Abstract
Objective
Systemic lupus erythematosus (SLE) is a heterogeneous autoimmune disease that predominantly affects females. SLE poses risks to the mother and fetus during pregnancy. Information on infection rates during pregnancy in patients with SLE is scarce. Therefore, this study assessed infection risk during pregnancy in women with and without SLE, and identified the factors influencing susceptibility to infections.
Methods
We conducted a retrospective cohort analysis using medical records from Tawam Hospital, Abu Dhabi, United Arab Emirates. Emirati women diagnosed with SLE who experienced one or more pregnancies between January 2010 and August 2023 were included in the study. Patients with SLE were compared with an age-matched control group of women without SLE. The control group was systematically selected from the hospital records of the same period. Demographic details, clinical data, and comprehensive infection histories during and before pregnancy were examined. Univariate and multivariate regression analyses were used to identify the factors contributing to the risk of infection in pregnant women with SLE.
Results
Data from 87 pregnancies in 41 women with SLE were analyzed. The mean age at conception was higher in women with SLE compared to the control group. The rates of infections (54.7% vs 29.8%), adverse fetal outcomes (48.3% vs 24.2%), and maternal outcomes (71.3% vs 43%) were significantly higher in the SLE group than in the control group. Respiratory infections were the most prevalent infection type. No variable was found to increase the risk of infection; however, hydroxychloroquine use during pregnancy was significantly associated with a reduced risk of infection.
Conclusions
The risks of infection and adverse outcomes are significantly higher in pregnant women with SLE than in the non-SLE population. Hydroxychloroquine use during pregnancy was crucial in reducing infection risk. These findings underscore the need for targeted interventions and a multidisciplinary care approach to improve pregnancy outcomes in women with SLE.
Key messages
What is already known on this topic
• Systemic lupus erythematosus (SLE) significantly impacts pregnancy outcomes; however, detailed data on infection rates during pregnancy compared to the non-SLE population are scarce.
What this study adds
• This study provides evidence that pregnant women with SLE have higher infection rates than those without SLE and demonstrates that hydroxychloroquine use during pregnancy significantly reduces the risk of infection.
How this study might affect research, practice, or policy
• The results of this study highlight the importance of hydroxychloroquine in reducing infection risk during pregnancy in women with SLE. Future research should focus on optimizing SLE management by incorporating infection surveillance and evaluating the protective role of medications like hydroxychloroquine. In clinical practice, revising care protocols to ensure consistent use of protective therapies and monitoring for infections may improve outcomes for pregnant women with SLE.
Introduction
Systemic lupus erythematosus (SLE) is a heterogeneous, multifactorial, multisystem autoimmune disease that mainly affects women. SLE is influenced by genetic factors, environmental triggers, and hormonal influences. 1 Given the multifaceted nature of SLE and its prevalence in females, the repercussions of the disease are particularly pronounced during pregnancy. Maternal and fetal outcomes are adversely affected by SLE. 2 The elevated risks to maternal and fetal health include increased maternal hypertension, pre-eclampsia, eclampsia, infections, preterm deliveries, and intrauterine growth retardation.3,4 These complications may be inherently linked to the disease pathology and may be influenced by immunomodulatory medications employed to manage SLE.5–7 In addition, these complications may be influenced by age, smoking history, and prior history of infection. 8
Patients with SLE are at an increased risk of infection compared with the general population. 9 However, the risks of pregnancy-associated infections in women with SLE are not well-documented. One study reported an increased risk of peripartum infection and antibiotic exposure in women with SLE; these complications may lead to neonatal infections and preterm births. 10 Given the limited data on this subject, the aims of this study were to ascertain the prevalence of infections in pregnant women with and without SLE, compare the risks of infection in SLE patients before and during pregnancy, and identify factors associated with increased susceptibility to infections in women with SLE. Enhancing our understanding of the impact of SLE on pregnancy-related complications will provide insights into the effects of medications, support targeted interventions, and underscore the necessity for multidisciplinary care to improve outcomes for this high-risk group.
Methods
We conducted a retrospective cohort analysis of medical records at Tawam Hospital, which integrates records from all Abu Dhabi Health Services (SEHA) hospitals and primary healthcare clinics across the Emirate of Abu Dhabi, United Arab Emirates (UAE). The diagnosis of SLE was based on the American College of Rheumatology criteria. The study included all women who were diagnosed with SLE, followed in the rheumatology clinics of Tawam Hospital, and were pregnant between January 2010 and August 2023. Exclusion criteria included incomplete records or pregnancies managed outside Tawam Hospital.
For the control group, a systematic sampling method was employed to ensure an unbiased comparison. We compiled a list of all pregnant women at Tawam Hospital during the study period. The records were organized by age to match the age distribution. Within each age cohort, the first patient in alphabetical order was selected.
Demographic and clinical data included age at diagnosis, disease duration, age at conception, comorbidities (using Charlson’s Comorbidity Index), medications used before and during pregnancies, and adverse maternal and fetal outcomes. Clinical variables included SLE organ involvement and disease activity, which were assessed using the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI). The SLEDAI provided a quantifiable measure of disease activity before pregnancy and during each trimester. For the comprehensive capture of infection data, all documented infections were extracted from medical records across all SEHA facilities. Infections were categorized by type (e.g., respiratory, genitourinary) and timing (specifying the trimester). Infections in SLE patients in the year before pregnancy were also included in the data analysis.
This study was approved by the Tawam Human Research Ethics Committee. Patient consent was waived due to the retrospective nature of the study and the use of de-identified data.
Data management
Data entry and management were facilitated using REDCap (Research Electronic Data Capture), a secure web-based application designed to support data capture for research studies. 11 This system was utilized to systematically collect, organize, and maintain all relevant study data, ensuring accuracy and accessibility for analysis.
Statistical analyses
The incidence rates of infections during pregnancy were calculated for both the SLE and non-SLE groups. For continuous variables, Student’s t-tests were used to compare the means between groups. For categorical variables, odds ratios were calculated using cross-tabulations to assess the strength of association between the groups; Chi-square tests or Fisher’s exact tests were used to determine the statistical significance of differences observed in the cross-tabulations.
Univariate regression analyses were conducted to identify variables significantly associated with infection risks during pregnancy, focusing on factors that were clinically relevant or supported by prior research. Variables that achieved a significance level of p < .05 were included in subsequent multivariate regression analyses to adjust for potential confounders and better understand the factors influencing infection risks. Additionally, variables with substantial clinical importance, such as “steroid use during pregnancy,” were included in the multivariate analysis to comprehensively assess their effects. Furthermore, age and comorbidity were included in the multivariable analysis despite not being significant in the univariate analysis, to ensure a thorough evaluation of all potential factors. Age and comorbidities are well-known determinants of health outcomes and help provide a comprehensive understanding of the risk factors for infection during pregnancy.8,12
Results
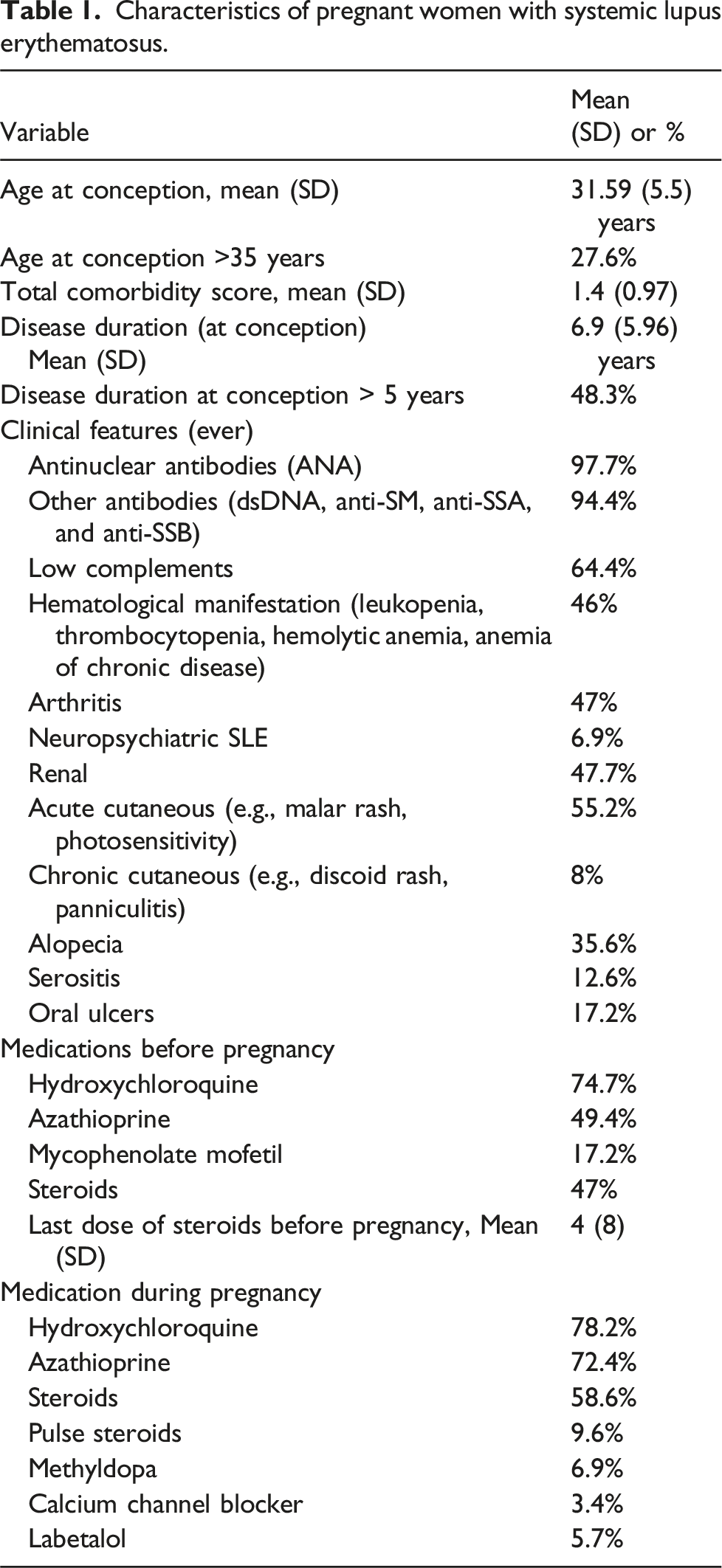
Characteristics of pregnant women with systemic lupus erythematosus.
Comparison of pregnancy outcomes between women with and without SLE.

N/A: Not applicable.
*Adverse fetal outcomes included: preterm delivery, low birth weight, stillbirth, miscarriage, and fetal anomalies.
**Adverse maternal outcomes included: pregnancy-induced hypertension; pre-eclampsia; gestational diabetes mellitus; and postpartum hemorrhage.
Infection types in patients with systemic lupus erythematosus during pregnancy and the year before pregnancy.

§Other infections include categories not listed such as neurological and musculoskeletal infections.
*indicates the count of antibiotic treatments administered across all reported infection episodes.
**indicates the total instances where infections led to hospital admissions.
Risk of infections per pregnancy among patients with systemic lupus erythematosus before and during pregnancy.

*Odds ratios compare the risk during pregnancy to the year before pregnancy.
Univariate analysis of factors influencing infection risk in pregnant women with systemic lupus erythematosus.
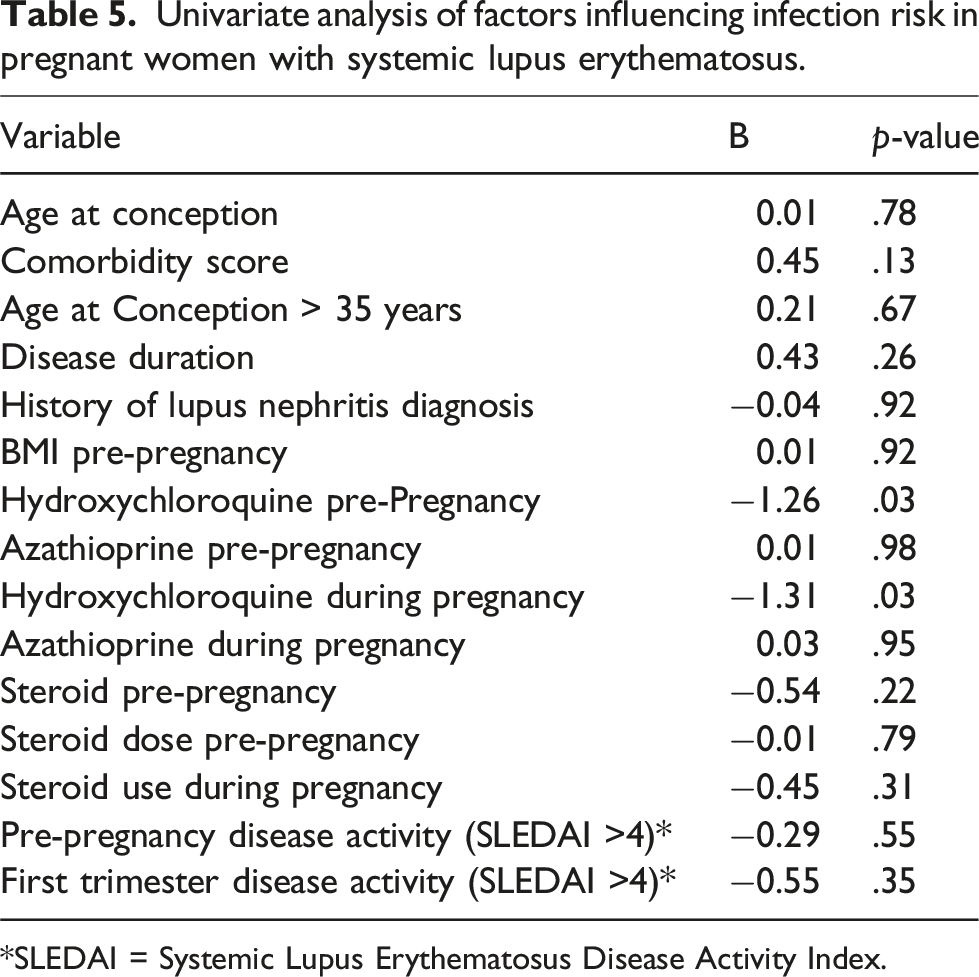
*SLEDAI = Systemic Lupus Erythematosus Disease Activity Index.
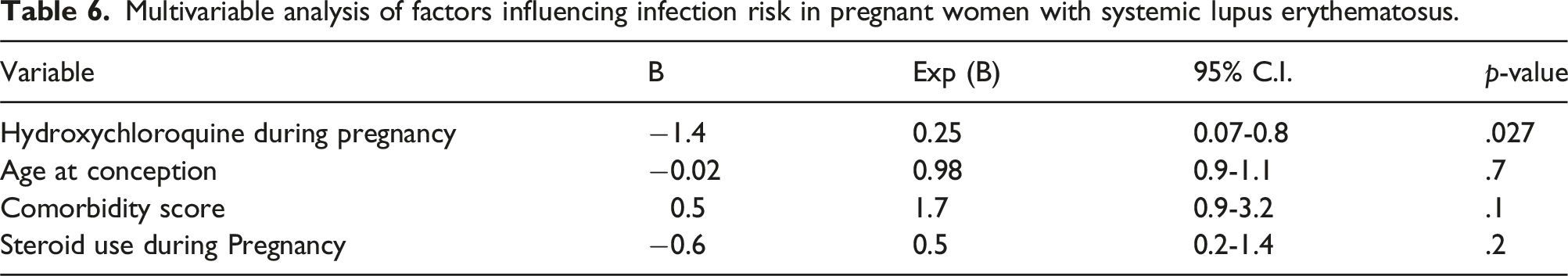
Multivariable analysis of factors influencing infection risk in pregnant women with systemic lupus erythematosus.
Discussion
The results from this retrospective cohort study from the UAE demonstrated that pregnant women with SLE have a higher incidence of infections than pregnant women of similar age without SLE. The risk was approximately twice as high in the SLE group, indicating significant susceptibility to infectious diseases during pregnancy. In particular, pregnant women with SLE had higher rates of respiratory and urinary tract infections compared to those without SLE. However, in SLE patients, infection rates remained stable before and during pregnancy, indicating that pregnancy itself did not increase infection susceptibility in this group. Furthermore, our data revealed that the use of hydroxychloroquine is associated with a protective effect against infections in pregnant women with SLE. Interestingly, we did not identify any factors that were significantly associated with an increased risk of infection in this population. These findings are critical for developing targeted strategies to manage infection risk and improve outcomes for both mothers and their infants.
Our research findings indicate that susceptibility to infections increased approximately twofold during pregnancy in women with SLE compared with women without SLE. This finding agrees with the National Inpatient Sample data, which documented a heightened risk of infections, especially sepsis and pneumonia, in women with SLE. 13 Moreover, our results align with various population studies. For instance, a retrospective cohort study from Brazil reported an infection incidence of 22.6% during pregnancy in women with SLE, supporting the finding of an elevated risk of infection in these women. 14 Conversely, a prospective multicenter study focusing on pregnant women with stable lupus nephritis found that 5.6% of the subjects experienced severe infections requiring hospitalization. 15 Similarly, Wu et al. reported a 0.9% prevalence of peripartum infections, which occurred between the onset of membrane rupture or labor and the 42nd postpartum day in pregnant women with SLE; the infection rate in women with SLE was only marginally higher than the infection rate in women without SLE counterparts (no infections). 16 Moreover, a meta-analysis by Bundhun et al. highlighted the significant post-partum infection risks for women with SLE, which greatly exceeded the risks in women without SLE. 2 Collectively, these studies emphasize a consistent pattern: women with SLE are at an elevated risk of infection during pregnancy and during peripartum and postpartum periods. These findings suggest that SLE may predispose the immune system to increased vulnerability to infections, reinforcing the need for vigilant monitoring and management of these patients throughout pregnancy and beyond. Although this was not accounted in the current study, the literature indicates that pregnancy in a population at high risk for infection may lead to recurrent pregnancy loss, premature rupture of membranes, and a high incidence of preterm deliveries.17,18 These complications underscore the critical need for diligent infection management to ensure healthier outcomes for mother and child.
During pregnancy, all women experience an increased risk of infection because of various immunological, hormonal, and structural changes. The maternal immune system shifts from T helper 1 to T helper 2 dominance to accommodate the fetus. These immune system changes reduce the mother’s ability to effectively combat infections. 19 Hormonally, elevated levels of progesterone and estrogens during pregnancy often suppress immune responses, thereby increasing susceptibility to infections.20,21 Structurally, the expansion of the uterine cavity during pregnancy can predispose women to ascending infections from the lower genital tract. 22 These factors collectively increase vulnerability to a wide array of pathogens that impact maternal and fetal health. 17
Our results show that the risk of infection during pregnancy in women with SLE is similar to that in the year before pregnancy, underscoring the heightened infection risk associated with SLE. Patients with SLE face a threefold greater risk of severe infections compared to the general population, largely due to immunosuppressive therapy and inherent immunological dysfunction. 8 Immunological dysfunction, exacerbated by disease activity, manifests as diminished T helper cell responses to viral antigens and frequent occurrences of neutropenia, lymphopenia, and hypocomplementemia, all of which heighten susceptibility to infections12,23. Moreover, chronic inflammation and corticosteroid use further impair immune responses, increasing the vulnerability of women with SLE to common and opportunistic pathogens. 8 Additionally, organ involvement and comorbidities in SLE patients complicate clinical management, resulting in a higher likelihood of severe infections, such as pneumonia and tuberculosis.8,23 Notably, the lower incidence of respiratory infections during pregnancy compared to the pre-pregnancy period suggests that disease activity, medication adjustments, or pregnancy-associated immune adaptations may influence infection risk in this population.
In our study, respiratory tract infections, followed by UTIs, were the most common infections during pregnancy in women with SLE. In the general population, UTIs are the most common bacterial infection during pregnancy. 24 According to a study from Brazil, UTIs were the most prevalent infections among pregnant women with SLE, accounting for 14.5% of infections, followed by respiratory tract infections at 6.8%. 14 Less frequent infections in other systems were also detected, including gastrointestinal and skin infections. In contrast, a national study using the Nationwide Inpatient Sample demonstrated that serious infections like sepsis and pneumonia are significant concerns; these infections were several times more likely among pregnant women with SLE than among pregnant women without SLE. 13 Furthermore, peripartum infections of the genital tract were also observed, underlining the need for the management of patients with SLE throughout and after pregnancy. 16 These findings illustrate a diverse infection profile in SLE pregnancies, highlighting the need for targeted monitoring and management strategies to effectively address these specific types of infections.
In our detailed examination of infection risks during pregnancy among women with SLE, no factors associated with elevated infection risks were identified. Our multivariable regression analyses revealed that hydroxychloroquine use during pregnancy, was the primary protective factors against infections in pregnant women with SLE. This protective effect aligns with previous research indicating that hydroxychloroquine reduces infection risk in patients with SLE due to its immunomodulatory properties and antimicrobial effects. 23 Barber and Clarke (2020) emphasize that hydroxychloroquine has antibacterial, antifungal, and antiviral activities, and its use is associated with a decreased risk of infections in SLE patients, 23 though most of the evidence pertains to non-pregnant populations. Although some studies in pregnant women with SLE did not find a significant association between hydroxychloroquine use and infection risk, 14 the overall evidence supports its role in decreasing infection susceptibility. These findings underscore the need for continued hydroxychloroquine use during pregnancy to optimize maternal health and reduce infection-related complications in this vulnerable group. Additionally, hydroxychloroquine use during pregnancy has been shown to reduce disease activity 25 and lower the risk of adverse pregnancy outcomes, such as preterm births and intrauterine growth restriction. 26
Steroids use did not emerge as significant factors contributing to infection risk in the multivariable analysis. This result conflicts with several studies demonstrating the role of higher steroid doses in increasing infection risks. For instance, in a study of 221 pregnant women with SLE, a higher prednisone dosage (≥10 mg) was associated with an increased likelihood of infections. 14 Furthermore, an investigation of infection risk during pregnancies in women with autoimmune conditions demonstrated that elevated steroid doses were significantly linked to serious infections, accentuating the importance of meticulous steroid management. 27 A meta-analysis by Bundhun et al. demonstrated that susceptibility to postpartum infections among patients with SLE was significantly influenced by comorbidity scores, 2 however, we did not observe a similar association in our study. Collectively, these studies underscore the interplay between SLE disease characteristics, therapeutic regimens, and infection risks during pregnancy. Rheumatologists should adopt an interdisciplinary approach to managing pregnant patients with SLE. This strategy should integrate specialized obstetric care, tailored immunosuppressive therapy adjustments, and proactive infection surveillance.
This study, conducted as a retrospective analysis of medical records at Tawam Hospital, offers important insights regarding infection risks and adverse pregnancy outcomes in Emirati women with SLE. However, several limitations of the study merit consideration. The retrospective design depends on the accuracy and completeness of the recorded data, which might lead to potential biases, including selection and information biases from excluded cases with incomplete documentation. Additionally, the single-center setting may limit the generalizability of the results to other populations, as patients treated at a tertiary referral center like Tawam Hospital may not represent the broader demographic of women with SLE. Future multicenter studies, including diverse populations, would help validate these findings and ensure broader applicability. Furthermore, disease activity was not incorporated into the multivariable analysis due to incomplete measurements, which could have influenced the findings. Despite these constraints, the findings contribute significantly to the limited literature on SLE in pregnancy available in the Middle East and emphasize the need for rigorous prenatal care in this high-risk group. Future multicenter, prospective studies are required to validate these findings in broader regional and global contexts.
Conclusions
The results of this study demonstrate that pregnant women with SLE have a twofold higher risk of infections compared to their non-SLE counterparts, with respiratory and urinary tract infections being particularly common. Importantly, hydroxychloroquine use during pregnancy was associated with a significant reduction in infection risk, underscoring its value as a protective therapy in this high-risk group. These findings highlight the urgent need for interdisciplinary collaboration between rheumatologists and obstetricians and the adoption of individualized therapeutic strategies, including the continuation of hydroxychloroquine, to preemptively address infection risks and optimize maternal and fetal health. Future research should focus on large, multicenter studies to validate these findings across diverse populations and to explore the long-term impact of such interventions on pregnancy outcomes.
Footnotes
Acknowledgments
Editorial assistance in English language and spelling corrections was provided by Enago, Crimson Interactive Inc. and supported by the United Arab Emirates University.
Author contributors
Ali M. Al Dhanhani acts as the guarantor and accepts full responsibility for the work and the conduct of the study, had access to the data, and controlled the decision to publish. Hiba I. Khogali and Ali M. Al Dhanhani conceptualized and designed the study, analyzed the data, and drafted the manuscript. Aaesha M. Alhebsi and Meera A. Altamimi contributed to data acquisition and drafting the manuscript. Ghada S. M. Al‐Bluwi and Virgie Guy Pedo contributed to data acquisition. All authors critically revised and approved the final manuscript for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by a grant from the Sure Plus program provided by the United Arab Emirates University. No additional specific funding was received from public, commercial, or not-for-profit sectors for the work presented in this article.
Ethical statement
Data Availability Statement
Data supporting the findings of this study will be made available upon reasonable request directed to the corresponding author.
