Abstract
Objective
This study explored whether modulating prefrontal alpha oscillations using transcranial alternating current stimulation (tACS) could alleviate depressive symptoms in individuals with systemic lupus erythematosus (SLE).
Methods
Over the course of 5 days, three individuals underwent daily 40-min sessions of 2 mA bifrontal individual alpha frequency tACS (IAF-tACS) while watching a relaxing video. Resting-state 128-channel electroencephalography (EEG) was recorded on Day 1 and Day 5 before each tACS session. Self-reported assessments of depression, anxiety, insomnia, and fatigue were administered on Day 1, Day 5, and at the 2- and 4-week follow-ups.
Results
Two out of three participants showed a reduction in left prefrontal alpha power after 5 days of IAF-tACS. Only those with this reduction reported short-term improvements in depression, insomnia, anxiety, and well-being.
Conclusion
These preliminary results suggest that modulating prefrontal alpha oscillations through tACS may offer a potential approach for alleviating depressive symptoms in individuals with SLE.
Keywords
Introduction
Depressive symptoms are highly prevalent in individuals with systematic lupus erythematosus (SLE),1,2 contributing to increased disease burden, poorer treatment outcome and significantly reduced quality of life. 3 A neurophysiological network-level feature of depressive symptoms is altered frontal alpha oscillations (8–12 Hz), particularly increased alpha activity in the left prefrontal cortex. 4 While alpha oscillations are generally considered inhibitory and aid cognitive processing, their heightened presence in depression may indicate hypoactivity in the prefrontal cortex. 4 This leads to maladaptive prefrontal top-down control, resulting in changes in affective processing and the engagement of circuits linked to reward and motivation. Treating depressive symptoms in the SLE population is particularly challenging due to a combination of factors, including diagnostic complexity, 5 as depressive symptoms often overlap with SLE-related symptoms such as fatigue and cognitive dysfunction, limited psychosocial support 6 and pharmacological constraints. Antidepressant medications offer a small to moderate effect size and are often associated with adverse effects that may be poorly tolerated or may interact with immunosuppressive therapies. 7 These limitations and the importance of effectively managing depressive symptoms in patients with SLE to improve overall disease burden have prompted a growing interest in non-pharmacological interventions that are both safe and well-tolerated.
In this context, non-invasive brain stimulation techniques such as transcranial magnetic stimulation (TMS) have already been FDA-approved and demonstrate effectiveness in reducing depressive symptoms in major depressive disorder. 8 Recently, transcranial alternating current stimulation (tACS) has emerged as a promising method to modulate prefrontal brain networks associated with mood regulation.9,10 Specifically, tACS was selected for this feasibility study due to its unique ability to non-invasively modulate endogenous brain oscillations in a frequency- and phase-specific manner. Targeting alpha oscillations is particularly relevant in the context of depression, where dysregulated frontal alpha power has been consistently observed.8,9 By targeting prefrontal alpha oscillations directly, our group previously developed a 10 Hz tACS treatment protocol for major depressive disorder, consisting of five daily 40-min sessions of bifrontal alpha tACS. 9 This approach has renormalized the aberrant alpha activity and has shown success in reducing depression symptoms. One approach to further enhance clinical effectiveness of tACS interventions is to optimize neural engagement by aligning exogenous tACS with an individual’s endogenous alpha frequency (IAF) oscillatory activity. 11
Compared to other non-pharmacological interventions such as TMS, which requires in-clinic equipment and is less feasible for immunocompromised patients, or transcranial direct current stimulation (tDCS), which lacks frequency specificity, tACS offers a portable, well-tolerated,9,10 and potentially home-based alternative. These characteristics make tACS a particularly attractive intervention for individuals with SLE, who often experience barriers to frequent clinic visits and may benefit from low-burden, brain-based treatments for depressive symptoms.
To our knowledge, tACS has not yet been evaluated in individuals with SLE. We therefore initiated a double-blind, placebo-controlled clinical trial to investigate the preliminary efficacy of individualized tACS in this population. Unfortunately, due to enrollment challenges posed by the COVID-19 pandemic, the study was concluded prematurely. Here, we present data from three individuals who completed the study. Our primary aim was to explore the link between prefrontal alpha oscillation changes and depression symptom relief after the 5-day IAF-tACS intervention, providing initial support for prefrontal alpha modulation as a potential therapeutic target in comorbid depression in SLE.
Methods
Study design
Although originally registered as a double-blind randomized controlled trial (RCT), the study was prematurely terminated due to significant recruitment challenges, particularly in enrolling immunocompromised individuals during the COVID-19 pandemic. As a result, only four participants were enrolled, and the current report is presented as a proof-of-concept feasibility study rather than a completed RCT. The study was approved by the Institutional Review Board of the University of North Carolina at Chapel Hill (IRB #19-0763) and registered on the International Clinical Trials Registry Platform (NCT04141046). Written informed consent was obtained before enrollment. Recruitment occurred at UNC’s Rheumatology Lupus Clinic with stimulation performed at the Carolina Center for Neurostimulation. Inclusion criteria required meeting at least four American College of Rheumatology (ACR) or the Systemic Lupus International Collaborating Clinics (SLICC) SLE criteria, testing positive for relevant antibodies (e.g., Antinuclear Antibodies; ANA, Anti-double-stranded DNA Antibodies; Anti-dsDNA), and low suicide risk (HDRS-17 item 3). Exclusion criteria included benzodiazepines or anticonvulsant medication, nursing or pregnancy, and medication changes within 6 weeks. A detailed overview of participant screening, enrollment, and follow-up is provided in the CONSORT flow diagram (Supplemental Figure S3).
Sample size rationale
This pilot study was designed to assess the feasibility, safety, and tolerability of individualized alpha-frequency tACS in individuals with SLE. No formal power calculation was conducted, as the study was not intended to test hypotheses or detect statistically significant effects. Instead, the goal was to generate preliminary procedural and neurophysiological insights to inform the design of future adequately powered RCTs.
Experimental procedures
Participants completed 5 days of individualized alpha frequency tACS (IAF-tACS) or sham tACS after informed consent and screening, including a neuroradiologist-reviewed non-contrast cranial MRI. Sham tACS consisted of 20 seconds ramp-in, 40 seconds of 10 Hz tACS and a subsequent ramp-out of 20 seconds. This procedure mimicked the sensation of active stimulation to maintain participant blinding. The same procedures and equipment were used in both sham and active sessions, and participants were unaware of their group assignment. Clinical symptoms were assessed on Day 1 and Day 5, and at 2- and 4-week follow-ups, using the Inventory of Depression and Anxiety Symptoms (IDAS) 12 and the Fatigue Scale for Motor and Cognitive Functions (FSMC) 13 by trained research staff following standardized written protocols. Assessors were blinded to stimulation condition. SLE activity was assessed with the SLE Disease Activity Index 2000 (SLEDAI-2K) 14 by a board-certified rheumatologist experienced in clinical SLE evaluation. Resting-state EEG (128-channel, eyes-open) was recorded on Day 1 and 5 (Electrical Geodesic Inc., Eugene, OR, USA). Day 1 EEG data determined each participant’s personalized stimulation frequency.
TACS electrodes (5 × 5 cm carbon-silicone electrodes) were placed at the left (F3) and right (F4) prefrontal cortex and a 5 × 7 cm electrode at the vertex (Cz; 10–20 EEG system). Using the XCSITE 100 device (Pulvinar Neuro LLC), 2 mA zero-to-peak, in-phase, IAF-tACS was applied to the prefrontal cortices for 40 minutes while participants watched a relaxing video. Fourteen potential adverse stimulation effects were assessed post-session using a scale from 1 to 4, where 1 indicated the effect was absent and 4 indicated it was severe (headache, neck pain, scalp pain, tingling, itching, ringing or buzzing noise, burning sensation, local redness, sleepiness, trouble concentrating, improved mood, worsening of mood, dizziness, and flickering lights).
EEG analysis
Three minutes of eyes-open resting state EEG data before the study intervention on Day 1 and Day 5 was analyzed using semi-automized preprocessing with custom scripts in MATLAB and EEGlab. 15 Data were high-pass and low-pass filtered (1–40 Hz), common average re-referenced, and cleaned of noisy channels, followed by Independent Component Analysis with combined automatic/manual artifact rejection. Power spectral density (PSD) was estimated using Welch’s method and z-transformed for spatial normalization. Average IAF PSD for our predefined region of interest in the left prefrontal cortex was calculated. Due to the small sample size, analyses focused on descriptive trends rather than inferential statistics to provide preliminary insights.
Results
Participants
We enrolled five participants, one of whom was excluded due to abnormal MRI findings. Four participants with SLE (Age: M = 44.5, all female, HDRS-17: M = 13, SLEDAI-2K: M = 3.5) completed the study. Of these, one received sham tACS, and three received IAF-tACS. We report on the results from participants who received IAF-tACS.
Alpha power change and clinical improvement
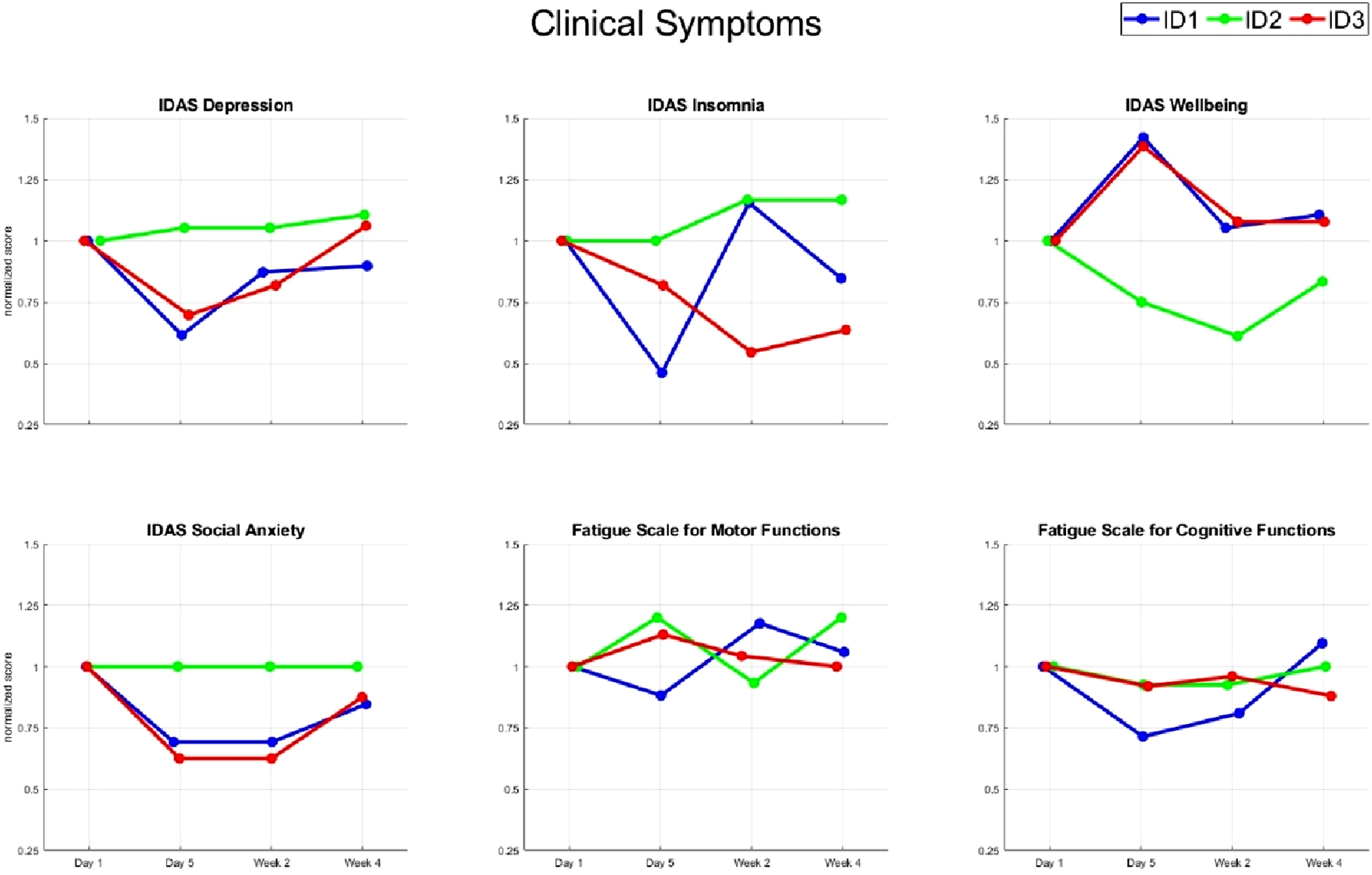
After five days of IAF-tACS, two of three participants (IDs 1 and 3) showed the expected reduction in left prefrontal alpha power (Figure 1). Notably, only these participants reported improvements in IDAS depression, insomnia and social anxiety scores, whereas the third participant (ID 2), who did not show a reduction in alpha power, experienced no symptom relief (Figure 2). However, this clinical improvement was transient, with symptoms gradually returning toward baseline after two and four weeks. IDAS well-being scores exhibited a similar trajectory alongside depression improvement. There was no noticeable impact of tACS on either cognitive or motor fatigue. Individual alpha frequency (IAF) tACS was administered to individuals with systemic lupus erythematosus (SLE). (a) 40 minutes of IAF tACS over five days resulted in a modulation of alpha power. Topoplots show IAF power difference scores (IAF power Day 5–IAF power Day 1) for the three participants. (b) Average individual alpha power for the three participants in the IAF tACS condition on Day 1 and Day 5 for the left dorsolateral prefrontal cortex (DLPFC; circled in black). The y-axis represents the spatially normalized alpha amplitude via z-transformation. Individual participant scores for different clinical symptoms. Scores are baseline normalized and displayed as ratio.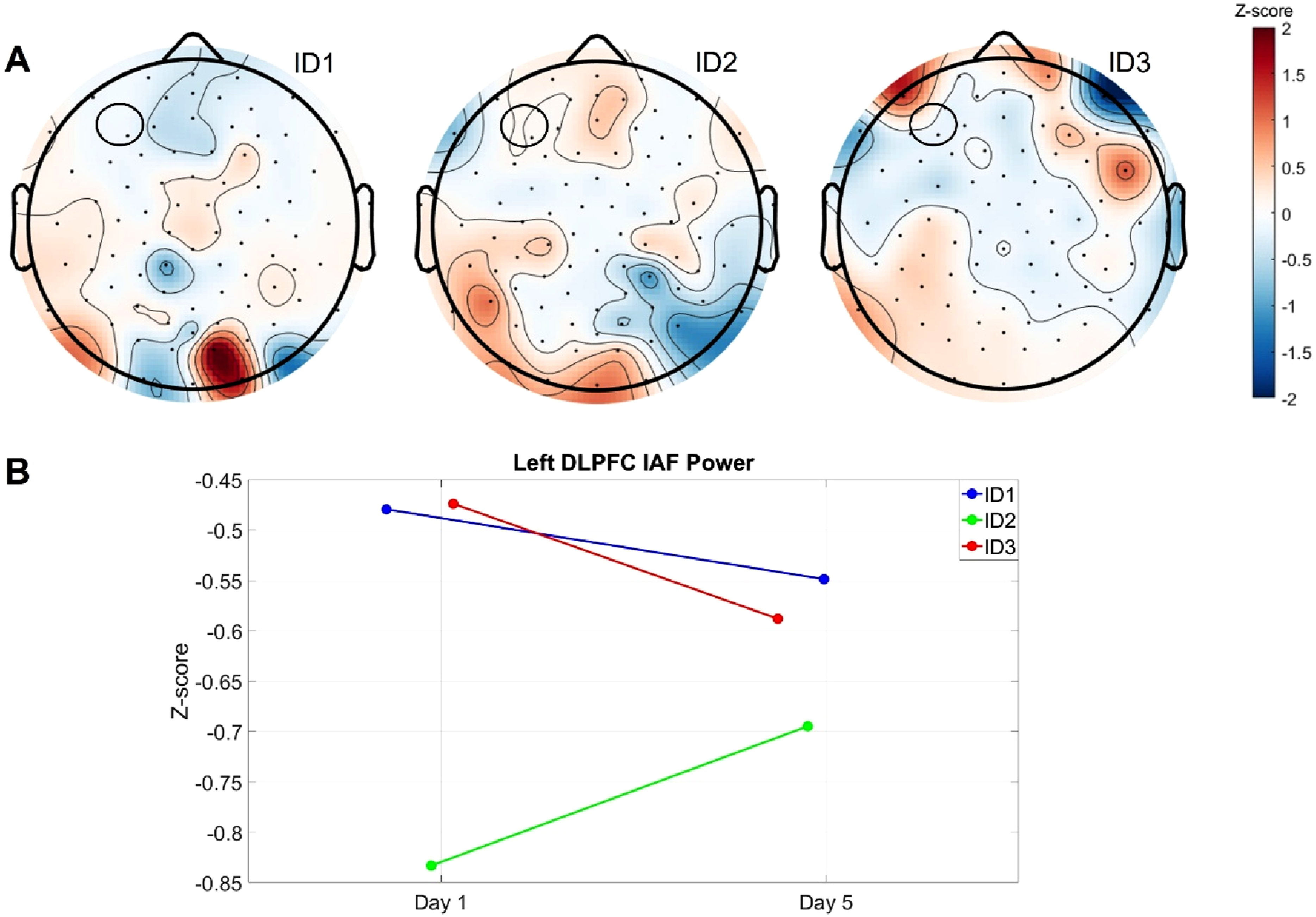
Tolerability
No serious adverse events and no unanticipated adverse events occurred. On average, participants rated adverse stimulation effects as mild (ID1: 1.53, ID2: 1.60, ID3: 1.23). The sham participant also reported mild adverse stimulation effects, with an average rating of 1.37.
Discussion
In line with our hypothesis, two out of three participants demonstrated successful target engagement, evidenced by a reduction in left prefrontal alpha power following five days of IAF-tACS. Notably, only these participants also showed improvements in depression, insomnia, and anxiety symptoms, suggesting preliminary target validation. While these observations are consistent with the proposed link between prefrontal alpha modulation and symptom improvement in SLE, they should be viewed as preliminary evidence from an exploratory, proof-of-principle study. To our knowledge, this is the first investigation of IAF-tACS in individuals with SLE, highlighting the need for further research in larger samples.
The observed clinical improvements were transient, with scores returning close to baseline by the 2-week follow-up. This pattern contrasts with findings from previous depression studies, where peak improvements often emerged 2 weeks post-intervention9,10 and were even maintained up to 3 months in a recent open label trial in MDD. 16 One possible explanation lies in the neuroinflammatory nature of SLE, which is known to affect brain function and structure, including disruptions in large-scale network connectivity. 17 Such altered functional connectivity in SLE—particularly reduced network integration—may diminish the brain’s capacity to sustai externally induced modulation through tACS. In addition to these neural factors, the broader clinical context of SLE may also limit the durability of therapeutic gains. While tACS may have temporarily alleviated depressive symptoms by modulating prefrontal alpha activity, its effects may have been insufficient to sustain improvement against SLE-related chronic somatic symptoms and immune dysregulation. The lack of improvement in core lupus symptoms may have contributed to a return of mood-related distress over time. These observations underscore the importance of addressing both neuropsychiatric and systemic aspects of SLE in future interventions, possibly through longer or combined treatment approaches.
Insomnia scores showed the greatest improvement by Day 5 in two of the three participants. All participants, including the one receiving sham stimulation, watched a relaxing 40-min video prior to each stimulation session, which may have contributed to a general relaxation response. It is possible that the combination of this relaxation period with active tACS produced a synergistic effect.
Participant 2 did not show a reduction in prefrontal alpha power nor clinical improvement. This may be explained by the absence of significant SLE disease activity at the time of participation, suggesting limited expression of the neural pattern that tACS was designed to target. This variability shows the importance of individualized baseline assessment and potential stratification based on clinical or neurophysiological markers.
Importantly, the sham participant showed behavioral improvements comparable to those seen in the active group (Figure S2) but did not exhibit a reduction in prefrontal alpha activity (Figure S1). This dissociation raises the possibility that non-specific factors such as placebo effects, participant expectations, or spontaneous symptom fluctuations may have contributed to the observed changes. To help control for such influences, all participants, including the sham participant, watched a standardized 40-min relaxation video prior to each session. Nevertheless, without a larger sample and appropriate statistical controls, it is not possible to determine whether the effects in the active group were specifically due to tACS.
Despite its early termination, this study provides important preliminary evidence supporting the safety, feasibility, and tolerability of IAF-tACS in individuals with SLE and comorbid depression. The intervention was well tolerated, with no adverse events reported, and all participants completed the stimulation sessions and follow-up assessments. Notably, reductions in prefrontal alpha power were observed following stimulation, and symptom improvements were noted in a subset of participants, indicating that neurophysiological modulation via tACS is achievable in this population. Given the scarcity of non-pharmacological treatment options for neuropsychiatric symptoms in SLE, tACS represents a promising, low-burden, and neurophysiologically targeted intervention with the potential for future home-based applications.
Limitations of this case series should be acknowledged. The study design does not allow firm conclusions about the efficacy of prefrontal alpha modulation for lupus-associated depression. The small sample size, constrained by recruitment challenges during the COVID-19 pandemic, precludes inferential testing and limits generalizability. Accordingly, no statistical analyses were conducted, and findings should be interpreted as preliminary and exploratory. It is not possible to distinguish specific stimulation effects from non-specific influences such as concurrent medication changes, placebo or expectation effects, regression to the mean, and spontaneous fluctuations in SLE disease activity or fatigue. The behavioral improvement observed in the sham participant further underscores the importance of future controlled studies using appropriate statistical models (e.g., ANCOVA, linear mixed-effects) to adjust for potential confounders such as baseline symptom severity, fatigue, and disease activity. Based on prior tACS research in depression, 9 a sample size of approximately 20 participants per group would be required to detect a meaningful change in alpha power with 80% power (assuming d ≈ 1.6, SD = 1.2, α = 0.05). These future studies will be essential for disentangling specific stimulation effects from non-specific placebo responses or natural symptom fluctuations. Given these limitations, the present findings should be regarded solely as preliminary evidence of safety, feasibility, and tolerability, pending replication in adequately powered, randomized, sham-controlled trials with pre-specified statistical models and adjustment for baseline symptom severity, medication use, and disease activity.
As the first study to explore IAF-tACS in individuals with SLE, our findings lay the groundwork for larger, adequately powered trials and provide important preliminary evidence supporting the safety, feasibility, and tolerability of this intervention in a complex and understudied population.
Supplemental Material
Supplemental Material - Exploring prefrontal alpha modulation as a mechanistic treatment target in lupus-associated depression: A transcranial alternating current stimulation study
Supplemental Material for Exploring prefrontal alpha modulation as a mechanistic treatment target in lupus-associated depression: A transcranial alternating current stimulation study by Magdalena Camenzind, Tobias Schwippel, Shruti Saxena-Beem, Yueh Z. Lee, Anthony Trujillo, Roger Huamani, Justin Riddle, Agnieszka Zuberer, Flavio Frohlich, Saira Z. Sheikh in Lupus
Footnotes
Acknowledgments
Generative AI (ChatGPT 4.0) was used for light editing and language improvement. A human author has reviewed and integrated editing suggestions provided by AI to ensure accuracy and appropriateness. The authors would like to acknowledge the study participants for their contributions to advancing science, and all individuals living with lupus for their courage and determination. This project was supported by the Fund for Excellence in Lupus & Sjogren’s and the Virginia A. and Douglas R. Ware Fund for Excellence in Autoimmune Diseases at the UNC Thurston Arthritis Research Center. This project utilized the NC TraCS DSMB, which is funded by the NCATS Clinical and Translational Science Award under CTSA grant Award Number UM1TR004406.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FF receives payments as a consultant from Electromedical Products International (EPI), Insel Spital, and the University of Michigan. SZS has served as a consultant for GSK, Biogen, AstraZeneca, Lilly USA, Cabaletta Bio and Aurinia Pharmaceuticals. The study described here is unrelated to these activities. All other authors report no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Fund for Excellence in Lupus & Sjogren’s and the Virginia A. and Douglas R. Ware Fund for Excellence in Autoimmune Diseases at the UNC Thurston Arthritis Research Center. This project utilized the NC TraCS DSMB, which is funded by the NCATS Clinical and Translational Science Award under CTSA grant Award Number UM1TR004406.
Ethical considerations
The study was approved by the Institutional Review Board of the University of North Carolina at Chapel Hill (IRB #19-0763).
Consent to participate
Written informed consent was obtained from all participants before enrollment.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
