Abstract
Objective
To investigate the effects of belimumab (BEL) on systemic lupus erythematosus (SLE) mucocutaneous and vasculitis manifestations.
Methods
This post hoc, integrated Belimumab Summary of Lupus Efficacy (Be-SLE) analysis pooled data from five international Phase 3, randomized, placebo (PBO)-controlled BEL trials (BLISS-52 [NCT00424476; conducted in 2007–2009], BLISS-76 [NCT00410384; 2007–2009], BLISS-SC [NCT01484496; 2011–2015], North East Asia [NCT01345253; 2011–2015], EMBRACE [NCT01632241; 2013–2018]). Adults with active SLE and Safety of Estrogens in Lupus Erythematosus National Assessment-SLE Disease Activity Index (SELENA-SLEDAI) score ≥6 (BLISS-52, BLISS-76) or ≥8 (BLISS-SC, North East Asia, EMBRACE), randomized to BEL (10 mg/kg/month intravenously or 200 mg/week subcutaneously) or PBO, plus standard therapy (ST) were included. Mucocutaneous and vasculitis manifestations (listed below) were measured (baseline and every 4 weeks) for 52 weeks using SELENA-SLEDAI and British Isles Lupus Assessment Group (BILAG).
Results
Of 3086 patients (BEL, n = 1869; PBO, n = 1217), 85% (BEL and PBO) by SELENA-SLEDAI and 58% (BEL) and 62% (PBO) by BILAG (moderate or severe activity) had mucocutaneous manifestations, and <10% had vasculitis at baseline. At Week 52, significantly more BEL-treated than PBO-treated patients demonstrated improvements in SELENA-SLEDAI (59% vs 49%; p < .0001) and BILAG (54% vs 43%; p < .0001) mucocutaneous domains. Significant differences between-treatment favored BEL at Week 52 for resolution of all SELENA-SLEDAI items (vasculitis, rash, alopecia, and mucosal ulcers), and nine of 20 BILAG items (mild maculopapular eruption, localized active discoid lesions, mild alopecia, small mucosal ulceration, malar erythema, subcutaneous nodules, swollen fingers, major cutaneous vasculitis including ulcers, and minor cutaneous vasculitis).
Conclusion
Patients with SLE treated with BEL plus ST experienced significant improvements in most mucocutaneous and vasculitis manifestations compared with patients receiving PBO plus ST. These data provide additional support for the use of BEL in SLE and show that it is associated with skin improvements.
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disorder characterized by abnormal immune function and autoantibody production, which leads to clinical presentations involving multiple organ systems.1,2 Several types of mucocutaneous manifestations, including lupus-specific (malar rash, discoid rash, photosensitivity, oral mucosal lesions) and nonspecific (such as alopecia and secondary atrophie blanche) lesions, affect up to 85% of patients. 3 Quality of life (QoL) in patients with SLE with mucocutaneous manifestations is poor; these manifestations contribute to feelings of unwanted attention, self-consciousness, emotional symptoms, and functional decline.4,5
Due to their impact, it is important to treat these mucocutaneous manifestations to minimize their short-term impact and long-term damage. 6 Of the 24 items that assess disease activity with the Safety of Estrogens in Lupus Erythematosus National Assessment-Systemic Lupus Erythematosus Disease Activity Index (SELENA-SLEDAI), three are for mucocutaneous (rash, alopecia, mucosal ulcers) and one for vasculitic manifestations.7,8 The classic British Isles Lupus Assessment Group (BILAG) index, published in 1988, measures SLE disease activity in eight organ-based domains including the mucocutaneous system (18 items) and vasculitis (2 items).9–11 BILAG was revised in 2004 to include nine organ systems instead of eight, removing vasculitis and adding ophthalmic and gastrointestinal systems. 12 In addition to these broad measures of SLE organ manifestations, the Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) was developed specifically to measure the activity and damage of mucocutaneous manifestations. 13 The CLASI, published in 2005, was relatively newly available when many of the early belimumab (BEL) trials were being designed; those trials only utilized SELENA-SLEDAI and BILAG measures to assess SLE activity for systemic manifestations and the mucocutaneous and vasculitic domains. However, the CLASI has been included in more recent real-world studies14–16 and will be utilized in patients with baseline mucocutaneous involvement in the international, Phase 4, BE-EARLY clinical trial (NCT06411249) of BEL’s efficacy and safety in early SLE. 17
Initial treatment of mucocutaneous manifestations of SLE include sun protection and the use of topical agents, antimalarials (e.g., hydroxychloroquine), and systemic corticosteroids. 18 However, almost 40% of patients do not respond to initial therapy and require the addition of immunosuppressants.18–20 Despite their routine use, there is limited evidence supporting the efficacy of these treatments and, to date, there have been few randomized controlled trials examining therapies for the treatment of mucocutaneous manifestations of SLE.21,22 In small studies, methotrexate (MTX) has been shown to resolve skin rashes more effectively than PBO at 6 months of treatment, and independently, chloroquine was shown to be as effective as MTX; however, both have known adverse effects. 21 MTX and mycophenolate mofetil (MMF) are efficacious in about half of patients, but frequently are not well tolerated. 23 Thalidomide or lenalidomide can be effective in some forms of refractory cutaneous disease but typically have not been used for SLE. 6 Biologics (BEL and anifrolumab) have also shown efficacy in mucocutaneous manifestations of SLE24–26 and are recommended by the 2023 EULAR guidelines for the treatment of active skin disease as second-line therapy. 18
BEL is a disease-modifying human IgG1λ monoclonal antibody that selectively targets soluble B lymphocyte stimulator (BLyS) and has been used to treat SLE for over 14 years. 27 BLyS plays a key role in the pathogenesis of SLE, and other autoimmune diseases, by promoting the differentiation and survival of B lymphocytes into Ig-producing plasma cells.27,28 BEL in combination with standard therapy is approved in many countries (including the US, European and Latin-American countries, Japan, and China) for the treatment of active SLE in patients ≥5 years of age as well as active lupus nephritis in adults (US and Europe) and children ≥5 years of age (US).29–32 In multiple studies worldwide, BEL in combination with standard therapy reduced SLE disease activity, autoantibody levels, and the risk of new severe flares when compared with PBO and standard therapy.25,33–37 BLyS levels have been shown to be elevated in the skin of patients with discoid lupus and other forms of cutaneous lupus erythematosus (CLE).38,39 Data from BEL trials indicated an improvement in the overall mucocutaneous system with BEL.40–42 To further investigate the clinical benefit of BEL on CLE and vasculitic manifestations of SLE, including a more in-depth analysis of individual symptoms, this post hoc, integrated analysis used a large dataset from five randomized Phase 3 clinical trials.25,33–36 These trials, published over the course of ∼10 years, evaluated the efficacy and safety of intravenous (IV) and subcutaneous (SC) BEL in geographically and ethnically diverse patient populations.
Methodology
Study design
Belimumab Summary of Lupus Efficacy (Be-SLE) was a post hoc, integrated analysis that pooled data from five, randomized, double-blind, PBO-controlled Phase 3 clinical trials in adult patients with SLE (BLISS-52 [NCT00424476], 34 BLISS-76 [NCT00410384], 33 North East Asia [NCT01345253], 35 BLISS-SC [NCT01484496], 25 and EMBRACE [NCT01632241]) 36 (Supplemental Figure 1). All five studies compared the efficacy and safety of IV BEL (10 mg/kg/4 weeks) or SC BEL (200 mg weekly) plus standard therapy versus PBO plus standard therapy. In BLISS-52, BLISS-76, North East Asia, and EMBRACE, patients received IV BEL infusions.33–36 In BLISS-SC, patients received weekly SC BEL. 25 These trials were conducted in a number of countries across Europe, Latin America, North and Central America, Asia, Australia, South Africa, and Asia-Pacific, representing multiple patient races and ethnicities. The individual trials were conducted in accordance with the ethical principles of the Declaration of Helsinki, International Council for Harmonisation on Good Clinical Practice and the applicable country-specific regulatory requirements. Additionally, the trials were approved by a central or local institutional review board or ethics committee, and all patients provided written informed consent.
Patients
Inclusion and exclusion criteria for each study have been reported previously.25,33–36 Briefly, participants were ≥18 years of age with a SELENA-SLEDAI score of either ≥6 (BLISS-52 and BLISS-76)33,34 or ≥8 (BLISS-SC, North East Asia, and EMBRACE)25,35,36 at screening. The current analysis included patients who received BEL 10 mg/kg/4 weeks IV or 200 mg/week SC or PBO. Across all studies, there were no inclusion or exclusion criteria for any specific type of mucocutaneous or vasculitic lesion.
Outcomes and endpoints
The effect of BEL versus PBO, both plus standard therapy, on mucocutaneous and vasculitic disease activity was assessed using SELENA-SLEDAI and classic 1988 BILAG. SELENA-SLEDAI was selected instead of SELENA-SLEDAI 2000 as it was collected in all studies, except EMBRACE, and there are no differences in scoring between these two measures for the domains reported in this analysis. Classic BILAG was used as it included the vasculitic domain; see Supplemental Material for the glossary and scoring guide used in the included studies.
In all five pooled studies, SELENA-SLEDAI and BILAG assessments were captured at baseline and every 4 weeks up to 52 weeks. The endpoints for this analysis were mucocutaneous and vasculitic domain improvement and individual item resolution. The presence (in EMBRACE presence or absence) of the SELENA-SLEDAI components related to CLE involvement (rash, alopecia, mucosal ulcers) and vasculitis7,43 within 10 days before assessment day were recorded. The proportions of patients with improvement in the CLE and vasculitis domains (in the subset of patients with involvement at baseline) and resolution in individual items present at baseline were summarized every 4 weeks up to Week 52. SELENA-SLEDAI domain improvement was defined as a decrease in score within the same organ system from baseline. SELENA-SLEDAI individual item resolution was defined as an item present at baseline and no longer present at the specified timepoint post-baseline.
For BILAG assessment of improvement in CLE and vasculitis domain scores, patients with severe (category A) or moderate (category B) scores at baseline were assessed for a category improvement (i.e., shift from A to B, C or D, or from B to C or D) at Week 52. In the analysis of the 18 mucocutaneous items and two vasculitis items of the classic BILAG 9 (Supplemental Figure 1), BILAG categories of improving, the same, worse, new/recurrent disease, and disease present, were combined and recorded as present to facilitate assessment of individual item resolution. Resolution of individual items was assessed in patients categorized as having the item present at baseline and defined as no longer having the item present at the specified timepoint post-baseline.
Change from baseline to Week 52 in organ damage accrual was examined using the Systemic Lupus International Collaborating Clinics (SLICC)/American College of Rheumatology (ACR) Damage Index (SDI), an instrument developed by international experts to measure organ damage. 44
Other endpoints included the proportion of patients achieving a prednisone-equivalent reduction to ≤5 mg/day during Weeks 40–52 and the mean change in prednisone dose from baseline at Week 52.
Statistical analysis
For summaries and analyses of improvement in SELENA-SLEDAI and BILAG domain scores, only patients with involvement at baseline were included. Improvements at Week 52 were analyzed using Fisher’s exact test. For summaries and analyses of individual item resolution, only patients with the item present at baseline were included. For items with ≥20 patients per arm at baseline, data are presented using spider plots and Week 52 data were analyzed using Fisher’s exact test. These analyses were limited by low power to detect statistically significant differences in the domains or individual items reported less frequently. Adjustments were not made for multiple comparisons. Nominal p-values are provided (at Week 52) for pairwise comparison of results between the BEL and PBO groups. As p-values were not available for all time points, and considering the large sample size of this analysis, a 5% cut off was selected as a clinically meaningful threshold for all analyses and interpretations. Additional subgroup analysis of SELENA-SLEDAI improvements in organ domains and individual item resolution categorized by race and ethnicity (self-reported, answered from a fixed set of categories) were analyzed at Week 52 using Fisher’s exact test (for all domains and items with at least 20 patients per arm with the item present at baseline). To account for dropouts over time, domain improvement and item resolution analyses used dropout/treatment failure = non-responder approach. Change in SDI score from baseline to Week 52 in BEL and PBO groups was analyzed using an analysis of covariance (ANCOVA) model with covariates for treatment group, study, baseline SDI score, and baseline SELENA-SLEDAI score (≤9 vs ≥10).
Results
Patient population
Patient demographics and clinical characteristics at baseline.
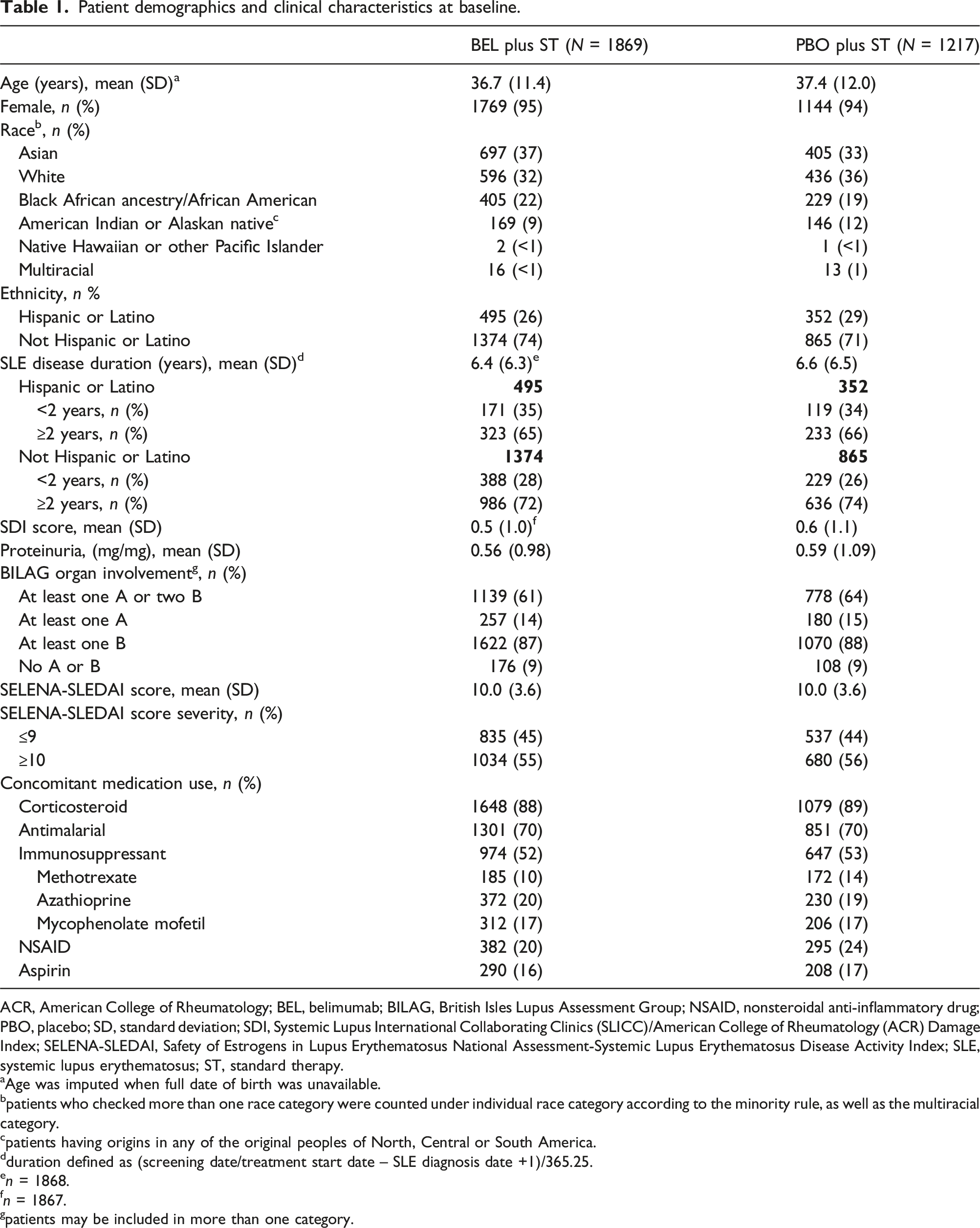
ACR, American College of Rheumatology; BEL, belimumab; BILAG, British Isles Lupus Assessment Group; NSAID, nonsteroidal anti-inflammatory drug; PBO, placebo; SD, standard deviation; SDI, Systemic Lupus International Collaborating Clinics (SLICC)/American College of Rheumatology (ACR) Damage Index; SELENA-SLEDAI, Safety of Estrogens in Lupus Erythematosus National Assessment-Systemic Lupus Erythematosus Disease Activity Index; SLE, systemic lupus erythematosus; ST, standard therapy.
aAge was imputed when full date of birth was unavailable.
bpatients who checked more than one race category were counted under individual race category according to the minority rule, as well as the multiracial category.
cpatients having origins in any of the original peoples of North, Central or South America.
dduration defined as (screening date/treatment start date – SLE diagnosis date +1)/365.25.
en = 1868.
fn = 1867.
gpatients may be included in more than one category.
Efficacy
SELENA-SLEDAI
At Week 52, a statistically significant greater proportion of patients treated with BEL compared with PBO experienced improvement in the SELENA-SLEDAI mucocutaneous (59% vs 49%; p < .0001) and vascular (67% vs 41%; p < .0001) domains in the subset of patients with mucocutaneous and vascular involvement at baseline, respectively (Figure 1(a) and (b)). There were also statistically significant treatment differences at Week 52 in favor of BEL in the proportion of patients with resolution of all individual SELENA-SLEDAI mucocutaneous and vasculitis items present at baseline (vasculitis, 25%, p < .0001; rash, 11%, p < .0001; alopecia, 7%, p = .0022; and mucosal ulcers, 12%, p = .0004) (Figure 2). Proportions of patients demonstrating improvement in (a) SELENA-SLEDAI mucocutaneous domain, (b) SELENA-SLEDAI vascular domain, (c) BILAG mucocutaneous domain, (d) BILAG vasculitis domain, over 52 weeks in the subset of patients with organ domain involvement at baseline Number of patients per treatment arm (n) remained the same throughout the analyses (dropout/treatment failure = non-responder). BEL, belimumab; BILAG, British Isles Lupus Assessment Group; PBO, placebo; SELENA-SLEDAI, Safety of Estrogens in Lupus Erythematosus National Assessment-Systemic Lupus Erythematosus Disease Activity Index; ST, standard therapy; Wk, week. Proportions of patients demonstrating resolution in (a) SELENA-SLEDAI vasculitis, and SELENA-SLEDAI mucocutaneous items: (b) rash, (c) alopecia, (d) mucosal ulcers, over 52 weeks in the subset of patients with the item present at baseline. Number of patients per treatment arm (n) remained the same throughout the analyses (dropout/treatment failure = non-responder). BEL, belimumab; PBO, placebo; SELENA-SLEDAI, Safety of Estrogens in Lupus Erythematosus National Assessment-Systemic Lupus Erythematosus Disease Activity Index; ST, standard therapy; Wk, week.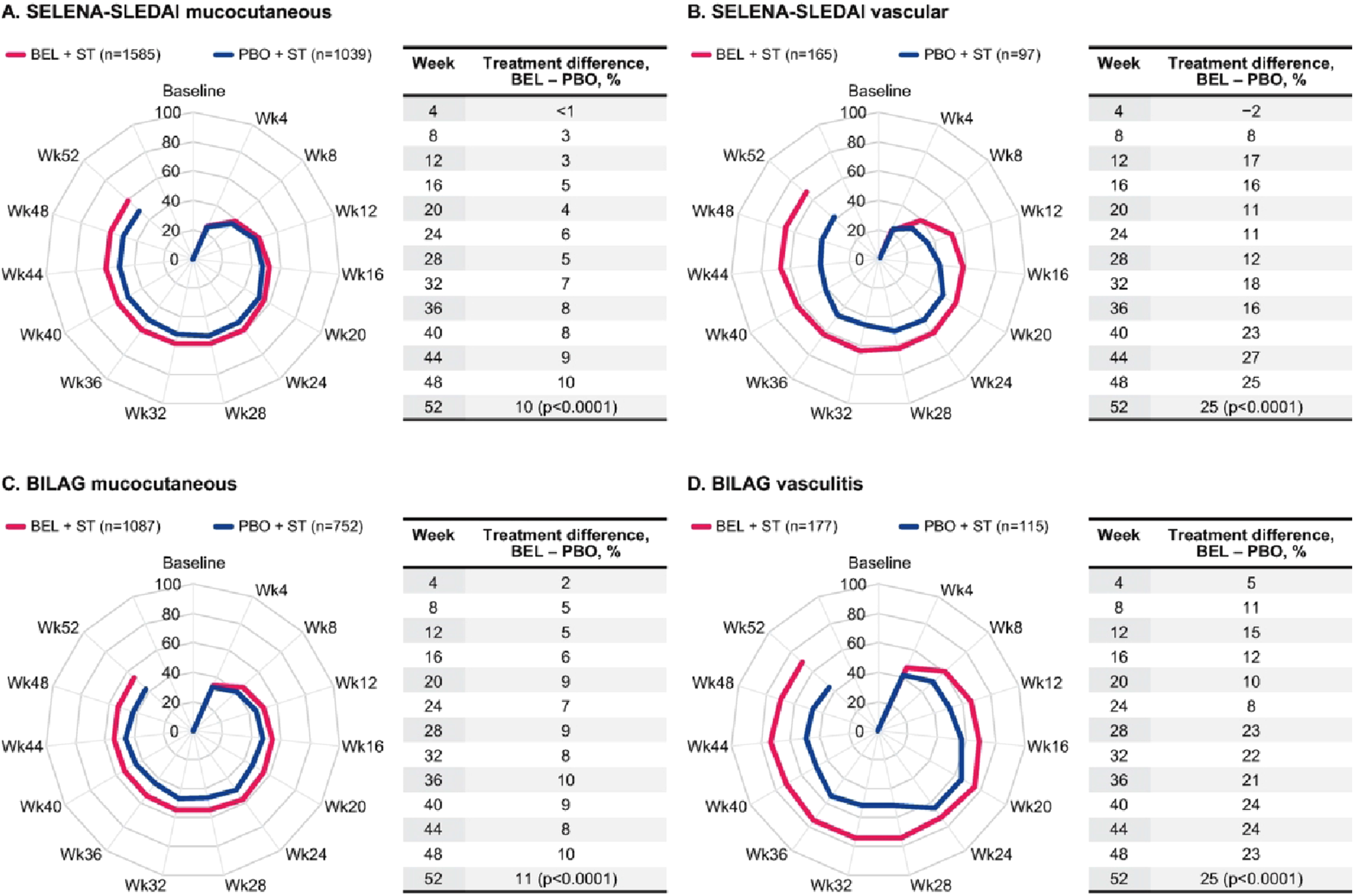
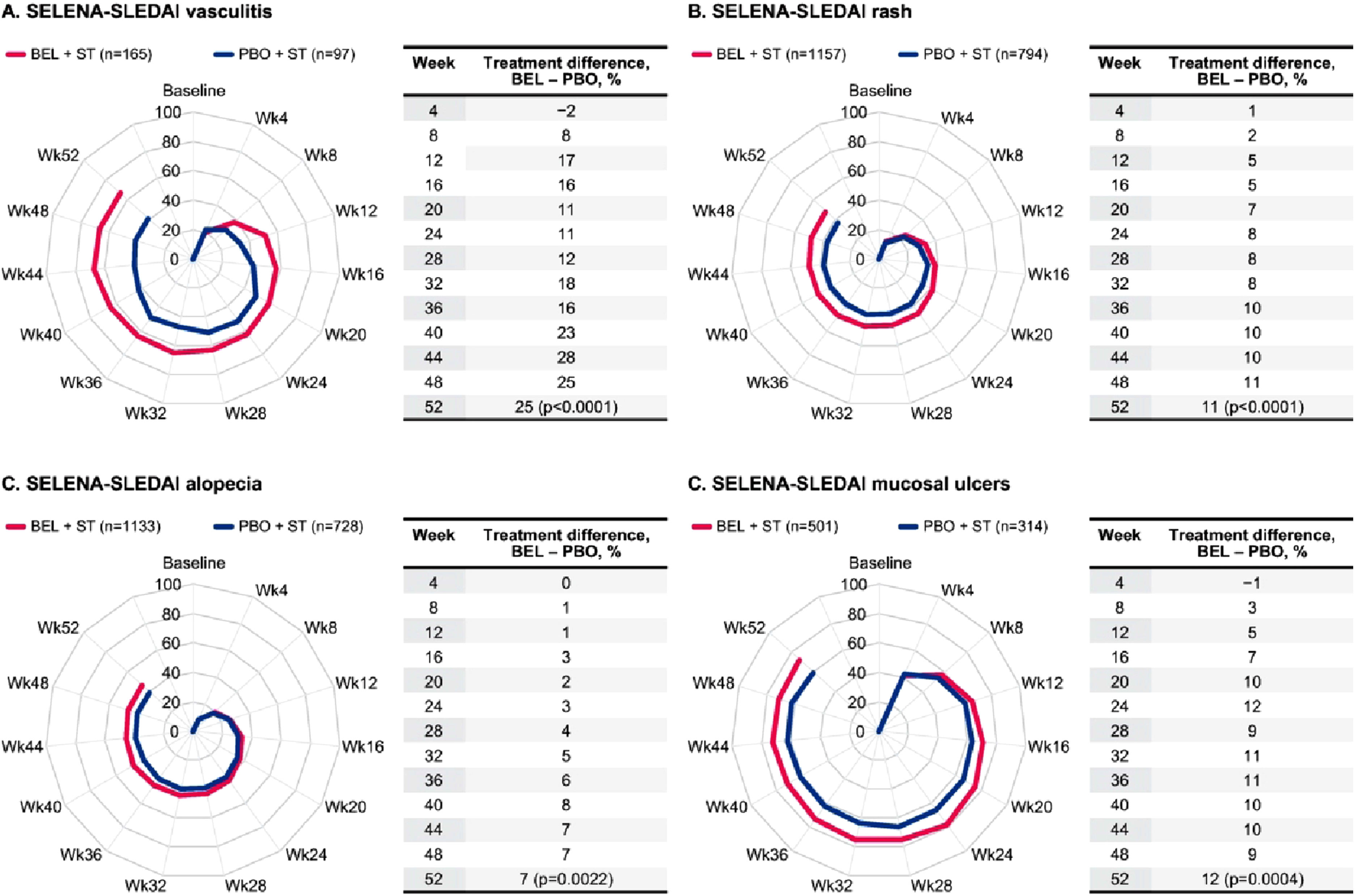
The most resolved item among patients who received BEL was mucosal ulcers (BEL, 72% vs PBO, 60%) and the greatest between-treatment difference in individual item resolution at Week 52 was observed for vasculitis (BEL, 67% vs PBO, 41%).
Treatment differences of at least 5% for the proportion of patients with resolution of the individual SELENA-SLEDAI mucocutaneous and vasculitis items through Week 52 were evident from Week 8 for vasculitis, Week 12 for rash, Week 32 for alopecia, and Week 12 for mucosal ulcers (Figure 2).
SELENA-SLEDAI by race and ethnicity
Mucocutaneous domain improvements at Week 52 by race and ethnicity are presented in Supplemental Figure 2(a). Statistically significant improvements with BEL versus PBO in SELENA-SLEDAI mucocutaneous domain were observed in patients of ‘Not Hispanic or Latino’ ethnicity (58% vs 46%; p < .0001) but not in patients of ‘Hispanic or Latino’ (63% vs 56%; p = .0505) origin. When analyzed by race, this trend in statistical significance was observed in patients of White (59% vs 48%; p = .0007) and Asian (63% vs 51%; p = .0011) race but not for patients who were of Black African ancestry/African American (52% vs 47%; p = .2555).
Similar to the overall analysis, a trend for treatment differences at Week 52 in favor of BEL was seen for all individual SELENA-SLEDAI mucocutaneous and vasculitis items when analyzed by race and ethnicity; however, treatment difference for the proportion of patients with resolution of the rash in Black African ancestry/African American patients was small (Supplemental Figure 2(b)). Statistically significant treatment differences were observed in all four SELENA-SLEDAI items (one vasculitis and three mucocutaneous [rash, alopecia, mucosal ulcers]) in patients of ‘Not Hispanic or Latino’ ethnicity; for patients who were Asian, statistically significant treatment differences were seen for all items except vasculitis. For patients of ‘Hispanic or Latino’ ethnicity or of Black African ancestry/African American, statistical significance in treatment difference was only reached for the mucosal ulcers item (14%, p = .0239; 20%, p = .0121, respectively); for patients who were White, statistical significance was reached for vasculitis (43%, p = .0015) and rash (14%, p = .0001) (Supplemental Figure 2(b)).
When analyzed by race and ethnicity, in most subgroups the most resolved item with BEL was mucosal ulcers; however, in White patients it was vasculitis (BEL, 71% vs PBO, 28%). As observed in the overall analysis, for all race and ethnicity groups except for Asian patients, the greatest between-treatment difference in individual item resolution at Week 52 was for vasculitis. In Asian patients, the greatest between-treatment difference at Week 52 was seen for mucosal ulcers (16%, p = .0150) (Supplemental Figure 2(b)).
BILAG
At Week 52, among patients with a BILAG A or B mucocutaneous or vasculitis score at baseline, a statistically significantly greater proportion of patients treated with BEL compared with PBO experienced improvements in the mucocutaneous domain (54% vs 43%; p < .0001) and vasculitis domain (70% vs 45%; p < .0001), respectively (Figure 1(c) and (d)).
Statistical comparisons of resolution between treatment groups were performed for 13 mucocutaneous BILAG items (where there were ≥20 patients per arm with the item present at baseline), while for five items (panniculitis─severe, angioedema, extensive mucosal ulceration, sclerodactyly, and calcinosis) too few patients had these manifestations at baseline for meaningful comparisons (Supplemental Table 1).
Of the 13 comparisons, seven items showed statistically significant treatment differences in favor of BEL at Week 52 (maculopapular eruption – mild, 9%, p = .0093; active discoid lesions – localized, 11%, p = .0483; alopecia – mild, 7%, p = .0016; small mucosal ulceration, 10%, p = .0032; malar erythema, 11%, p < .0001; subcutaneous nodules, 30%, p = .0088; swollen fingers, 15%, p = .0312; Figure 3). Of these, at Week 52, the most commonly resolved item among patients who received BEL was small mucosal ulceration (BEL, 69% vs PBO, 59%). The greatest between-treatment difference in individual item resolution was observed for subcutaneous nodules (BEL, 60% vs PBO, 29%). There were six mucocutaneous BILAG items that did not show significant treatment differences in favor of BEL at Week 52 (maculopapular eruption − severe, active, 5%, p = .7932; active discoid lesions – generalized, 4%, p = .8128; perniotic skin lesions, -17%, p = .1075; periungual erythema, 10%, p = .1205; telangiectasia, 17%, p = .1408; alopecia − severe, active, 15%, p = .0542; Supplemental Figure 3). Proportions of patients demonstrating resolution in BILAG mucocutaneous items over 52 weeks in the subset of patients with item present at baseline, for items with a statistically significant difference at Week 52 between BEL and PBO, (a) maculopapular eruption – mild, (b) active discoid lesions – localized, (c) alopecia – mild, (d) small mucosal ulceration, (e) malar erythema, (f) subcutaneous nodules, (g) swollen fingers. Number of patients per treatment arm (n) remained the same throughout the analyses (dropout/treatment failure = non-responder). BEL, belimumab; BILAG, British Isles Lupus Assessment Group; PBO, placebo; ST, standard therapy; Wk, week.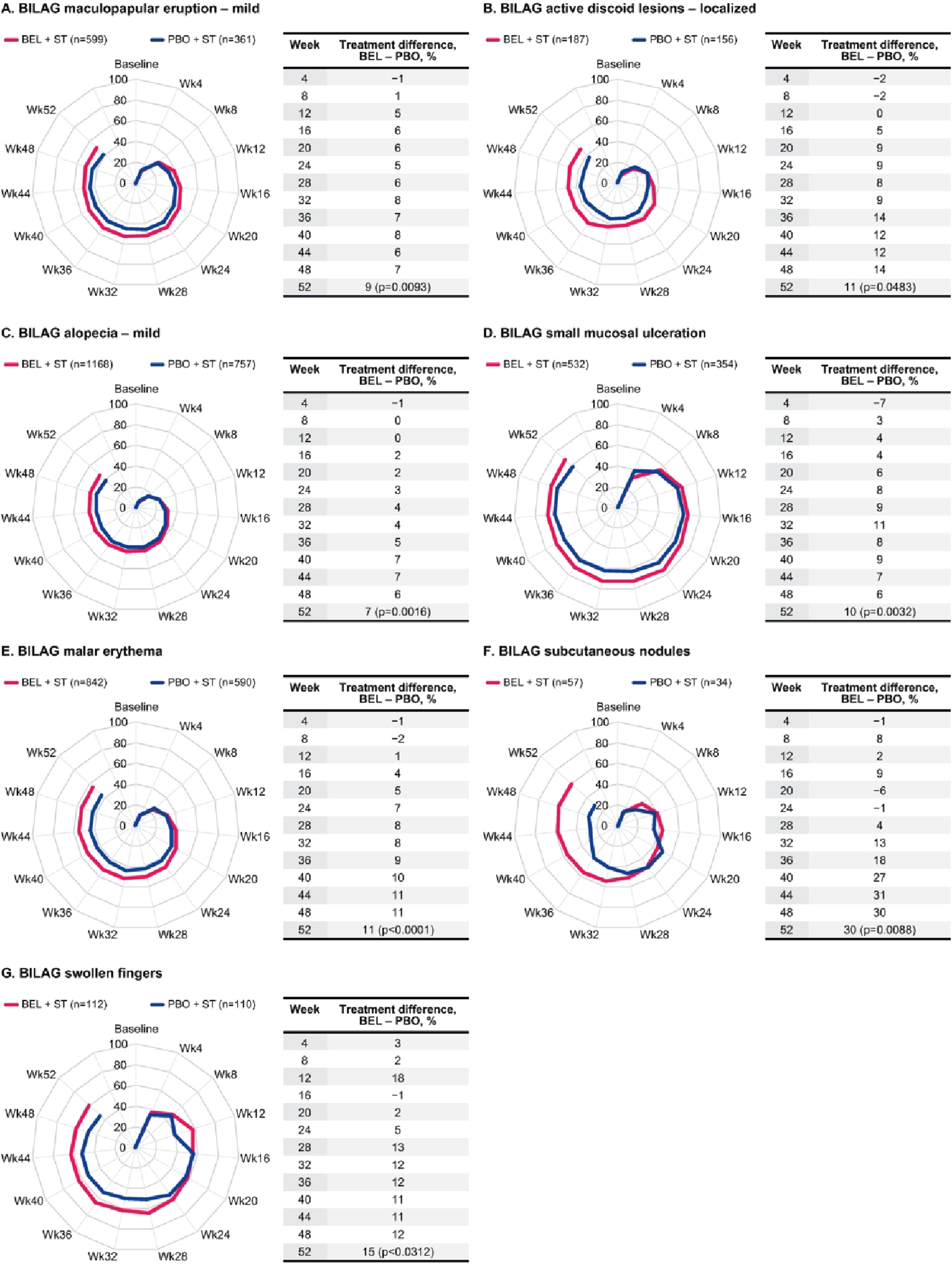
Treatment differences ≥5% for the proportion of patients with resolution of the individual BILAG mucocutaneous items through Week 52 were consistently evident from Week 12 for maculopapular eruption – mild; Week 16 for active discoid lesions – localized; Week 20 for alopecia – severe, active, small mucosal ulceration, and malar erythema; Week 24 for swollen fingers, maculopapular eruption – severe, active, and periungual erythema; Week 32 for subcutaneous nodules; Week 36 for telangiectasia; and Week 36 for alopecia – mild. Treatment difference for active discoid lesions – generalized did not consistently reach the 5% threshold and treatment difference for perniotic skin lesions favored PBO (Figure 3 and Supplemental Figure 3).
Both cutaneous vasculitis BILAG items showed statistically significant treatment differences in favor of BEL at Week 52 (major– including ulcers, 34%, p = .0039 and minor, 20%, p = .0003); major cutaneous vasculitis – including ulcers resolved in 75% of BEL-treated patients compared with 41% of PBO-treated patients (Figure 4). Treatment differences ≥5% for the proportion of patients with resolution of the individual BILAG vasculitis items through Week 52 were evident from Week 4 for major cutaneous vasculitis – including ulcers and Week 12 for minor cutaneous vasculitis (Figure 4). Proportions of patients demonstrating resolution in BILAG vasculitis items over 52 weeks in the subset of patients with item present at baseline (a) major cutaneous vasculitis – including ulcers, (b) minor cutaneous vasculitis. Number of patients per treatment arm (n) remained the same throughout the analyses (dropout/treatment failure = non-responder). BEL, belimumab; BILAG, British Isles Lupus Assessment Group; PBO, placebo; ST, standard therapy; Wk, week.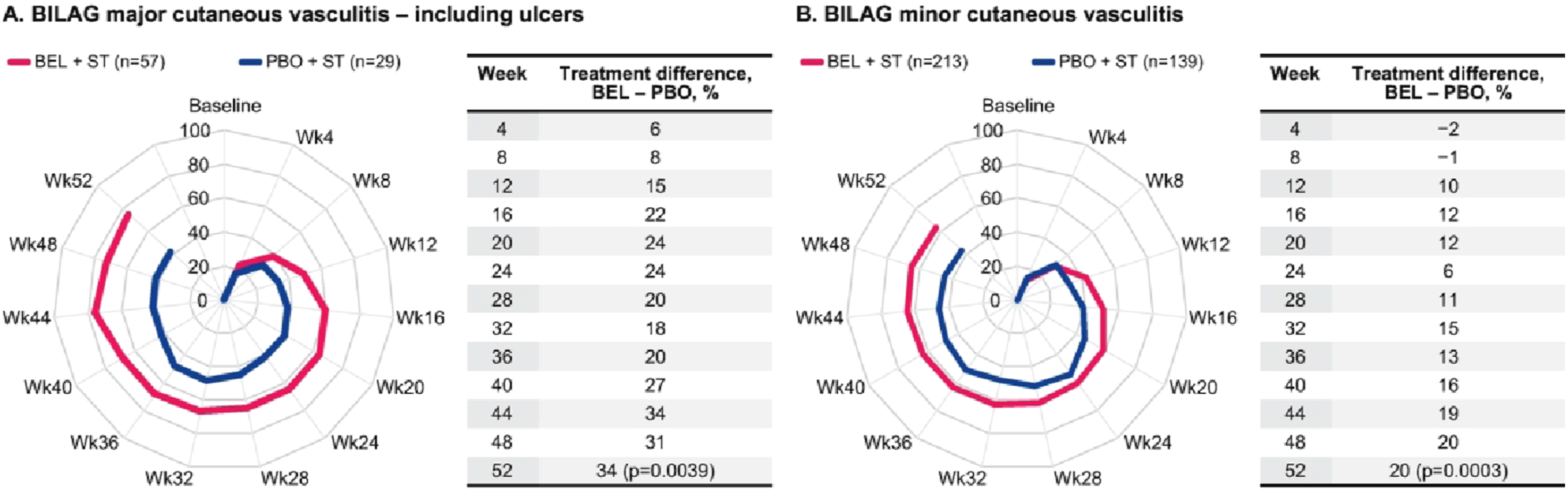
Corticosteroid use
At baseline, mean (standard deviation [SD]) corticosteroid dose was similar between patients receiving BEL (12.3 [9.8] mg/day) and patients receiving PBO (12.2 [9.6] mg/day). At Week 52, the treatment difference (95% confidence interval [CI]) of BEL versus PBO in mean absolute corticosteroid change from baseline was −0.82 mg/day (−1.70, 0.07; p = .0698). More patients treated with BEL (n = 125/1255, 10%) versus PBO (n = 62/820; 8%) achieved a corticosteroid dose reduction to ≤5 mg/day during Week 40 to Week 52 (OR [95% CI]: 1.40 [1.01, 1.93]; p = .0441) among patients with average corticosteroid dose >5 mg/day at baseline.
SDI
Overall, 66% of patients had an SDI score of 0 at baseline. There was no mean (SD) change in SDI score from baseline at Week 52 in patients receiving BEL (0.0 [0.18]) or PBO (0.0 [0.25]).
Discussion
This post hoc, integrated Be-SLE analysis of five randomized, controlled Phase 3 BEL clinical trials demonstrated that in patients with SLE, BEL plus standard therapy led to statistically significant improvements in CLE and cutaneous vasculitis, assessed by SELENA-SLEDAI and BILAG at Week 52, compared with patients receiving PBO plus standard therapy. At Week 52, more BEL-treated patients resolved most of the individual mucocutaneous and vasculitis items, in addition to greater corticosteroid dose reductions, than patients receiving PBO plus standard therapy.
The highest proportion of patients with resolution of an item at Week 52 was for BILAG major cutaneous vasculitis. Several other manifestations also demonstrated statistically significant higher resolution in patients treated with BEL versus PBO. These findings demonstrate that the use of BEL plus standard therapy in SLE was associated with skin improvement. Furthermore, treatment differences (≥5%) in resolutions in favor of BEL versus PBO were observed as early as Week 4 for major cutaneous vasculitis BILAG item and Week 8 for vasculitis SELENA-SLEDAI item.
Statistically significant improvement in SELENA-SLEDAI mucocutaneous domain for BEL compared with PBO was observed in patients of ‘Not Hispanic or Latino’ ethnicity, and in patients of White and Asian race but not for those of ‘Hispanic or Latino’ ethnicity or Black African ancestry/African American patients. Analyses by race and ethnicity also showed differences in resolution of individual items. Patients of different race or ethnicity may respond differently to BEL treatment, and this may be related to multiple factors, including greater disease activity at baseline or longer disease duration due to delays in diagnosis or access to care;36,45 however, in our study, similar proportions of ‘Hispanic or Latino’ patients (66%) and ‘Not Hispanic or Latino’ patients (72%) had disease duration ≥2 years. Of note, we defined disease duration at baseline as time since SLE diagnosis for consistency with previously published studies, rather than time since symptom onset. Thus, it is unclear if there were any differences in diagnosis timings among different ethnic groups. A previous study demonstrated that cutaneous lupus lesions can have differing inflammatory infiltrate profiles, which can be dominated by plasmacytoid dendritic cells, B cells or interferons. 46 Since BEL is known to reduce autoantibody levels and B-cell subsets, while preserving memory B cells and T cells, patients with SLE with predominantly B-cell infiltrated lesions may be more likely to respond to BEL.37,46
The current findings extend available evidence regarding the effectiveness of BEL for mucocutaneous and vasculitis manifestations in SLE and are in agreement with the findings of a previous post hoc analysis of the BLISS-52 and BLISS-76 studies, which found that the addition of BEL to standard therapy resolved both rashes and vasculitis in a significantly higher proportion of patients with SLE compared with standard therapy. 40 However, because a cutaneous-specific assessment tool such as CLASI was newly available and not established when the original studies were designed, the current results are derived from SELENA-SLEDAI and BILAG, measures that are not specific to cutaneous manifestations alone. In more recent observational studies that used CLASI, 48% and 62% of BEL-treated patients achieved CLASI remission over 12 months and 24 months, respectively 47 and 5 BEL-treated patients with recalcitrant cutaneous lupus had a CLASI activity score decrease from median (range) 17 (9–31) to 3 (2–14) over 16 weeks (p = .043). 15 Another small study (N = 7) demonstrated clinical improvement based on CLASI scores in the majority of patients with CLE who received BEL. 16
SELENA-SLEDAI and BILAG may be less sensitive to capturing CLE changes than CLASI; however, in this post hoc analysis of the largest SLE population to date, BEL demonstrated a statistically significant and consistent improvement/resolution in various CLE manifestations measured by both SELENA-SLEDAI and BILAG compared with PBO. Furthermore, more recent and future studies have been and will utilise CLASI to continue to investigate the efficacy of BEL on CLE.14–16,47 Importantly, in the 2023 EULAR guidelines for the management of SLE, BEL is recommended as second-line therapy for the treatment of active CLE. 18
Observed improvement in most but not all mucocutaneous manifestations with BEL may be related to differences in sample size (the numbers of patients with the item present at baseline) and the chronic and/or irreversible lesions seen in perniosis, chilblains, and telangiectasias. Overall, 66% of patients had an SDI score of 0 at baseline and there was no mean change in SDI score from baseline at Week 52 in either treatment group.
Strengths of this post hoc analysis include the large population size, a randomized controlled group, and use of validated measures to assess outcomes on CLE and vasculitis manifestations. Both the SELENA-SLEDAI and BILAG have been shown to be reliable, valid, and sensitive to changes in disease activity over time and correlate well with each other. 48
Limitations of this pooled analysis include the low prevalence of some manifestations, which reduced the power to detect a statistically significant difference, precluding meaningful comparison of treatment response between groups. As such, some results should be interpreted with caution. Furthermore, some BILAG items such as telangiectasia are likely to be permanent/irreversible, so no improvement or resolution was expected. The BILAG index used in this analysis was the classic 1988 version, as opposed to the revised 2004 version, which removed vasculitis. 48 As such, comparisons with other clinical studies that used the revised index should be interpreted with caution. A glossary and scoring guide for 1988 BILAG have been provided in the Supplemental Material to aid the readers less familiar with the classic BILAG with interpretation of our results. In addition, no skin-specific measures such as the CLASI were used because it was newly available and not well established when the original studies were designed. The findings of this pooled analysis should be interpreted in the context of original study designs and assessment tools used, with the understanding that more specific measures of CLE have been developed since. The studies included in this post hoc analysis were neither designed nor powered to demonstrate the efficacy of BEL in individual organ systems or for specific items.
Conclusion
In conclusion, compared with PBO plus standard therapy, patients with SLE treated with BEL plus standard therapy experienced statistically significant improvements in mucocutaneous and vasculitis manifestations and experienced resolution of some individual items as early as Week 4, with statistically significant between-treatment differences and corticosteroid dose reductions at Week 52. The benefit of the clinical improvements observed were aligned with a greater corticosteroid tapering in the BEL group. This post hoc, pooled analysis of five randomized, PBO-controlled Phase 3 clinical trials (Be-SLE; N = 3086) supports the use of BEL plus standard therapy in the treatment of mucocutaneous and vasculitis manifestations of SLE.
Supplemental Material
Supplemental Material - The effect of belimumab on mucocutaneous and vasculitis manifestations in patients with systemic lupus erythematosus: A large pooled post hoc analysis
Supplemental Material for The effect of belimumab on mucocutaneous and vasculitis manifestations in patients with systemic lupus erythematosus: A large pooled post hoc analysis by Susan Manzi, Jorge Sánchez-Guerrero, Naoto Yokogawa, Joerg Wenzel, Josephine C Ocran-Appiah, Julia HN Harris, Munther Khamashta, Bernard Rubin, Norma Lynn Fox, Roger A Levy and Victoria P Werth in Lupus
Footnotes
Acknowledgments
The authors would like to thank the participating patients and their families, clinicians, and study investigators. Medical writing and submission support was provided by Claire Barron, MSc, and Madeline Thomas, MRes, Fishawack Indicia Ltd, UK, part of Avalere Health, and was funded by GSK.
Author Contributions
SM, JS-G, NY, JW, JCO-A, JHNH, MK, BR, NLF, RAL, and VW contributed to the acquisition of the data. JCO-A, MK, BR, NLF, and RAL contributed to the conception or design of the study. All authors contributed to the analysis or interpretation of the data.
Declaration of Conflicting Interests
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by GSK, for the analysis of GSK Studies (BEL110752 (NCT00424476)), (BEL110751 (NCT00410384)), (BEL112341 (NCT01484496)), (BEL113750 (NCT01345253)), and (BEL115471 (NCT01632241)].
Correction (May 2025):
Minor corrections have been made throughout the article for clarity and accuracy.
Ethical statement
Data availability statement
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
