Abstract
Objective
While oral glucocorticoids (GC) have improved the prognosis of lupus nephritis (LN), dose reduction to ≤5.0 mg/day is recommended, considering their toxicity. We investigated the real-world prescription status following dose reduction of oral GC after starting initial therapy in patients with LN.
Methods
Data were extracted from the JMDC insurance claims database between May 2016 and March 2023 with the following inclusion criteria: patients with ≥2 LN diagnoses; follow-up for ≥90 days pre-index and ≥540 days post-index; oral GC dose ≥20.0 mg/day (prednisolone equivalent) or intravenous glucocorticoid pulse; ≥2 urinalyses, anti-double stranded DNA antibody, and complement tests within 360 days post-index. Descriptive analyses were performed by characterization as dose reduction target (GC ≤5.0 mg/day at Day 540) ‘achieved’ or ‘non-achieved’ groups.
Results
Mean oral GC baseline dose for the overall population (n = 295) was 36.1 mg/day. The two most prescribed immunosuppressants were mycophenolate mofetil (MMF; 41.7%) and tacrolimus (TAC; 16.9%). The proportion of patients with prescribed GC doses ≤5.0 mg/day was 1.0% on Day 1 and 48.1% on Day 540. In the achieved and non-achieved groups, the baseline concomitant immunosuppressants were MMF: 51.4% and 32.7% and TAC: 15.5% and 18.3%, respectively, while the proportion of patients with GC ≤7.5 mg/day was 31.0% and 12.4% on Day 180, respectively.
Conclusion
These real-world administrative data show the actual oral GC prescription status after starting initial LN therapy in Japan. Approximately half of the patients with LN were prescribed GC ≤5.0 mg/day within 1.5 years after starting the initial therapy.
Introduction
Lupus nephritis (LN), a frequent and serious complication of systemic lupus erythematosus (SLE), is a major cause of morbidity and mortality, characterized by a relapsing and remitting clinical course.1–3 In Japan, there are more than 64,000 registered patients with SLE, as per the Japan intractable diseases center; the patients are often diagnosed between the ages of 20 and 40 years, with the prevalence in women being 9 times that in men. 4
The treatment goals for LN mainly focus on prevention of LN flares, preserving kidney function, and limiting disease progression to avoid the need for dialysis and transplantation. 5 Treatment in class III/IV LN (as per the International Society of Nephrology and Renal Pathology Society, 2003) typically involves two phases: initial therapy, which aims to control renal inflammation and achieve remission of nephritis, and subsequent therapy, to sustain the response obtained during the initial phase and prevent systemic and renal relapses. 6
Current guidelines recommend a combination of glucocorticoids (GC; oral GCs with or without intravenous GC (ivGC) pulse therapy) with cyclophosphamide (CYC) or mycophenolate mofetil (MMF) for initial therapy, followed by subsequent therapy to stabilize the symptoms, with dose reduction. 7 However, despite such aggressive treatment, up to 25% of the patients who achieve remission have been reported to experience a flare within 3 to 4 years, with 5–20% of patients progressing to end-stage kidney disease (ESKD).5,8
Although the use of GCs apparently improves the outcomes of SLE, it can also lead to organ damage, resulting in comorbidities such as diabetes mellitus, vertebral fracture, cataract, and cardiovascular or cerebrovascular damage (GC-related adverse events [AEs]).9–12 The European League Against Rheumatism (EULAR) therefore recommends a reduction in cumulative GC exposure in the initial treatment phase, and also tapering the GC dose (prednisolone equivalent) to ≤5 mg/day in the subsequent phase, to minimize adverse reactions and complications associated with high-dose or long-term GC administration. 13 Japanese Clinical Guidelines (2019) for SLE also consider it appropriate to reduce the dose of GC to 10 mg/day after initial therapy and then maintain the dose at approximately 5 mg/day, with reference to the Lupus Low Disease Activity State and other indices in LN. 7
With the emergence of new therapies and given the need to improve outcomes, an understanding of existing treatment patterns, disease burden, and management becomes increasingly important. Accordingly, the achievement of sustained reduction of GC dose has been measured in various clinical trials in recent years.1,14,15 However, limited data are available regarding the prescription status of GCs after initial therapy.
In this study, our primary purpose is to describe the changes in oral GC dose (measured as prednisolone equivalent hereafter) in patients with LN treated with initial therapy. We also focus on the prescription status of immunomodulators and immunosuppressive agents concomitant with initial therapy and describe the characteristics of patients who achieved or did not achieve the dose reduction target of oral GC ≤5.0 mg/day (referred to as ‘achieved’ and ‘non-achieved’ groups, respectively, hereafter). We also evaluated the relationship between the cumulative GC dose and the occurrence of GC-related AEs.
Patients and methods
Study design and data source
This retrospective, non-interventional study (UMIN000053093) was conducted using data from the JMDC claims database (JMDC, Tokyo, Japan), 16 which provides healthcare insurance claims data with high traceability for those aged 0–74 years. The data extraction period for the present study was from May 2016 to March 2023.
Patients and analysis sets
Cases of LN were identified based on the following pre-defined algorithm developed with the medical experts in this study. Individuals who had a disease code of LN diagnosis (Japanese standard disease code ‘7100007’) as a confirmed diagnosis for ≥2 months during the study period were identified as eligible patients with LN. Patients were included in the primary population if they met all of the following criteria: (1) prescribed an oral GC at a dose of ≥20.0 mg/day or ivGC pulse therapy, and assigned a disease code 7100007 as confirmed diagnosis in the same month (medications were considered to be oral or intravenous medications on basis of their product names); (2) were traceable for ≥90 days before the primary index date; (3) were traceable for ≥540 days after the primary index date; (4) had not received oral GCs ≥20.0 mg/day or ivGC pulse therapy within 90 days before the primary index date; and (5) the following tests must have been performed during the same month as the confirmed diagnosis of LN within 360 days after the primary index date: ≥2 urinalyses; ≥2 anti-double stranded DNA antibody tests; and ≥2 complement tests. The primary index date (Day 1) was defined as the first prescription date of oral GC ≥20.0 mg/day or a course of ivGC pulse therapy.
The exploratory population was established for analysis of the relationship between the GC dose and the incidence of AEs. Patients were included in the exploratory population if they had a confirmed diagnosis of LN (disease code 7100007) and (a) ≥2 urinalyses, (b) ≥2 anti-double stranded DNA antibody tests, (c) ≥2 complement tests, and (d) ≥1 percutaneous needle biopsy. Specifically, for meeting each inclusion criterion (a, b, c, and d), each of the tests should have been performed within 1 month from recording the diagnosis code (7100007), and all the inclusion criteria (a, b, and c) should have been met within 360 days prior to meeting the last of these inclusion criteria.
Furthermore, patients had to have a follow-up period of ≥1800 days after the exploratory index date. The exploratory index date was defined as the first day GCs (oral or intravenous) were prescribed.
Definition of flares
Flares were defined as any of the following moderate or severe flares using the International Classification of Diseases, Tenth Revision (ICD-10) codes (Module 32 excluding Module 32.0) and frequency of drug prescriptions. The criteria for flares were defined in consideration of the current treatment status in Japan and with reference to previous reports. 17 If a patient experienced more than one of the treatments listed within 30 days, the treatment was counted as one flare.
A severe flare was defined as when any of the following conditions (1) to (3) were met after Day 180: (1) Patient had no prescription of oral GCs at doses greater than or equal to the baseline oral GC dose within the past 60 days and was prescribed oral GC at a dose greater than or equal to the baseline oral GC dose; (2) Patient had no prescription of CYC within the past 60 days and was prescribed CYC; (3) Patient had no prescription of ivGC pulse therapy within the past 60 days and was prescribed ivGC pulse therapy.
A moderate flare was defined as when any of the conditions (1) to (2) below were met after Day 180: (1) Patient had no prescription of oral GC ≥7.5 mg/day within the past 60 days and was prescribed oral GC ≥7.5 mg/day but less than or equal to baseline oral GC dose; (2) Patient had no prescription of immunosuppressants or immunomodulators including CYC within the past 60 days and was prescribed immunosuppressants excluding CYC (MMF, tacrolimus [TAC], azathioprine [AZA], mizoribine [MZB], cyclosporin A [CYA], rituximab [RTX], anifrolumab, belimumab [BLM]) or an immunomodulator (hydroxychloroquine [HCQ]).
Outcomes
The primary outcome was the proportion of patients with prescription of oral GC dose ≤5.0 mg/day until Day 540. The daily GC dosage was calculated as the amount prescribed/the number of days of prescription. Secondary outcomes included the proportion of concomitant immunosuppressants or immunomodulators, the frequency and proportion of flares, proportion with ivGC pulse therapy and the mean dose of oral GCs. These outcomes were assessed in the overall population, as well as in the achieved and non-achieved groups. The incidence of comorbidities by Day 1800 was analyzed as an exploratory outcome in the exploratory population.
Statistical analysis
Analyses were descriptive in nature. Frequencies (n) and proportions (%) were calculated for categorical data, and a chi-square test was used as a test of association, where applicable. The summary statistics (mean, standard deviation [SD], median, minimum [min], and maximum [max]) were calculated for quantitative data. No imputation of missing values was carried out. No calculations for sample size were done, as descriptive statistics were used. All analyses were carried out using the SAS® System Release 9.4 software.
Ethics statement
This retrospective study was carried out in accordance with the Declaration of Helsinki. De-identified, anonymized claims data were utilized for this study. Anonymization of these data was conducted in line with the Japanese Act on the Protection of Personal Information. The study was reviewed and approved by the Non-Profit Organization MINS Institutional Review Board.
Results
Study population and index date characteristics
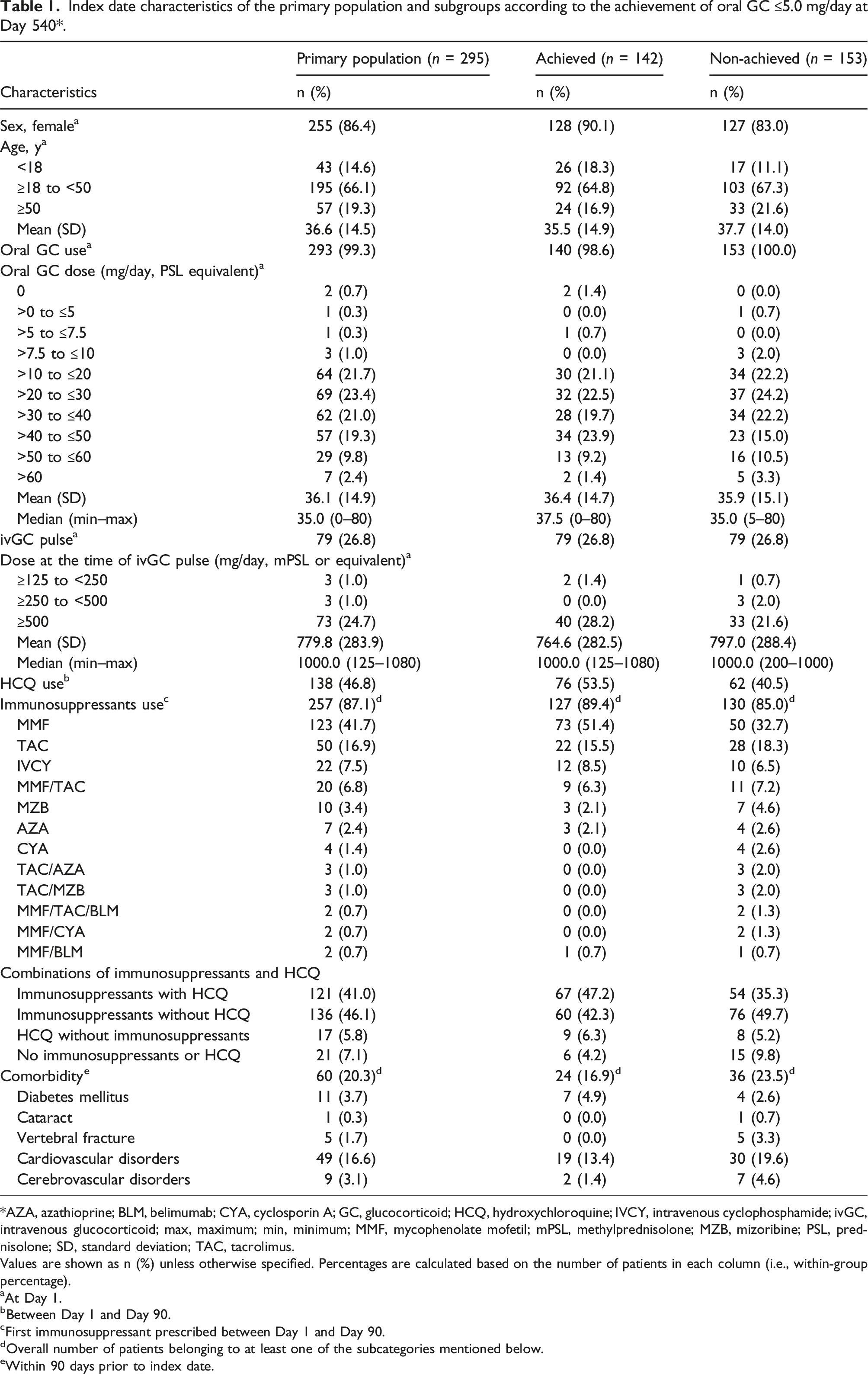
Of 6565 eligible patients identified from the database, 295 patients were included in the primary analysis and 120 in the exploratory analysis, as shown in Figure 1. For the primary population, the mean (SD) age was 36.6 (14.5) years, with a female:male patient ratio of ∼9:1 (Table 1). The median (min–max) oral GC dose was 35.0 (0–80) mg/day. At Day 1, 26.8% of patients had received ivGC pulse (Table 1). Among concomitant medications, 87.1% patients received immunosuppressants, with MMF (41.7%) and TAC (16.9%) being the two most prescribed concomitant immunosuppressants by Day 90, while 46.8% patients had a prescription of HCQ, an immunomodulator, by Day 90 (Table 1). One-fifth of patients (60, 20.3%) had comorbidities, with cardiovascular disorders (49, 16.6%) being the most common (Table 1). Patient flow. * GC, glucocorticoid; ivGC, intravenous glucocorticoid; LN, lupus nephritis. Index date characteristics of the primary population and subgroups according to the achievement of oral GC ≤5.0 mg/day at Day 540*. *AZA, azathioprine; BLM, belimumab; CYA, cyclosporin A; GC, glucocorticoid; HCQ, hydroxychloroquine; IVCY, intravenous cyclophosphamide; ivGC, intravenous glucocorticoid; max, maximum; min, minimum; MMF, mycophenolate mofetil; mPSL, methylprednisolone; MZB, mizoribine; PSL, prednisolone; SD, standard deviation; TAC, tacrolimus. Values are shown as n (%) unless otherwise specified. Percentages are calculated based on the number of patients in each column (i.e., within-group percentage). aAt Day 1. bBetween Day 1 and Day 90. cFirst immunosuppressant prescribed between Day 1 and Day 90. dOverall number of patients belonging to at least one of the subcategories mentioned below. eWithin 90 days prior to index date.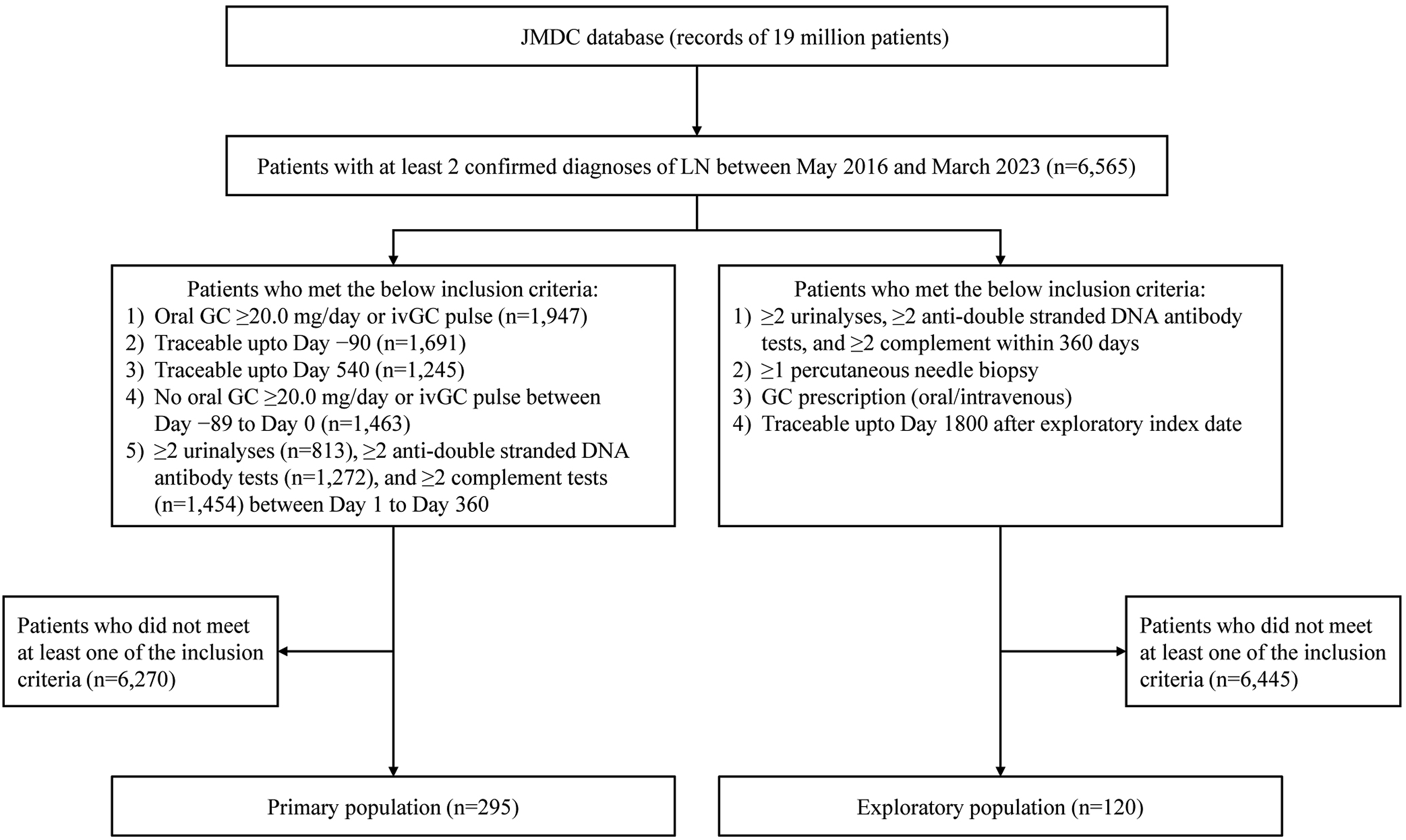
Glucocorticoid dose reduction
The proportion of patients who received oral GC prescription of ≤5.0 mg/day was 1.0% at Day 1 and 48.1% at Day 540 (Figure 2(a)). The mean (SD) prescribed oral GC dose showed a continuous decline from 36.1 (14.9) mg/day at Day 1, reducing markedly to 11.9 (6.4) mg/day at Day 180 and gradually thereafter, to 6.8 (4.2) mg/day at Day 540 (Figure 2(b)). The reduction in oral GC dose to ≤5.0 mg/day and the oral GC dose trends according to concomitant immunosuppressants received by Day 90 are shown in Supplemental Table 1 and Supplemental Figure 1, respectively. All groups showed a reduction in GC dose over time, with a faster reduction by Day 180 and a slower but continuous reduction thereafter (Supplemental Figure 1). Changes in oral GC ≤5.0 mg/day by Day 540 among the primary population. (a) Proportion of patients with oral GC ≤5.0 mg/day by Day 540. Patients in whom oral GC never exceeded 5.0 mg/day throughout the observation period were excluded; (b) The mean (SD) GC dose by Day 540 in the primary population. * GC, glucocorticoid; max, maximum; min, minimum; SD, standard deviation.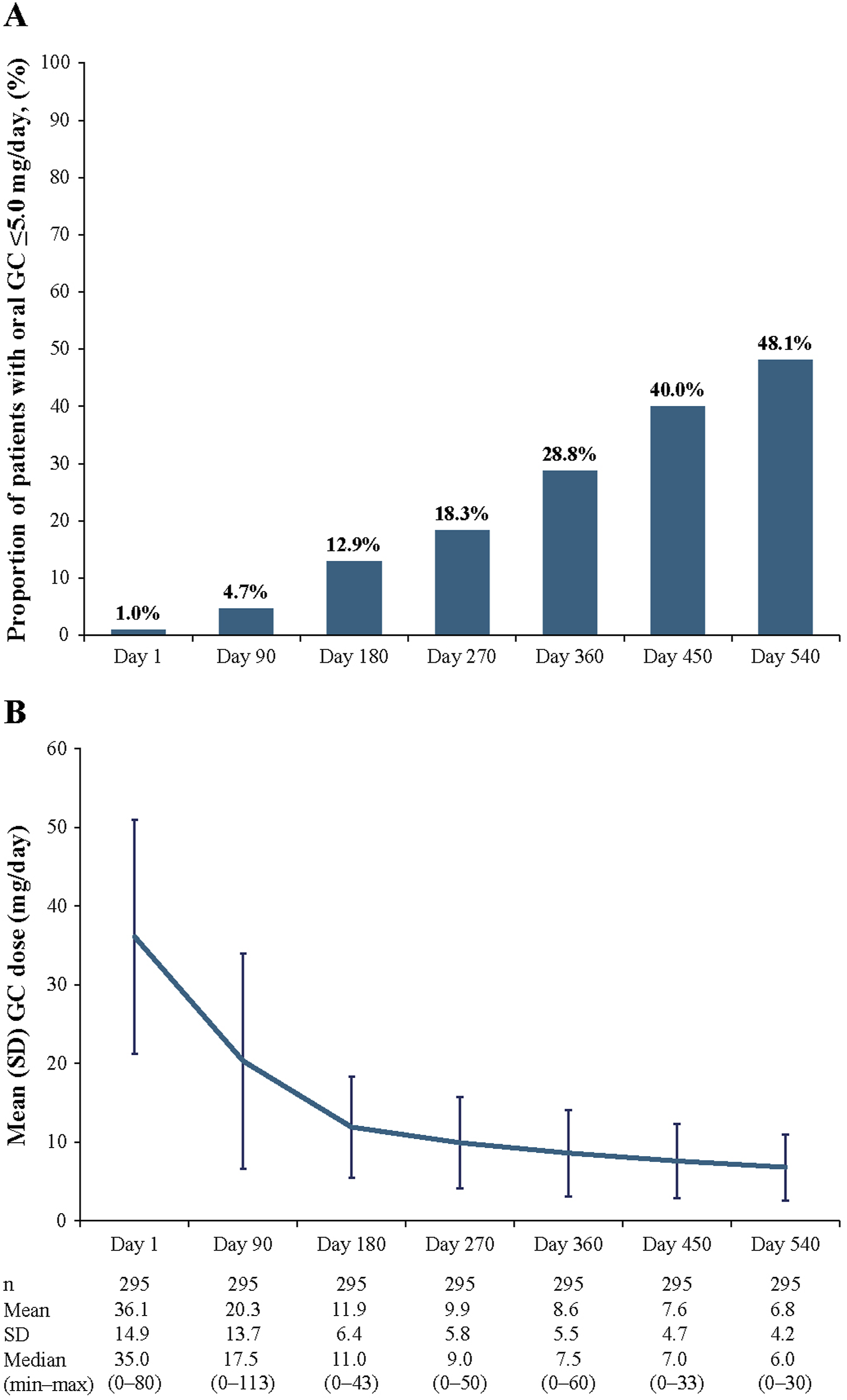
Concomitant immunomodulator/immunosuppressants
The proportion of patients receiving concomitant immunomodulator HCQ was 47.8% between Day 1 and Day 90, and 52.9% between Day 451 and Day 540 (Supplemental Table 2). Of the immunosuppressants, MMF was prescribed to the greatest proportion of patients, followed by the MMF/TAC group, throughout the period up to Day 540. AZA was used concomitantly in only a limited number of patients (Figure 3). The proportion receiving MMF continued to increase after Day 1 in association with the transition from the MMF/TAC group, the MMF/IVCY group and intravenous CYC (IVCY) (Figure 3). Use of immunosuppressants up to Day 540. Sankey diagram showing the change in proportion of concomitant immunosuppressant prescriptions in the primary population over 540 days. Others includes MMF/TAC/AZA, TAC/IVCY/AZA, TAC/BLM, MMF/TAC/IVCY, MMF/TAC/MZB, MMF/IVCY/BLM, MMF/CYA/MZB, MMF/AZA/BLM, TAC/CYC/IVCY, TAC/IVCY/MZB, TAC/IVCY, TAC/CYA/MZB, TAC/CYA/BLM, IVCY/CYA/BLM, IVCY/CYA, IVCY/AZA, IVCY/MZB, IVCY/BLM, AZA/MZB, RTX, MMF/TAC/CYA, MMF/IVCY/CYA/AZA/BLM, MMF/anifrolumab, TAC/IVCY/RTX, TAC/AZA/BLM, TAC/RTX, CYC/IVCY/AZA, CYC, IVCY/RTX, CYA/AZA, CYA/MZB, CYA/RTX, BLM. * AZA, azathioprine; BLM, belimumab; CYA, cyclosporin A; CYC, cyclophosphamide; IVCY, intravenous cyclophosphamide; MMF, mycophenolate mofetil; MZB, mizoribine; RTX, rituximab; TAC, tacrolimus.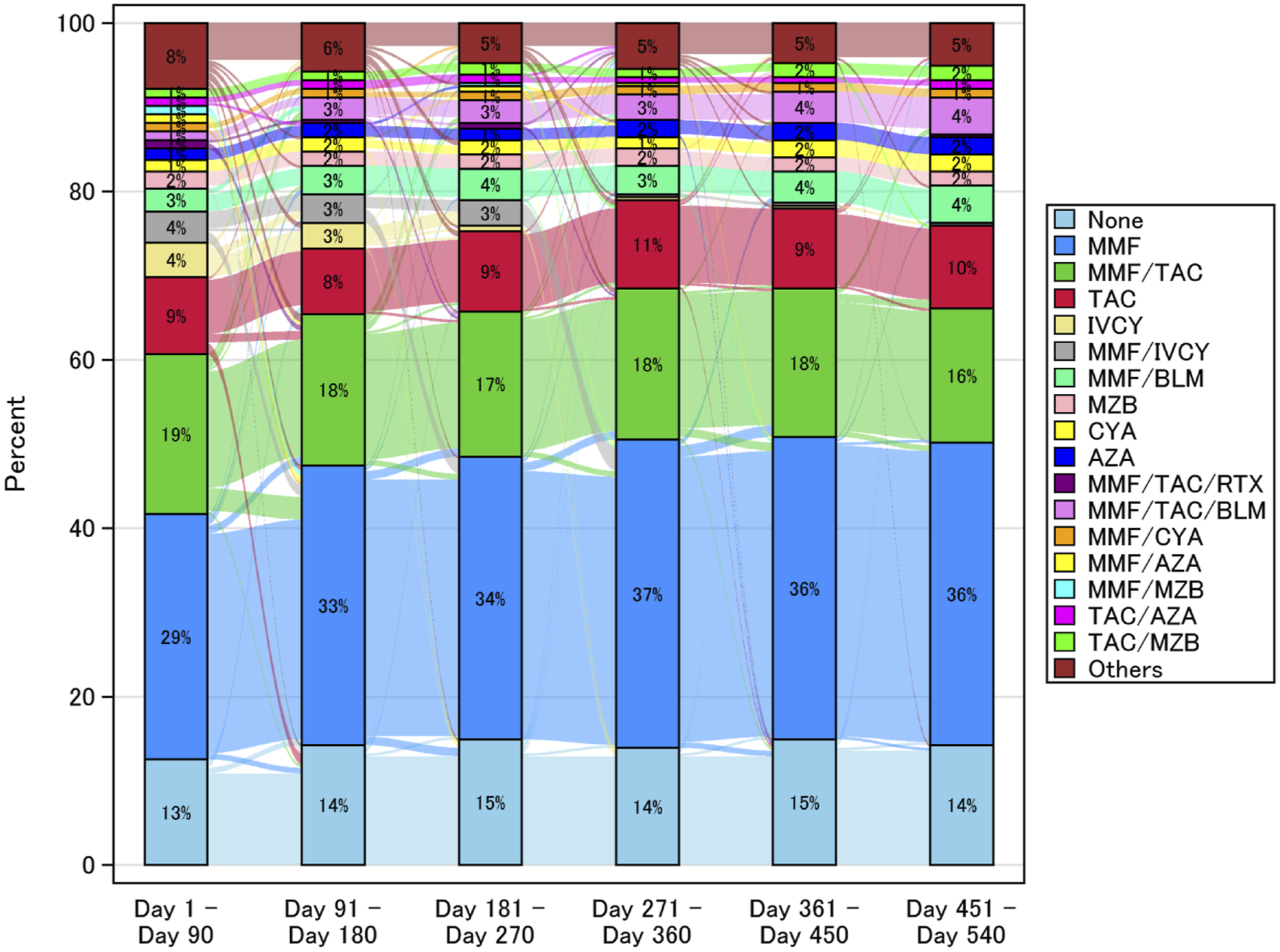
Incidence of flares
Incidence of flares between Day 180 and Day 540 (in primary population, n = 295)*.

*CYC, cyclophosphamide; GC, glucocorticoid; HCQ, hydroxychloroquine; ivGC, intravenous glucocorticoid.
aThe breakdown of flares is as described in detail in the Methods section.
bSevere flares were defined as the additional prescription of CYC; moderate flares were defined as the additional prescription of immunosuppressants or an immunomodulator (HCQ), excluding CYC.
cSix patients had both moderate and severe flares.
dTwo patients met the definition for multiple types of moderate flares on the same day.
eTwo patients met the definition for multiple types of severe flares on the same day.
Index date characteristics among GC ≤5.0 mg/day achieved and non-achieved groups
At Day 540, 142/295 patients achieved GC reduction to ≤5.0 mg/day (achieved group), while the remaining 153 did not (non-achieved group) (Table 1). In the achieved and non-achieved groups, the mean (SD) age was 35.5 (14.9) and 37.7 (14.0) years and the median GC dose at baseline was 37.5 and 35.0 mg/day, respectively. The proportion of HCQ use between Day 1 and Day 90 was 53.5% in the achieved and 40.5% in the non-achieved groups (Table 1). The two most prescribed immunosuppressants in the achieved and non-achieved groups were MMF (51.4% and 32.7%), and TAC (15.5% and 18.3%), respectively. Among patients in the achieved group, 47.2% were treated with HCQ in combination with immunosuppressants, whereas 35.3% of those in the non-achieved group received such combination therapy. In contrast, immunosuppressants without HCQ were prescribed in 42.3% and 49.7% of patients in the achieved and non-achieved groups, respectively.
Clinical course by Day 540 in GC ≤5.0 mg/day achieved and non-achieved groups
As the EULAR 2019 recommendations 18 suggest a dose reduction to ≤7.5 mg/day within 3–6 months after starting initial therapy, we also analyzed the proportion of patients achieving ≤7.5 mg/day in the overall primary population, as well as in the achieved and non-achieved groups. In the primary population, 8.5% and 21.4% of patients achieved ≤7.5 mg/day at Day 90 and Day 180, respectively (Supplemental Figure 2A). In the achieved group, the proportion achieving ≤7.5 mg/day was 11.3% at Day 90 and 31.0% at Day 180, whereas the proportion was 5.9% at Day 90 and 12.4% at Day 180 in the non-achieved group (Supplemental Figure 2B).
The incidence of flares was 23.9% achieved and 25.5% in non-achieved groups, with severe flares in 6.3% and 15.0% patients, between Day 180 and Day 540 (Supplemental Table 3).
Incidence of GC-related diseases by cumulative GC dose
Incidence of specific diseases by cumulative GC dose (in exploratory population, n = 120)*.
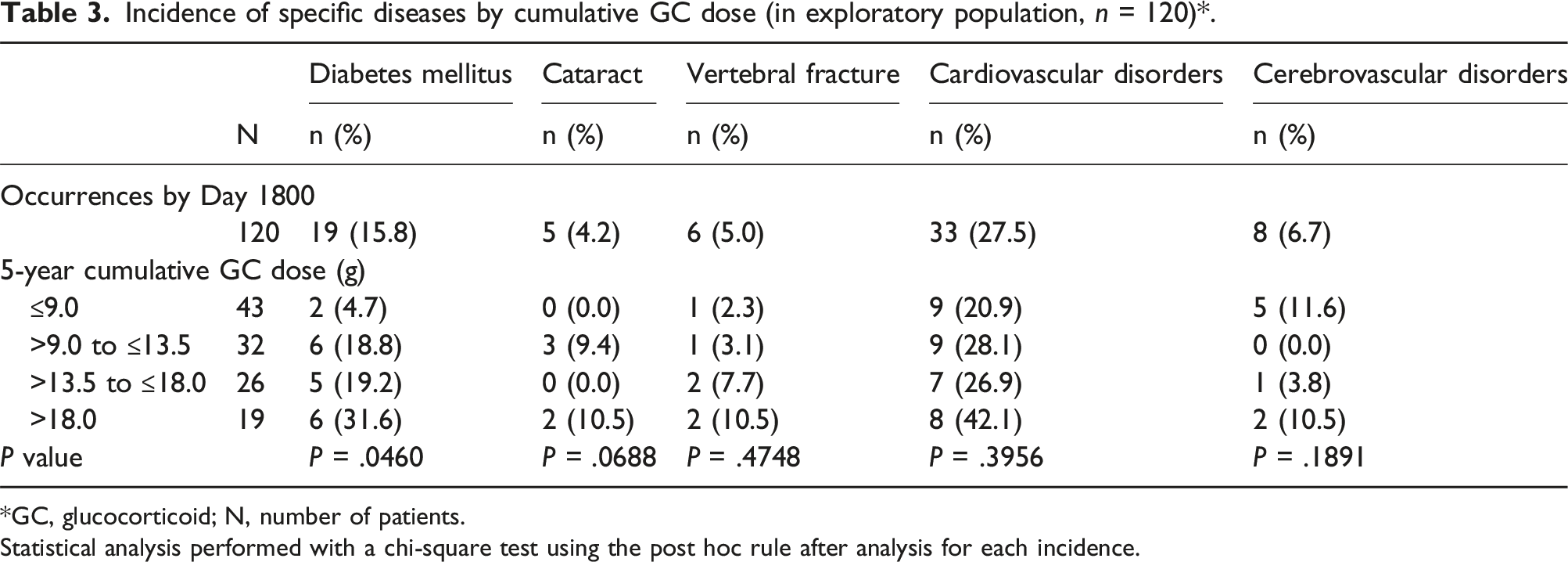
*GC, glucocorticoid; N, number of patients.
Statistical analysis performed with a chi-square test using the post hoc rule after analysis for each incidence.
Discussion
In this retrospective study, we investigated the real-world patterns and feasibility of oral GC dose reduction, after starting initial therapy, to the target dose (≤5.0 mg/day, prednisolone equivalent) recommended by clinical guidelines 13 in Japanese patients with LN. Of 295 patients in the primary population, 48.1% of patients showed a reduction of oral GC to ≤5.0 mg/day at Day 540. An intensive initial therapy with high dose of GC combined with immunosuppressant therapy has shown improvement in remission rates in patients with LN. 19 However, as the impact of cumulative GC-related side effects has become apparent,20,21 strategies which involve the reduction of GC dose are warranted.
GC dose reduction, although advisable, is not always achieved in real-world practice. A recent retrospective database study from Germany characterized the treatment patterns among adult patients with LN receiving immunosuppressant therapy with MMF, IVCY, AZA, TAC, CYA, or RTX. This real-world study reported an increase in median corticosteroid dose, with the proportion of patients with ≥5.0 mg/day increasing from 40.1% to 61.1% after 1 year. 22 Another study in patients with LN with onset of ESKD reported that >50% received corticosteroid doses of >10 mg/day, after median (min–max) follow-up of 761 (374–1375) days. 23 In contrast, we observed an almost six-fold reduction in the median prednisolone-equivalent GC dose over a period of 540 days, while the proportion of patients prescribed with oral GC ≤5.0 mg/day increased from 1.0% to 48.1% in this Japanese real-world analysis. The importance of the reduction of the oral GC is recognized by national and international guidelines.7,13 Although about half of the patients reached the target GC dose at 1.5 years, multiple reasons may underly the frequent use of higher doses of oral GC during subsequent therapy, including the occurrence of flares, the discontinuation of immunosuppressants due to adverse reactions, and the limited efficacy of immunosuppressant therapies.
In this study, the prescription status of concomitant immunosuppressive therapy was also described. MMF, alone or in combination with other immunosuppressants, was the most frequently prescribed agent throughout the observation period, in line with national and international guidelines.7,18 In contrast to studies investigating the prescription patterns in the United States of America and Europe,22,24 AZA was used less than TAC in our study, possibly because TAC is approved for LN in Japan. Use of IVCY decreased markedly after Day 180, consistent with the recommendations of prescribing the drug for 6 months during remission initial therapy. 25 Although BLM is a newly approved biologic therapy in SLE and has shown a sustained dose reduction of oral GC,1,26 the limited timeframe of the data collection period resulted in few cases of BLM use, which limited full assessment of its effectiveness.
In our study, patients in achieved and non-achieved groups showed no specific trend in their baseline GC dose level or status of ivGC pulse prescription. However, baseline MMF-only administration was found to be more frequent in the achieved group than the non-achieved group (51.4% and 32.7% respectively), as compared to the proportion of TAC (15.5% and 18.3%, respectively). This may be because MMF/IVCY are preferred drugs during initial treatment while TAC is often used in patients with an inadequate response to these drugs. 25 Thus, those in the non-achieved group, who possibly had inadequate responses, would have received TAC more frequently than the achieved group. A high initial dose of IVCY might have been used for severe cases as recommended, with TAC used for milder cases. Furthermore, most patients prescribed with MMF/IVCY therapy tended to achieve GC dose reduction, consistent with a previous randomized trial investigating the efficacy of MMF as a subsequent therapy for preventing relapses, following initial monotherapy with IVCY or MMF. 27
In the primary population, the incidence of flares was 24.7%, with moderate flares in 15.9% patients and severe in 10.8%. Furthermore, the incidence of severe flares was 6.3% in the achieved and 15.0% in the non-achieved group. A higher incidence of flares/relapses, especially severe flares, or longer cumulative duration of time spent in flares is associated with increased risk of renal function deterioration or progression to ESKD.28–33 Higher flare frequency has also been reported to have a negative impact on quality of life. 34 Our results emphasize the still unmet need of accelerating the achievement of GC dose reduction while avoiding severe flares.
Long-term use of GCs is known to be associated with AEs. 20 However, in our study, after a follow-up of ≥5 years, a clear trend between the incidence of GC-related diseases and cumulative GC dose was not seen for comorbid diseases other than diabetes mellitus. Possible reasons for this may include an insufficient observation period, fewer events of comorbidities, and not allowing for adjustment with other factors such as smoking history, that may affect the incidence of cardiovascular and cerebrovascular diseases. In contrast, a clear dose-response effect was evident for the occurrence of diabetes mellitus, in relation to cumulative GC dose, which strongly supports the importance of GC dose reduction.
Previous literature suggests that early achievement of complete renal response predicts good long-term renal outcomes.35,36 Taken together, our results also suggest that it is important to aim for earlier reduction of GC doses in patients whenever possible, with the long-term goal of reducing cumulative GC doses. However, GC dose reduction is a slow process in the real-world scenario: despite the EULAR 2019 recommendations, 18 only 31.0% of patients who achieved ≤5.0 mg/day at Day 540 were able to reduce the dose to ≤7.5 mg/day at Day 180 of initial therapy (Supplemental Figure 2B). Thus, our results suggest the need for new treatment strategies that enable early reduction of GC dose after initial therapy, while taking into account the toxicity of cumulative GC prescriptions. Further intensification of initial therapy to enable achievement of early GC dose reduction is required.
Limitations
This study has some limitations, mostly intrinsic to the study design and the administrative database source. Although there are limited data on elderly patients aged ≥75 years in the JMDC database, in view of the age distribution for onset of SLE and LN, this database with high traceability across sites was considered to be the most suitable for the analysis of long-term investigation of prescription patterns. Next, among the target population, a large proportion could not be followed up till Day 540, as their index date was within 540 days before March 31st, 2023, and those with an insufficient observation period could not be included in the long-term analysis. Additionally, there were discontinuations because of death or retirement (Supplemental Table 4). However, as the dropout rate in this study is close to the general withdrawal rate for JMDC, 37 we consider the impact of survival bias to be limited. Also, the JMDC database only captures prescription records, not whether the patient actually received the medication. Furthermore, we lacked access to data from all laboratory tests, including serology and urinalysis, which are important for understanding the diagnosis and pathology of SLE and LN. Although this could lead to potential misclassification, we addressed this issue as far as possible by incorporating multiple LN-related examinations into the inclusion criteria. Notably, the inability to adjust for the important covariates of disease activity made any confounding adjustments such as propensity score matching and multivariate analyses difficult to perform. However, the potential for residual confounding exists in any observational study despite applying the best practices to control it. This being a descriptive epidemiological study precludes comparisons of efficacy between immunosuppressants and treatment with or without HCQ. Lastly, the JMDC includes data for Japanese population, the external validity of this study may be limited.
Conclusion
These real-world administrative claims data from Japan were beneficial in showing the actual status of oral GC prescription after starting initial therapy for LN. We recognized an important and unmet need for treatment strategies that can enable achievement of early GC dose reduction while inhibiting the occurrence of flares, as suggested by the status of oral GC prescription, implementation of ivGC pulse in initial therapy and the concomitant use of immunomodulatory or immunosuppressive drugs at baseline in Japanese patients with LN.
Supplemental Material
Supplemental Material - Real-world glucocorticoid prescription patterns in patients with lupus nephritis: A retrospective study using a healthcare insurance claims database
Supplemental Material for Real-world glucocorticoid prescription patterns in patients with lupus nephritis: A retrospective study using a healthcare insurance claims database by Tatsuya Atsumi, Hironari Hanaoka, Nobuo Nishijima, Kohji Murakami, Mariko Nio, Tsutomu Urakawa, Takaaki Fujimura, Hiroki Hayashi in Lupus
Footnotes
Acknowledgments
We would like to thank INTAGE Healthcare Inc. for statistical analysis, funded by Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd. Medical writing assistance was provided by Shivani Niphadkar, MSc and Vidula Bhole, MD, MHSc of MedPro Clinical Research and was funded by Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd
Author contribution
Conceptualization: All authors; Methodology: TA, HHanaoka, NN, KM, MN, TU, HHayashi; Investigation: TA, HHanaoka, NN, KM, TU, HHayashi; Project administration: TU, TF; Supervision: TA, HHanaoka, HHayashi; Writing–review and editing: All authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tatsuya Atsumi reports grants from GlaxoSmithKline plc., and consultation fees from AstraZeneca plc, Idorsia Pharmaceuticals Ltd, Otsuka Pharmaceutical Co., Gilead Sciences, Inc., GlaxoSmithKline plc., Sanofi K.K., Eli Lilly Japan K.K., Nippon Boehringer Ingelheim Co., Ltd, Janssen Pharmaceutical K.K., UCB Japan Co. Ltd, Kissei Pharmaceutical Co. Tatsuya Atsumi also reports speaker engagement and honoraria with Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co. Ltd, Janssen Pharmaceutical K.K., Mitsubishi Tanabe Pharma Co., Daiichi Sankyo Co. Ltd, Pfizer Inc., Alexion Inc., Novartis Pharma K.K., Eli Lilly Japan K.K., Kyowa Kirin Co., Ltd, AbbVie Inc., Nippon Boehringer Ingelheim Co., Ltd, Amgen Inc., UCB Japan Co. Ltd, AstraZeneca plc, Asahi Kasei Pharma Co. Ltd, Eisai Co., Ltd, Takeda Pharmaceutical Co., Ltd, Astellas Pharma Inc., Sanofi K.K. Hironari Hanaoka reports speaker engagement and honoraria with Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd. Nobuo Nishijima, Mariko Nio, Tsutomu Urakawa, and Takaaki Fujimura are employees of Chugai Pharmaceutical Co., Ltd. Kohji Murakami is an employee of Nippon Shinyaku Co., Ltd. Hiroki Hayashi reports speaker engagement and honoraria with Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd and payment for lectures from Otsuka Pharmaceutical Co., Ltd, AstraZeneca K.K., Asahi Kasei Pharma Corporation, GlaxoSmithKline K.K., and Nippon Boehringer Ingelheim Co., Ltd
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd. Statistical analysis and medical writing support were also funded by Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd
Ethical consideration
This retrospective study was carried out in accordance with the Declaration of Helsinki. De-identified, anonymized claims data were utilized for this study. Anonymization of these data was conducted in line with the Japanese Act on the Protection of Personal Information. The study was reviewed and approved by the Non-Profit Organization MINS Institutional Review Board.
Data Availability Statement
No data other than what is included in this article or supplementary material will be shared. Chugai Pharmaceutical Co., Ltd and Nippon Shinyaku Co., Ltd have a contract with JMDC to use this database and publish the results. Please note that JMDC makes its databases of data from medical institutions widely available, on a fee-paying basis, for use of surveys, research, and commercial purposes. Inquiries about these databases can be made via JMDC’s website [![]() ], and it can be accessed after completing the contract for use of specific data.
], and it can be accessed after completing the contract for use of specific data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
