabian Gulf
Supplemental Material for LUNELORD: A descriptive, prospective study on the demographics, disease characteristics and health-related quality of life of patients with LUpus NEphritis and long-term ORgan damage in rheumatology clinics in the Arabian Gulf by Jamal Al-Saleh1, Faisal Elbadawi, Rajaie Namas, Mohamed Elarabi, Samar Al-Emadi, Miral H. Gharib, Khalid A. Alnaqbi, Hiba Khogali, Humaid A. Al Wahshi, Sadiq Abdul Baqi, Amjad Alkadi, Suad Hannawi, Samah Allam, Reem Al-Jayyousi, Mohamed Hamouda, Averyan Vasylyev, Evelina Zimovetz, Munther Khamashta in Lupus
Supplemental material
Supplemental Material - LUNELORD: A descriptive, prospective study on the demographics, disease characteristics and health-related quality of life of patients with LUpus NEphritis and long-term ORgan damage in rheumatology clinics in the Arabian Gulf
Supplemental Material for LUNELORD: A descriptive, prospective study on the demographics, disease characteristics and health-related quality of life of patients with LUpus NEphritis and long-term ORgan damage in rheumatology clinics in the Arabian Gulf by Jamal Al-Saleh1, Faisal Elbadawi, Rajaie Namas, Mohamed Elarabi, Samar Al-Emadi, Miral H. Gharib, Khalid A. Alnaqbi, Hiba Khogali, Humaid A. Al Wahshi, Sadiq Abdul Baqi, Amjad Alkadi, Suad Hannawi, Samah Allam, Reem Al-Jayyousi, Mohamed Hamouda, Averyan Vasylyev, Evelina Zimovetz, Munther Khamashta in Lupus
Footnotes
Acknowledgments
The authors acknowledge Phoenix CR for substantial contributions to study conduct and design. The authors also thank Costello Medical for editorial assistance and publication coordination, on behalf of GSK, and acknowledge Ellie Fung, Costello Medical, UK for medical writing and editorial assistance based on authors’ input and direction.
Author contributions
Substantial contributions to study conception and design:
Declaration of conflicting interests
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by GlaxoSmithKline Biologicals SA (oneCDP identifier: 216989). Support for third-party writing assistance, editorial assistance and publication coordination for this article, provided by Ellie Fung, Costello Medical, UK was funded by GSK in accordance with Good Publication Practice (GPP 2022) guidelines (![]() ).
).
Ethical considerations
This study was performed according to the legal requirements of each participating country and principles outlined in the Good Pharmacoepidemiological Practice guidelines and Declaration of Helsinki. The study protocol and any amendments were approved by the relevant Ethics Committee/Institutional Review Board/Regulatory Authorities for each site prior to study commencement (![]() ).
).
Consent to participate
All patients, or their legally acceptable representative, provided written informed consent before participating in the study. This study complied with all applicable laws regarding participant privacy.
ORCID iDs
Data Availability Statement
Supplemental material
Supplemental material for this article is available online.
