Abstract
Objectives
SLE diagnostic journeys can be protracted, with negative impacts on long-term health. This study explored the role of diagnostic overshadowing (DOS) in delaying SLE diagnoses.
Methods
A qualitative analysis of 268 completed SLE patient surveys and 25 in-depth interviews purposively selected from the 2018-2021 Cambridge University Systemic Autoimmune Rheumatic Disease (SARD) studies.
Results
The majority of participants appear to have experienced DOS and there were indications that sustained DOS (S-DOS) may add years to some SLE diagnostic journeys. Symptom misattributions which contributed to S-DOS included: (1) “Medical mystery”, particularly when the clinician indicated that it was too expensive to keep investigating. (2) Negative misattributions (e.g. “nothing seriously wrong”), often due to a failure to connect multiple symptoms as possible indicators of an underlying condition. (3) Diagnostic roadblocks, including, in the case of some participants, a mental health, psychosomatic, ME/CFS or fibromyalgia (mis)diagnosis. (4) Moral misattributions, such as to “malingering”, which could undermine patient help-seeking and/or clinician help-giving.
Conclusion
Our data suggests that DOS may be an important factor in diagnostic delay in patients with SLE.
Introduction
Early diagnosis and treatment of Systemic Lupus Erythematosus (SLE) can slow disease progression and limit long-term damage. 1 Missteps in the medical system, however, can thwart opportunities for early intervention. Studies indicate a mean time to SLE diagnosis in the UK of between 6 and 7 years from first experiencing symptoms.2,3 Misdiagnoses and other symptom misattributions can contribute to these delays. 4 SLE symptoms have been misattributed to a wide range of medical conditions, 3 as well as to non-clinical characteristics, such as to “growing pains”. 5 Nonetheless, early diagnosis does occur. Nightingale et al., 6 for instance, found a median time to SLE diagnosis (from first musculoskeletal symptoms) of 6 months among 10–19 year olds with mild to moderate disease in the UK, albeit using a different methodology to the studies2,3 cited above. With improvements, including rapid referrals, 1 timely diagnoses such as these could arguably become the norm.
Symptom misattributions and diagnostic delay
Piecing together findings from SLE studies,2–5,7,8 and case reports,9,10 symptom misattributions appear to contribute to diagnostic delay through two principal related processes.
First, a symptom of the undiagnosed disease (in this case SLE) is misattributed during a medical consultation to a previously not suspected patient characteristic or condition, and this reduces or removes the perceived need to continue seeking an explanation for the symptom. Fatigue arising from SLE might, for example, be taken to be a symptom of depression and contribute to a misdiagnosis of Major Depression. Second, the now established belief that the patient has or could have the characteristic/condition overshadows future clinical encounters. In particular, additional symptoms of the still undiagnosed disease may be misattributed to it. This could, for instance, entail the clinician misattributing SLE joint pains to the misdiagnosed Major Depression. The reasoning might be that the pains are a psychosomatic sequela of the depression.
The second described process is ‘diagnostic overshadowing’. Whilst there are a wide range of definitions of ‘diagnostic overshadowing’,11–13 the term is most often used to refer to the misattribution of the symptoms of an undiagnosed condition to a diagnosed one. 13 A limitation of this usage/definition is that the more impactful forms of symptom misattribution can be to undiagnosed but suspected conditions 12 ; non-clinical characteristics 3 ; or assumed behaviours, such as feigning symptoms. 14
Diagnostic overshadowing (DOS) in SLE
Where DOS has been investigated – principally among those taken to have intellectual disabilities or severe mental health conditions – it has been found to be a substantial problem.15,12 It also seems possible that DOS could be a major problem across a wide range of health conditions, including SLE. Our literature review indicated that there may be no published studies which address (with reference to “diagnostic overshadowing” and on the basis of empirical findings) DOS in SLE. There are, however, SLE studies3–5,16 that, whilst not referring to DOS, include a focus on phenomena entailing the misattribution of the symptoms of undiagnosed SLE. Neuro-psychiatric (NP) SLE symptoms are particularly susceptible to misattribution, 3 and so also perhaps to DOS. This appears to arise in part from clinicians underestimating NP symptom prevalence in SLE. 17
Whilst these SLE studies link misattribution to diagnostic delay,3–5,16 they leave largely unaddressed the question of how the misattributions survive multiple clinical encounters, often with senior consultants. With this in mind, and to address the DOS definitional limitations referred to above, we developed an operationalised definition of sustained DOS (S-DOS) (conceived as a DOS subtype) – with a set of requirements (Figure 1) and indicators (Table 5) – and applied it to the analysis of qualitative data from the Cambridge University SARD studies. Operationalised definition of Sustained DOS.
The study aims were: (1) Identify and categorise, from the qualitative survey and interview data, the principal types of misattribution of SLE symptoms. (2) Identify, from among these principal types, some of the more sustained and impactful forms of DOS, along with some of their causes and characteristics.
Materials and methods
Research strategy
Qualitative methods, drawing-upon constructivist grounded theory, 18 were used to identify causal processes. 19 Our approach was pragmatic, with a focus on informing practice rather than on building higher-level theories.
Data collection and analysis
Principal data collection and analysis stages.

Ethical approval
The Cambridge University Psychology Research Ethics Committee approved the primary research studies (PRE 2018-84 and PRE 2019.099) and subsequent analyses of the data, including that in the current study. Participants gave electronic (for surveys) and audio-recorded oral (for interviews) informed consent.
Results
Interviewee characteristics and participant mean times to diagnosis.
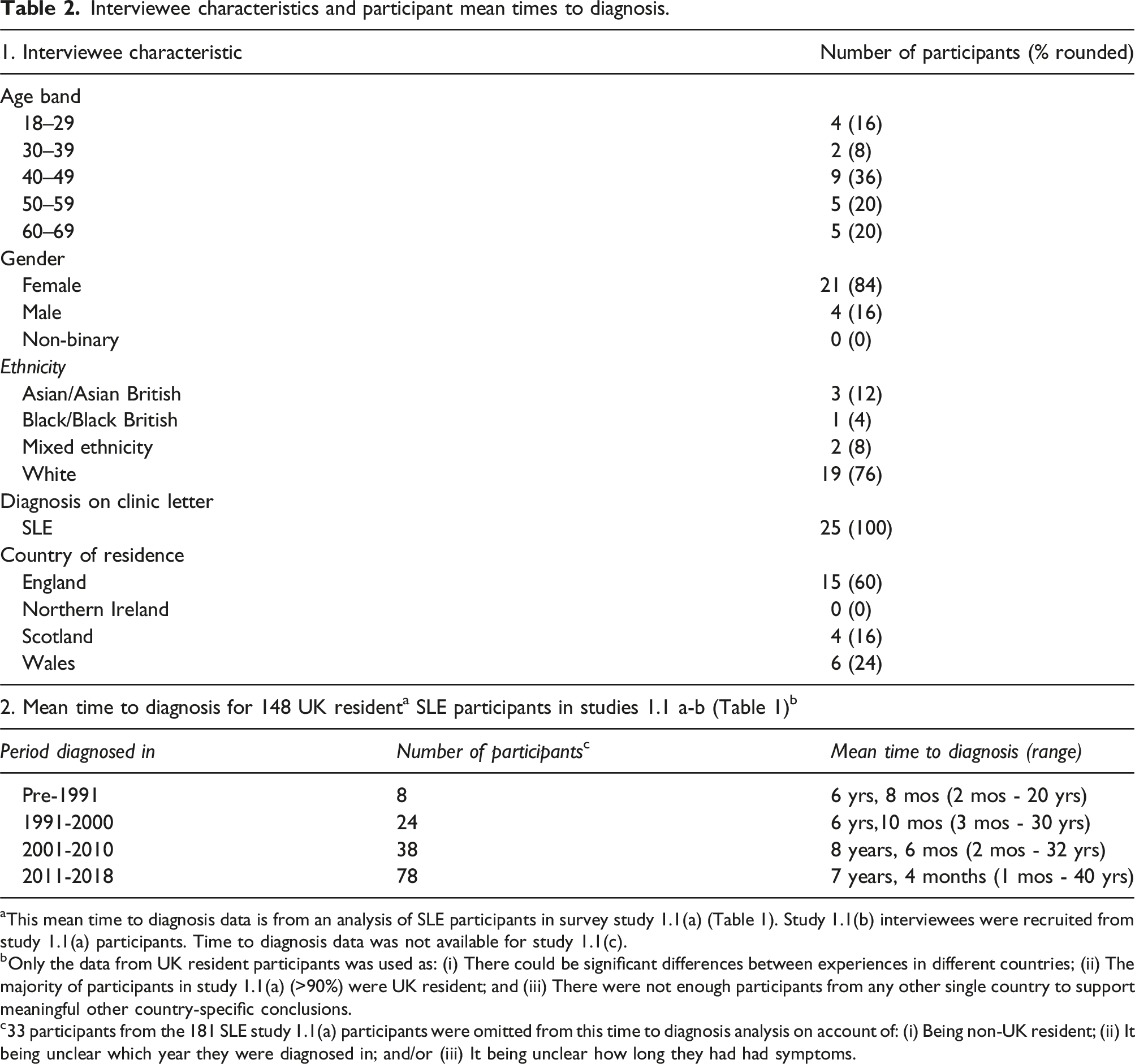
aThis mean time to diagnosis data is from an analysis of SLE participants in survey study 1.1(a) (Table 1). Study 1.1(b) interviewees were recruited from study 1.1(a) participants. Time to diagnosis data was not available for study 1.1(c).
bOnly the data from UK resident participants was used as: (i) There could be significant differences between experiences in different countries; (ii) The majority of participants in study 1.1(a) (>90%) were UK resident; and (iii) There were not enough participants from any other single country to support meaningful other country-specific conclusions.
c33 participants from the 181 SLE study 1.1(a) participants were omitted from this time to diagnosis analysis on account of: (i) Being non-UK resident; (ii) It being unclear which year they were diagnosed in; and/or (iii) It being unclear how long they had had symptoms.
Patient reported misdiagnoses and other misattributions (other than ‘moral misattributions’) with the potential to constitute DOS.

aIt was not possible to verify with clinicians whether they had made these patient reported attributions or whether for instance patients had misinterpreted their intended meanings.
‘Moral misattributions’. a

aMoral misattributions entail clinicians erroneously attributing disease symptoms, symptom reports, and/or other patient conduct, to presumed blameworthy patient motivations or behaviour.
bAs with the attributions in Table 3, there was no opportunity to ask the referred to clinicians about the ‘moral misattributions' which patients reported them as having made.
Some of the principal indicators of possible sustained DOS. a
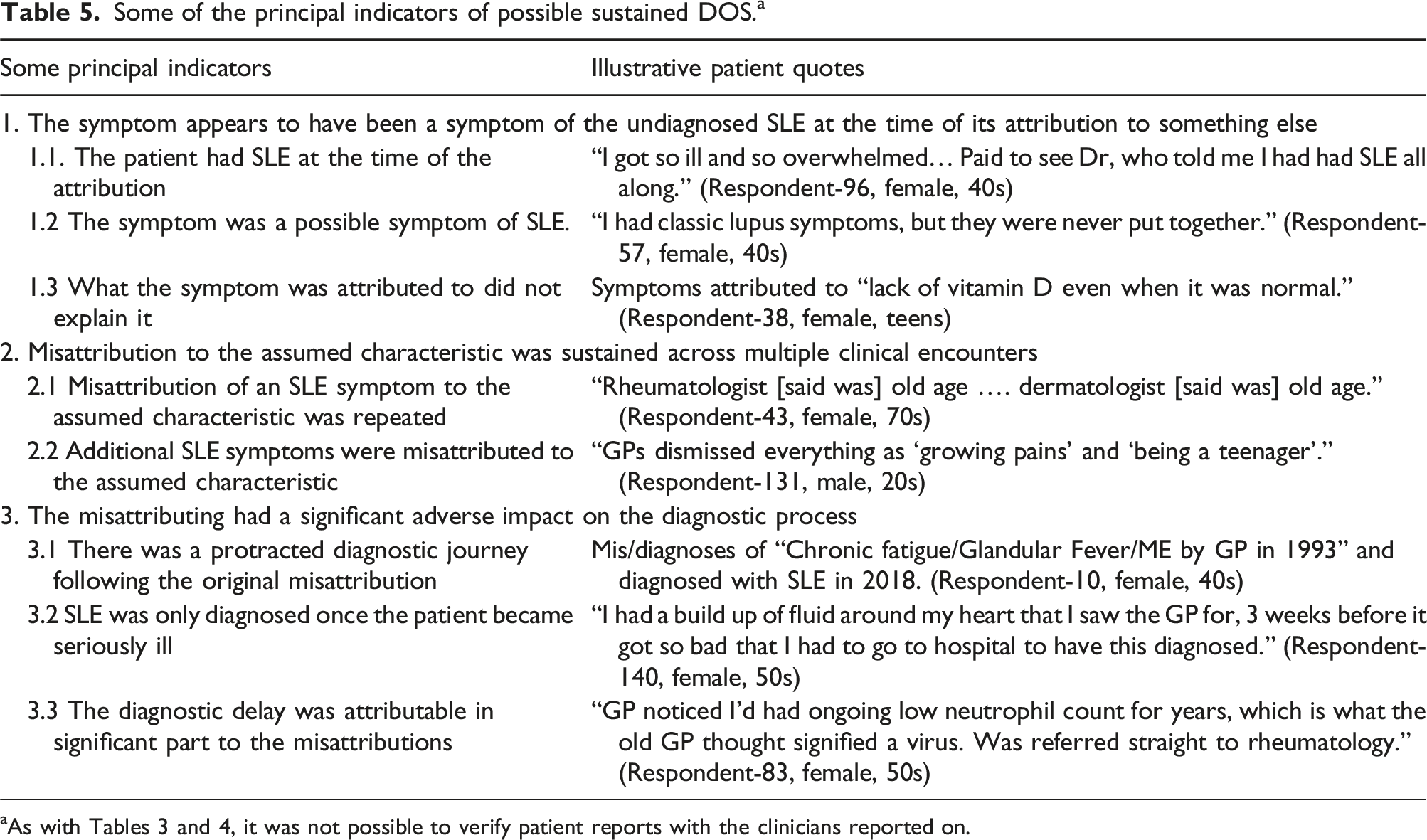
Not all reported instances of misattribution seem to have involved S-DOS. In particular, some attributions were taken to be provisional and investigations continued, and so the attribution did not have (as required in our S-DOS definition) “a significant adverse impact on the diagnostic process” (Figure 1). Respondent-86 (female, 30s), for example, reported: Initially I was diagnosed with juvenile arthritis, but only a couple of days later they diagnosed lupus.
To help assess the likelihood of there having been a misattribution, and of any misattribution having contributed to S-DOS, a set of S-DOS indicators (Table 5) was developed. For instance, Respondent-96 (female, 40s) reported that a lupus expert had told her that she had had SLE at the time of the possible misattributions (matching Indicator:1.1, Table 5); her symptoms (such as joint inflammation) were possible SLE symptoms (Indicator:1.2); and the dermatological condition, to which these symptoms had initially been attributed, did not explain them (Indicator:1.3). These indicators also assisted in the identification of factors that had contributed to S-DOS, such as the misinterpretation of blood results (Table 5/Indicator:3.3), as well as highlighting some DOS impacts.
Misattributions with the greatest impact
Certain types of misattribution, including those described below, appear to have quite often led to S-DOS with a substantial detrimental impact on the diagnostic process. (a) “Medical mystery” and negative misattributions
Some participants reported that their clinicians had labelled symptoms as inexplicable or indicated that it was too expensive to keep investigating them (Table 3/Rows:10.1-10.4). It was, however, the assumed absence of a condition – “nothing seriously wrong” or not a specified disease (Table 3/Rows:1.1-1.3) – which could most powerfully overshadow the diagnostic process. For example, Respondent-89 (female, 30s) wrote: Was being investigated for MS [multiple sclerosis]… and after confirming I didn’t have it, was told by GP it must be stress.
In this and comparable instances, the ascertained absence of one serious condition seems to have been equated with there being no serious condition. In other cases, the investigation appears to have been used to reassure patients, rather than as part of a differential diagnostic process.
(b) Symptoms viewed in isolation
Conflating symptoms and conditions, such as diagnosing “rash” when presented with an SLE indicative “malar rash” (Respondent-113, female, 30s), and not connecting multiple symptoms as possible indicators of an underlying systemic disease, were among the principal problems across the different types of S-DOS. One participant’s advice to clinicians was to “join the dots… if a patient is describing a set of symptoms on a number of visits” (Respondent-35, female, 60s). Patients reported that these unconnected “dots” were attributed to a wide range of clinical conditions and non-clinical characteristics, such as being “too active, not active enough” (Respondent-57, female, 40s). Patients reported having been correctly diagnosed with some of the conditions to which SLE symptoms had been attributed but that these conditions had left some “dots” unexplained. For example, one patient with cystic fibrosis (CF) had felt that some of her symptoms were “very odd for CF, which eventually led to a rheumatology screen revealing lupus specific markers” (Respondent-90, female, 40s).
(c) Psychosomatic and mental health attributions
As opposed to no connection being made between symptoms, they were quite often connected as the assumed manifestations of a psychosomatic or mental health condition (Table 3/Rows:4.1-5.3). These psychological misattributions could be resilient in the face of visible physical symptoms, as Respondent-212 (female, 30s) found: GP told me I was suffering with anxiety... when I went with swollen legs.
In addition, what the clinician labelled as “an element of anxiety” (Interviewee-14, female, 30s) could in combination with patient-clinician interactions – such as the patient insisting that there is an undiagnosed illness – lead to a more formal diagnosis of health anxiety. (d) SLE ‘diagnostic roadblocks’
Certain diagnoses had a particular tendency to stall the SLE diagnostic process. It is recognised that a substantial number of participants had, and believed that they had, some of these conditions (see Supplemental Material on fibromyalgia). Nonetheless, many participants indicated that the misattribution of SLE symptoms to these conditions (whether extant or not) had substantially delayed their diagnostic journey. Among these ‘diagnostic roadblocks’ were mental health (MH) and psychosomatic diagnoses, but also a number of conditions – including fibromyalgia and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) – which were said to have been conceptualised by some clinicians as comparable to MH and/or psychosomatic conditions. For example, Respondent-173 (female, 40s) reported that she was diagnosed with ME in 2005 and added: nothing then looked at properly because of this ME diagnosis… Was not diagnosed [with SLE] until 2013.
In addition, diagnosis of these conditions appeared to quite often be problematic, such as diagnosing fibromyalgia “when no exam had been done…” (Respondent-103, female, 40s). Patients pointed to a number of factors (other than their symptom presentations) that may have contributed to these diagnoses. These included clinicians being confident to make them without a referral, and treatment for them being associated with limited perceived risk, such as “go to the gym” (Respondent-131, female, 20s). In addition, there appeared to be a willingness to proceed without a clear diagnosis. For example, Respondent-135 (female, 50s) reported: GP diagnosed me as ‘nervous housewife’... Prescribed a sedative.
In contrast, clinicians were reported to be “reluctant to diagnose lupus” (Respondent-237, female, 40s), which several respondents suggested may have been because of the risks associated with autoimmune medications. It was also hard for patients to challenge some of these ‘roadblock’ diagnoses, including when there was no formal diagnosis to challenge, such as “anxiety issues” (Respondent-173, female, 40s).
Moral misattributions
For the purposes of our study, ‘moral misattributions’ entail clinicians erroneously attributing symptoms and/or patient conduct to presumed blameworthy patient motivations and/or behaviour. A number of principal categories of moral misattributions were identified, including: • Dishonesty (Table 4/Row:1.2). Patients quite often felt that they were being accused of exaggerating symptoms or even “faking it to some degree” (Interviewee-17, female, 50s). Motivations clinicians were said to have ascribed to these presumed behaviours (Table 4/Rows:2.1-2.2) included attention seeking or avoiding anxieties or effort, such as being “school-shy” (Respondent-52, female, 40s). Patients with neurological symptoms, such as “vertigo” (Respondent-169, female, 20s), appeared to be particularly vulnerable to clinician disbelief. • Mental illness conceptualised as moral weakness (Table 4/Row:2.3). This included clinicians being reported to have implied that patients should get a grip and be less depressed. • Being difficult (Table 4/Rows:3.1-3.3). Patient behaviour that clinicians were reported to have taken to be “difficult” included pushing for tests/referrals, disagreeing with the clinician, and/or in-putting their own diagnostic suggestions. Being “difficult” appears, however, to have often led towards the correct SLE diagnosis. • Being to blame for the illness (Table 4/Rows:4.1-4.3). This included patients being blamed for contributing to the illness, such as through an unhealthy lifestyle (Respondent-142, female, 50s), or through not treating it, such as not taking anti-depressants for SLE symptoms misattributed to depression.
Moral misattributions could have a negative impact on the diagnostic process through providing incorrect explanations for symptoms that might otherwise have led to the SLE diagnosis. Moral misattributions could also undermine patient help-seeking and/or clinician help-giving. This included the impact of articulated moral misattributions on patient self-concept, and/or on patient-clinician relationships, reducing patient engagement with clinicians or its forcefulness. In addition, moral misattributions, such as taking the patient to be in part to blame for their illness, could reduce the help that clinicians were prepared to provide. Clinicians could also be less willing to engage with patients taken to be difficult, whilst being difficult could in itself be interpreted as evidence of a MH disorder. However, whether being ‘difficult’ – as in pushing for answers – slowed-down or speeded-up the diagnostic process seemed to depend in part upon the patient’s social position. For example, Respondent-245 (female, 50s) reported: a friend of a friend who’s a GP had a long chat with my GP who then agreed my symptoms were serious and I was referred to a neurologist.
Moral misattributions could also influence social status and reinforce power imbalances. For example, with several (at the time) children (Respondent-52, female, 40s; Interviewee-27, male, 20s), the misattribution of SLE symptoms to “laziness” seems to have reduced their social standing with, and support from, the medical system and/or family. This slowed down their diagnostic journeys and rendered them more distressing.
Discussion
As far as we are aware, this is the first published empirical study which addresses “diagnostic overshadowing” (DOS) in SLE. We applied an operationalised definition of sustained DOS (S-DOS) to the qualitative answers from 268 completed patient surveys and the transcripts of 25 in-depth interviews. Experiences of S-DOS were common among study participants. Misattributions which contributed to S-DOS appear to have had a particularly detrimental impact on the diagnostic process, principally because the misattributions influenced multiple subsequent clinical encounters (in some cases for over a decade). For instance, once the early symptoms of some participants’ undiagnosed SLE had been misattributed to fibromyalgia, the consequent fibromyalgia diagnosis provided an explanation for some of their later SLE symptoms. This could help explain Cornet et al.’s finding that an initial mis/diagnosis of fibromyalgia was associated with a 5 year increase in the median SLE diagnostic delay. 16 We acknowledge that early childhood trauma may be a shared risk factor for fibromyalgia 25 and SLE. 26 Hence, there exists the possibility of developing fibromyalgia many years prior to developing SLE.
Whilst the literature focuses on DOS among individuals with a serious mental illness, 27 or with intellectual disabilities, 28 our study indicates that DOS could be an equally powerful force among patients without these conditions/characteristics. Furthermore, whereas much of the literature frames DOS as something which happens in one location at one time, and in particular during a visit to the emergency department, 12 our findings suggest that DOS tends to be a process over time and across settings. This is why the perspective of the patient – who is in general the only one ‘there’ across the whole diagnostic journey – can be critical. With a few exceptions,28,29 this perspective has been neglected in DOS studies. Our findings also suggest that one form of DOS (such as attributing joint pains to anxiety) can morph into a more durable form (such as a diagnosis of health anxiety) if the patient fails to accept the clinician’s misattribution as a sufficient explanation for their symptoms.
Typology of S-DOS
We identified circumstances and types of misattribution which appear to have quite often led to S-DOS with a substantial detrimental impact on the diagnostic process. These include: (1) Negative misattributions, unconnected symptoms, and medical mysteries. A principal facilitator of negative misattributions, such as “nothing seriously wrong”, was a failure to connect multiple symptoms as possible indicators of an underlying systemic condition, a failure also noted in other studies.
30
Even when the symptoms were connected, it was often as the assumed manifestations of a psychological illness. In other cases, clinicians were reported to have been content to leave the symptoms and/or the patients as an insoluble “medical mystery”. (2) Diagnostic roadblocks. Mental health, psychosomatic, ME/CFS and fibromyalgia diagnoses - sometimes based upon the misattribution of the symptoms of undiagnosed SLE - had a particular tendency to stall the SLE diagnostic process. That these diagnoses were difficult to challenge, but relatively easy to make, contributed to their power to overshadow. For example, consistent with the literature,
31
fibromyalgia was frequently diagnosed without a full history or exam. (3) Moral misattributions. These entail clinicians erroneously attributing symptoms or patient conduct to presumed blameworthy patient motivations and/or behaviour (such as being “difficult” or “faking” symptoms).
Moral misattributions can disrupt the diagnostic process through undermining patient help-seeking and/or clinician help-giving. Some patients, for instance, reported being treated as less deserving of help on account of being regarded by clinicians as in part to blame for their symptoms. This seems consistent with Weiner’s attribution-emotion-action model. 32 In addition, consistent with studies of self-stigma, 33 some patients appear to have internalised moral misattributions (such as thinking that maybe they were “malingering”), which left them feeling less able to push for further investigations. Moral misattributions could also draw-on or add to power imbalances. For example, with several (at the time) children, the misattribution of SLE symptoms to “laziness” seems to have reduced their social standing with and support from the medical system and family.
These findings highlight the negative impact of DOS on quality of care and outcomes, as well as providing examples of DOS entailing “epistemic injustice”. 34
Participant insights and the literature suggest some possibilities for addressing S-DOS. For instance, the reported clinician reluctance to make an SLE diagnosis, for fear of autoimmune medication related risks, seems consistent with an omission/inaction bias, 35 and so may be amenable to “cognitive debiasing”. 36 Other problems and related solutions – such as improving clinician autoimmune knowledge 3 and longer consultations 37 – could also be pertinent. In addition, there appeared to be important medical system issues. Of particular note, whilst GPs as gatekeepers can be critical in reaching a correct diagnosis, 38 patient reports indicated that GPs may not in general actively plan, coordinate or drive forward the diagnostic process. This may, for instance, help explain why momentum tended to stall with a negative Multiple Sclerosis test, rather than it leading to the next step in the differential diagnosis. It may also be why patients were often left to advance their own diagnostic journeys, which could lead to clinician perceptions of them as difficult and/or to a health anxiety diagnosis.
Strengths and limitations
Whilst qualitative methods were well suited to identifying S-DOS processes, 19 it would be useful if future research could explore their frequency across a more representative sample of SLE patients. Purposive sampling of participants to interview (from survey respondents) helped to ensure a reasonable range of interviewee characteristics, including length of diagnostic journey. Nonetheless, shortfalls in survey representativeness could (even from a qualitative perspective) limit the “transferability” 39 of our findings. These shortfalls included sampling biases arising from having recruited through online support groups and there being a considerable under-representation of survey respondents who were male or non-white. This is of particular importance, as SLE symptomology and healthcare experiences can vary with gender and ethnicity. 40 In addition, whilst participants reporting an SLE diagnosis on their clinic letter was an inclusion criteria, there was in most cases insufficient symptom information to verify the SLE diagnoses. There may also have been significant recall bias.
A major limitation of our S-DOS analysis was that it was based on patient perceptions alone. Some participants may have misinterpreted clinician attributions and some reported misattributions of SLE symptoms could have been correct attributions of non-SLE symptoms. Further, no one can be an entirely neutral observer of their own diagnostic journey. This, however, appears to have been more of an issue with broader patient judgements than with event specific information (such as whether fibromyalgia was diagnosed without a physical exam), and it was the latter type of information that our analysis focussed on. It is also important to acknowledge the potential for bias arising from researcher “positionality”. 41 This was mitigated by the study team having a broad mix of characteristics (e.g. clinicians, academics and autoimmune patients), and through reflexivity practices. 41 Nonetheless, other groups of authors may have interpreted the data differently. Another limitation is that we did not explore in any detail whether the experiences of participants diagnosed more than a decade ago differed from those with a more recent diagnosis. In addition, we had limited data on comorbidities. More details on the data collection limitations are reported elsewhere3,21,22 and more on the analysis limitations are provided in Supplemental Material (section 3).
Our study provided valuable patient-data derived insights into some of the principal causes and consequences of SLE symptom misattributions across our study cohort. However, further research – including with clinician interviews and more representative samples – is needed before our findings can be regarded as other than provisional if generalised/transferred to different and larger groups of SLE patients.
Conclusions
Our study suggests that diagnostic delay and sustained DOS (S-DOS), which appear to be common in SLE, need to be understood as in part the result of patient-clinician attitudes, motivations and interactions, and that misattributions can contribute to and arise from all of these. In addition, the factors that seem to contribute to S-DOS in SLE, such as the prevalence of neuro-psychiatric symptoms, also occur in other autoimmune diseases. Therefore, whilst the literature tends to focus on DOS in mental illness, our study suggests that it could be an equally powerful force across a wide range of autoimmune diseases.
Supplemental Material
Supplemental Material - Diagnostic overshadowing in systemic lupus erythematosus (SLE): A qualitative study
Supplemental Material for Diagnostic overshadowing in systemic lupus erythematosus (SLE): A qualitative study by Rupert Harwood, Chris Wincup, David D’Cruz and Melanie Sloan in Lupus
Footnotes
Acknowledgments
We would like to thank all the study participants; and thank the following people for their work in relation to data collection for the original studies and/or for providing feed-back on draft manuscripts: Colette Barrere, Eva (Bic Shan) Liu, Michael Bosley, James Brimicombe, Jenny Dimond, Wendy Dimont, Dr Bridie Evans, Professor Caroline Gordon, Paul Howard, Les James, Dr Ashra Khanom, Dr Elliott Lever, Jude Machin-Smith, Professor Felix Naughton, Dr Alison Porter, Peter Smith, Chanpreet Walia, and Angela Williams. We would also like to thank Lupus UK for funding the studies which produced the data that we analysed; and thank the academic reviewers, Lupus editors and Sage editorial staff.
Author contributions
MS conceived and planned the 2018-2021 Cambridge University SARD studies (with contributions from DC, RH, CW and others); MS obtained funding and ethics approval; MS conducted the interviews and distributed/administered the surveys; MS, RH, DC, CW and others designed the surveys; RH conceived the diagnostic overshadowing study; RH, MS, DC, and CW developed the study aims; RH and MS sampled the data; RH analysed the data, with MS second-coding a sample; DC and CW contributed clinical advice on the analysis; RH wrote the first and subsequent drafts of the manuscript; MS, DC and CW reviewed and edited the manuscript; and all authors approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the LUPUS UK provided funding towards the collection of the data (2018-2021) which this 2023 study analysed a purposely selected sample of.
Data Availability Statement
Additional anonymised data, including that showing the development of concepts, is available on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
