Abstract
Objectives
To assess the real-world efficacy and safety of telitacicept in Chinese systemic lupus erythematosus (SLE) patients.
Methods
This retrospective single-center observational study enrolled 41 SLE patients treated with telitacicept between April 2021 and March 2022. Baseline characteristics and 24-week follow-up clinical outcomes were collected for analysis.
Results
A total of 41 SLE patients who received telitacicept at least once were included. Compared with baseline (10.0 ± 6.85), SLEDAI-2K scores significantly decreased at 12 weeks (6.25 ± 5.46, p < .001) and 24 weeks (4.18 ± 4.78, p < .001). The proportion of organ involvement, as assessed by SLEDAI-2K, significantly decreased in skin/mucosa and musculoskeletal involvement. The mean glucocorticoid dose (prednisone equivalent) was significantly reduced at 12 weeks (14.19 ± 8.88 mg/d, p < .001) and 24 weeks (9.86 ± 4.25 mg/d, p = .001) compared with baseline (20.87 ± 16.71 mg/d). C3 and C4 levels showed an increasing trend. In patients with lupus nephritis, 24-h urinary protein levels significantly reduced from 2.81 ± 3.83 g/day at baseline to 1.00 ± 1.32 g/day after 24 weeks of treatment (p = .005), with a 68.66% decline (IQR, 35.61%, 76.30%). Albumin levels also significantly improved (p = .003). Nine treatment-emergent adverse events (TEAEs) were recorded in six patients, all of which were mild and moderate.
Conclusions
In this real-world study, telitacicept combined with conventional therapy showed good onset efficacy and safety for treating SLE patients.
Introduction
Systemic lupus erythematosus (SLE) is a chronic, multisystemic autoimmune disease. 1 Its manifestations can range from mild skin conditions to life-threatening organ failures. The treatment goal of SLE is remission or low disease activity, with the prevention of flares in all organs, maintained with the lowest possible dose of glucocorticoids.2,3 According to the European Alliance of Associations for Rheumatology (EULAR) Recommendations for the Management of SLE, 4 the ideal treatment target is remission, defined by the definition Of Remission In SLE (DORIS) criteria as clinical SLE disease activity index 2000 (SLEDAI-2K) score of zero, no symptoms of lupus activity, a Physician’s Global Assessment (PGA) score of less than 0.5, and a maximum glucocorticoids dose of 5 mg/day or other immunosuppressive drugs.5,6 An alternative target is the Lupus Low Disease Activity State (LLDAS). 7 which is achieved when the patient’s SLEDAI-2K scores is four or less, with no new lupus disease activity, a PGA score of one or less, and a current prednisone (or equivalent) dose not exceeding 7.5 mg/day, while the patient may be on anti-malarial drugs or immunosuppressive therapy. LLDAS is associated with a reduction in flares and accrual of damage-related morbidity. 8 Therefore, it is an important endpoint to evaluate clinical benefit.
The prevalence of SLE in China was 30
Telitacicept, a novel recombinant fusion protein targeting B-cell lymphocyte stimulator (BLyS) and a proliferation-inducing ligand (APRIL), showed promising efficacy and safety in phase IIb trial and phase III trial for SLE, and was approved in China in 2021.11–14 However, in the real world, the clinical value of telitacicept is still unclear. Our study aims to explore the efficacy and safety of telitacicept in real-world clinical practice.
Patients and methods
Patients
A total of 41 SLE patients, diagnosed according to the American College of Rheumatology (ACR) or Systemic Lupus International Collaborating Clinics (SLICC) criteria, and received telitacicept at 160 mg or 80 mg weekly at least once in the Department of Rheumatology and Immunology of Shengjing Hospital Affiliated to China Medical University, China, from April 2021 to March 2022, were included.
Study design
This study was a retrospective, observational study. The efficacy outcomes were the change in the SLEDAI-2K scores from baseline to 12 weeks and 24 weeks and the change in the proportion of organ involvement via the SLEDAI-2K scores. DsDNA, complements, immunoglobulin, and 24-h urinary protein level changes were also analyzed as efficacy outcomes. We also used an alternative composite endpoint: “SLEDAI-2K ≤4 plus prednisolone (or equivalent) dose ≤7.5 mg daily” and attempted to evaluate the possibility of LLDAS with telitacicept treatment. Treatment-emergent adverse events (TEAEs) were collected and analyzed as safety outcomes.
Data collection
The data collection process encompassed a comprehensive range of patient information and clinical data. Baseline demographic information such as age, gender, and disease duration were systematically recorded. Clinical data included an assessment of SLE disease activity using the SLEDAI-2K scores and determination of organ involvement as defined by the SLEDAI-2K criteria. The laboratory tests included routine blood and urine tests, detection of antinuclear antibody (ANA), measurement of anti-double-stranded DNA (anti-dsDNA) antibody levels, complement (C)3, C4, serum albumin, immunoglobulin G (IgG), immunoglobulin A (IgA), immunoglobulin M (IgM), and 24-h urine protein quantification was performed. Treatments administered, including doses of telitacicept (RemeGen Co., Ltd), glucocorticoids, and immunosuppressants, were meticulously documented to ensure a thorough analysis of efficacy and safety outcomes.
Statistical analysis
Statistical analysis was conducted using SPSS 23.0 and GraphPad Prism 8. The variables were described using mean and standard deviation (SD). Categorical variables were described using percentages. A paired samples t test was used to compare continuous variables at different time points (baseline, 12 weeks, and 24 weeks). A chi-square test was applied to compare categorical variables. All tests were two-sided, and a p-value of < .05 was considered statistically significant.
Informed consent was obtained from all patients before receiving telitacept treatment. The Medical Ethics Committee of Shengjing Hospital, Affiliated with China Medical University approved this study protocol (approval number 2022PS081K).
Results
Patient’s characteristics
Baseline patient demographics and disease characteristics (n = 41).

Efficacy outcomes
Forty-one patients had consistent telitacicept treatment for 12 weeks. The SLEDAI-2K scores at 12 weeks was 6.25 ± 5.46, with a significant decrease compared with baseline (10.02 ± 6.85, p < .001) (Figure 1(a)). However, by 24 weeks, three patients withdrew telitacicept due to adverse events (injection site pruritus, fever, and upper respiratory tract infection), one due to pregnancy planning, and three because of favorable clinical outcomes. So, 34 patients received consistent telitacicept treatment for 24 weeks. The SLEDAI-2K scores at 24 weeks was 4.18 ± 4.78, with a significant decrease compared with the baseline (p < .001) (Figure 1(a)). In the subgroup of the SLEDAI-2K score ≥10, the SLEDAI-2K scores was 9.95 ± 5.58 at 12 weeks and 6.47 ± 5.04 at 24 weeks, with significant decreases compared with the baseline (15.70 ± 5.08, both p < .001). Parallel trends were observed in the subgroup with baseline SLEDAI-2K scores below 10 (Figure 1(b)). SLEDAI-2K scores and system involvement. (a) SLEDAI-2K scores at baseline, 12 weeks and 24 weeks; (b) SLEDAI-2K scores in the subgroup of SLEDAI-2K ≥10 and <10; (c) Proportion of SLEDAI-2K scores decreased ≥ 4; (d) Percentages of patients with different system involvement at baseline, 12 weeks and 24 weeks, assessed by SLEDAI-2K scores. CNS, central nervous system. *p < .05 vs. baseline; **p < .01 vs. baseline; ***p < .001 vs. baseline.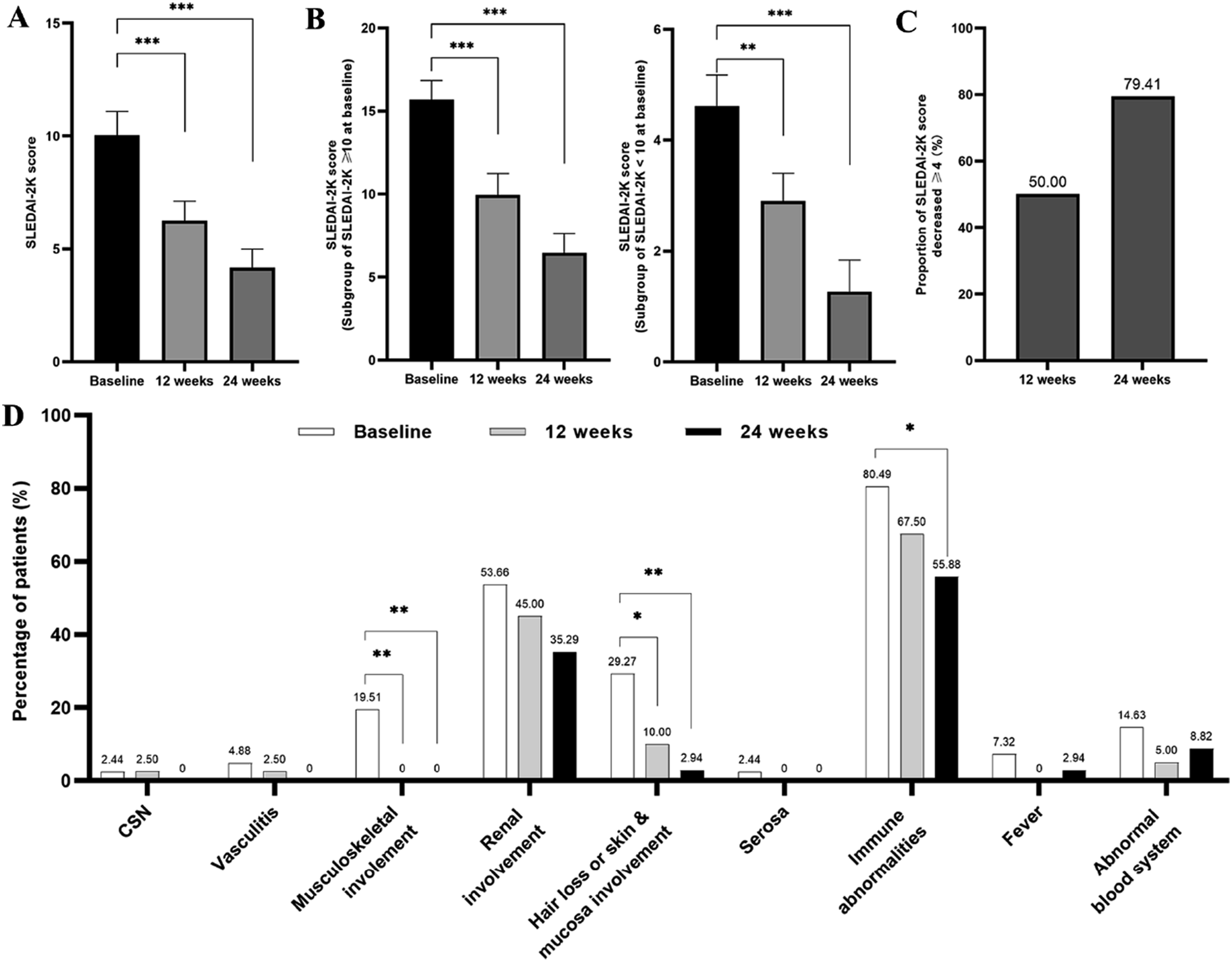
SLEDAI-2K scores and serologic parameters at baseline, 12 weeks and 24 weeks.
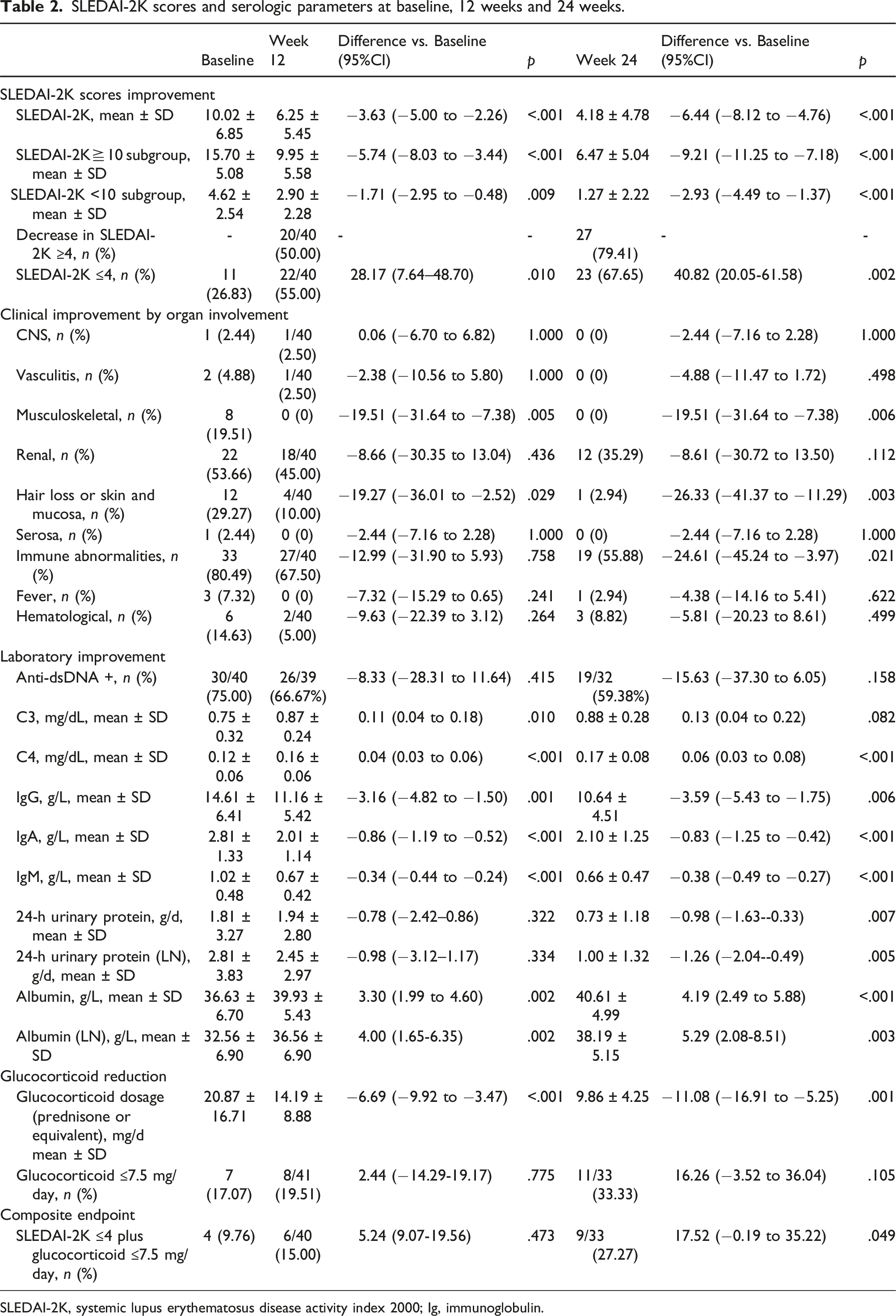
SLEDAI-2K, systemic lupus erythematosus disease activity index 2000; Ig, immunoglobulin.
Furthermore, significant increases in complement components were observed, with C3 rising to 0.11 mg/dl (0.04 to 0.18, p = .010) and C4 to 0.04 mg/dl (0.03 to 0.06, p < .001) at 12 weeks, and further to 0.06 mg/dl (0.03 to 0.08, p < .001) at 24 weeks from a baseline of 0.12 ± 0.06 mg/dl (Table 2 and Figure 2(a)). Immunoglobulin levels (IgG, IgA, and IgM) decreased persistently from baseline to 24 weeks, remaining within normal ranges (Table 2 & Figure 2(b)). The rate of positive anti-dsDNA showed a declining trend (Figure 2(c)). Serologic parameters at baseline, 12 weeks and 24 weeks. (a) C3 and C4; (b) IgG, IgA and IgM; (c) Anti-dsDNA titer (upper limit of detection 300 IU/mL); (d) 24-h urinary protein. *p < .05 vs. baseline; **p < .01 vs. baseline; ***p < .001 vs. baseline; NS means no significant.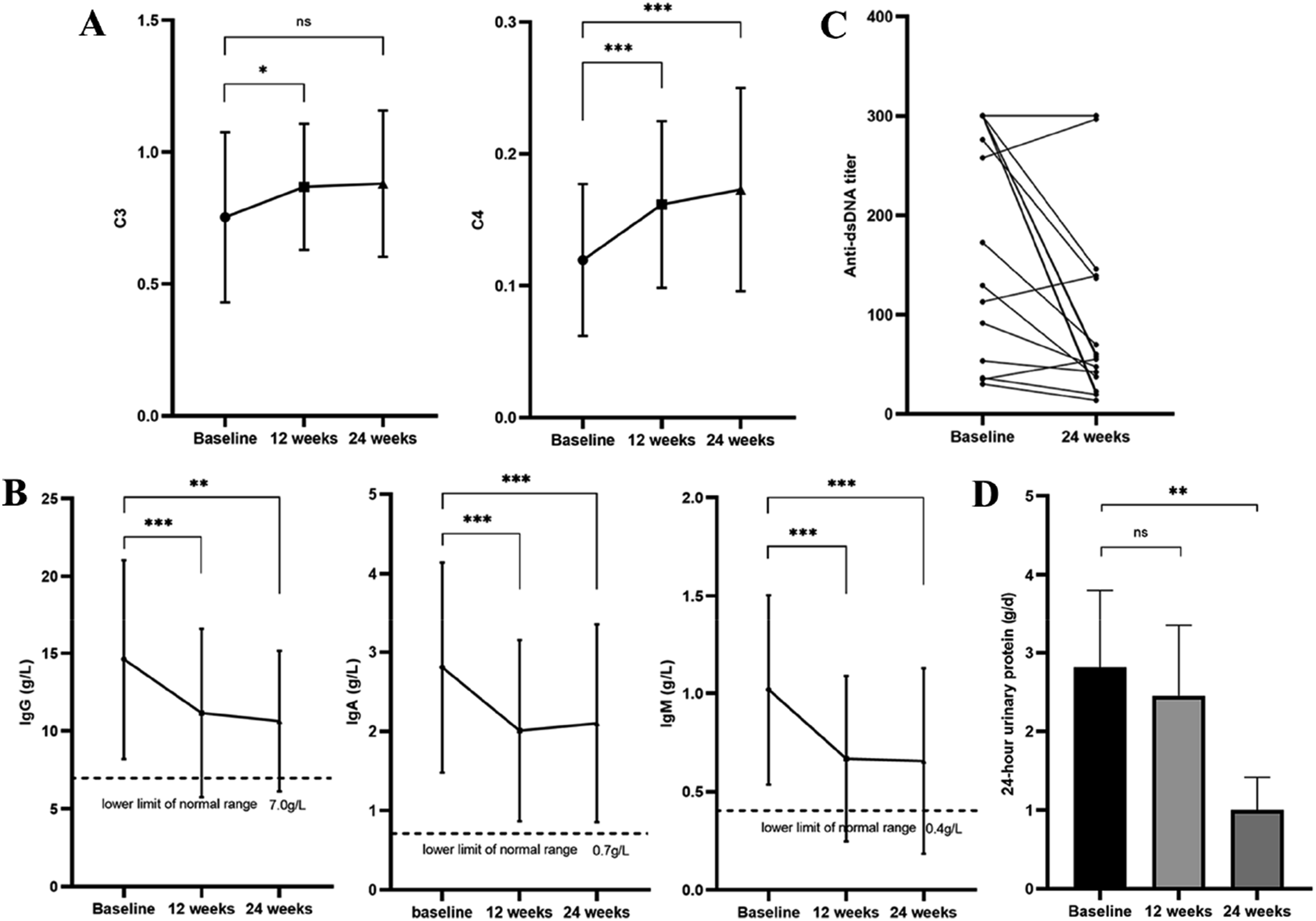
There were 16 (39.02%) patients diagnosed with lupus nephritis. The levels of 24-h urinary protein were significantly reduced from 2.81 ± 3.83 g/day at baseline to 1.00 ± 1.32 g/day after 24 weeks of treatment (p = .005) (Figure 2(d)), with a decline in 24-h urinary protein levels by 68.66% (IQR, 35.61%, 76.30%). Additionally, serum albumin levels significantly improved from 32.56 ± 6.90 g/L at baseline to 36.56 ± 6.90 g/L and 38.19 ± 5.15 g/L after 12 and 24 weeks of treatment, respectively (p < .01) (Table 2).
The glucocorticoid (prednisone or equivalent) dose reduced from baseline (20.87 ± 16.71 mg/d) to 12 weeks (14.19 ± 8.88 mg/d, p < .001) and 24 weeks (9.86 ± 4.25 mg/d, p = .001) significantly (Figure 3(a)). The proportion of glucocorticoid ≤7.5 mg/day increased from baseline (17.07%) to 12 weeks (19.51%, p = .775) and 24 weeks (33.33%, p = .105) (Figure 3(b)). And the proportion of SLEDAI-2K ≤4 increased from baseline (26.83%) to 12 weeks (55.00%, p = .010) and 24 weeks (67.65%, p = .002) (Figure 3(c)). Thus, the proportion of composite endpoint, SLEDAI-2K ≤4 plus glucocorticoid ≤7.5 mg/day, increased from baseline (9.76%) to 12 weeks (15.00%, p = .473) and 24 weeks (27.27%, p = .049) (Table 2 and Figure 3(d)). Glucocorticoid dosage and composite endpoint. (a) Glucocorticoid dosage; (b) Proportion of glucocorticoid ≤7.5 mg/day; (c) Proportion of SLEDAI-2K ≤4; (d) Proportion of composite endpoint, SLEDAI-2K ≤4 plus glucocorticoid ≤7.5 mg/day. *p < .05 vs. baseline; **p < .01 vs. baseline; ***p < .001 vs. baseline.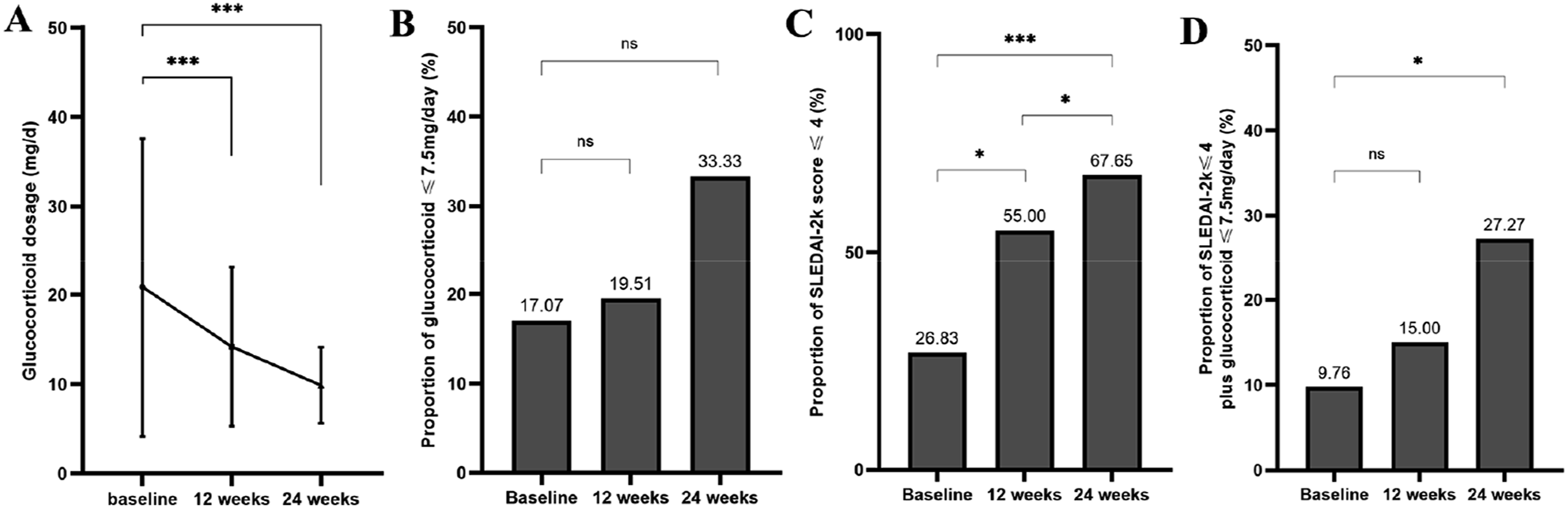
Safety
Treatment-emergent adverse events (TEAEs) that occurred during the period of telitacicept treatment.

aAppeared in the same patient.
bAppeared in the same patient.
cAppeared in the same patient.
dAppeared in the same patient.
eAppeared in the same patient.
Discussion
One of the goals of SLE treatment is remission or low disease activity and prevention of flares in all organs, maintained with the lowest possible dose of glucocorticoids. 15 LLDAS has been generated and validated by a panel of lupus experts from the Asia-Pacific region. 8 Patients who spent over 50% of their observed time in LLDAS had significantly reduced organ damage accrual 8. A study of the Chinese SLE cohort also proved a highly protective effect for preventing flares and damage accrual of LLDAS. 16 However, in real practice, only 7.85% of Chinese SLE patients could reach DORIS “low disease activity,” and 2.47% were in “clinical remission on therapy”. 10 An Italy SLE cohort data showed that failure to achieve LLDAS 6 months after diagnosis is associated with early damage accrual. 17 New evidence showed that novel biologics added value to conventional therapy in LLDAS achievement.2,3,18
Telitacicept, a fusion protein, marries transmembrane activator and calmodulin ligand interactor (TACI) with the Fc component of human IgG1, strategically targeting BLyS and APRIL.19,20 This design thwarts their interaction with the three B cell receptors—TACI, B cell maturation antigen (BCMA), and B cell activating factor receptor (BAFF-R). The latter, BAFF-R, with its high affinity for BLyS, is instrumental in the maturation of transitional B cells to naïve B cells, as delineated by Dörner et al. 21 Belimumab, targeting BLyS, depletes mature B cells, curtailing the production of autoantibodies, a phenomenon documented by Cancro et al. 22 TACI’s dual affinity for both BLyS and APRIL facilitates the differentiation of mature B cells to plasmablasts and short-lived plasma cells, thereby inducing class switching from IgD to IgM, IgA, and IgG, a process resulting in a surge of autoantibodies. 23 BCMA’s selective affinity for APRIL underpins the survival of long-lived plasma cells, perpetuating the synthesis of autoantibodies. In its role as a dual inhibitor, telitacicept imposes a blockade on B cell maturation and plasma cell transition, fostering apoptosis in long-lived plasma cells and attenuating the production of autoantibodies, as illustrated by Shi et al. 24 and Fan et al. 19 These mechanisms underscore the therapeutic potential of telitacicept in SLE management. Clinical trials, including a phase IIb study and a phase III randomized controlled trial, have affirmed telitacicept’s efficacy and safety in SLE treatment.11,12 Despite these advances, further research must delineate its practical benefits in routine clinical applications.
Our study aimed to explore the effectiveness of telitacicept in the real world, especially its onset efficacy and safety, and the efficacy of treat-to-target, reduction of glucocorticoid, and improvement of lupus nephritis. This study enrolled 41 SLE patients treated with telitacicept at least once in our hospital. Forty-one patients have been treated with telitacicept for 12 weeks at least, and 34 for 24 weeks. The mean SLEDAI-2K scores at 24 weeks was 4.18 ± 4.78 and 6.25 ± 5.46 at 12 weeks, significantly decreasing compared with the baseline. This is especially true in the subgroup of SLEDAI-2K ≥10 (6.47 ± 5.04 at 24 weeks and 9.95 ± 5.58 at 12 weeks vs. 15.70 ± 5.08 in baseline). Significant improvements in organ involvement via the skin/mucosa, musculoskeletal, and immune systems were found in this study as well. This result was consistent with the Phase IIb and III studies.11,12 Treatment compliance is a crucial factor for chronic disease; data showed that poor treatment compliance is one of the key issues in the poor outcomes of Chinese SLE patients. 10 Our study had relatively good patient management; 100% of patients had treatment duration of telitacicept in 12 weeks, which guaranteed the good onset effect of telitacicept treatment. SLE patients receiving long-term glucocorticoid therapy were at significant risk of morbidity due to permanent organ damage. 25 In a cohort study, patients receiving >7.5 mg/d of prednisone exhibited a higher risk of cumulative organ damage and steroid-related harm after 5 years of treatment. 26 Finally, a cut-off of ≤7.5 mg/day of prednisolone or equivalent glucocorticoid was most recommended in the LLDAS definition. 27 Our study showed that via telitacicept treatment, the proportion of glucocorticoid ≤7.5 mg/day increased from baseline (17.07%) to 12 weeks (19.51%) and 24 weeks (33.33%). SLEDAI-2 K ≤4 is another component in the LLDAS definition. 27 We evaluated the proportion of SLEDAI-2K ≤4 in our study. Compared to the baseline, the proportion of SLEDAI-2K ≤4 increased from baseline (26.83%) to 12 weeks (55.00%) and 24 weeks (67.65%). The PGA score can allow physicians to avoid underestimating disease severity for some important systems not measured in the SLEDAI-2K, such as gastrointestinal involvement and hemolytic anemia. Hence, PGA is included in the composite measures of LLDAS. In our data, no PGA was recorded. However, no other important systems excluded from SLEDAI-2K were found in our data. We used an alternative composite endpoint: “SLEDAI-2K ≤4 plus prednisolone (or equivalent) dose ≤7.5 mg daily” to reflect the LLDAS endpoint. The proportion of the composite endpoint increased significantly from baseline (9.76%) to 12 weeks (15.00%) and 24 weeks (27.27%). This result is likely the post-hoc analysis of the ADRESS II study, 2 the TULIP extension trial, 18 and the BLESS-52/76 study. 3
In our study, nearly 40% of patients had lupus nephritis. Following a 6-month therapeutic regimen, we observed a significant reduction in 24-h urinary protein levels from an initial 2.81 ± 3.83 g/day to 1.00 ± 1.32 g/day at week 24, with a decline rate of 68.66% (IQR, 35.61%, 76.30%), which was in accordance with the targets (reduction in proteinuria ≥50% after 6 months treatment) according to the 2023 update of EULAR recommendations for the management of SLE. 28 Meanwhile, serum albumin concentrations significantly improved, increasing from 32.56 ± 6.90 g/L at baseline to 36.56 ± 6.90 g/L and 38.19 ± 5.15 g/L after 12 and 24 weeks of treatment, respectively. These findings underscore the efficacy of combining telitacicept with conventional treatments in managing lupus nephritis. Considering the 2023 Update of EULAR recommendations for the management of SLE, 28 an intensive treatment strategy of early combination of belimumab plus MMF or low-dose CYC in LN treatment is endorsed. Nevertheless, the optimal telitacicept treatment protocol for lupus nephritis patients remains to be further explored.
This study carefully monitored laboratory measurements, including complement components and immunoglobulin levels, during telitacicept treatment. We observed a significant elevation in the levels of C3 and C4, which are important markers of disease activity in SLE. Notably, treatment with B cell-targeted biologics, such as belimumab, rituximab, and atacicept, has been frequently associated with a decline in immunoglobulin levels.29–31 Atacicept, which has the same mechanism as telitacicept, had an unexpected decline in serum immunoglobulin G, especially serious infections in a trial in patients with lupus nephritis.29–31 However, although the mean serum IgG, IgA, and IgM levels significantly declined in our study, they were all above the bottom line of normal value. This is also consistent with the Phase IIb and III studies. The comparably high affinity of atacicept to APRIL might be related to its safety concern. 32
This study showed an acceptable safety of telitacicept. All AEs were mild or moderate. Although after 24 weeks of treatment, 17 mild hypo-immunoglobulin events (5 of IgG, 2 of IgA, and 10 of IgM) were found, no serious infection event was reported. These results suggest that telitacicept is well-tolerated and does not increase the risk of serious infections.
This study does have some limitations. The number of subjects was small, and there was no control group to compare telitacicept with conventional treatment and relevant short observational time, which was only 24 weeks. Despite these limitations, our study showed the potential value of telitacicept onset treatment in SLE patients combined with traditional therapy. To the best of our knowledge, this is the first observational study that explores the potential benefits of telitacicept in treat-to-target strategy and lupus nephritis patients. Real-world studies, which have large sample sizes, long-term observational time, and focus on treat-to-target treatment strategy and long-term outcomes of SLE patients, should be conducted in the future to assess the comprehensive effectiveness and safety of telitacicept in clinical practice. Moreover, the studies of the mechanism of action of telitacicept are also encouraged to be added to clinical studies to explore the precise utilization of telitacicept and ensure patient value maximization.
In conclusion, our study showed that telitacicept combined with conventional therapy has good onset efficacy and safety for treating SLE patients.
Footnotes
Acknowledgments
We are grateful to all patients and their families, as well as all members of the study group.
Author contributions
Conceptualization: Jing Zhang, Yun Guo; Data collection: Yue Sun, Na Zhang; Data analysis: Jing Zhang, Li-Hua Chang; Data interpretation: Lei Jin; Writing-original draft: Jing Zhang, Yun Sun, Na Zhang, Li-Hua Chang, Lei Jin; Writing-review & editing: Yun Guo.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Basic Scientific Research Project of Colleges and Universities of Liaoning Province Education Department (Surface project, No. LJKMZ20221212).
Ethical statement
Data Availability Statement
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
