Abstract
Objective
Belimumab, a B-cell modulator, targets the central immunopathogenic pathway in systemic lupus erythematosus (SLE) by selectively inhibiting B lymphocyte stimulator (BLyS) and reducing the autoreactive B cells that drive disease activity. Its effectiveness in reducing disease activity and its steroid-sparing potential have been well-documented in both clinical trials and real-world studies. This study evaluated belimumab’s long-term effectiveness in reducing oral glucocorticoid (OGC) use based on updated European Alliance of Associations for Rheumatology (EULAR) recommendations, and in attaining low disease activity and remission in adults with SLE.
Methods
This post hoc descriptive analysis (GSK Study 219649) utilized pooled data from individual OBSErve studies conducted in eight countries, collected at belimumab initiation and 6 months (all countries), and 6 to 24 months (USA and Argentina) post-initiation. Endpoints included percentages of patients achieving ≤5 mg/day OGC; maintaining 0 and ≤5 mg/day OGC beyond 6 months; attaining low disease activity (modified Lupus Low Disease Activity State [mLLDAS]: SLE Disease Activity Index [SLEDAI] score ≤4, OGC dose ≤7.5 mg/day) and remission (modified Definition Of Remission In SLE [mDORIS]: SLEDAI score = 0, OGC dose ≤5 mg/day) over time. The sample size in this study was fixed by available data from the OBSErve studies.
Results
Data from 959 patients were included (mean [SD] age: 41.5 [12.4] years; 89.5% female; 52.2% from the USA). Of patients prescribed OGC at index, percentages receiving ≤5 mg/day increased from 16.1% at belimumab initiation to 51.6% at 6 months and 87.5% at 24 months; 8.2% discontinued OGC at 6 months and 44.4% at 24 months post-initiation. Of patients achieving 0 and ≤5 mg/day OGC at 6 months, 87.9% and 92.9% maintained this dose for 24 months. Percentage attaining mLLDAS/mDORIS increased from 0.4%/0.2% at belimumab initiation to 11.1%/7.4% at 6 months and 18.2%/12.9% at 24 months post-initiation.
Conclusions
These results from a real-world clinical setting suggest that belimumab treatment supports patients in achieving EULAR-recommended OGC taper goals, with sustained OGC dose reductions observed alongside an increased percentage of patients attaining low disease activity and remission as early as 6 months following belimumab initiation.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease associated with decreased quality of life, organ damage, and increased mortality.1–5 While oral and intravenous (IV) glucocorticoids (GC), antimalarials, and immunosuppressants are the mainstay treatments in SLE, 6 prolonged and cumulative GC use is associated with an increased risk of infections and irreversible organ damage accrual.7,8 Therefore, SLE management necessitates balancing therapeutic efficacy and reducing reliance on GC, warranting individualized treatment strategies. The European Alliance of Associations for Rheumatology (EULAR) 2023 recommendations for the management of SLE advise using GCs as a bridging therapy during periods of high disease activity and, where used as a maintenance therapy, the aim should be to reduce to ≤5 mg/day or withdraw entirely. 9 Furthermore, the EULAR 2023 recommendations suggest achieving a state of low disease activity (measured by the Lupus Low Disease Activity State [LLDAS] criteria) or remission (measured by the Definition Of Remission In SLE [DORIS] criteria) as key SLE treatment goals. 9 The LLDAS criteria are defined as SLE Disease Activity Index (SLEDAI)-2K score ≤4, with no activity in major organ systems (renal, central nervous system, cardiopulmonary, vasculitis, fever) and no hemolytic anemia or gastrointestinal activity; no new lupus disease activity compared with the previous assessment; Physical Global Assessment (PGA) score ≤1 (on a 0–3 scale); current prednisone (or equivalent) GC dose ≤7.5 mg/day; and standard maintenance doses of immunosuppressive drugs and approved biological agents. 10 The DORIS criteria are defined as clinical SLEDAI score = 0, irrespective of serology; PGA score <0.5; prednisone-equivalent GC dose ≤5 mg/day and/or stable immunosuppressives including biologics. 11 The use of disease-modifying therapies that target underlying SLE pathogenesis could therefore aid in achieving these targets. 12
Belimumab, a B-cell modulator, targets the central immunopathogenic pathway in SLE by selectively inhibiting soluble B lymphocyte stimulator (BLyS) and reducing the autoreactive B cells that drive disease activity.13,14 By modulating autoreactive B cells, belimumab can reduce ongoing disease activity while preserving the immune system’s key functions, and it has been approved for treating SLE in more than 75 countries.
6
Previous clinical trials15,16 and real-world studies,17–19 including those conducted through a global Evaluation
Previous OBSErve studies (conducted in the USA, Canada, Argentina, Spain, Germany, and Switzerland) reported improved clinical outcomes and OGC use following 6–24 months of belimumab treatment. 26 In this descriptive post hoc analysis, we incorporated more recent real-world data from Saudi Arabia and Russia, and aimed to re-evaluate belimumab’s effectiveness in reducing OGC dose and maintaining lower OGC dose thresholds (≤5 mg/day of prednisone equivalent), in line with updated 2023 EULAR recommendations. We also evaluated the status of the key EULAR 2023-recommended treatment goals, low disease activity and remission, in this diverse group of patients with SLE for up to 24 months following belimumab initiation.
Methods
Study design
This was a pooled, descriptive, post hoc analysis (GSK Study 219649) of patient-level data from eight observational cohort OBSErve studies conducted in the USA (GSK Study 117295),
20
Argentina (GSK Study 201282),
25
Canada (GSK Study 117300),
23
Germany (GSK Study 117214),
22
Spain (GSK Study 200883),
21
Switzerland (GSK Study 201232),
24
Saudi Arabia,
27
and Russia
28
(Saudi Arabia and Russia: GSK Study 215349) (Figure 1). Overview of global OBSErve program studies. aFor patients in the USA and Argentina only, who were followed up for 24 months; bmLLDAS was defined as SLEDAI score ≤4 with no worsening of clinical manifestations (i.e., renal, central nervous system, cardiopulmonary, fever, hemolytic anemia, vasculitis, or gastrointestinal activity, determined by clinical judgment of the investigators) since belimumab initiation and OGC dose ≤7.5 mg/day;
29
cmDORIS was defined as total SLEDAI score = 0 and OGC dose ≤5 mg/day.
11
IV, intravenous; mDORIS, modified Definition Of Remission In SLE; mLLDAS, modified Lupus Low Disease Activity State; OGC, oral glucocorticoid; SLE, systemic lupus erythematosus; SLEDAI, SLE Disease Activity Index.
Data were collected between 2011 and 2017 (Argentina, Canada, Germany, Spain, Switzerland and USA), and between 2021 and 2022 (Russia and Saudi Arabia). Follow-up periods varied between the original OBSErve studies; a 6-month follow-up was available for Canada, Germany, Spain, Switzerland, Saudi Arabia, and Russia, and 6–24 months (in 6-monthly intervals) for the USA and Argentina. Steroid-tapering protocol was not mandated in the included studies, and decisions regarding GC dosing were made at physicians’ discretion according to routine clinical practices within each country. The current analysis utilized demographic and clinical characteristics data (such as OGC usage and disease activity) collected at belimumab initiation and during 6-monthly follow-ups. Disease severity at diagnosis and at belimumab initiation was assessed based on the physician’s clinical judgment. All patients from the original studies with available demographic and clinical characteristics data at each follow-up were included in the analyses, including those lost to follow-up after each interval. There were some exceptions: patients who discontinued within 6 months of initiating belimumab treatment were not included in the 6-month follow-up (apart from patients from Argentina, Germany, and Switzerland who were included in the 6-month follow-up); patients in the USA and Argentina who discontinued belimumab after 6 or 12 months from initiating were not included in subsequent follow-ups. This post hoc analysis was conducted for: (1) the overall population, encompassing all the assessed countries; (2) the USA and Argentina population combined; (3) the USA population alone; and (4) patient subpopulation with baseline anti-dsDNA and/or complement alterations (immunologic activity).
Physicians and patients
Full physician and patient eligibility criteria for the OBSErve studies have been published.20–25 Briefly, rheumatologists/internal medicine specialists who prescribed belimumab to patients with SLE as part of their usual clinical practice and had experience managing and treating ≥10 patients with SLE, treating SLE for ≥5 years, along with treating ≥2 patients with belimumab as part of standard care, were invited to participate.
Eligible patients were ≥18 years of age with a diagnosis of SLE, who were prescribed belimumab as an add-on to their standard SLE treatment, initiated belimumab 6 months (Germany, Spain, Switzerland, Saudi Arabia, Russia, and n = 217/501 of patients in the USA) or 12 months (Argentina and n = 284/501 of patients in the USA) before enrollment (period of identification for inclusion in the study), or had received ≥8 belimumab infusions equivalent to 6 months of IV belimumab treatment before enrollment (USA and Canada only), and had a documented reason for belimumab initiation.
Endpoints
The primary endpoint for this descriptive post hoc analysis was the percentage of patients achieving a ≤10 mg, ≤7.5 mg, ≤5 mg, or 0 mg daily OGC dose from belimumab initiation through the follow-up periods.
Secondary endpoints included the mean absolute and percent changes in daily OGC dose from belimumab initiation, the percentage of patients maintaining the same daily OGC dose threshold at 12, 18, and 24 months as achieved at 6 months post-initiation (≤10 mg, ≤7.5 mg, ≤5 mg, or 0 mg) and the mean average absolute change in daily OGC dose from 6 to 12, 18 and 24 months post-initiation, and the percentage of patients achieving low disease activity state and remission, measured by modified LLDAS (mLLDAS) and modified DORIS (mDORIS). mLLDAS was defined as SLEDAI score ≤4 with no worsening of clinical manifestations (i.e., renal, central nervous system, cardiopulmonary, fever, hemolytic anemia, vasculitis, or gastrointestinal activity, as determined by the clinical judgment of the investigating physician) since belimumab initiation and OGC dose ≤7.5 mg/day. 29 mDORIS was defined as SLEDAI score = 0 and OGC dose ≤5 mg/day 11 ; the PGA component of mDORIS was not captured fully in the original OBSErve studies and it was excluded from the mDORIS definition used in this study. SLEDAI scores captured at each follow-up were total scores only (individual domains were not recorded), and both, SLEDAI-2K and Safety of Estrogens in Systemic Lupus Erythematosus National Assessment (SELENA)-SLEDAI scores, were used. The attainment of mLLDAS and mDORIS was calculated only for patients with complete data (including available SLEDAI). Separate analyses for mLLDAS and mDORIS attainment over time were also conducted by clinical manifestations at belimumab initiation.
All endpoints were analyzed at 6, 12, 18, and 24 months after belimumab initiation (as appropriate; for details see Figure 1). For all endpoints, additional analyses were conducted for a subgroup of patients with immunologic/serologic activity, defined as increased anti-dsDNA antibody level, low complement levels, and/or elevated erythrocyte sedimentation rate at belimumab initiation.
Statistical analysis
No formal sample size calculations were performed; this descriptive analysis utilized a fixed sample size determined by the available data from the original OBSErve studies. No imputation of missing data was performed.
Descriptive statistics were used to summarize continuous data (mean, median, standard deviation [SD]) and categorical data (counts and frequencies).
Patients who discontinued belimumab between the follow-up time points were lost to follow-up after the time point for which discontinuation was confirmed. Therefore, any data captured at the same follow-up time point as belimumab discontinuation were noted (i.e., OGC use and mLLDAS/mDORIS components), were considered valid, and included in the assessment of study endpoints. Changes in OGC dose and disease activity between follow-up periods were not captured; data were only collected at specified 6-monthly time periods. Patients prescribed no OGC at initiation and/or follow-up were included in the 0 mg/day OGC dose category. For the OGC dose, prednisone equivalency OGC was assumed based on prednisone being the most common OGC used in lupus management. 30 OGC dose per administration was captured as mg, mg/kg, or other, with doses given in mg/kg converted to mg using available weight data. OGC administration frequencies were captured ranging from at least twice daily to 6-monthly, with all doses converted to daily.
Results
Patient population
Patient characteristics.

CNS, central nervous system; OGC, oral glucocorticoid; SD, standard deviation; SLE, systemic lupus erythematosus; SLEDAI, SLE Disease Activity Index.
aN = 878, data for Argentina were unavailable.
bPatients were enrolled (identification for inclusion) 6 months (Germany, Spain, Switzerland, Saudi Arabia, Russia, and n = 217/501 of patients in the USA) or 12 months (Argentina and n = 284/501 of patients in the USA) after belimumab initiation, or after ≥8 belimumab infusions equivalent to 6 months of treatment (USA and Canada only).
cRecord-level data were not available from Argentina, resulting in a pooled SD.
dPhysician assessed at belimumab initiation for all countries except Russia and Saudi Arabia and assessed at diagnosis only for patients in Russia and Saudi Arabia.
eN = 853, data for Russia and Saudi Arabia were unavailable.
fN = 106.
gN = 760, patients on any steroid, including non-OGC, and excluding patients on 0 mg/day.
hN = 738, among patients with OGC >0 mg/day at belimumab initiation, with known dose, unit, and frequency.
iN = 937, excluding patients on non-OGC and unknown dose, unit, or frequency.
Among those receiving any steroid (including non-oral formulations, such as IV and intramuscular) at belimumab initiation (79.2%; n = 760/959), 98.0% (n = 745/760) of patients received OGC. Of those patients, 99.1% (n = 738/745) received OGC of known dose, unit, and frequency (>0 mg/day) at belimumab initiation, with a mean (SD) OGC dose of 16.6 (13.2) mg/day (Table 1). At belimumab initiation (N = 738), 16.1% (n = 119), 22.0% (n = 162), 48.0% (n = 354), and 52.0% (n = 384) received ≤5, ≤7.5, ≤10, and >10 mg/day OGC, respectively (Figure 2(a)). The percentage of patients prescribed lower daily OGC doses (≤10 mg, ≤7.5 mg, ≤5 mg, and 0 mg) increased over time (Figure 2(a)). The percentage of patients with an OGC dose ≤5 mg/day changed from 16.1% (n = 119/738) at belimumab initiation to 51.6% (n = 379/734) at 6 months post-initiation and to 87.5% (n = 225/257) at 24 months post-initiation. The percentage of patients who discontinued OGC was 8.2% (n = 60/734) at 6 months and 44.4% (n = 114/257) at 24 months following belimumab initiation. Percentage of patients achieving each OGC dose thresholda at 6 months (all countries) and up to 24 months (USA/Argentina) among those receiving any dose of OGC (>0 mg/day) (a) or >10 mg/day OGC (b) at belimumab initiation. aDose categories are cumulative and not mutually exclusive; bin the USA and Argentina population. OGC, oral glucocorticoid.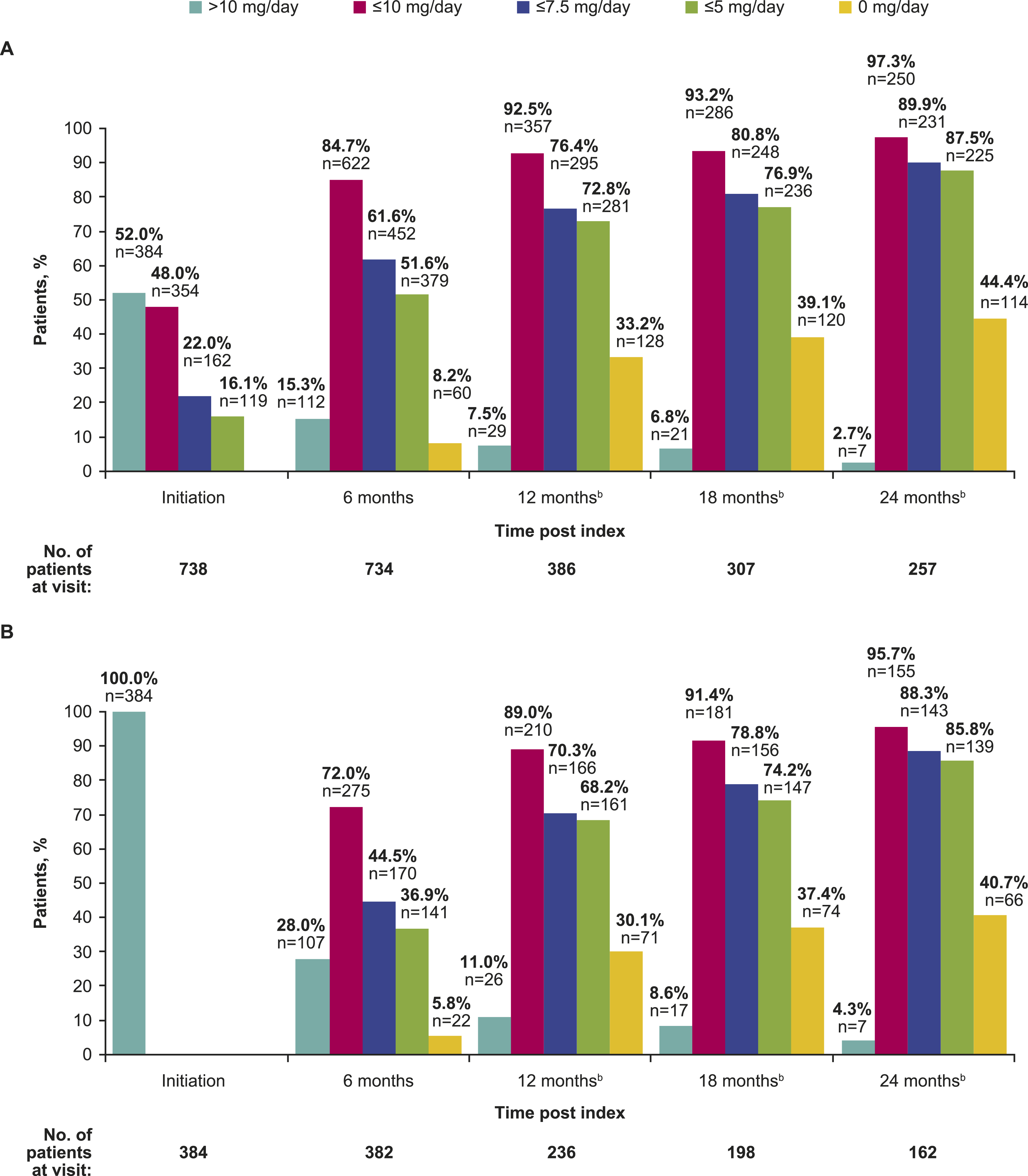
Among patients receiving >10 mg/day OGC at belimumab initiation (n = 384), the percentage of patients prescribed lower daily OGC doses (≤10 mg, ≤7.5 mg, ≤5 mg, and 0 mg) also increased over time (Figure 2(b)). The percentage of patients receiving ≤5 mg/day OGC changed from 36.9% (n = 141/382) at 6 months to 85.8% (n = 139/162) at 24 months after belimumab initiation. The percentage of patients who discontinued OGC changed from 5.8% (n = 22/382) at 6 months to 40.7% (n = 66/162) at 24 months following belimumab initiation.
Among the USA patients receiving OGC at belimumab initiation, the percentage of patients receiving ≤5 mg/day continued to increase throughout the 24-month follow-up period (Supplemental Figure 2). A gradual OGC discontinuation in the USA population and among those receiving OGC >10 mg/day at belimumab initiation was also observed between 6 and 24 months following belimumab initiation.
The percentage of patients with immunologic activity at belimumab initiation who reduced or discontinued OGC use were consistent with the findings above (Supplemental Table 1).
Over half of the overall pooled population (n = 532/933, 57.0%) reduced their mean absolute OGC dose from belimumab initiation to 6 months, while fewer patients experienced no change (n = 370/933, 39.7%) or a dose increase (n = 31/933, 3.3%) (Figure 3(a)). The overall mean relative change in OGC dose was −41.2% following 6 months of belimumab treatment (Figure 3(b)). Dose decreases were also observed among those followed up beyond 6 months (USA/Argentina population), from belimumab initiation throughout the 24-month follow-up period (Figure 3(a)), with the overall mean relative change in OGC dose of −50.2% at 6 months, to −78.0% at 24 months (Figure 3(b)). The USA population alone also showed dose decreases throughout the 24-month follow-up period (Supplemental Figure 3A), with the overall mean relative change in OGC dose ranging between −50.4% and −78.8% following 6 to 24 months of belimumab treatment (Supplemental Figure 3B). Absolute changes in daily OGC dose (a) and relative percent changes in OGC dose (b) from initiation over time in all patientsa and patients in the USA and Argentinaa.aPatients with available data and with known OGC dose, unit, and frequency were included in the analysis. Patients on 0 mg/day OGC at baseline were also included. OGC, oral glucocorticoid.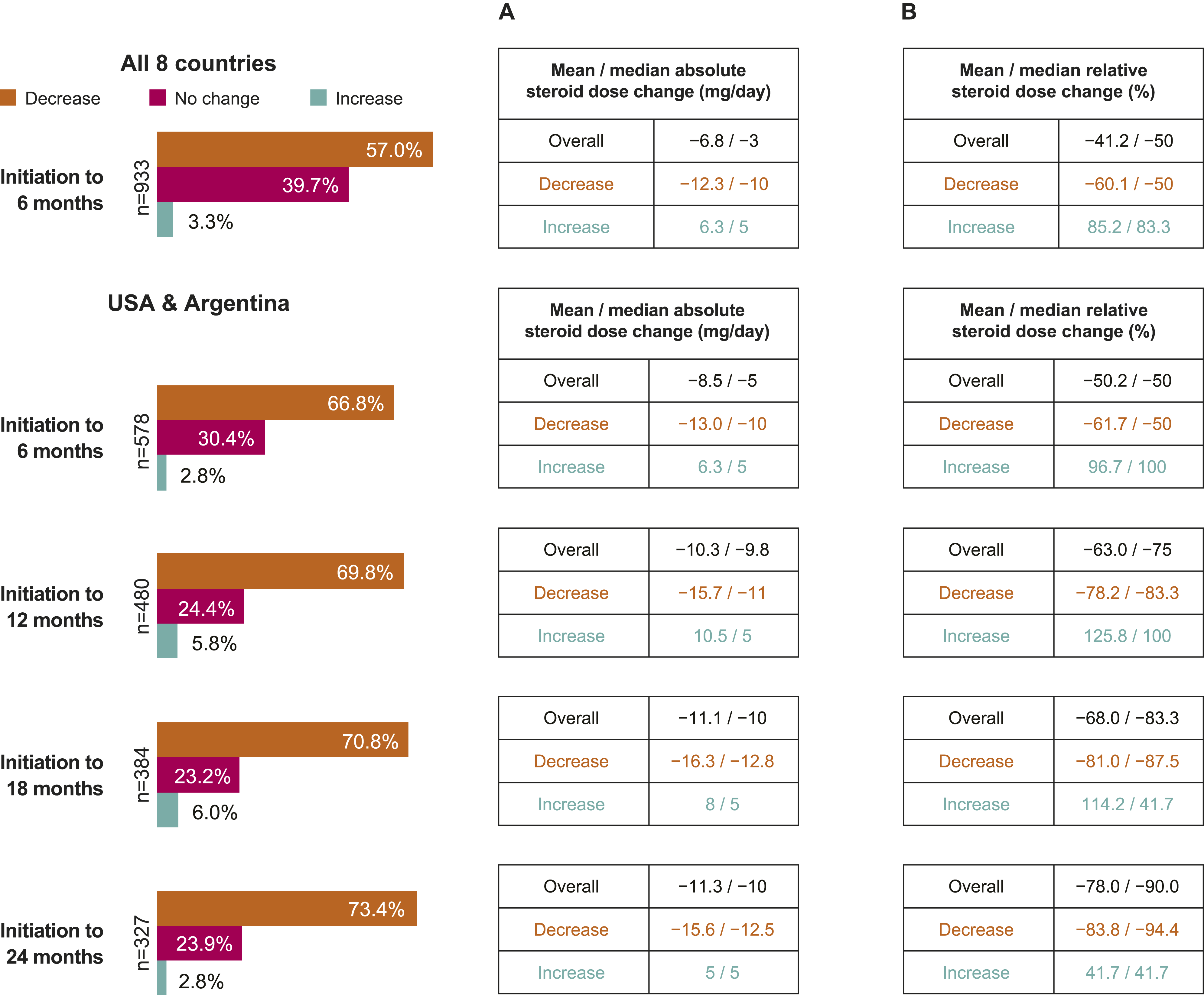
Of those followed up beyond 6 months (USA/Argentina population), at least 87.9% maintained an OGC dose of 0 mg/day between 6 and 24 months following belimumab initiation (Supplementary Figure 4A). Similarly, at least 92.9% of patients who achieved a daily OGC dose of ≤5 mg sustained this dose between 6 and 24 months after belimumab initiation (Supplementary Figure 4A). Among patients in the USA, at least 87.1% and 91.8% of patients continued to maintain an OGC dose of 0 mg/day and ≤5 mg/day, respectively, from 6 to 24 months following belimumab initiation (Supplementary Figure 4B).
Mean absolute and relative percent changes in OGC dose and for the OGC dose maintenance beyond 6 months of belimumab initiation in patients with immunologic activity at initiation are in line with the findings above (Supplemental Tables 2 and 3).
Attainment of low disease activity state and remission
In the overall pooled population with complete data available (including SLEDAI), the percentage of patients attaining mLLDAS increased from 0.4% (n = 2/447) at belimumab initiation to 11.1% (n = 49/440) at 6 months following belimumab initiation (Figure 4(a)). Of those followed up beyond 6 months (USA/Argentina population), the percentage of patients attaining mLLDAS was 19.5% (n = 39/200) at 6 months, 26.9% (n = 45/167) at 12 months, 10.3% (n = 11/107) at 18 months, and 18.2% (n = 18/99) at 24 months (Figure 4(a)). A number of patients who achieved mLLDAS at 12 months had missing SLEDAI data (n = 6/45) and were lost to follow-up (n = 12/45) at 18 months (Figure 4(a)). Percentage of patients attaining mLLDAS (a) and mDORIS (b), and complementary patient journeys in USA and Argentina. Note. The attainment of mLLDAS and mDORIS was calculated only for patients with complete data (including available SLEDAI). mDORIS, modified definition Of Remission In SLE; mLLDAS, modified Lupus Low Disease Activity State; SLEDAI, SLE Disease Activity Index.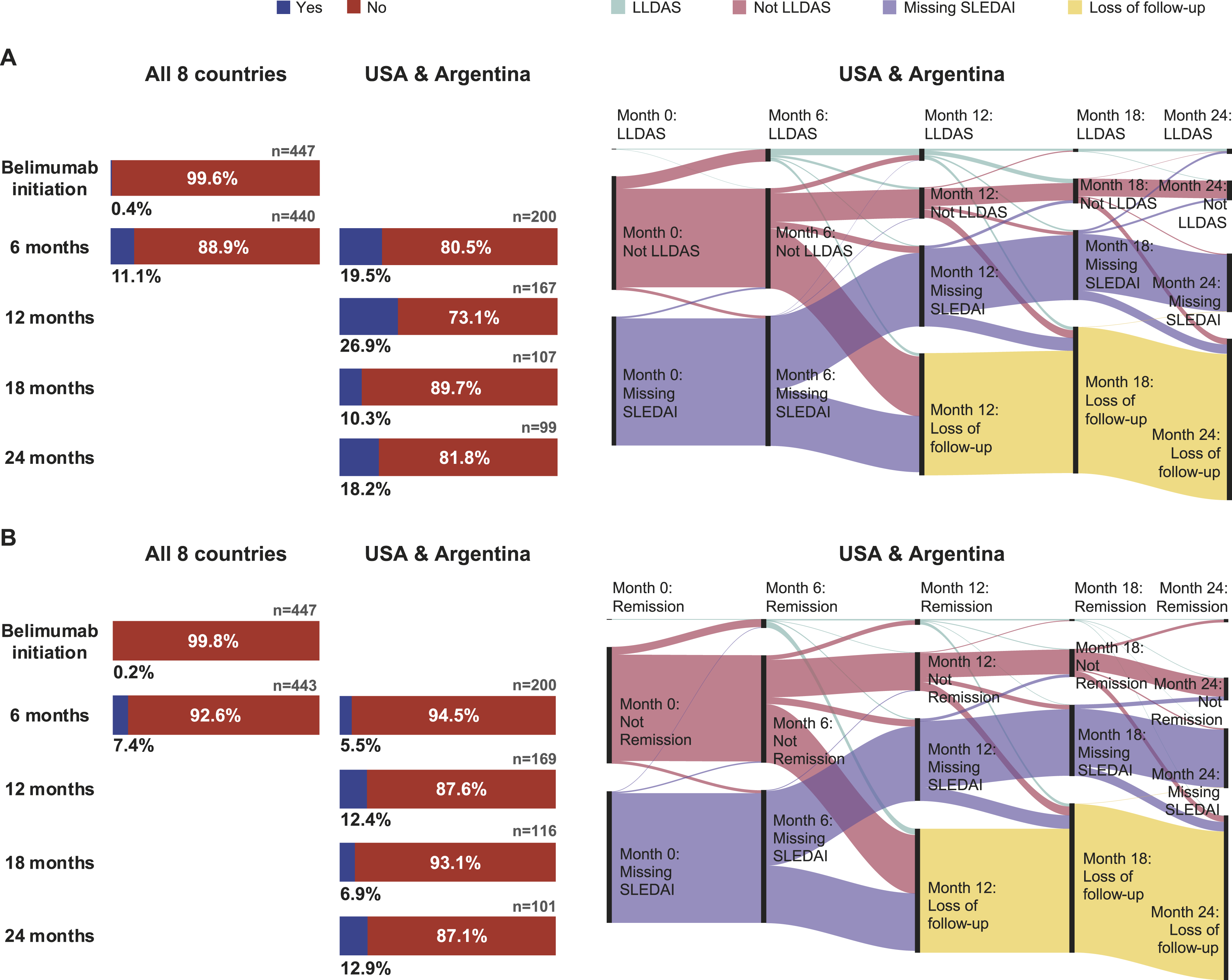
Overall, among the patients with complete data and available SLEDAI, the percentage of patients in mDORIS remission increased from 0.2% (n = 1/447) at belimumab initiation to 7.4% (n = 33/443) at 6 months following belimumab initiation (Figure 4(b)). Among the pooled USA/Argentina population, the percentage of patients attaining mDORIS was 5.5% (n = 11/200) at 6 months, 12.4% (n = 21/169) at 12 months, 6.9% (n = 8/116) at 18 months, and 12.9% (n = 13/101) at 24 months (Figure 4(b)). A proportion of patients who achieved mDORIS at 12 months were lost to follow-up (n = 11/21) and had missing SLEDAI (n = 2/21) at 18 months (Figure 4(b)).
Results for mLLDAS and mDORIS attainment in patients categorized by clinical manifestation at belimumab initiation are summarized in Supplemental Table 4. Briefly, the number of patients with immunologic activity attaining mLLDAS at 6 and 12 months following belimumab initiation was consistent with the findings above for the larger population; however, fewer patients achieved mLLDAS at 18 and 24 months (5.9%, n = 5/85, and 7.2%, n = 5/69, respectively) relative to the USA and Argentina pooled population. Overall, the attainment of mLLDAS by clinical manifestation varied at different follow-ups but numerically higher trends were observed among patients with cardiopulmonary and central nervous system (CNS) involvement at belimumab initiation (Supplemental Table 4).
Results of mDORIS attainment in the subgroup of patients with immunologic activity were comparable to the findings from USA and Argentina populations, except for 24 months, where a higher number of patients (17.1%, n = 12/70) achieved mDORIS. Similarly to mLLDAS, the attainment of mDORIS by clinical manifestation at belimumab initiation also varied at different follow-ups (Supplemental Table 4).
Discussion
This descriptive, pooled analysis assessed the impact of belimumab use on OGC use and disease activity over time in a large and diverse population, predominantly with moderate or severe SLE activity at belimumab initiation. To align with the 2023 EULAR recommendations for the management of SLE, 9 our analyses focused on lower OGC dosing thresholds (i.e., ≤5 mg/day) and explored the short (6 months follow-up from belimumab initiation) and long-term (>6–24 months follow-up) implications of belimumab treatment on OGC use in a broad population of patients with SLE. Further, this study also evaluated the impact of belimumab use on disease activity and achieving remission, as measured by mLLDAS and mDORIS, respectively.
We demonstrated that approximately half of patients treated with belimumab in real-world practice were able to achieve the 2023 EULAR-recommended OGC dose threshold of ≤5 mg/day as early as 6 months after belimumab initiation, and maintain this OGC dose through 24 months of follow-up. We noted a substantial shift in patients receiving ≤5 mg/day OGC, from 16.1% (n = 119/738) at index to 87.5% (n = 225/257) at 24 months after belimumab initiation, among patients on >0 mg/day OGC at index, as well as a shift in the proportion of patients receiving other dose thresholds (>10, ≤10, ≤7.5, 0 mg/day). Although higher baseline OGC doses might be expected in correlation to the reported disease severity at belimumab initiation (with 71.3% and 20.5% of patients presenting with moderate and severe disease, respectively), the observed mean (SD) dose was 16.6 (13.2) mg/day. While a substantial percentage of patients (52% [n = 384/738]) was receiving OGC doses greater than 10 mg/day at the start of belimumab treatment, variability in dosing practices across the included studies may have influenced these findings. Furthermore, more than half of the cohort had a longstanding SLE (>5 years), suggesting that many patients were beyond the early, more aggressive phase of OGC therapy. In such patients, OGC doses are often tapered over time to minimize cumulative toxicity, even when disease activity remains moderate or severe. 9 Despite our additional analysis of mean absolute changes in daily OGC dose showing that 39.7% (n = 370/933) of patients had no change in their mean OGC dose from belimumab initiation to 6 months post-initiation, it is worth noting that this particular analysis was inclusive of 21.2% (n = 199/937) of patients who received no OGC (0 mg/day) at belimumab initiation. Moreover, 33.9% (n = 318/937) of patients were already receiving ≤5 mg/day OGC (including those on 0 mg/day) at belimumab initiation, and further reductions may have been unlikely. Although the original OBSErve studies were not designed to assess steroid tapering, nor were there any guidelines about steroid tapering at the time of those studies, the effect of belimumab treatment on the progressive reduction of OGC dosing aligns with the EULAR 2023 recommendations. 9
The impact of long-term GC usage on the development/progression of organ damage in patients with SLE has been well-established.31,32 Belimumab may further mitigate this effect by decreasing OGC reliance. Previous studies using USA healthcare claims support this premise by demonstrating that patients with SLE treated with belimumab early in their disease experienced favorable outcomes in a real-life setting. However, the likelihood of favorable outcomes decreased with increasing organ damage.33,34
This analysis demonstrated that sustained belimumab treatment contributed to attaining low disease activity and remission, measured by mLLDAS and mDORIS. A trend for a higher proportion of patients with vital organ involvement (such as cardiopulmonary, CNS, and renal manifestations) achieving mLLDAS compared to other subgroups was observed (Supplemental Table 4). This finding was unexpected and could be explained by factors such as more aggressive management strategies in these patients, small subgroup sizes influencing proportions, and potential selection bias in the cohort, and warrants further future investigation. Overall, missing SLEDAI data in a large portion of patients in this real-world cohort contributed to observed fluctuations over the 24-month follow-up period. Since the mLLDAS and mDORIS definitions used in this analysis required a patient to have a low OGC dose (≤7.5 mg/day and ≤5 mg/day, respectively) and a SLEDAI score indicative of minimal disease activity (≤4 and 0, respectively), missing SLEDAI data would affect the ability to classify patients according to these criteria. The retrospective nature of the studies included in this analysis meant that SLEDAI assessments were not mandated during the follow-up visits; thus, data missingness could not be controlled. The drop in the percentage of patients achieving mLLDAS or mDORIS at 12 and 18 months post-initiation was also associated with the loss to follow-up after 12 months. The use of LLDAS and DORIS endpoints in clinical and real-world studies is a relatively new concept. As a result, analyses of past data on the effect of belimumab on these outcomes are subject to certain limitations, and may have resulted in the underrepresentation of the actual numbers of patients achieving mLLDAS and mDORIS. Nevertheless, the results of this study align with an integrated post hoc analysis of data pooled from five phase 3 belimumab studies, which demonstrated a significantly greater probability of attaining LLDAS and DORIS with belimumab than with placebo added to standard therapy. 35 It is worth noting that none of these clinical studies mandated an attempt to taper steroids. 35 Collectively, these results provide further support that belimumab may help patients achieve the EULAR 2023-recommended SLE treatment targets: low disease activity state and remission. 9
The major strength of this analysis lies in the coverage of data from multiple countries and the use of data from real-world clinical settings from multiple countries. The results build upon previous findings by incorporating data from two additional studies, despite none of the studies being designed with steroid tapering in mind, and present strong evidence for the use of belimumab in all assessed countries. The inclusion of up to 24 months of follow-up data from the USA and Argentina further strengthens the analysis by allowing a more detailed evaluation of outcomes seen with belimumab therapy over time. The long-term follow-up period provided insights into the durability of treatment response, demonstrating that the improvements in disease activity and reductions of OGC use associated with belimumab are maintained over an extended period in a real-world clinical setting. In addition, data from the OBSErve populations provide an additional source of clinical information related to OGC use, mLLDAS, and mDORIS, addressing critical gaps that exist in data from real-world clinical settings. Unlike randomized controlled trials with stringent eligibility criteria and closely monitored treatment protocols, the analysis of OBSErve study data reflects the complexities and nuances of everyday clinical practice, enhancing the relevance and applicability of these findings to clinicians and patients, who make treatment decisions in routine care settings. Overall, these results complement the findings of the individual OBSErve studies, which demonstrated overall clinical improvement and reduced risk of new flares in patients with SLE.20–25
Limitations possibly affecting the generalizability of the results to a broader SLE population include selection bias, as patients with SLE were sampled from a subset of sites managing SLE as part of the original OBSErve studies, under various eligibility criteria. While some clinical characteristics relied on physician judgement and recall, available data in medical records supported retrospective assessments, reducing missingness and risk of recall bias. OGC dose and disease activity were captured at each 6-monthly follow-up visit; however, any changes occurring between these intervals or cumulative dosages were not recorded, potentially leading to interval bias. In addition, the open-label design of the OBSErve studies and the lack of a comparator cohort and data on concurrent medication, such as immunosuppressant use during the observation period, may limit conclusions about belimumab’s relative effectiveness compared with other therapeutic options. Due to limitations in available data across included cohorts, the study was unable to control or adjust outcomes for confounding variables such as concurrent immunosuppressive therapy. Of note, previously published pooled analysis of OBSErve studies (excluding Saudi Arabia and Russia) reported immunosuppressant use in 59% (n = 488/830) of patients at index. 26 In OBSErve Saudi Arabia, 70% (n = 33/47) of patients received immunosuppressants at/prior to index, 27 and 51% (n = 30/59) of patients in OBSErve Russia (GSK data on file; not published). The lack of the PGA component, as outlined in the full definition of LLDAS and DORIS, poses another limitation to this study, as it is essential to capture disease severity not reflected by SLEDAI.10,11 Another potential limitation is that the historical data on OGC use may not reflect the current clinical practice; most of the OBSErve studies were conducted before EULAR 2019 and all before EULAR 2023 recommendations, advocating to reduce GC doses to ≤7.5 mg/day or ≤5 mg/day, respectively, or withdrawing when possible.9,36 Finally, patients’ non-adherence to the prescribed treatment could also have influenced the assessed outcomes, as patients could modify or omit self-administered SLE treatments, such as OGC, without these adjustments being recorded. This, in turn, may lead to a potential over- and underestimation of study outcomes. Despite these limitations, many of which are inherent to real-world studies, our descriptive analysis of data from clinical practice provides compelling evidence of the beneficial impact of belimumab treatment in patients with SLE.
Conclusions
Consistent with EULAR 2023 recommendations for SLE treatment, OGC dose reductions to ≤5 mg/day were attained as early as 6 months and sustained up to 24 months following belimumab initiation, thus demonstrating belimumab’s steroid-sparing potential. Moreover, disease activity improvements were seen following belimumab use, as evidenced by achieving mLLDAS and mDORIS, further emphasizing the therapeutic effectiveness of belimumab in mitigating disease burden and improving patient outcomes in SLE management.
Supplemental material
Supplemental Material - Long-term steroid-sparing effect of belimumab in systemic lupus erythematosus: Post hoc pooled analysis of OBSErve multi-country cohort data
Supplemental Material for Long-term steroid-sparing effect of belimumab in systemic lupus erythematosus: Post hoc pooled analysis of OBSErve multi-country cohort data by Daniel Moldaver, Seth Anderson, Marguerite Bracher, Holly A Quasny, Robert Wood, Elke Rottier, Rosie Wild, Alexandra Cusmano, Roger A Levy in Lupus
Consent to participate
Informed consent was obtained from all participants in the original OBSErve studies. No direct subject contact or primary collection of individual human subject data occurred in this study, and study results are in tabular form and aggregate analyses that omit subject identification, therefore informed consent was not required (a waiver granted by relevant IRB).
Footnotes
Acknowledgments
We would like to thank all the patients and healthcare professionals involved in the OBSErve studies. Medical writing support was provided by Olga Conn, PhD, and Marta Budzinska, PhD, Fishawack Indicia Ltd, UK, part of Avalere Health, and was funded by GSK. Submission support was provided by Marta Budzinska, PhD, Fishawack Indicia Ltd, UK, part of Avalere Health, and was funded by GSK. Authors authorized manuscript submission via third party and any statements or declarations have been approved.
Author contributions
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by GSK (GSK Study 219649).
Correction (March 2026):
Ethical considerations
All original studies were approved by relevant national authorities, ethics committees and/or institutional review board (IRB), and were conducted in accordance with the Declaration of Helsinki 2008 and its later amendments, and the International Society for Pharmacoeconomics and Outcomes Research Guidelines/Guidelines for Good Pharmacoepidemiology Practices/Guidelines for Good Clinical Practice. An ethics committee’s or IRB’s approval was not required for this analysis, as it used previously published data.
Data Availability Statement
Supplemental material
Supplemental material for this article is available online.
