Abstract
Introduction
Lupus podocytopathy (LP) is an under-recognized pathological manifestation in patients with systemic lupus erythematosus (SLE). Despite being a distinct entity, current American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) guidelines do not include specific recommendations about LP, contributing to uncertainty regarding its diagnosis and treatment. This systematic review aims to synthesize the available literature on LP from case reports, case series and retrospective cohort studies to better characterize its clinical course, thereby informing clinical decision-making.
Material and methods
A systematic search of EMBASE and MEDLINE was conducted to identify relevant studies. Eligible studies included case reports, case series, and cohort studies reporting SLE patients who had biopsy-proven lupus podocytopathy without features of class III, IV, or V lupus nephritis. Demographic characteristics, clinical presentations, relevant laboratory and pathology results, treatment and outcomes were studied.
Results
This systematic review included 26 studies (18 case reports/small series and 8 cohorts), analyzing 19 individual cases and 240 cohort patients with LP. Most patients were young females, and LP was often part of the initial lupus manifestation with nephrotic-range proteinuria. Minimal change disease (MCD) was the predominant pathology. Patients with LP had an overall favorable outcome with treatment employing systemic steroid and steroid-sparing agents.
Conclusions
LP is an uncommon but distinct manifestation of SLE with overall favorable outcome with treatment using systemic steroid and steroid-sparing agents.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease characterized by autoantibody production and immune-complex deposition leading to multiorgan inflammation. 1 Kidney involvement in SLE, known as lupus nephritis (LN), is one of the most severe complications, affecting a large number of patients and significantly impacting prognosis. 2 Approximately 50% of patients with SLE develop LN, and up to 10% progress to end-stage renal disease (ESRD).3–5 LN typically presents with hematuria, proteinuria, and declining renal function, and is a major contributor to SLE morbidity and mortality. Immune-mediated renal injury in LN is usually diagnosed by classic biopsy findings of immune complex-mediated glomerulonephritis with a “full house” pattern on immunofluorescence (IgG, IgA, IgM, C3, and C1q). The Society of Nephrology/Renal Pathology Society (ISN/RPS) classification divides lupus nephritis into six classes based on the location and extent of immune complex deposition and glomerular injury. 6 Nephrotic syndrome in SLE is typically associated with focal, diffuse (Class III/IV) or membranous (Class V) LN. However, in rare cases, patients with SLE develops nephrotic syndrome without typical immune-complex deposition. Over the years, scattered reports have described SLE patients with nephrotic syndrome whose kidney biopsies reveal pathology resembling minimal change disease (MCD) and/or focal segmental glomerulosclerosis (FSGS). These observations prompt speculation into whether such pathology represents coincidental finding or a lupus-related mechanism. By the early 1980s, researchers began to identify these cases as a unique type of lupus-related kidney disease, distinct from common lupus nephritis. 7 This led to the concept of lupus podocytopathy (LP), a term now used to describe a unique renal pathology in SLE characterized clinically by nephrotic syndrome with pathological features resembling primary MCD or FSGS rather than classic immune-complex LN.8–10 In LP, light microscopy of the renal biopsy usually shows normal glomeruli, minimal mesangial proliferation, or FSGS lesions, typically without the endocapillary proliferation, necrosis, or crescents seen in active LN.9,11,12 Immunofluorescence is negative or shows only mesangial immune complex deposition, and no subendothelial or subepithelial immune complex deposition. Electronic microscopic examination reveals diffuse podocyte foot process effacement, similar to changes in MCD. 13 LP is relatively rare, accounting for roughly 1 to 2%13,14 of nephrotic patients with SLE. It seems to represent a non-immune-complex podocyte injury in the setting of SLE. The severe foot process effacement observed in LP suggests a mechanism similar to idiopathic podocytopathy (as seen in primary MCD/FSGS). It has been hypothesized that SLE-related immune dysregulation such as circulating permeability factors, cytokine dysregulation (notably interleukin-13 and other lymphokines), and T-cell dysfunction could trigger podocyte damage, mirroring the pathogenesis of idiopathic MCD.12,15–20 This contrasts with proliferative LN (classes III/IV) and membranous LN (class V), where immune complexes drive leukocyte infiltration, capillary wall damage, and complement-mediated injury.
Due to its rarity, diagnosing LP can be challenging and is often delayed or missed. Clinicians might incorrectly attribute severe proteinuria to more common proliferative or mambranous LN or to unrelated primary kidney diseases, delaying accurate diagnosis and appropriate care. To accurately diagnose LP, clinicians need to have a high suspicion and obtain a kidney biopsy with electron microscopy. A further challenge is that current official guidelines and classification systems did not officially include LP. For instance, the widely used ISN/RPS lupus nephritis classification and the ACR/EULAR SLE criteria include various types of LN but not lupus podocytopathy as a distinct class.6,21 Without clear guidelines, clinicians rely mainly on individual case reports and expert opinions, leading to uncertainty in how to best manage LP patients.
Current treatment approaches for LP primarily involve glucocorticoids, which often successfully reduce proteinuria22,23 and can lead to remission. However, relapses are common, and there is no standard recommendation on the use of other immunosuppressants, particularly for steroid-resistant or relapsing cases.22,24
Due to these gaps, a systematic review of existing literature on LP is essential. Consolidating clinical evidence from various reports will enhance awareness, improve diagnostic accuracy, and guide more effective treatment. Ultimately, a clearer understanding of LP can lead to its inclusion in official guidelines, ensuring better patient care and outcomes.
Material and methods
This systematic review was registered on PROSPERO with identifier CRD420251050644. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 25
Search strategy
Two reviewers (QW, BB) systematically searched electronic databases (MEDLINE and EMBASE) for articles published in English before May 12th, 2025, about lupus podocytopathy. The keywords and the search strategy used for the literature search are provided in Supplemental Table S1 and S2. Two reviewers (QW, BB) independently screened titles and abstracts of the articles identified in the literature search. Then, the reviewers retrieved the full texts and assessed for inclusion based on the eligibility criteria. A third reviewer (KS) reviewed the articles when there was uncertainty, and disagreements were resolved after discussion between the three reviewers.
Eligibility criteria
The inclusion criteria were 1 : Patients of any age or sex with a diagnosis of SLE 2 ; Kidney biopsy-confirmed lupus podocytopathy which was defined as MCD/FSGS on light microscopy with or without mesangial proliferation, with electron microscopy (EM) showing foot process effacement (FPE), and absence of proliferative features typical of lupus nephritis (LN). The exclusion criteria were 1 : Articles with patients not meeting ACR/EULAR or Systemic Lupus International Collaborating Clinics (SLICC) criteria for SLE 2 ; Renal biopsy with significant immune deposits in subepithelial or subendothelial regions 3 ; Studies in which EM was not performed; However, studies in which EM data were available for most patients but missing in a small minority were eligible for inclusion, provided that those cases met predefined substitute criteria 4 ; LP with co-existing LN class III, IV or V on the same biopsy; Cases classified as class II LN were included only if meeting LP criteria (EM with FPE, unless EM was unavailable) 5 ; Cases with no follow-up data 6 ; Review articles, animal studies, non-English articles. We included case reports, case series, and cohort studies fulfilling our criteria. Letters to editors were also included if fulfilled the criteria and reported information for the patients. Cohort studies would partially be excluded if some of the cases do not meet the criteria – for example, if some of the patients had pure LP and some had co-existing LV class III, IV or V, then only patients with pure LP would be included in this review.
Quality assessment
Two reviewers (QW, RS) independently reviewed each included study to assess the quality using Joanna Briggs Institute (JBI) tools. We used JBI case report checklists and JBI case series checklists 26 for the included case reports, case series and cohort studies. Since our study focused on the characteristics of the LP population without a comparator, we determined that the JBI checklist for cohort studies was not appropriate. Any disagreements that occurred during the assessment were resolved after mutual discussion.
Data extraction
Data extraction was performed by using Rayyan. 27 The reviewers (QW, RS, BB, KS, MJ) extracted data from the included studies into two separate standardized spreadsheets for the case reports and case series/cohort studies.
The following data were extracted for case reports: gender, age, history of prior LN, initial presentation, whether LP was part of the initial presentation, SLE history at the time of LP diagnosis, baseline kidney function, prior SLE treatment, extra-renal features, laboratory test results, renal biopsy findings, treatment, response, relapse, and outcomes.
The following data were extracted for case series and cohort studies: total patients number, age (mean or range), percentage of female patients, percentage of nephrotic syndrome, laboratory tests, history of prior LN, renal biopsy findings, treatment, response, relapse rate, and outcomes.
Data analysis
Data were summarized descriptively. Meta-analysis was not performed. Relevant clinical information was summarized in tables. To address variations in proteinuria from different studies, we integrated both 24-h urine protein measurements and spot urine protein creatinine ratio (UPCR) values with standardized conversion to gram/day, in order to estimate a weighted average daily proteinuria.
For case reports, data from pediatric and adult patients’ data were analyzed separately. For cohort studies, data from pediatric and adult populations were not analyzed separately as only one cohort consisted solely of pediatric patients, while two other cohorts included a mix of both age groups. We used Rstudio to calculate p-values for pooled data when necessary. Categorical variables, were compared using Fisher’s exact test in this study.
To address variations in proteinuria from different studies, we integrated both 24-h urine protein measurements and spot urine protein creatinine ratio (UPCR) values with standadized conversion to gram/day, in order to estimate weighted average daily proteinuria.
Results
Data selection
Our search protocol identified 307 records. One additional study was identified through manual searching based on its thematic connection to a previously included article. After removing duplicates, 223 records remained. We excluded 142 records based on title and abstract review. The full texts of 35 articles were assessed for eligibility. Among these reports, 3 studies were reviews of a few cases lacking sufficient detail on LP, 3 lacked treatment or renal outcome data, 1 was published in a non-English language, 1 did not meet the SLE classification criteria, 1 described dense deposits in the capillary wall suggestive of subendothelial or subepithelial pathology rather than LP, and 1 was a duplicate study of the same cohort focusing on different aspects. Ultimately, 26 studies met the inclusion criteria and were included in the final systematic review.11,12,15,24,28–49 Among these 26 studies, 18 were case reports, letter to editor with sufficient data, or case series with 2 cases, 8 studies were retrospective cohort studies or case series of more than 5 cases with aggregated data.
The steps of literature review and selection was generated by PRISMA Flow Diagram
50
and is shown in Figure 1. PRISMA flow diagram for literature review and selection process.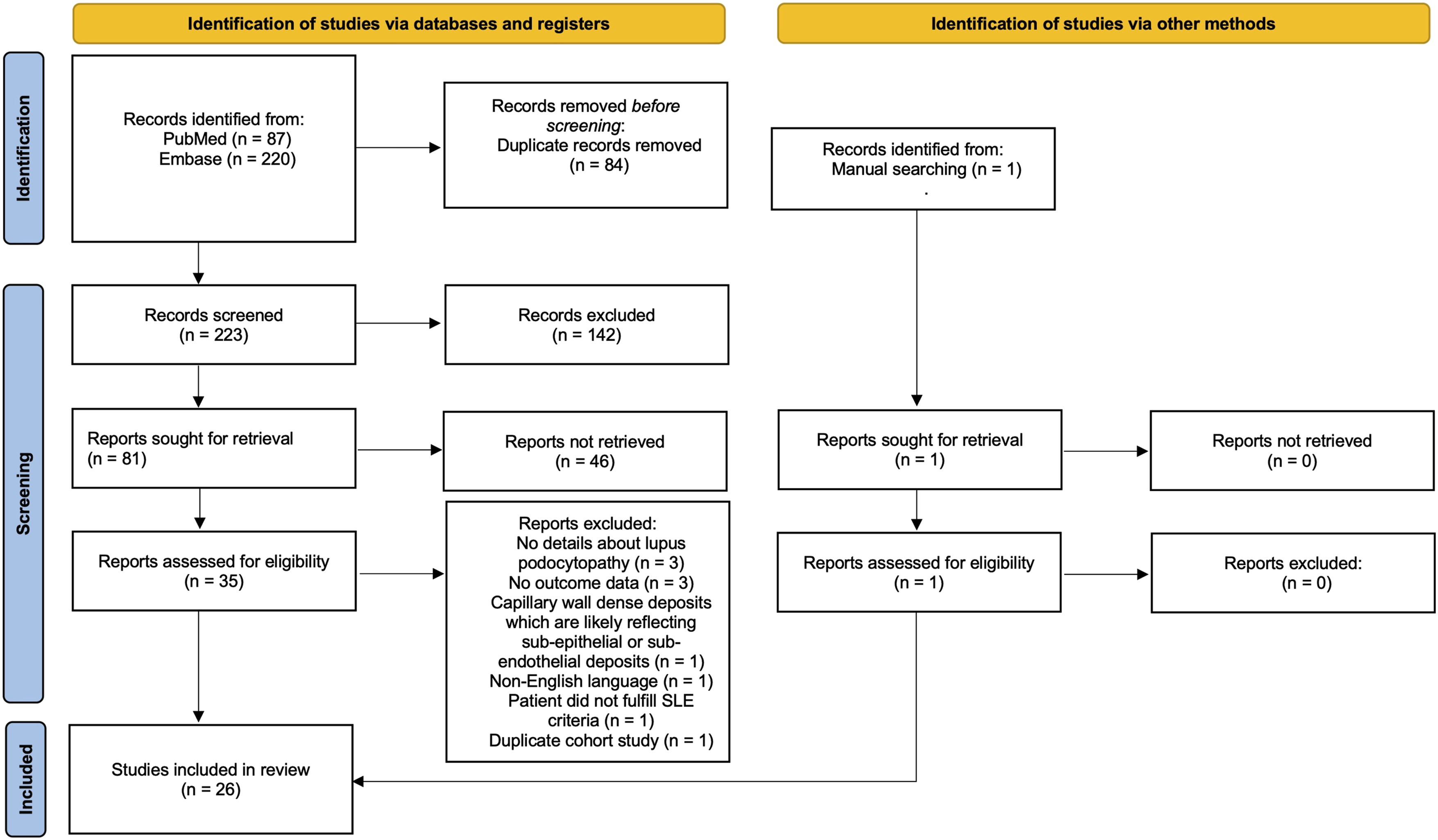
Case reports
A total of 19 patients were identified from case reports and small case series. The median age was 23 years (range 3–46) and 84% were female. Six out of 19 were pediatric patients and 13 were adult patients. Baseline characteristics are summarized in Supplemental Table S3.
Presentation
Baseline characteristics, clinical manifestations, diagnostic findings, management, and outcomes of lupus podocytopathy in individual case reports, stratified by age groups.
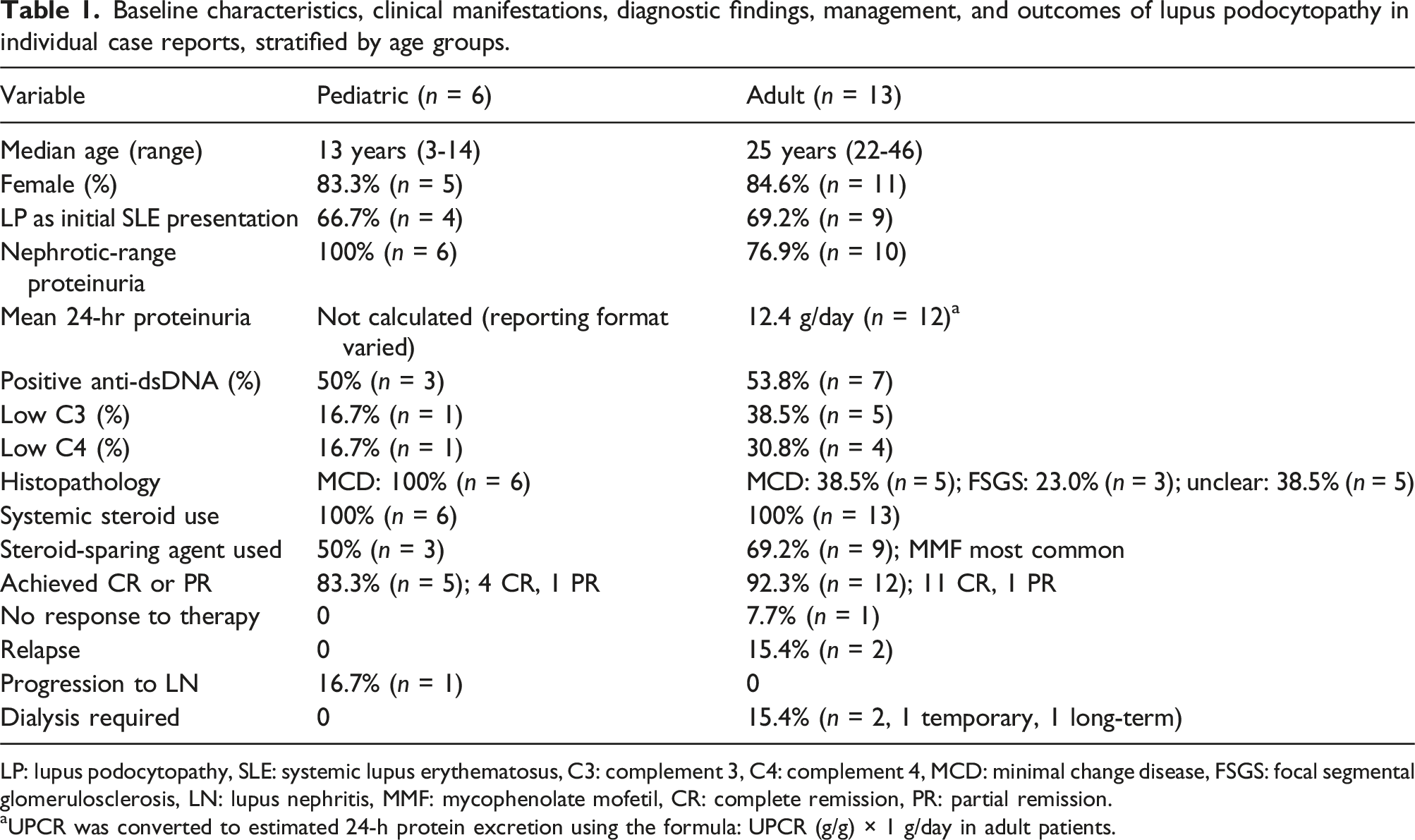
LP: lupus podocytopathy, SLE: systemic lupus erythematosus, C3: complement 3, C4: complement 4, MCD: minimal change disease, FSGS: focal segmental glomerulosclerosis, LN: lupus nephritis, MMF: mycophenolate mofetil, CR: complete remission, PR: partial remission.
aUPCR was converted to estimated 24-h protein excretion using the formula: UPCR (g/g) × 1 g/day in adult patients.
Among the 6 pediatric patients, 66.7% (n = 4) patients had LP as the initial presentation of lupus. The other 2 patients had LP 3 and 7 years after diagnosis of lupus respectively. All patients had nephrotic range proteinuria. DsDNA was positive in 50% (n = 3) patients, 2 had negative dsDNA and 1 was not mentioned. The mean 24-h urine protein was not calculated as some reports used spot urine protein creatinine ratio (UPCR) and some used gram per day. Low C3 was observed in 16.7% (n = 1) patients and low C4 was observed in 16.7% (n = 1) patients.
Among the 13 adult patients, 69.2% (n = 9) patients had LP as the initial presentation of lupus. Of the remaining 4 patients, one developed LP 10 years after a diagnosis of SLE, one after 6 months, and another after 3 years.36,39,42 The fourth patient had a history of discoid lupus diagnosed 10 years earlier and developed features suggestive of SLE during follow-up, although a formal diagnosis was not established. 45 DsDNA was positive in 53.8% (n = 7) of the patients. Low C3 and C4 were observed in 38.5% (n = 5) and 30.8% (n = 4) of the patients, respectively (Table 1). Mean 24-h proteinuria was 11.58 g. UPCR and 24-h urine protein data were appropriately converted and pooled for analysis.
Pathology
Histopathologic patterns were consistent with MCD or MCD-like in 57.9% of the patients, FSGS in 15.8%, and unclear or unspecified patterns in 26.3% due to lack of detail or ambiguous descriptions in biopsy reports (Table 1).
In the pediatric population, all patients (n = 6) had MCD pathology, whereas in the adult population, only 38% (n = 5) had MCD.
Treatment and outcome
All patients received systemic steroids. A total of 69% (n = 13) were treated with at least one steroid-sparing agent, excluding those who had received such agents prior to the diagnosis of LP, among which 61% (n = 8) received mycophenolate.
In the pediatric population, all patients (n = 6) received systemic steroids, and 50% (n = 3) received at least one steroid-sparing agent in the initial presentation. 83.3% (n = 5) of the patients achieved complete or partial remission, among which 66.7% (n = 4) achieved complete remission (CR) and 16.7% (n = 1) achieved partial remission (PR) while 1 patient was marked as “clinical stable” without enough details to specify. Time to remission varied among patients and is summarized in Table S3. None of the 6 patients experienced clinical relapse. One patient developed into LN class IV after 2 years which required systemic steroid and mycophenolate mofetil and the histology of this patient was MCD. No patients ended up requiring dialysis (Table 1).
In the adult population, all patients (n = 13) received systemic steroids, 69.2% (n = 9) received and at least one steroid-sparing agent in the initial presentation. 92.3% (n = 12) of the patients achieved CR or PR, among which 84.6% (n = 11) patients achieved CR and 7.7% (n = 1) achieved PR while 7.7% (n = 1) had no response. Time to remission was summarized in Table S3. Relapse occurred in 15.4% (n = 2) patients. 15.4% (n = 2) required dialysis, one temporary and one long-term (Table 1).
For both pediatric and adult patients, the time course of response to therapy, when available, was relatively rapid, with most patients experiencing CR or PR within several weeks up to 3 to 5 months. Unfortunately, the length of remission is difficult to assess, since duration of follow up was only up to 1 year in most reported patients (Table S3).
Cohort studies
Eight cohort studies were included. We excluded patients with biopsy findings consistent with ISN/RPS class III, IV, or V lupus nephritis at the time of LP diagnosis. For some cohorts, only a subset of patients met the inclusion criteria for this systematic review, and these data were subsequently re-aggregated. Nearly all the patients had EM demonstrating FPE. In one cohort study, EM was not available for a small number of patients (n = 9). In these cases, the presence of mesangial immune deposits on immunofluorescence, and the presence of nephrotic syndrome were used as a substitute. 30
Altogether 240 patients were included. The median age was at 29 with 87.7% females from 209 patients of 7 studies. One cohort did not specify age and gender in podocytopathy group.
Among the 8 cohorts, 1 consisted only of pediatric population, 5 included only adult patients, and the other 2 cohorts had a combination of adult and pediatric patients. Baseline characteristics are summarized in Supplemental Table S4.
Presentation
Baseline characteristics, clinical manifestations, diagnostic findings, management, and outcomes of lupus podocytopathy in retrospective cohort studies.

MCD: Minimal change disease, FSGS: focal segmental glomerulosclerosis, MsP: mesangial proliferation, LN: lupus nephritis, MMF: Mycophenolate Mofetil, CYC: cyclophosphamide, AZA: azathioprine, CNI: Calcineurin Inhibitor, CR: complete remission, PR: partial remission, ESRD: end-stage renal disease, HD: hemodialysis.
Pathology
Among these 240 cases, MCD-like lesions were reported in 42.5% (n = 102), FSGS in 17.9% (n = 43), and mesangial proliferation in 37.9% (n = 91) of the patients. Eleven patients were reported as having LN class I (Table 2). Notably, the included studies lacked uniformity in histologic classification. While some referred to findings as lupus nephritis (LN) class II, others used the broader term mesangial proliferation (MsP) to describe similar features. Certain studies categorized LN class I as a distinct subtype, whereas others interpreted such findings as minimal change disease (MCD) or MCD-like pathology. Additionally, while some studies considered MCD and MsP as separate histologic patterns, others suggested that they may coexist within the same biopsy specimen. These overlapping and inconsistently defined classifications were often not clearly described in the original reports. Moreover, in the study by Wang et al. 46 only LP patients with mesangial proliferative (MsP) patterns were included, while those with other histologic subtypes such as FSGS were excluded. As a result, the overall proportions summarized in this review may not accurately reflect the true distribution of histologic patterns in the overall LP population.
Treatment and outcomes
All studies reported glucocorticoid therapy as the mainstay. 98.8% (n = 237) of the patients received different kinds of systemic steroids. Steroid sparing agents include mycophenolate mofetil (MMF), cyclophosphamide (CYC), azathioprine (AZA), cyclosporine, tacrolimus, and rituximab. We were not able to aggregate data regarding the percentage of patients who received steroid sparing agents for their LP due to the heterogeneity of the studies. Details of the use of steroid-sparing agents for each study were summarized in Supplemental Table S4.
Among the 240 patients, 94.6% (n = 227) achieved complete or partial response and 5.4% (n = 13) patients had no response. Four studies reported relapse rates. Among the 208 patients, 52.9% (n = 110) patients experienced relapse during the follow-up time period. Four out of eight studies reported remission rates in MCD and FSGS separately and among these patients, 33 out of 39 FSGS patients achieved CR or PR with remission rate of 84.6%, while 91 out of 92 MCD patients achieved CR or PR with 98.9% remission rate (p = .003) (Table 2).
Among these 240 patients, 20 patients developed into LN. Three patients developed into end stage renal disease (ESRD)/advanced renal failure/hemodialysis (HD) dependent eventually.
Discussion
This systematic review summarized data from case reports, case series and retrospective cohort studies focusing on LP. Among the 19 patients from case reports, more than half had LP as part of the initial presentation of SLE and most of them had a favorable outcome with only one patient developing LN and two requiring dialysis. Compared with LN, LP appears to have a more benign course and outcome.
Proposed criteria for diagnosis of lupus podocytopathy.
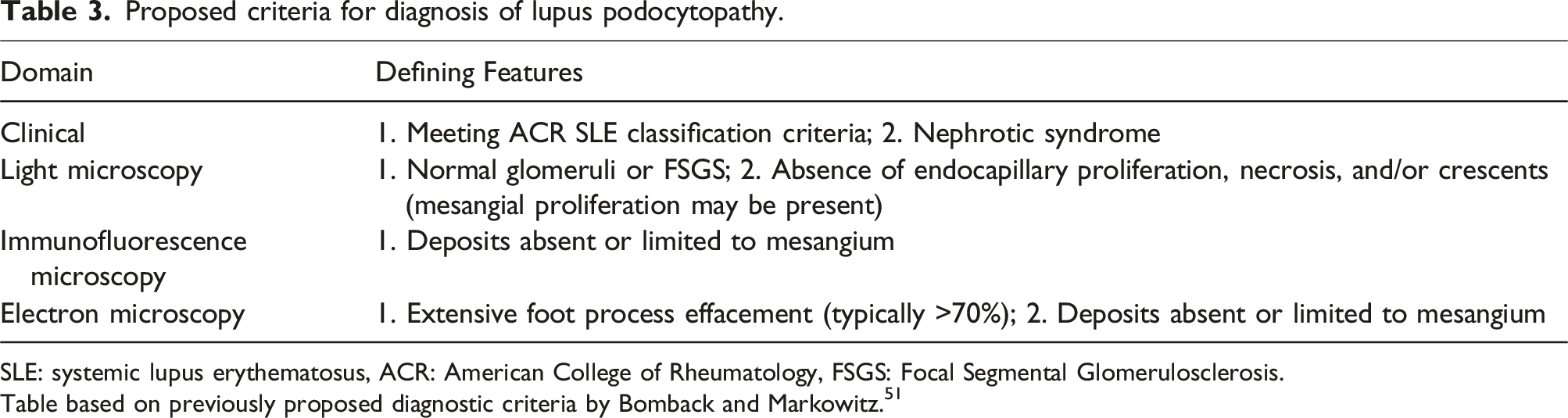
SLE: systemic lupus erythematosus, ACR: American College of Rheumatology, FSGS: Focal Segmental Glomerulosclerosis.
Table based on previously proposed diagnostic criteria by Bomback and Markowitz. 51
Podocytes are specialized epithelial cells that serve as a critical filtration barrier in glomeruli. 52 When injured, they lose their actin cytoskeleton structure and undergo effacement of foot processes which leads to filtration impairment and result in significant proteinuria. Key proteins such as nephrin anchor the cytoskeleton to the slit diaphragm and maintain podocyte shape and adhesion. Pathways including nephrin phosphorylation and actin polymerization have been shown to be central to podocyte motility and injury response. 53 Wiggins 54 proposed the podocyte depletion hypothesis in 2007, suggesting that the outcome of glomerular injury depends on the extent of podocyte depletion, regardless of the nature of the initial insult (immune, toxic, infectious, ischemic, or otherwise).
Meliambro et al. 55 summarized different mechanisms of podocyte injury, including genetic causes; circulating factors such as nephrin autoantibodies; immune-mediated pathways including tumor necrosis factor (TNF) signaling, inflammatory cytokines (e.g., interleukins), and complement activation; as well as mechanical and metabolic stress and viral infections. More specifically, the study by Bruggeman et al. 56 identified tumor necrosis factor receptor 2 (TNFR2) expression on podocytes and its association of podocyte and kidney injury in mice. Complement activation has been shown to play a role in podocyte injury by activating cytokines, proteases, oxidants, altering cytoskeleton and slit diaphragm proteins. 57
Watts et al. (2022) 12 identified autoantibodies against nephrin, which is a component of the podocyte slit diaphragm in approximately 30% of patients with MCD. These antibodies were present during active disease and diminished with remission. Renal biopsies also showed punctate IgG deposits colocalizing with nephrin, supporting an autoimmune mechanism of MCD. Hengel et al. 58 later confirmed that anti-nephrin autoantibodies can cause actin and cytoskeletal alterations via phosphorylation of nephrin, which suggested the direct pathogenicity of these autoantibodies. Notably, in their study, 44% of MCD patients and 9% of primary FSGS patients had antinephrin autoantibodies. No patient with LN was found to have anti-nephrin autoantibodies. However, Chugh and Clement 59 proposed a different perspective, as these antibodies have not induced foot process effacement in animal models. They argued that antinephrin autoantibodies may reflect a secondary immune response to podocyte injury rather than a primary cause. Epitope mapping would be needed to prove this concept.
SLE is a systemic autoimmune disorder associated with dysregulation of both the innate immune system and the adaptive immune system with autoantibody production and cytokine activation. 60 Previous studies have shown that the podocytopathy rate was higher in lupus population compared to general population, 17 suggesting a potential pathophysiological link between lupus-related immune dysregulation and podocyte injury. 17 However, the exact mechanism remains unclear.
We propose several potential mechanisms linking SLE to podocyte injury. First, SLE has been shown to increase the production of reactive oxygen species (ROS), 61 which can contribute to downstream podocyte stress and damage. 62 Second, complement activation, a known feature of SLE, 60 has also been implicated in podocyte injury. 57 Finally, the presence of diverse autoantibodies and proinflammatory cytokines in SLE may interact with podocyte surface receptors, triggering signaling cascades that lead to further cellular injury. 62
The predominant renal manifestation of LP is nephrotic range proteinuria. In this review, almost all patients presented with nephrotic range proteinuria with average 24-h protein of 6.2 g/day. In the study by Wang et al., 46 patients with podocytopathy had significantly higher proteinuria than those without, suggesting that proteinuria severity is linked to the degree of podocyte effacement—a hallmark of LP. They also commented that podocytopathy is more likely to be the initial lupus presentation than LN without podocytopathy, which aligns with the findings of our systematic review.
In our study, patients with LP overall showed a favorable response to steroids and steroid sparing agents, with a high remission rate. Notably, the FSGS subtypes tended to have a lower remission rate compared to the MCD subtypes. While there are no current guidelines for the treatment of LP, the studies involved have used systemic steroid, and steroid-sparing agents including mycophenolate, cyclophosphamide, azathioprine, cyclosporin, tacrolimus, and rituximab with good response.
Additionally, more than half of the patients experienced at least one relapse of LP requiring treatment. Relapse data were reported in 4 of the 8 included studies. In the study of Bonelli et al. 30 the researchers analyzed therapies at the time of diagnosis and did not identify any specific agent associated with a reduced risk of relapse. In another study, Xia et al. 24 was able to identify acute kidney injury (AKI) as an independent risk factor for kidney relapse. Overall, the available data were insufficient to draw meaningful conclusions regarding the impact of specific therapies on relapse prevention because most studies did not report outcomes explicitly for these agents.
Nevertheless, emerging studies have demonstrated that certain immunosuppressive agents may have protective effects on podocytes, thereby reducing proteinuria. Calcineurin Inhibitors (CNI) have been demonstrated to block the phosphatase activity of calcineurin, which dephosphorylates synaptopodin, and thus maintain the stability of the actin cytoskeleton in podocytes and reduce proteinuria.63–65 Among these agents, volclosporin has a more sustainable profile with no requirement to monitor blood levels. 65 Additionally, MMF has also been shown to have a direct effect on the transcriptome of podocytes via non-immunologic pathways, potentially reducing proteinuria. 66 However, whether these agents can reduce relapse rate warrants further clinical investigation.
Futher more, the review by Meliambro et al. 55 pointed out several emerging targets and interventions for podocytopathies, although not specifically for LP. These include Apolipoprotein L (APOL)-targeted therapies (Inaxaplin), JAK–STAT pathway inhibitors (Baricitinib), endothelin receptor antagonists (Sparsentan), TRPC channel inhibitors, complement inhibitors and mitochondrial-targeted therapies.
There are several limitations of this systematic review. First, there is inconsistency in the definition of lupus podocytopathy (LP). While LP is generally characterized by proteinuria with significant podocyte foot process effacement in the absence of endocapillary proliferation or substantial subendothelial or subepithelial immune complex deposits, there is no universally accepted cutoff for the degree of foot process effacement required for diagnosis. As a result, although most patients included had diffuse FPE, the extent varied. In a small subset of patients from a single study, patients with any degree of PFE were included. While this may reflect real-world reporting, it introduced variability into the study. Second, while mesangial proliferation and mesangial immune complex deposition are typically considered acceptable within the spectrum of LP, some studies also included cases with mild subepithelial deposits, further contributing to variability in histologic definitions. Third, as mentioned above, studies lacked uniformity in histologic classification and there were overlaps between different subtypes. Fourth, one study specifically included masengial proliferative LP which makes the proportion of each subtype not represent the real population. Fifth, although nearly all patients had EM confirmed FPE, there was a small subset of patients (n = 9) whose EM was not available and the study used substitute clinical criteria, which may introduce potential misclassification. Sixth, two of the studies mixed pediatric and adult populations, which may have introduced heterogeneity due to age-related differences in disease presentation, treatment response, and histologic patterns. Seventh, in some studies, the use of steroid-sparing agents was not clearly reported. For instance, while the authors listed the number of patients who received each specific agent, it was unclear whether these treatments overlapped in individual patients. As a result, the total number of patients who received any steroid-sparing agent could not be reliably determined. Eighth, while some studies defined complete and partial remission according to established criteria, for example, proteinuria <0.5 g/day with near-normal GFR for complete remission; and ≥50% reduction in proteinuria to <3 g/day for partial remission, many did not specify the criteria used or used different criterias. Ninth, while some studies specified whether or not patients were off steroid-sparing agents during and after remission, others provided less detailed information.
Conclusions
LP is a distinct and under-recognized renal manifestation of SLE. It is characterized clinically by nephrotic-range proteinuria and pathologically by podocyte effacement in the absence of proliferative lupus nephritis features. Our systematic review, which included published case reports, case series and retrospective cohort studies, demonstrated that LP tends to be the initial presentation of SLE and generally responds well to treatment with systemic corticosteroids and steroid-sparing agents.
Patients with MCD pathology tend to achieve higher remission rates compared to those with FSGS. Although relapse is common, only a small proportion of patients progress to classical LN or ESRD requiring dialysis.
The pathogenesis of LP remains poorly understood, but podocyte injury and effacement appear to be driven by immunologic pathways, inflammatory cytokines, and complement activation. While current treatment strategies remains unclear, overall patients responded to systemic steroid and steroid-sparing agents. There are emerging therapies targeting podocytes for podocytopathies, although not specifically for LP.
Given the difference in clinical presentation and outcomes between LP and traditional LN, early histological diagnosis through kidney biopsy is essential to guide appropriate treatment strategies.
Supplemental material
Supplemental material - Lupus podocytopathy: A systematic review of clinical evidence from cases and cohorts
Supplemental material for Lupus podocytopathy: A systematic review of clinical evidence from cases and cohorts by Qi Wang, Bismah Basharat, Kefang Sun, Roshan Subedi, Massiel Jimenez Artiles and Stanley Ballou in Lupus
Footnotes
Author contributions
Qi Wang designed the search strategy, performed literature screening, quality assessment, data extraction, and wrote the original draft of the manuscript. Bismah Basharat contributed to literature screening and data extraction, and participated in manuscript review and editing. Kefang Sun assisted with literature screening and data extraction, drafted the introduction section, and contributed to manuscript review and editing. Roshan Subedi contributed to quality assessment and data extraction, and participated in manuscript review and editing. Massiel Jimenez Artiles participated in data extraction and manuscript review and editing. Stanley Ballou provided senior supervision and critical revision of the manuscript. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
This study is based on previously published literature and does not involve any new studies with human participants or animals performed by any of the authors.
We confirm that the manuscript is original, has not been submitted elsewhere, and complies with the journal’s guidelines. All authors meet authorship criteria.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
