Abstract
Background
Healthcare disparities in SLE randomized clinical trials (RCTs) are well known, with minoritized persons being under-represented as trial participants despite facing more significant SLE morbidity and mortality. There is currently limited information as to how to best recruit a diverse group of participants to SLE clinical trials. Barriers to clinical trial participation can originate from either the patients or their clinicians. We therefore sought to investigate the effectiveness of educational curriculum in decreasing such barriers.
Methods
We utilized materials from the American College of Rheumatology, the Lupus Research Alliance, and the Lupus Foundation to develop educational curriculums for both clinicians and patients to see if we could improve their knowledge, attitudes, and skills regarding clinical trial participation.
Results
Attitudes towards clinical trials were highly favorable among patients and clinicians prior to the workshop and remained positive afterwards. The workshops increased the patients’ comfort level regarding clinical trial participation, and increased clinician comfort with the clinical trial process and how to refer patients to clinical trials. Regarding knowledge about clinical trials, the results were mixed, with certain knowledge aspects such as the protections afforded to patients in RCTs and SLE disparities improving, but knowledge about the more logistical and regulatory aspects of RCTs not significantly improving. However, these improvements did not translate into increasing patient enrollment into RCTs or research registries.
Conclusion
Although our study did increase both patients’ and clinicians’ skill and comfort level regarding RCT participation, its small size did not allow us to observe concrete gains in RCT enrollment.
Introduction
SLE is currently an active area of drug development, with over 50 drugs and targets having been evaluated in the last 13 years, 1 leading to the recent FDA approval of anifrolumab and voclosporin in 2021. 2 Despite these advances, significant disparities exist in SLE outcomes, particularly among patients of color who experience more severe disease. 3
To assure greatest validity, clinical trials should include participants whose demography reflects the population with the disease. 51% of participants in SLE RCT were White, while comprising only 33% of patients with SLE. 14% of SLE RCT participants were Black while comprising 43% of people with SLE. 3 Limited diversity in RCT participants decreases understanding of new therapeutics. For example, physicians were urged to use caution when prescribing belimumab to Black patients due to lack of representation in the RCTs. 4 Further studies proved the drug’s efficacy for Black patients 5 but this led to their delayed access to the drug. 6
RCT participation is reduced by concerns about placebo, strict study inclusion and exclusion criteria, complex protocols, and concern for an SLE flare and logistical challenges like lack of transportation, lack of health insurance, and frequent blood draws and office visits. 3 Motivating factors include altruism, physician recommendation, culturally sensitive marketing, access to high quality healthcare, increased disease knowledge, and hope for SLE improvement.7,8 Solutions to provide patient support throughout the study have been proposed but evidenced-based best practices for patient recruitment remain lacking. 9
Patient, physician, and patient-physician relational factors individually or collectively influence clinical trial participation. One key motivating factor is a trusting physician-patient relationship.10–12 Unfortunately, patients of color report mistrust of the medical community.13,14 Referring clinicians and research staff viewed minority participants as less-promising candidates, with some withholding RCT opportunities. 15 Discordance between physician and patient attitudes towards risk, benefits, and patient understanding regarding RCTs can exacerbate disparities. 16 Other barriers include lack of human and technical resources, perceived lack of patient interest, lack of trial awareness, restrictive eligibility criteria, and documentation burden.17–21 Adequate reimbursement to build the infrastructure could help. 22
Critically, prior research indicates a critical role for educational interventions in improving clinical trial participation. In a survey of the Lupus Clinical Investigators Network (LuCIN) members, 49.1% of respondents reported no prior training in recruiting diverse participants. 23 One solution targeting clinicians is the MIMICT (Materials to Increase Minority Involvement in Clinical Trials) from the American College of Rheumatology. 24 Patient educational initiatives have also been studied, such as Lupus Therapeutics’ Patient Advocates for Lupus Studies (PALS) peer education program. 25 Both were shown to be successful.
There is currently limited research in rheumatology field on the impact of didactic curriculums on educating patients and their clinicians about RCTs and the importance of diversity in clinical research. Our study addresses this knowledge gap by assessing the effectiveness of educational initiatives in shaping knowledge, skills, and attitudes regarding clinical trial participation and recruitment. Our goal is to establish a rapid, low cost, and easily implementable intervention to improve the recruitment of underrepresented groups into SLE clinical trials.
Methods
For this study, 589 patients were contacted via email from a database of SLE patients who attend the rheumatology clinics at UT Southwestern Medical Center. 800 patients from Parkland Memorial Hospital were contacted via letter, and 235 patients were called 5 days later. The methods across both institutions differed due to institution-specific patient recruitment rules. Both English and Spanish-speaking patients were contacted, utilizing hospital interpreters when necessary. We captured the demographic information from the medical records of the 824 patients contacted. A total of 45 patients and family members (non-clinician participants) participated in the workshops, which were held both virtually and in person. These workshops educated non-clinician participants about participating in a clinical trial and measured their knowledge and attitudes toward research studies. Three workshops were held at UT Southwestern during November and December 2022 (2 in person and 1 virtual). Four workshops were held throughout March and April 2023 at Parkland (2 in person (one in English, one in Spanish), and 2 virtual (one in English, one in Spanish)).
Simultaneously, rheumatology clinicians were approached during UTSW Grand Rounds to attend similar educational workshops and complete pre-intervention and post-intervention surveys. 32 clinicians, ranging from nursing staff to advanced practice providers to physicians, were recruited to the study. Clinicians from both Parkland and UT Southwestern were in attendance.
Ethical considerations
The institutional review board at UT Southwestern Medical Center approved the study. Non-clinician participants were assured both during the initial recruitment process and the workshop that their private health information and survey data would be kept confidential and would not affect their employment or clinical care.
Non-clinician participant curriculum
For non-clinician participants, we utilized materials from the Lupus Research Alliance and the Lupus Foundation to create a didactic curriculum. This curriculum was based on written patient education materials from the Lupus Foundation and the Lupus Research Alliance PALS peer education program. We adapted these materials into a didactic patient-centered curriculum lasting 60 minutes with an additional 30 minutes allotted for discussion. We then added additional material aimed at eliminating common clinical trial misconceptions. These materials are available on request. In addition to dispelling misconceptions, participants learned about the clinical trial process, protections afforded to patients in clinical trials, and the importance of these trials to the care of patients with SLE. The curriculum also educated non-clinician participants about their SLE diagnosis and treatment, the causes of SLE, and self-management techniques. The workshops were facilitated by Black and Hispanic clinicians. These workshops were conducted in English and Spanish, with in-person and virtual options. Each participant attended one workshop session.
Clinician curriculum
We utilized the online Materials from the American College of Rheumatology to Increase Minority Involvement in Clinical Trials (MIMICT) (https://thelupusinitiative.org/mimict-2/providers/online-education-course/) as the basis for our project. We condensed the four modules into one 90-min workshop and edited them to fit our needs. The modules provided practical information about the clinical trial process, how to speak with patients about clinical trials, and how to make a referral. Participants also learned about disparities in clinical trial participation, barriers to referring underrepresented participants to clinical trials, protections afforded to clinical trial participants, and a brief overview of the history of clinical trials in the US. The added material provided information about local clinical trials and added more historical context to the barriers faced by underrepresented groups regarding clinical trial participation. The subsequent materials were delivered as part of our departmental grand rounds in a hybrid format.
Survey design and administration
For both clinical and non-clinician participants, we administered a survey before the workshop, immediately after the workshop, and then an optional survey 1 month later. The 1-month survey was made optional to decrease participant burden. In-person participants filled out the first 2 surveys on paper, and the 1-month follow-up was administered online via REDCap.26,27 Records from paper surveys were manually added to the REDCap database. Virtual participants completed all 3 surveys on REDCap. The survey was designed to measure three domains: knowledge, attitudes, and skills regarding clinical trial participation. The surveys contained a mixture of Likert scale, multiple choice and free entry questions.
The knowledge questions for non-clinician participants assessed how knowledgeable they were regarding the clinical trial process and the protections afforded to clinical research participants. The skills questions assessed non-clinician participants’ comfort level regarding clinical trial participation and what sources they would trust for information about clinical trials. The attitude questions evaluated how participants viewed the importance of clinical trials in the care of patients with SLE, the risks and benefits of clinical trial participation, and the effectiveness of current protections in place.
For the clinician survey, the knowledge questions assessed how knowledgeable they were regarding clinical trial disparities, enrollment barriers, and protections afforded to research participants. The skills questions evaluated the clinicians’ comfort level regarding clinical trial referrals, and their self-assessed familiarity with the clinical trial process and disparities in clinical trial enrollment. The attitude questions assessed how clinicians viewed the importance of clinical trials in the care of patients with SLE and what barriers they faced when referring patients to clinical trials.
The follow-up surveys for both groups directly judged changes in attitude regarding clinical trial participation after the workshop. The pre-intervention survey also captured demographic information and information about their prior experience with clinical trials participation. The surveys were designed specifically for our study and were reviewed by experts in mixed-methods research but were not formally validated. Overall, missing survey data was 1% for clinician and 1.4% for non-clinician participants, respectively.
Statistical analysis
Mean values of participant responses as well as differences between individual pre- and both the immediate and 1-month post-intervention responses were calculated. SAS was used to calculate the p-value using the Wilcoxon signed-rank test to accommodate the observed skew of the data. Due to the poor response rate on the 1-month follow-up surveys, we were unable to calculate p values to determine the statistical significance of the noted changes from the initial survey.
The knowledge questions were then scored as correct or incorrect. These ordinate variables were then the p values were calculated using the McNemar’s test.
Results
Participant demographics
Of the 824 patients who were contacted, 42% identified as Black, 43% as White, 5% as Asian, 3% as other, and 8% unknown. 26% identified as Hispanic, 70% as non-Hispanic, and 4% unknown. 92% were female and 8% were male.
Non-clinician participant demographics.

Clinician participant demographics.
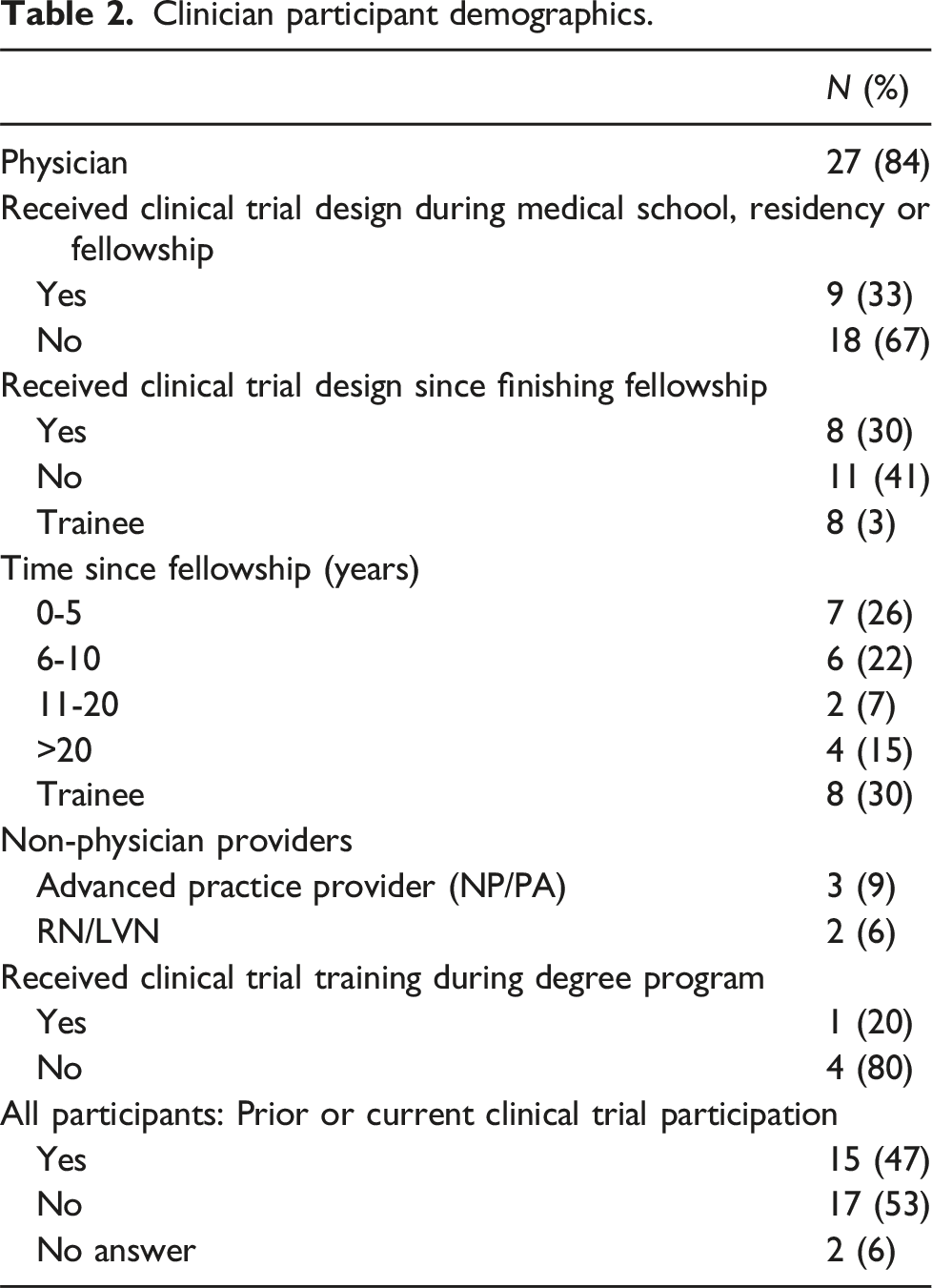
Non-clinician participant survey results
Skills
Non-clinician participants were asked before and after the workshop about the factors that would increase their comfort level with participating in a clinical trial. Multiple answers were allowed. Both pre- and post-workshop, the most chosen reason was if “my rheumatologist recommended the study,” with 69% participants answering this before the workshop and 47% after the workshop. The second most common reason pre-workshop was if “my primary care doctor recommended the study”, while the second most common reason post-workshop was if “my family supported the decision” (answered by 36% and 24% of participants respectively). Additionally, at the 1-month follow-up, participants were queried about the sources whom they trust for information about clinical trials. Again, multiple answers were allowed to be selected. The most chosen options were “other lupus patients” and “social media,” answered by 16% and 7% of participants respectively.
Attitudes
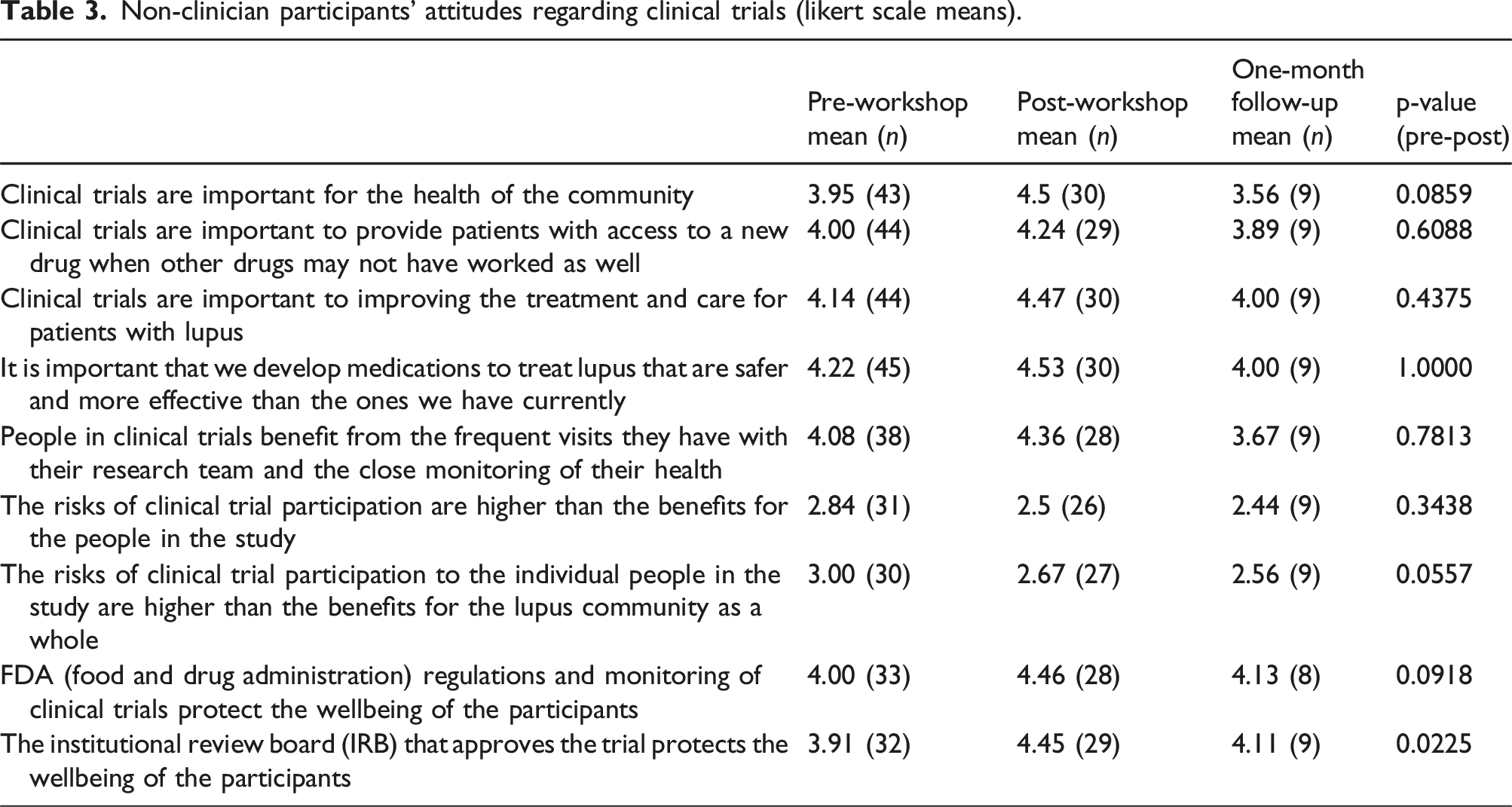
Non-clinician participants' attitudes regarding clinical trials (likert scale means).
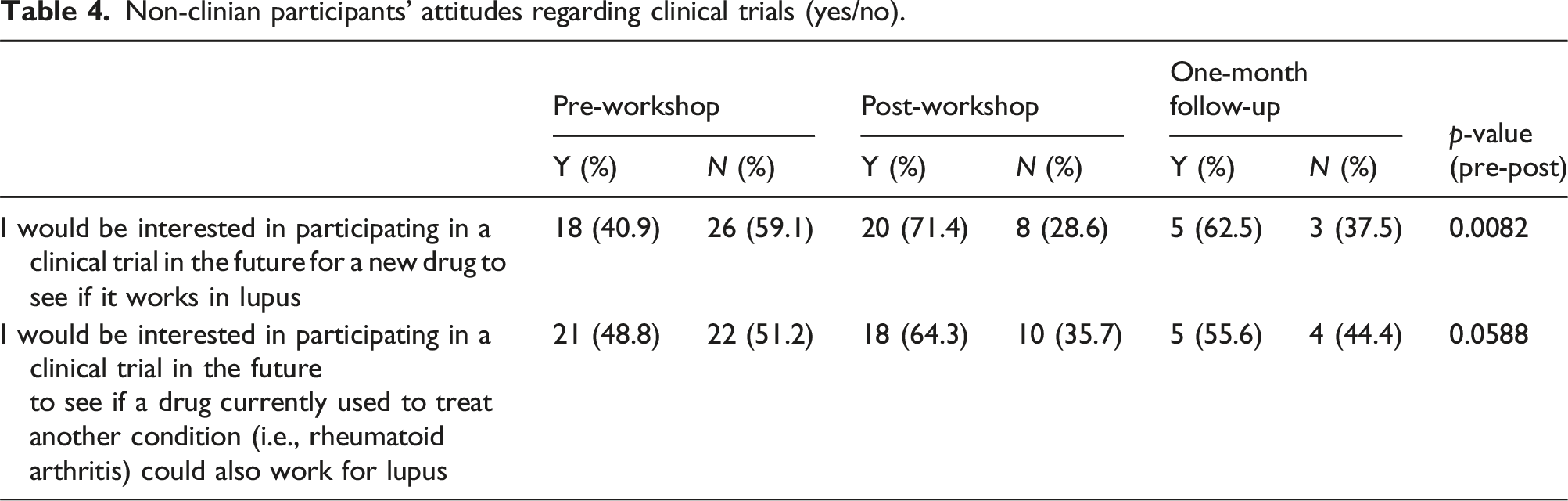
Non-clinian participants’ attitudes regarding clinical trials (yes/no).
Knowledge: There was a significant increase in non-clinician participants’ knowledge that participants in clinical trials can exit the trial at any time for any reason, from pre-to post-workshop (Appendix 2). Additionally, there was a statistically significant difference in the number of participants who correctly identified that researchers cannot remove participants from the study, if necessary, when comparing before and after the workshop. There was also a statistically significant difference in the number of participants who correctly identified that insurance would not be billed for receiving care from a clinical trial. However, from pre-to post-workshop, there was no significant change in participants’ knowledge involving the purpose of clinical trials, alternative treatments, receiving a placebo, and valuing patients’ well-being.
Intention to Change: At the 1-month follow-up, 44% of non-clinician participants agreed that they felt more comfortable participating in a clinical trial after the workshop. However, none of the surveyed participants reported having been asked to join a study, signed up for a study, or registered with a research database.
Clinician survey results
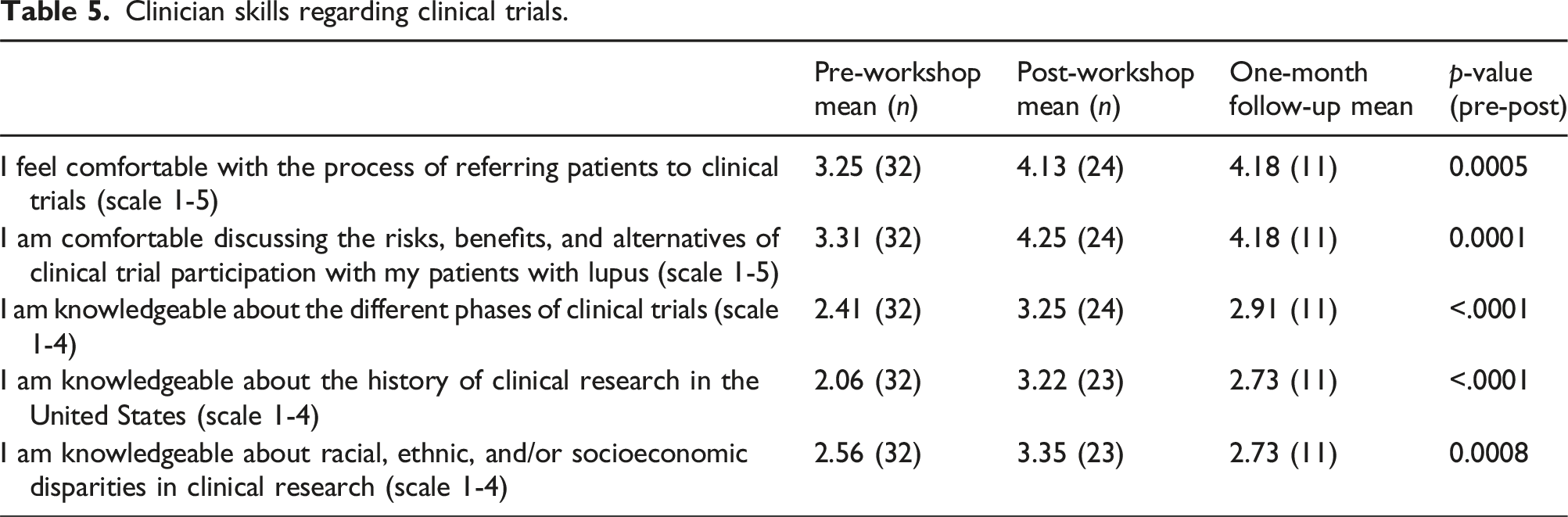
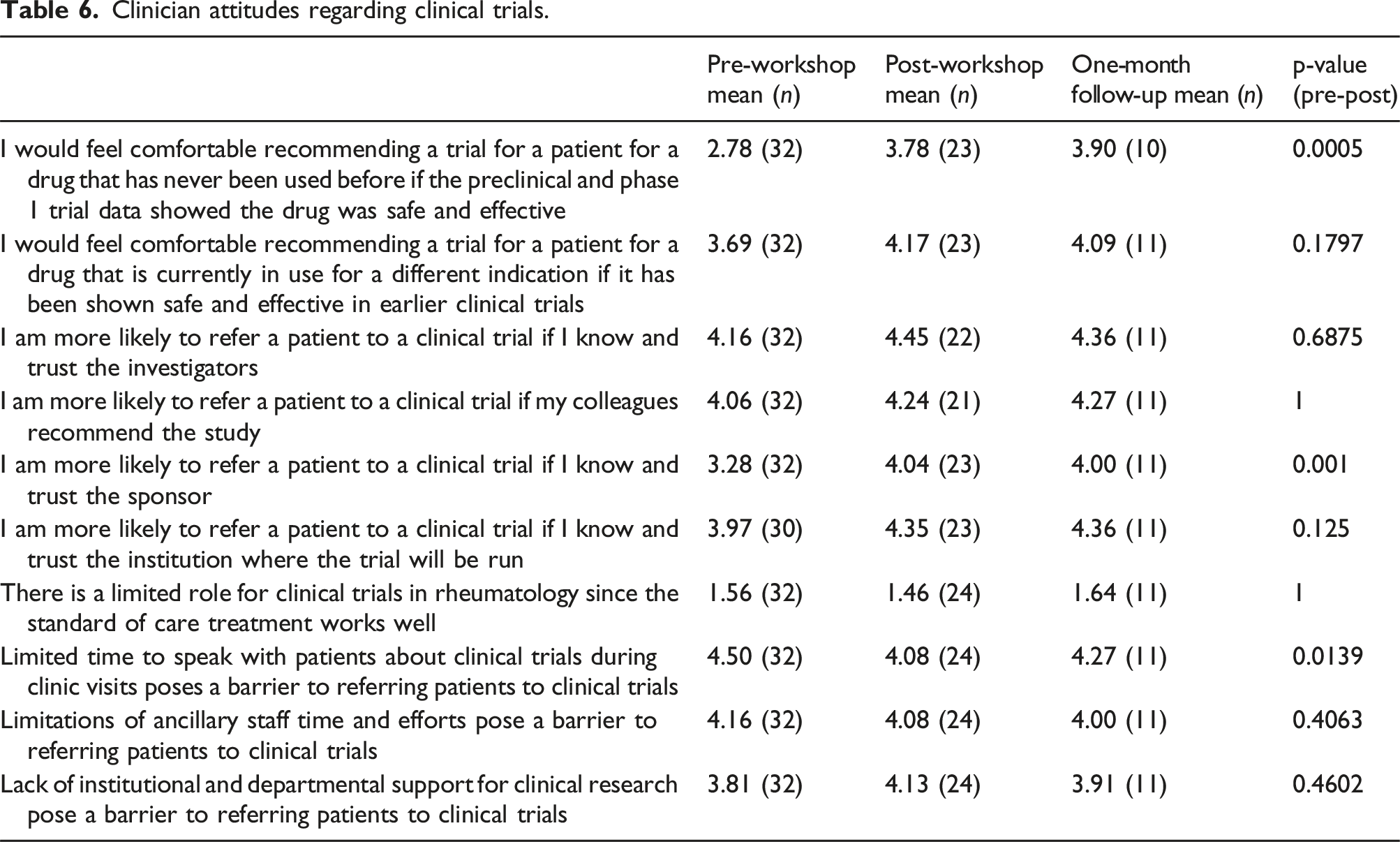
Skills: The comfort level of clinicians regarding clinical trial participation improved following the workshop (Table 5). There was a statistically significant increase in how knowledgeable the participants felt regarding the different phases of clinical trials, the history of clinical research in the United States, and the racial, ethnic, and/or socioeconomic disparities in clinical research. This gain in confidence is somewhat attenuated at the 1-month follow-up.
Clinician skills regarding clinical trials.
Clinician attitudes regarding clinical trials.
Knowledge
More than twice as many participants were able to identify correctly that approximately 5% of clinical trial participants in the US are African American after the workshop than before, but these gains were decreased at the 1-month follow-up. Twice as many participants could also correctly identify the principles of the Belmont Report after the workshop (see Appendix 1). Very few participants knew that Asian patients are more likely to develop lupus nephritis (6.3%), but this was significantly higher post workshop (54.2%). However, few participants were able to identify that they should consider factors pertaining to the trial site, such as accommodations and protections provided by the site, rather than patient-specific factors such as health literacy.
Intention to change
Clinicians agreed that they intend to discuss clinical trials with patients more frequently after the workshop (4.1 on 1-5 scale both post-workshop and 1-month follow-up). They also reported feeling more prepared to discuss clinical trials with their patients (4.1 post-workshop and 3.9 at 1-month follow-up). Participants also reported a greater understanding of the clinical trial process and how to refer patients to clinical trials (4.2 post workshop and 4 at 1-month follow-up).
Discussion
The results indicate that educational workshops can successfully influence both clinicians and non-clinicians’ perceptions of clinical trials. Along with changing non-clinician participants’ skills, these initiatives were also effective in changing attitudes about clinical trials, specifically with regards to the risks of clinical trial participation and the protective role of the IRB. Furthermore, post-workshop, non-clinician participants were significantly more interesed joining a clinical trial in the future. However, not all attitudes about clinical trials were changed. Non-clinician participant beliefs about the importance and benefits of clinical trials, developing new lupus medications, and the role of the FDA, were not significantly changed but these were largely favorable. Non-clinician participants overall had very positive attitudes regarding clinical trials, which were unchanged on follow-up.
Clinicians reported more comfort with referring patients to clinical trials and discussing clinical trials with their patients. They also reported feeling more knowledgeable about the clinical trial process and its history, and a better understanding of trial disparities. Their attitudes were also very favorable regarding clinical trials both before and after the workshop.
Regarding non-clinician participants’ knowledge, the educational workshops were successful in some areas. Specifically, knowledge about exiting the trial, the researchers’ inability to remove participants, and the role of insurance in clinical trials significantly improved after the workshop. However, some concepts were not correctly understood after the educational initiatives. For instance, there was no significant change in knowledge involving the purpose of clinical trials, alternative treatments, receiving a placebo, and the value of patients’ well-being after the workshop.
Overall, this project indicated that educating clinicians and non-clinicians about clinical trials is effective in changing their attitudes and knowledge toward trials. However, these initiatives are less effective in eliciting actual patient intentions to join a trial. With regards to the initial goals, this study answered the question of why many patients, especially those from an underrepresented background, do not join clinical trials. By elucidating the reasons behind decreased participation – hesitant attitudes and inaccurate knowledge about trials – future researchers can better understand how to improve patient involvement in clinical projects. Regarding recruitment, this study did not tangibly increase the number of patients who registered for a clinical trial despite the non-clinician participants reporting increased motivation post-workshop. Ultimately, this project contributes a clearer understanding of patients’ underlying motivation to join research trials. Most importantly, it highlights how providing patient education through workshops does improve patient beliefs about clinical trials.
There were some limitations to this study. Firstly, due to the small sample size, the results may not be generalizable to the greater population of interest. The limited sample size along with the short follow up may have also partially contributed to the low intention to change despite improved patient attitudes and knowledge. Secondly, educational initiatives are important but not sufficient in isolation in order to effect change. 28 A multifaceted approach is necessary for lasting results.
Response bias was present throughout each stage of the project. With each subsequent survey, fewer non-clinician participants and clinicians responded. Furthermore, even those participants that responded to the survey did not answer every item, causing an even lower sample size for certain questions. Originally, we did not require mandatory selections in our surveys to promote ease of completion, but perhaps future surveys should require mandatory selections for each question before submission is allowed. We could also employ other methods to promote later survey completion besides email reminders, such as calling participants.
Going forward, to increase local and national patient participation in clinical trials, similar educational initiatives can be implemented in other medical centers as a key part of a multidimensional approach to improve clinical trial recruitment. Creating more programs will ideally not only improve the number of patients enrolled but will provide more insight into patient perceptions toward clinical trials. Additionally, utilizing peer teaching can improve trial registration rates with patients. Helping patients recruit each other, like in the PALS 25 program, can build trust and rapport among patients. Similar approaches could be employed among clinicians, in which interested healthcare professionals could be paired up with an experienced clinical trial investigator to learn more about clinical trials to promote participation among their patients.
Supplemental Material
Supplemental Material - Evaluating the effectiveness of educational initiatives on recruiting underrepresented participants into SLE clinical trials
Supplemental Material for Evaluating the effectiveness of educational initiatives on recruiting underrepresented participants into SLE clinical trials by Shivani Kottur, Marie Maitre, David Karp, Maria Bacalao in Lupus.
Footnotes
Acknowledgments
The authors wish to thank Dr Una Makris and Dr Simon Craddock Lee for their help in editing and formulating the surveys. We also wish to thank Ang Cao for statistical analysis
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S. Kottur: None M. Maitre: None M. Bacalao: GSK SLE Educator’s network D. Karp: none
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The projected was funded by a grant from the Robert A Winn Diversity in Clinical Trials Award Program. Statistical analysis was funded by the UT Southwestern CTSA Grant UL1TR003163.
Declaration
All authors contributed to the conception, execution of the project and authoring the manuscript.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
