Abstract
Aims
Lupus nephritis (LN) is common in systemic lupus erythematosus (SLE) and is associated with adverse renal outcomes and premature mortality. There is limited data examining LN outcomes in Aotearoa New Zealand and none examining outcomes since mycophenolate mofetil (MMF) became subsidised for use in class III/IV LN induction. We describe a cohort of adults with biopsy-confirmed LN over an 18-year period in two regions of Aotearoa New Zealand, including LN characteristics, treatment, and outcomes.
Methods
Cases were identified from laboratory databases and relevant data extracted from patient records. Response was defined per Kidney Disease, Improving Global Outcomes (KDIGO) with overall renal response (ORR) defined as at least partial response (PR). Outcomes among patients with class III/IV LN were explored by induction treatment and timing of MMF restrictions.
Results
One hundred cases were identified, including 74 with class III/IV LN. Most (85/100) were women, living in urban areas (78%), with ethnicities including Māori (25%), Pacific (13%) and NZ European (38%). The median age at LN diagnosis was 38 years (range 18-74) and the median time between SLE diagnosis and renal biopsy was 2 years. In the MMF-restricted period, MMF was used for induction in class III/IV LN in 43% (12/28) of cases, and in 72% (33/46) of cases in the MMF-unrestricted period (p = .01). In the MMF-unrestricted period, use of high dose cyclophosphamide stopped (18% to 0%), complete response (CR) rates doubled (14% to 33%, p = .08) whereas rates of ORR did not show statistically significant change (39% to 50%, p = .37). At last-observed follow up (mean 7 years from biopsy) 26/74 (36% cases) had poor outcomes with no renal response.
Conclusion
In this LN cohort in Aotearoa New Zealand, half of people with LN class III/IV do not achieve early renal response and over one-third have poor outcomes over less than a decade of follow up. Subsidy of MMF substantively increased its use and patients in this time period had better rates of good LN outcomes. These data suggest considerable unmet need for effective treatments for LN and that funding of effective medicine for LN increases their use and improves LN outcomes.
Introduction
Lupus nephritis (LN) is a common and potentially severe manifestation of systemic lupus erythematosus (SLE) associated with significant risk of kidney failure (KDIGO CKD G5) and premature mortality. 1 Treatments can improve outcomes but have a significant potential for treatment-related adverse effects. Fortunately, treatment of LN has evolved so cyclophosphamide (CYC) induction regimens can be replaced by less toxic mycophenolate mofetil (MMF), which has similar efficacy and is indicated for LN class III/IV induction therapy.2–4 Achieving even a partial response of LN is associated with better long-term renal outcomes and reduced mortality. 5 Despite widespread use of hydroxychloroquine and increasing therapeutic options, a recent US population based study suggests that incidence of LN has not changed over the last four decades and unfortunately patient outcomes have not improved. 6 Therefore, LN remains a key manifestation of concern in SLE.
There is a paucity of data examining LN outcomes for people in Aotearoa New Zealand where there is a government-funded healthcare system with universal access. A 2007 case series from two hospitals in Auckland included 170 patients with SLE and reported 14% of patients had LN at presentation, which increased to 32% at last follow up. 7 A higher risk of LN was reported in Māori (odds ratio (OR) 8.5) and Pacific Peoples (OR 3.1). A retrospective single hospital case series from Auckland in 2017 described outcomes for 41 people with LN, 53% of Pacific Island ethnicity. 8 There were no differences in response or long term outcomes between people with difference ethnicities, with an overall renal response (ORR) achieved for 67% (10/15) of Pacific patients receiving MMF or CYC induction. More recent comprehensive national data examining long-term SLE outcomes from the Australia New Zealand Transplant and Dialysis Registry (ANZDATA) included 2837 patients with SLE, of which 210 (7.4%) developed kidney failure, 182 (86.7%) being attributed to LN. 9 Overall, these data suggest LN is a concern in Aotearoa New Zealand, particularly with increased risks of poor outcomes in Māori and Pacific peoples.
Aotearoa New Zealand has a socialised healthcare system, being government-funded with universal access. The Pharmaceutical Management Agency (Pharmac) is responsible for regulating funding and access to pharmaceuticals. Pharmac applied full subsidy to MMF on 1st October 2014, meaning prescribers did not have the potential barrier of application for MMF subsidy for LN treatment, requiring demonstration that specific, restrictive criteria were met. Around this time, the evidence base was accumulating to support MMF as an effective first line induction agent for class III/IV LN. There is a little work investigating induction therapy outcomes in LN since the improved access to MMF in Aotearoa New Zealand. We used laboratory data to identify people with LN in two regions of Aotearoa New Zealand (Wellington and Waikato), which have a combined population of approximately 965,000, around 20% of New Zealand’s population. 10 In this retrospective case series we describe 100 adults with biopsy confirmed LN over an 18-year period, class III/IV induction treatments, and outcomes including LN response at 6-12 months. We specifically examined for differences in induction therapy and disease outcomes since MMF became easily accessible in 2014, as well as describing disease characteristics and outcomes.
Methods
To identify adults with LN, we searched renal biopsy reports from 2006 to 2023 in the Omnilab (Waikato) and Awanui (Wellington) Laboratory anatomic pathology databases using keyword search “lupus” and “nephritis”. Reports were reviewed and those not consistent with LN were excluded. Inclusion criteria were (a) age ≥18 years at time of renal biopsy, (b) biopsy consistent with LN and (c) resident in catchment of Health New Zealand Capital, Coast and Hutt Valley (CCHV – Wellington region) or Health New Zealand Waikato (Waikato region) at the time of biopsy. Where multiple biopsies for a single patient were identified, the first biopsy within the study period meeting inclusion criteria was selected for analysis and referred to as ‘index biopsy’. Where LN class V co-existed with another LN class, we classified the biopsy as the co-existing LN class in the data set.
Using the national health index (NHI) as the patient identifier, we extracted demographic and clinical data from the hospital electronic records system (Clinical Workstation (Waikato) and Medical App Portal (Wellington)). Electronic documents accessed included clinic letters, discharge summaries, community dispensed medications, renal biopsy reports, and regional laboratory results. Data were entered into a password protected Excel spreadsheet and de-identified before analysis.
Demographic data included age, gender, ethnicity (per Stats NZ prioritisation for level 1 codes), rurality (per geographic classification of health) and NZ deprivation index ((NZDep18), based on domicile address at time of index biopsy).11–13 Baseline SLE clinical characteristics extracted included: date of SLE and LN diagnosis, current treatment, presence of double-stranded DNA (dsDNA) antibodies, anti-cardiolipin antibodies, hypocomplementaemia, leukopenia, thrombocytopaenia, suppressed haptoglobin or blood film evidence of haemolysis. 14 Histopathological data included LN class per International Society of Nephrology/Renal Pathology Society (ISN/PRS) 2016 and National Institutes of Health (NIH) histological activity and chronicity scores.15–17 Induction treatment agent was classified as MMF, low dose CYC (500 mg every 2 weeks for six doses, euro-lupus regimen), high dose CYC (0.5-1 g/m2 monthly for 6 months, NIH regimen) or ‘other’ if dosage not consistent with euro-lupus or NIH regimen. 18
Data points used to derive response status included urine protein: creatinine ratio (uPCR) (as a surrogate for 24 hr urine protein collection given this is rarely used in local clinical practice), and estimated glomerular filtration rate (eGFR) as per chronic kidney disease epidemiology collaboration (CKD-EPI). uPCR and eGFR were collected prior to index biopsy (closest date), in the period between 6 and 12 months post biopsy (highest uPCR/lowest eGFR), and at most recent follow-up testing. These results were used to derive response and CKD G5 status following induction and at most recent follow up testing per Kidney Disease, Improving Global Outcomes (KDIGO) definitions including complete response (CR), primary efficacy renal response (PERR) and partial response (PR). 4 Overall renal response (ORR) was defined as being achieved when at least partial response was obtained. We also report on dialysis dependence, renal transplantation and death at last-observed face-to-face follow up in the clinical record. Two patients had missing data for last-observed face-to-face follow up; where no follow-up date was evident but patient was deceased, date of death was used as date of last follow up. In the case of missing uPCR or eGFR, outcome was defined as no response.
Patient characteristics (age, SLE duration and presence of LN at SLE presentation) and renal data (including LN class, NIH activity index, chronicity index, uPCR and eGFR) at index biopsy were compared by ethnicity. Differences by ethnic group were examined with one-way ANNOVA for continuous variables and Chi-square test for categorical variables. For patients with LN class III/IV, their baseline characteristics and induction treatment were explored. The six to 12-month response (CR, PERR and PR) rates were compared by induction treatment and MMF-unrestricted period versus restricted period (pre 1st October 2014). For patients with LN class III/IV, we also examined the outcomes at long-term follow-up, including CR, PERR, PR, ORR, KDIGO CKD G5, dialysis, transplant, death and no response. We used last-observed laboratory data (uPCR/eGFR) and last-observed clinic review information regarding use of dialysis, transplant and death to derive long-term outcome data. Where missing data created ambiguity regarding outcome measures, patients were classified as the inferior outcome. All data analyses were conducted with R (R Institute, Vienna, Austria).
This study has ethical approval from the Health and Disability Ethics Committee (Ref 2024 EXP 19500) New Zealand as well as locality approval from Health New Zealand Capital Coast and Hutt Valley (Wellington) and Waikato, and underwent Māori consultation with Interim-Research Advisory Group Māori (Wellington) and Te Puna Oranga Māori Research Review Committee (Waikato).
Results
Baseline characteristics.
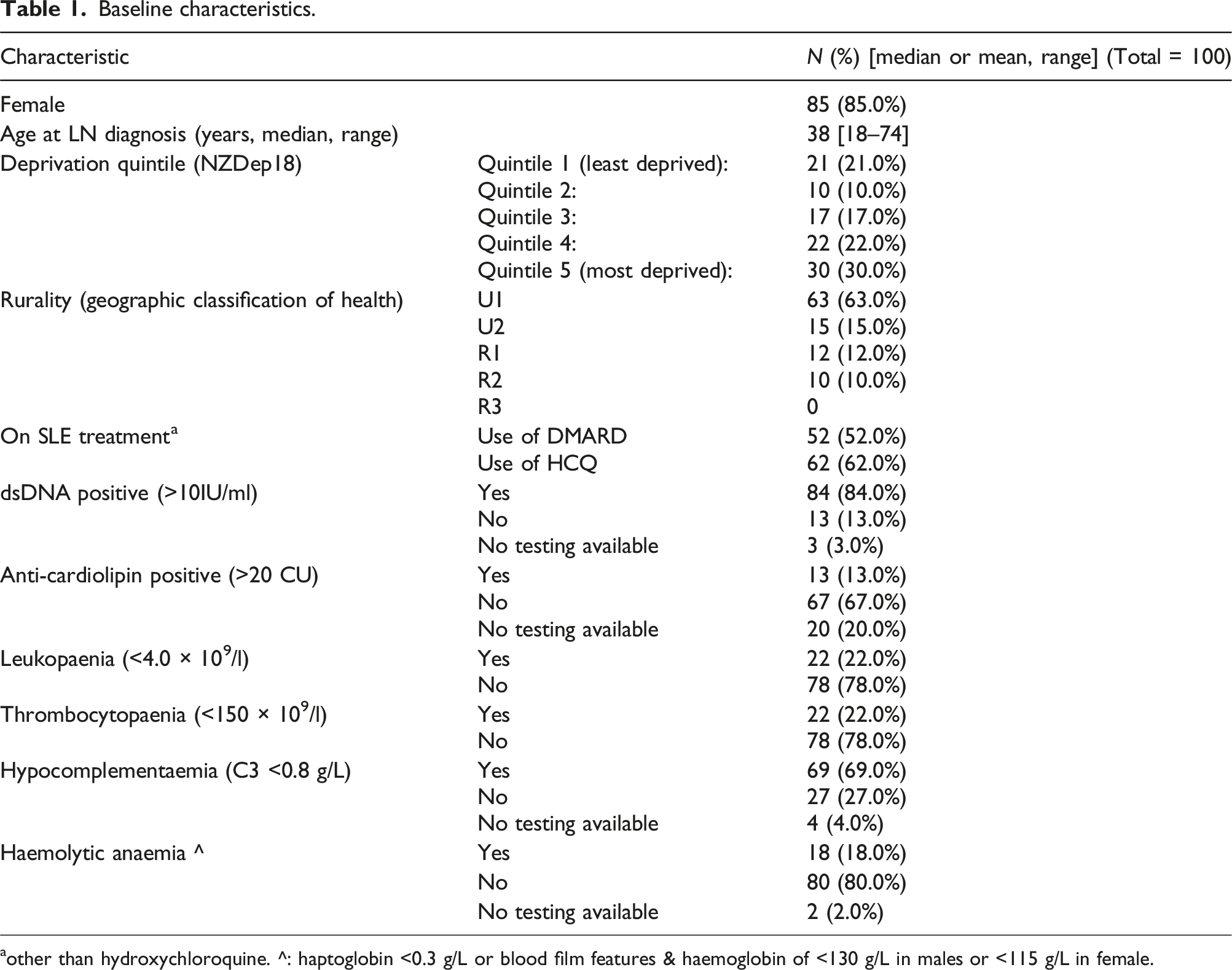
aother than hydroxychloroquine. ^: haptoglobin <0.3 g/L or blood film features & haemoglobin of <130 g/L in males or <115 g/L in female.
Renal baseline characteristics by ethnicity.
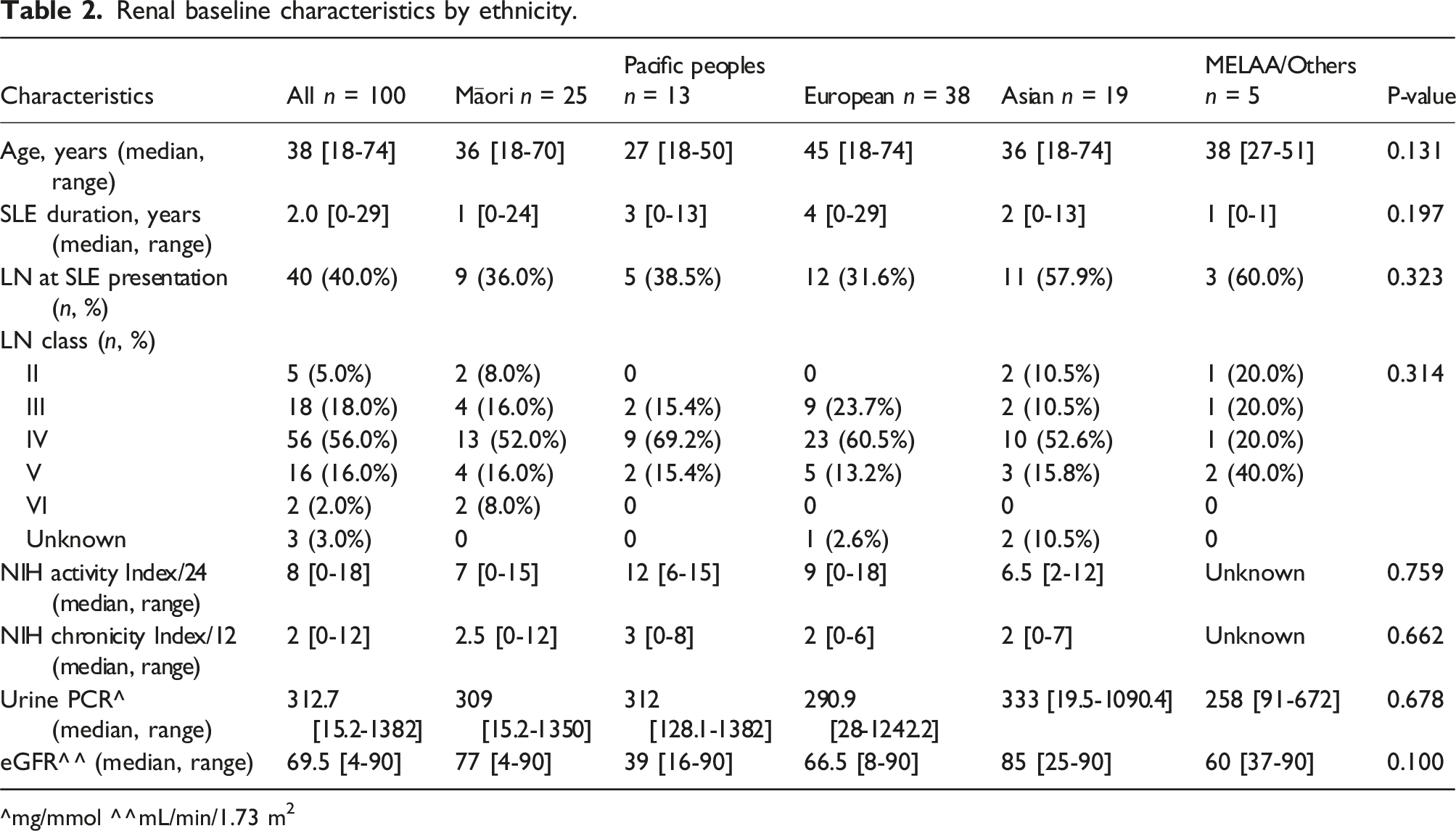
^mg/mmol ^^mL/min/1.73 m2
LN class III/IV (+/− V) Baseline characteristics at index biopsy, N = 74.
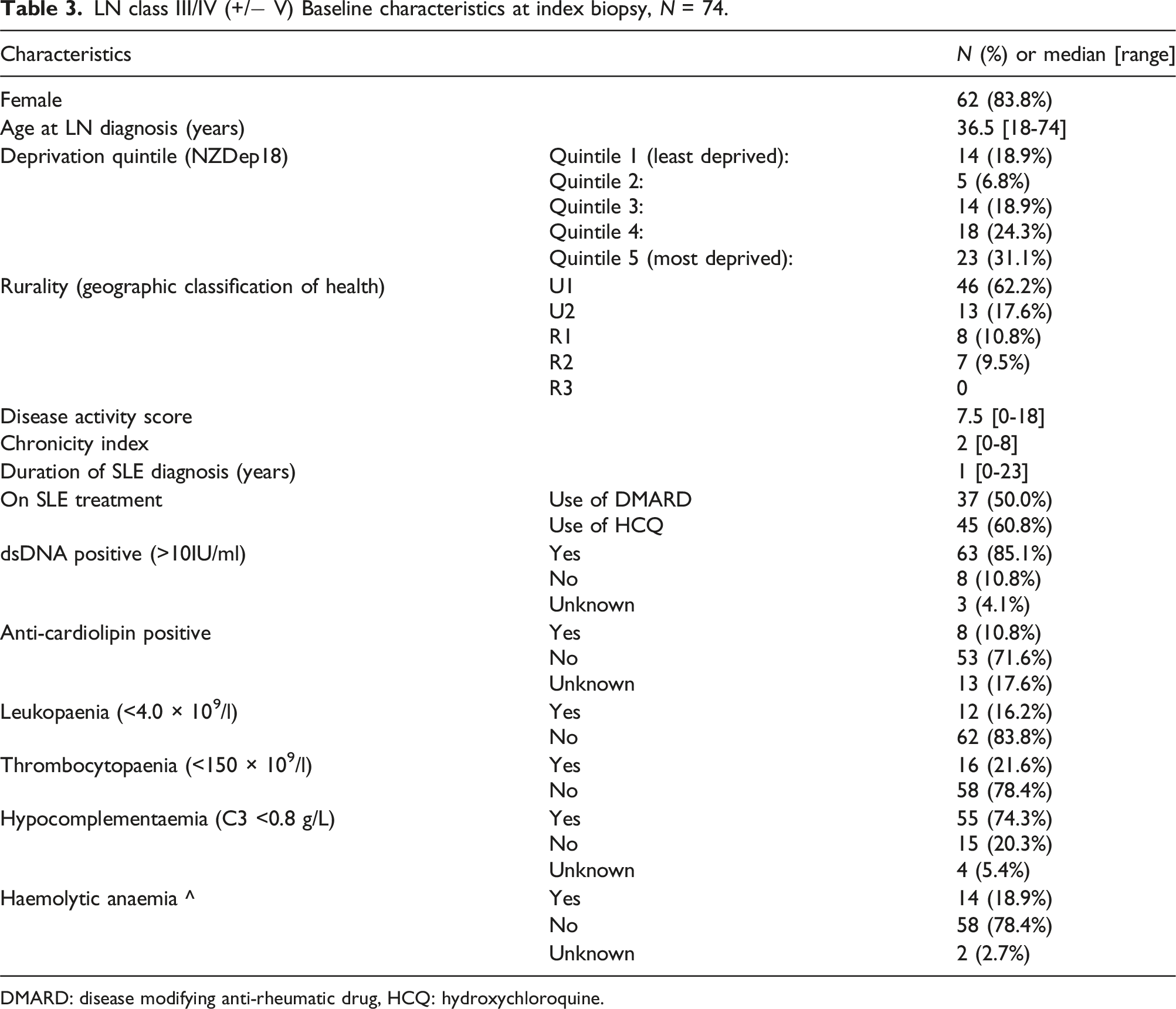
DMARD: disease modifying anti-rheumatic drug, HCQ: hydroxychloroquine.
LN class III/IV (+/− V) – Induction therapy & 6-12 month response rate.
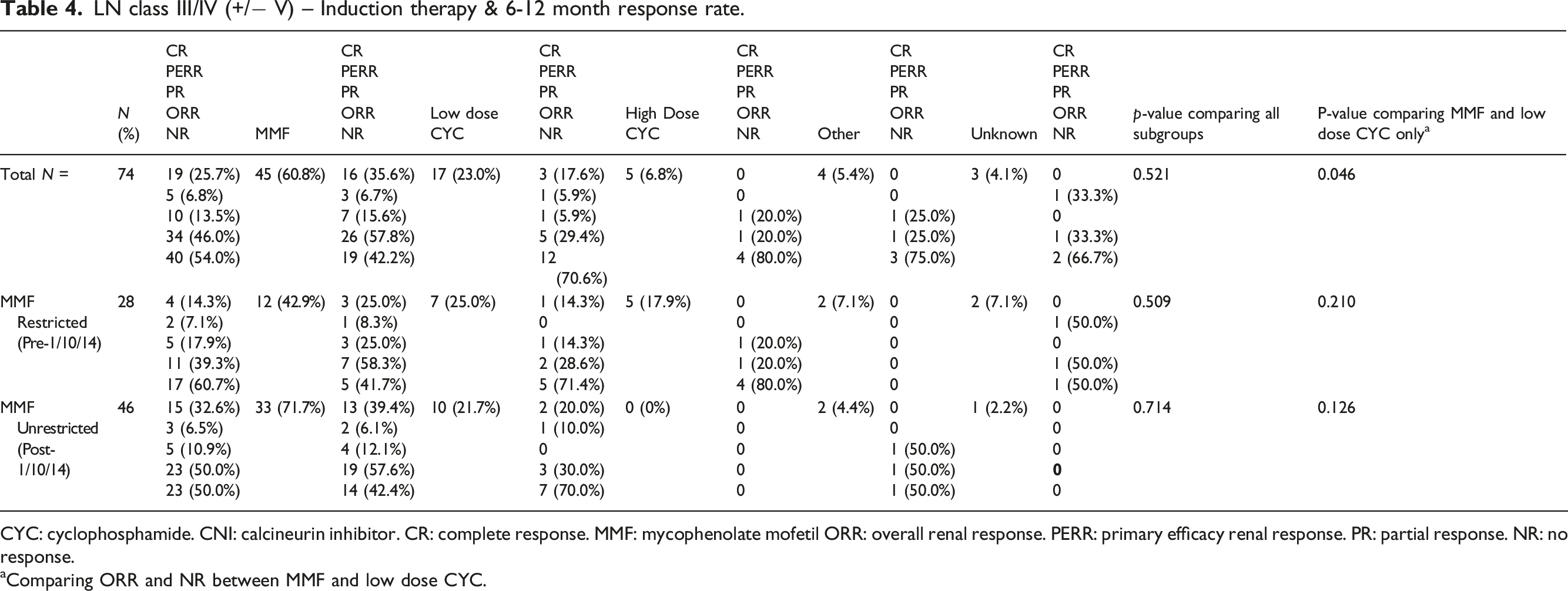
CYC: cyclophosphamide. CNI: calcineurin inhibitor. CR: complete response. MMF: mycophenolate mofetil ORR: overall renal response. PERR: primary efficacy renal response. PR: partial response. NR: no response.
aComparing ORR and NR between MMF and low dose CYC.
LN class III/IV (+/− V) - Long term outcomes.
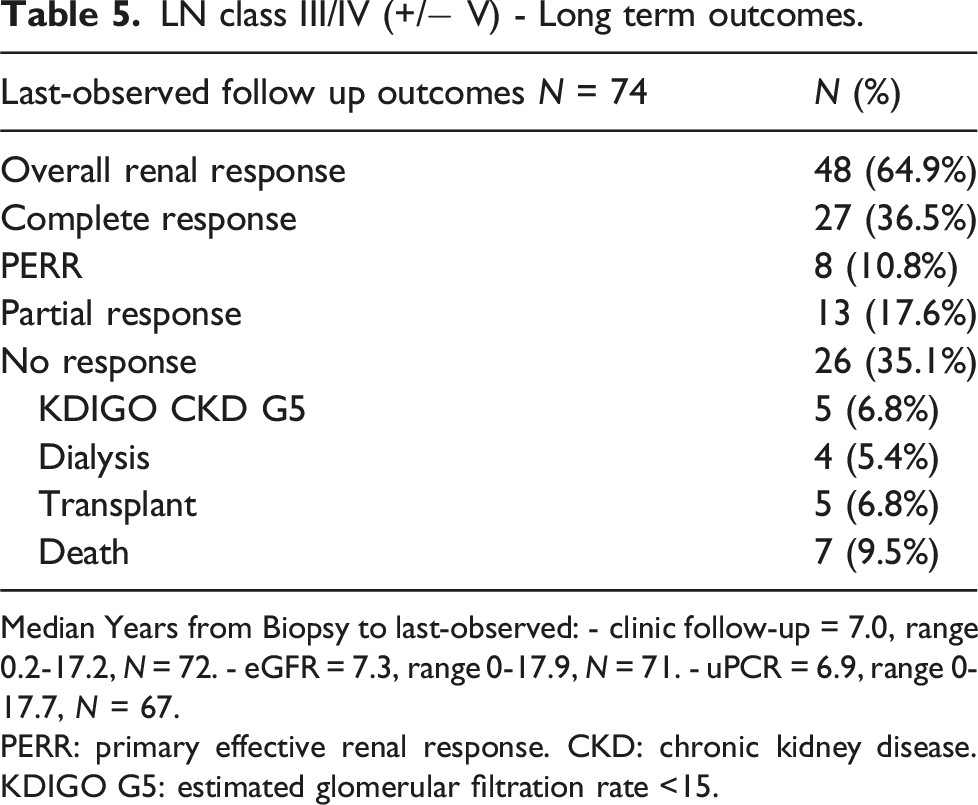
Median Years from Biopsy to last-observed: - clinic follow-up = 7.0, range 0.2-17.2, N = 72. - eGFR = 7.3, range 0-17.9, N = 71. - uPCR = 6.9, range 0-17.7, N = 67.
PERR: primary effective renal response. CKD: chronic kidney disease. KDIGO G5: estimated glomerular filtration rate <15.
Discussion
Our cohort of 100 people with biopsy confirmed LN in two hospitals over an 18-year period were young, had LN onset soon after diagnosis of SLE, and less than half achieved any form of LN response within 6-12 months of biopsy. After MMF had unrestricted funding, its use for class III/IV LN induction increased and induction with high-dose CYC stopped, with rates of renal response unchanged.
Our cohort had proportions of Māori (25%) and pacific peoples (13%) above those of the population which is consistent with the higher incidence of SLE in these ethnicities. 8 ,19 Consistent with previous smaller studies in Aotearoa New Zealand we saw no statistically significant differences in eGFR and proteinuria at LN presentation or histopathological subtype between ethnicities (except class II, where numbers were small). This contrasts with variation in LN characteristics and severity between ethnicities reported internationally.20,21 It is important to note that our study was not designed to describe incidence of LN at a population level nor in different ethnic groups. Longitudinal nation-level data would be required to describe LN epidemiology and assess if there is variation in LN class between ethnicities.
Previous studies investigating LN class III/IV induction with CYC or MMF have shown 6-12 month CR in 30-66% and ORR in 65-88%. 22 Even acknowledging unavoidable bias and study comparability, our cohort CR proportion of 26% and ORR of 46% seem low. Proportion of individuals achieving renal response improve in the MMF-unrestricted funding period (CR 39%, ORR 58%) suggesting that the overall lower response rates may be in part due to suboptimal treatment when CYC was the only readily available induction option. Calcineurin and B-cell activating factor (BRAF) inhibition are now recommended in KDIGO and the American College of Rheumatology (ACR) guidelines as first line agents to be used alongside MMF and glucocorticoids for LN class III/IV.4,23 Currently the only subsidised first line calcineurin inhibitor for use in LN in Aotearoa New Zealand is cyclosporin, which has an inferior side effect profile and evidence base in LN compared to tacrolimus and voclosporin. Lack of licencing and subsidy means use of currently recommended first line agents is likely to be limited in Aotearoa New Zealand. Newer therapies mostly obviate the need for attendance at infusion centres which holds the potential for improve access in priority/geographically remote populations. We propose that improved access to guideline driven therapies has the potential to improve LN outcomes in Aotearoa.
We suggest removal to barriers for MMF prescribing, alongside the evidence for MMF efficacy for induction therapy, largely explain the 29% increase in MMF as induction therapy after October 2014. The reduced use of CYC after MMF funding became unrestricted is likely to have led to lower treatment-related toxicity for patients, although safety data were not collected in our study. After MMF became subsidised, CR rates more than doubled. The short and long term benefits of achieving response of any form, in particular CR, is well described and include improved renal outcomes and reduced mortality. 5 While there may be confounders to the improved LN response rates after easier access to MMF, such as improved supportive care, earlier diagnosis, changing population demographics and service provision, it would seem likely that ready access to MMF was a key influence. Overall, our data suggest Pharmac’s decision to apply a subsidy to MMF, with proven efficacy, favourable safety profile and less burdensome administration, improved LN outcomes.
KDIGO outline the importance of renal biopsy in identifying and classifying LN, to inform prognosis and select treatment. 4 Almost three-quarters of our cohort’s biopsies showed LN class III/IV with relatively high median NIH histopathological activity and chronicity Indices. This suggests that good clinical judgement was shown in selecting biopsy candidates. The use of hydroxychloroquine in just under two-thirds of patients at the time of biopsy probably reflects that 40% of patients had LN at presentation, LN being the organ involvement leading to SLE diagnosis. This finding is in keeping with other epidemiological data showing LN to be an early SLE disease manifestation, seen at diagnosis in approximately 31% of cases. 24
Despite relatively favourable rates of LN ORR/CR at last-observed follow up, over one third had no renal response, nine patients required dialysis or transplant and seven had died. This highlights the severe nature of LN and that longer term outcomes are poor. LN mortality rates vary by study population, with one large US-population based study reporting a 10 year mortality rate of 30% whereas others report as low as 4.3%.6,25–28 Our data show a fairly similar proportion of people with LN progressing to KDIGO CKD G5 (6.8%) as the 5.7% seen the ANZDATA analysis. 9 Our data emphasise the significant morbidity and mortality associated with LN and the need for early and effective management with a goal of early and sustained CR.
Median time to last clinic follow-up was just 7 years, which is relatively short-term follow up in a chronic disease occurring in young people. Our cohort’s median age of 38 highlights that LN, which has a high relapse rate, presents a large disease burden on both individuals and society. We did not collect data on treatment beyond induction, or incidence of relapse. These, longer term outcomes for LN across a larger patient cohort as well as cost-effectiveness of evidence based novel therapeutics could be foci for future studies.
Our study has a number of limitations. While our cohort came from two large hospitals in Aotearoa New Zealand over an 18-year period, our sample size of 100 is modest and is insufficient to report on outcomes by ethnicity. We did not report on use or dosing of glucocorticoids at induction. All induction regimens use concomitant glucocorticoids and variable dosing regimens likely influence outcomes, including treatment-related morbidity. Given the need for a large cohort for validity of detailed exploratory analysis, collecting and reporting glucocorticoid data was deemed inappropriate for this study. We did not report on co-morbidities, and given LN response is defined by eGFR and proteinuria our results may have been affected by common co-morbidities like diabetes and hypertension. Even though the rate of poor outcomes at last follow up was over one in three for those with class III/IV LN, it is possible that this proportion is higher due to incomplete follow up. Finally we must emphasize that retrospective studies like this are prone to bias and cannot be used to suggest rates of health outcomes at a population level.
Our study does add new and valuable data on LN outcomes in Aotearoa New Zealand and is the only study to our knowledge to examine how these have changed since the MMF became easier to prescribe for induction therapy. Furthermore, full ascertainment of cases via centralised laboratory databases is a strength of our study. We also have relatively few missing data across our 12 month follow up period.
Conclusion
Our study adds to the limited LN data in Aotearoa New Zealand and shows considerable unmet need for effective treatment. Over half of patients with LN class III/IV showed no early treatment response and over one third experienced poor outcomes over less than a decade of follow up. There was increased use of MMF for induction and associated improved outcomes after MMF was fully subsidised by Pharmac, highlighting the benefits of improved access to effective therapies for New Zealanders.
Ethical consideration
This study has ethical approval from the Health and Disability Ethics Committee (Ref 2024 EXP 19500) New Zealand as well as locality approval from Health New Zealand Capital Coast and Hutt Valley (Wellington) and Waikato, and underwent Māori consultation with Interim-Research Advisory Group Māori (Wellington) and Te Puna Oranga Māori Research Review Committee (Waikato).
Consent to participate
The application for ethical approval to Health and Disability Ethics Committee New Zealand requested that consent for access to health informed was waived based on practical and ethical reasons. The request for waiver was approved.
Consent for publication
Not applicable as no individual data presented.
Footnotes
Acknowledgments
The authors thank Dr Daniel Ninin and Dr Fouzia Ziad, pathologists, for assisting with case ascertainment.
Author contributions
RG and JB designed the overall study with DW, CH, DK, CC, and CC all contributing to practical aspects of study design. JB and DW acquired the data. JB, DW, CH and RG all contributed to data analysis. JB wrote the first draft of the manuscript and JB, DW, DK, CH, CC, CC and RG all critically revised it for intellectual content. JB, DW, DK, CH, CC, CC and RG all approved the final manuscript for publication and all agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data management plan approved by the Health and Disability Ethics Committee (Ref 2024 EXP 19500) New Zealand does not allow us to share our data in a repository or with other researchers. This is required as part of Māori data sovereignty.
