Abstract
Objective
Although belimumab has been widely used in patients with systemic lupus erythematosus (SLE) globally, real-world safety data among Chinese patients are limited, particularly for children. This study assessed the safety and tolerability of belimumab in adult and paediatric patients with SLE in China in real-world clinical practice.
Methods
This Phase 4, multicentre, prospective, observational study enrolled patients prescribed intravenous belimumab by their physicians in tertiary hospitals, independent of a clinical study, during routine clinical visits between May 2021 and May 2022. Patients could have been receiving belimumab prior to enrolment. The primary objective was to describe the incidence of adverse events (AEs), serious AEs (SAEs), adverse drug reactions (ADRs) and AEs of special interest (AESIs) over the 24-week follow-up period. Data were collected at enrolment and approximately 4, 12 and 24 weeks post-enrolment, during routine clinical visits. AEs, ADRs and SAEs were collected as independent events. The safety population comprised patients who received ≥1 dose of belimumab and completed ≥1 follow-up visit.
Results
Overall, 417 patients were included in the analysis (safety population); 89.2% were female and mean (standard deviation) age was 35.5 (11.9) years. AEs were reported in 158 patients (37.9%) and were mostly mild (79.1%). The most common AEs were upper respiratory tract infections (n = 19, 4.6%) and hypokalaemia (n = 18, 4.3%; all mild). Among 22 patients (5.3%) with SAEs, four patients (1.0%) had drug-related SAEs (pneumonia, respiratory tract infection, gingivitis and decreased white blood cell and neutrophil count). ADRs were experienced by 25 patients (6.0%), most commonly urinary tract infections (n = 5, 1.2%). There were no fatal SAEs. AESIs occurred in 14 patients (3.4%), including infections of interest (n = 5, 1.2% all herpes zoster), serious selected psychiatric events (n = 3, 0.7%) and infusion-related systemic and anaphylactic reactions (n = 7, 1.7%). Of 14 paediatric patients enrolled, six experienced AEs, zero ADRs, two SAEs and one AESI.
Conclusion
Belimumab was generally well tolerated in adult and paediatric patients with SLE in this real-world setting in China, with no new safety signals identified. Future assessment of long-term belimumab safety in China beyond 24 weeks will extend this current body of evidence.
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease that can affect various organs and systems in the body, often with severe manifestations leading to considerable morbidity and early mortality. 1 Available evidence suggests that the prevalence of SLE in China is in line with the estimated global prevalence, with 30–70/100,000 people having SLE in China versus an average of 44/100,000 globally.2,3 The symptomatology of SLE in the Chinese population differ from those in patients in the USA and Europe, particularly for those with haematological, neurological and kidney involvements. 4 Previous studies indicated that Asian patients with SLE exhibit higher rates of kidney involvement compared with other ethnic groups, notably Caucasians.5,6
Treatment options for SLE include antimalarials, glucocorticoids (GCs), immunosuppressants and biologics. 7 GC use should be minimised to the lowest dose possible, or used solely as bridging therapy due to their detrimental effects on patients’ health. 7 In 2020, the Chinese Rheumatology Association (CRA) published clinical practice guidelines specific to SLE recommending the use of biologics for patients with SLE who are refractory or intolerant to immunosuppressants and/or GCs, or have relapsed. 8
One such biologic is belimumab, a human IgG1λ monoclonal antibody that selectively binds to and inhibits soluble B-lymphocyte stimulator (BLyS). Belimumab was initially approved in 2011 for the treatment of adult patients with SLE, and was approved for paediatric patients with SLE in 2019 and for lupus nephritis in 2020. 9 Belimumab is approved in China for the treatment of active, autoantibody-positive SLE in adults and children, and was first approved in adults in 2019; belimumab is also licensed for treatment of lupus nephritis in China.10–12
Belimumab’s efficacy and safety in the treatment of SLE have been demonstrated in three global Phase 3 trials13–15; however, the number of Asian patients receiving marketed doses of belimumab varied between trials (Furie et al 2011: n = 11/273 [4.0%]; Navarra et al 2011: n = 116/290 [40.0%]; Stohl et al 2017: n = 111/556 [20.0%]).13–15 Specifically in the Asian population, the efficacy and safety of belimumab were demonstrated in a Phase 3 randomised, controlled trial in North East Asia (China, South Korea and Japan; n = 451/677 [66%] patients treated with belimumab), with an open-label extension study in China (425/547 [78%] patients from the initial Phase 3 North East Asia study).16,17 Global real-world evidence has demonstrated belimumab’s effectiveness at reducing SLE disease activity and its GC-sparing effect.18,19
The safety of belimumab has also been studied in a large Phase 4 global safety study, known as the BASE study. 20 This randomised controlled trial found that although the number of adverse events was small, there was an imbalance of serious infusion or hypersensitivity reactions, depression and suicidality among patients with SLE treated with belimumab compared with placebo, 20 identifying these events as adverse events of interest in further safety studies. All-cause mortality was similar between both treatment groups during the on-treatment period. 20
Despite the wealth of global data on belimumab, there is a lack of real-world safety data in Chinese patients with SLE,21,22 with a notable absence of evidence among the paediatric patient population. This post-authorisation safety study aimed to assess the safety and tolerability of belimumab in adult and paediatric patients with SLE in China in real-world clinical practice, consistent with the regulatory requirement of intensive drug monitoring for newly marketed drugs in China.
Methods
Study design
This was a Phase 4, multicentre, prospective, observational study (GSK Study 213210) to assess the safety and tolerability of belimumab in adult and paediatric Chinese patients with SLE receiving intravenous (IV) belimumab as part of their standard medical care. Investigators enrolled all eligible, consenting patients from May 2021 to May 2022 in the selected study sites, which were tertiary hospitals; investigators collected all data at routine clinical visits. The total duration of safety monitoring was 24 weeks (Supplemental Figure 1), and all safety data were collected prospectively, up to November 2022. The primary objective was to describe the incidence of safety events. At Visit 1 (onsite), information related to inclusion/exclusion, demographic details, belimumab treatment (if ongoing), medical history, concomitant medication use, adverse events (AEs), serious AEs (SAEs), adverse drug reactions (ADRs), AEs of special interest (AESIs) and pregnancy were collected. Visits 2–4 were completed at 4, 12 and 24 weeks, respectively, and could be carried out onsite if the patient had a routine visit scheduled, or via telephone if not. At these visits, information on AEs, SAEs, ADRs, AESIs, belimumab treatment, concomitant medication and pregnancy was collected, unless the patient withdrew consent. Patients who discontinued belimumab but consented to continue in the study had data collected via telephone until Week 24.
Patients
Patients were required to have a physician-confirmed diagnosis of SLE that met relevant SLE classification criteria (i.e. American College of Rheumatology [ACR] 1997 criteria, the Systemic Lupus International Collaborating Clinics [SLICC] 2012 criteria or the ACR/European Alliance of Associations for Rheumatology [EULAR] 2019 criteria).23–26 In China, belimumab is approved for use in patients ≥5 years of age with active, autoantibody-positive SLE with high disease activity (e.g., positive anti–double-stranded DNA and low complement; Safety of Estrogens in Lupus Erythematosus: National Assessment Version of the Systemic Lupus Erythematosus Disease Activity Index [SELENA-SLEDAI] score ≥8), who are receiving standard therapy.11,12 Patients were, according to the investigator’s judgment, prescribed IV belimumab for appropriate medical use, and the decision to prescribe belimumab was unrelated to participation in this observational study. Patients could have already received belimumab treatment (belimumab-treated) or be initiating belimumab treatment (belimumab-naïve) at the time of enrolment. Exclusion criteria included participation in clinical trials or initiation of belimumab as part of a clinical trial at the time of the informed consent process or being considered unsuitable to be included for any reason judged by the investigator. All patients provided written informed consent. For patients under 18 years of age, the parent or caregiver provided consent, but the patient could co-sign the form, if able.
Outcomes
Safety was assessed through 24 weeks by monitoring AEs, SAEs (including SAEs related to the study drug), ADRs and AESIs (including all-cause mortality). All were reported according to Medical Dictionary for Regulatory Activities (MedDRA) Version 24.0 system organ class and preferred terms. AEs, ADRs and SAEs in this study were collected independently, and there was no inclusion relationship among the three, i.e. an AE reported as an SAE was included only in SAEs, a non-serious ADR was included only in ADRs and a non-serious AE not reported as an ADR was included in AEs. Investigators assessed whether each AE or SAE was potentially related to treatment. AESIs included infections of special interest (including serious and non-serious opportunistic infections, tuberculosis or tuberculosis reactivation, hepatitis B, hepatitis C, herpes zoster), infusion-related systemic reactions and anaphylactic reactions, progressive multifocal leukoencephalopathy, selected serious psychiatric events (depression and negative thoughts/suicidal ideation, intent or behaviour), malignancies (excluding non-melanoma skin cancers), non-melanoma skin cancers and all-cause mortality. Safety outcomes were also evaluated among older patient (≥65 years of age) and paediatric patient (<18 years of age) subgroups. The tolerability of belimumab and reasons for discontinuation were recorded in the total safety population.
Statistical analysis
In this post-marketing observational study, no hypothesis testing was performed, and only descriptive data are presented. All enrolled patients who received belimumab treatment during the study period were included in analyses. All analyses were performed using SAS version 9.4 or higher (SAS Institute, Cary, NC). Unless otherwise specified, continuous data were summarised using descriptive statistics, and categorical data were summarised as the number and percentage of patients in each category. If a date was missing the day, but had a complete year and month, the date was imputed using the ‘15th’; if the month or year was incomplete then the date was recorded as missing. Missing relatedness data were handled using the worst-case scenario.
Ethics
Before study commencement, approval was obtained from the relevant Ethics Committees for each institution; the full name of each Ethics Committee and associated approval number are provided in Supplemental Table 1. The study was performed in accordance with International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use – Good Clinical Practices (GCP), China GCP and good pharmaco-epidemiological practice guidelines. 27
Results
Patient population
The study enrolled 417 patients: 109 (26.1%) belimumab-naïve patients and 308 (73.9%) belimumab-treated patients. All were included in the safety population, and 409 (98.1%) completed the study. Of the eight patients who did not complete the study, three were lost to follow-up and one withdrew consent; reasons for the remaining four were listed by the investigator as ‘other’.
Patient demographics, clinical characteristics and belimumab exposure (enrolled/safety population).
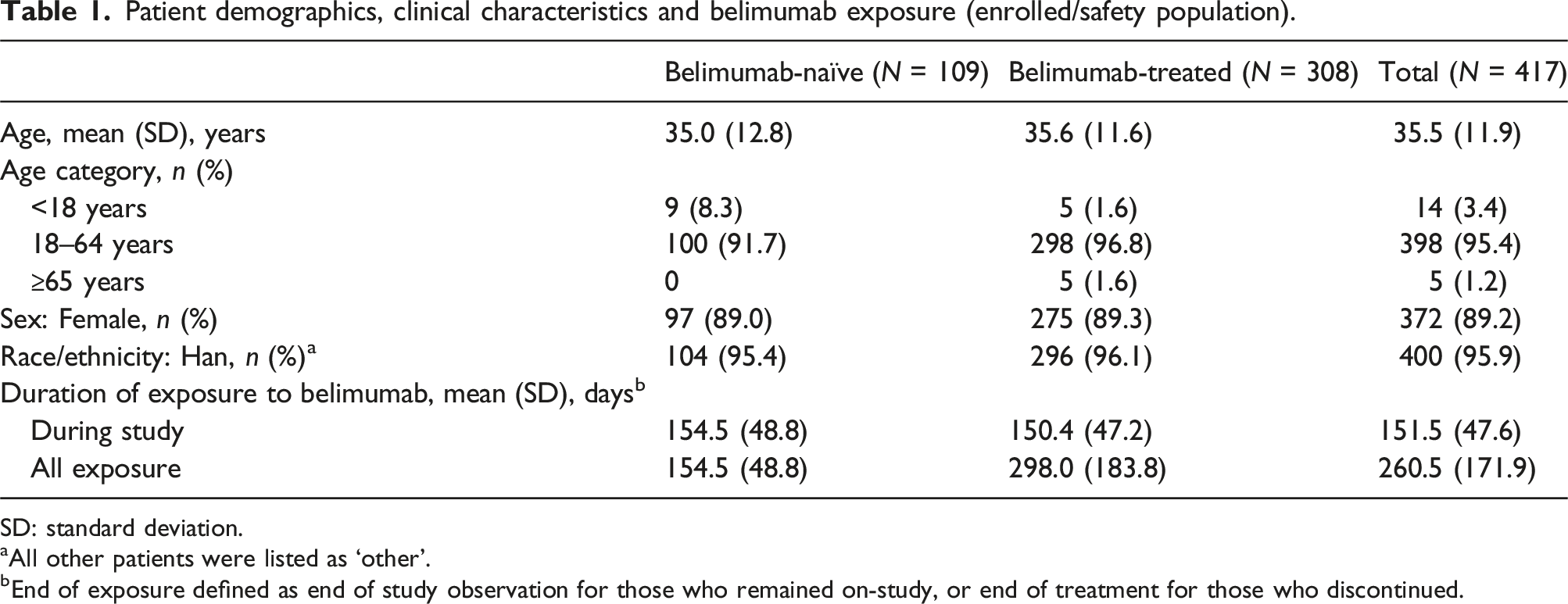
SD: standard deviation.
aAll other patients were listed as ‘other’.
bEnd of exposure defined as end of study observation for those who remained on-study, or end of treatment for those who discontinued.
All enrolled patients had received concomitant medications prior to the study, but details for one patient were unavailable due to study withdrawal. Of the 416 patients with data, the most commonly used concomitant medications were systemic corticosteroids (n = 387, 92.8%), antimalarials (n = 265, 63.5%) and immunosuppressants (n = 386, 92.6%). In addition, 74.6% of patients (n = 311) were taking vitamins. The three most common concomitant medications were hydroxychloroquine sulphate (n = 265, 63.5%), calcium carbonate-vitamin D3 (n = 214, 51.3%) and mycophenolate mofetil (n = 181, 43.4%).
Adverse events
Overview of AEs and severity (safety population).
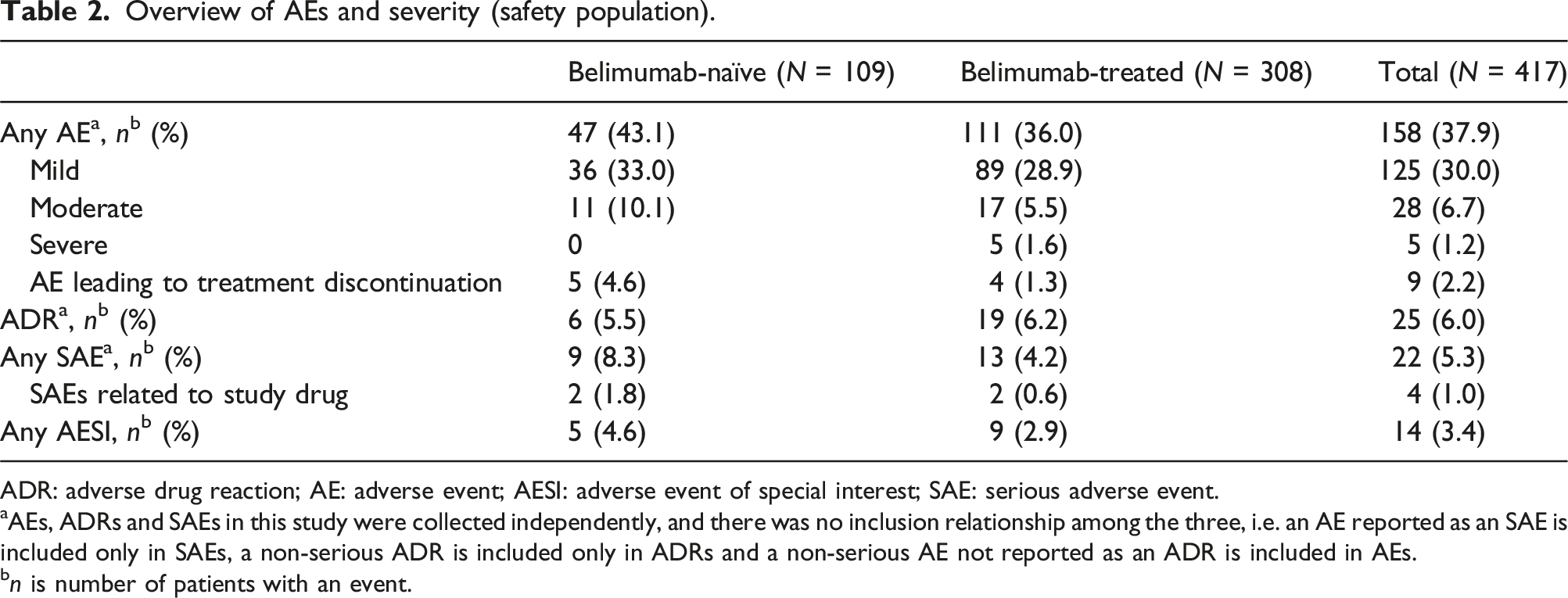
ADR: adverse drug reaction; AE: adverse event; AESI: adverse event of special interest; SAE: serious adverse event.
aAEs, ADRs and SAEs in this study were collected independently, and there was no inclusion relationship among the three, i.e. an AE reported as an SAE is included only in SAEs, a non-serious ADR is included only in ADRs and a non-serious AE not reported as an ADR is included in AEs.
bn is number of patients with an event.
AEs by system organ class and preferred term (occurring in ≥1% of patients in the safety population).
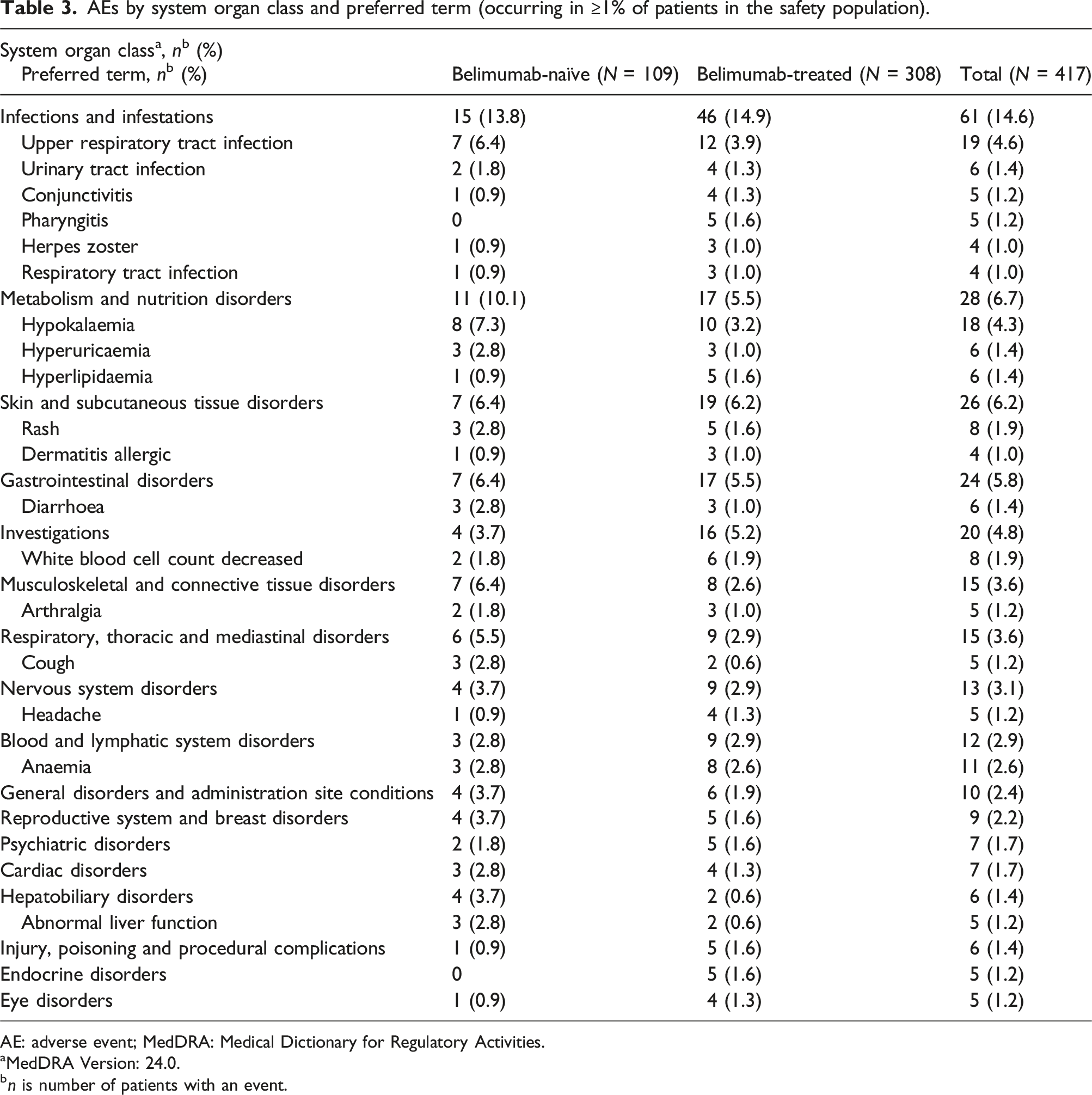
AE: adverse event; MedDRA: Medical Dictionary for Regulatory Activities.
aMedDRA Version: 24.0.
bn is number of patients with an event.
Serious adverse events
SAEs by system organ class and by preferred term (occurring in >1 patient in the total safety population).
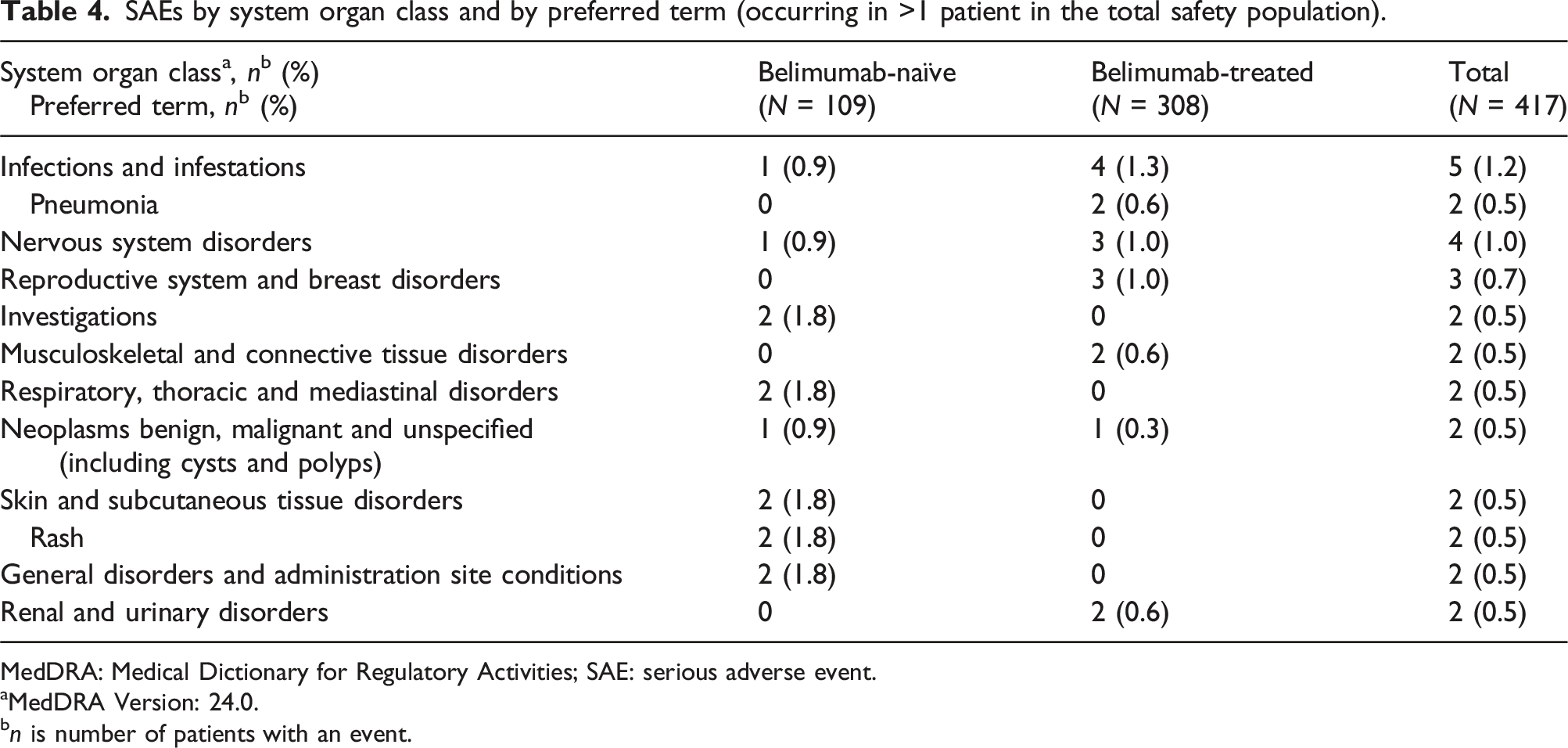
MedDRA: Medical Dictionary for Regulatory Activities; SAE: serious adverse event.
aMedDRA Version: 24.0.
bn is number of patients with an event.
Adverse drug reactions
ADRs by system organ class and by preferred term (occurring in >1 patient, in the safety population).
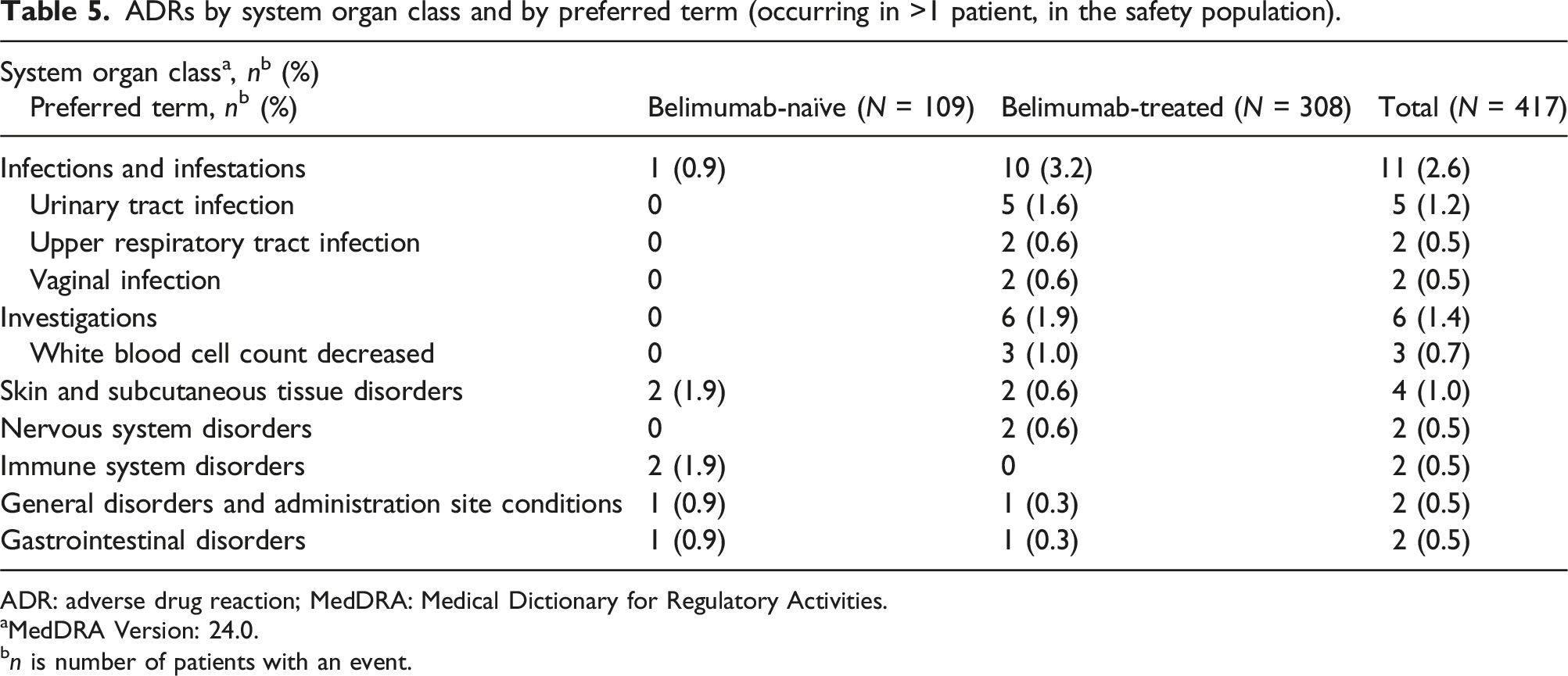
ADR: adverse drug reaction; MedDRA: Medical Dictionary for Regulatory Activities.
aMedDRA Version: 24.0.
bn is number of patients with an event.
Adverse events of special interest
Summary of AESIs, action taken and outcomes (safety population).
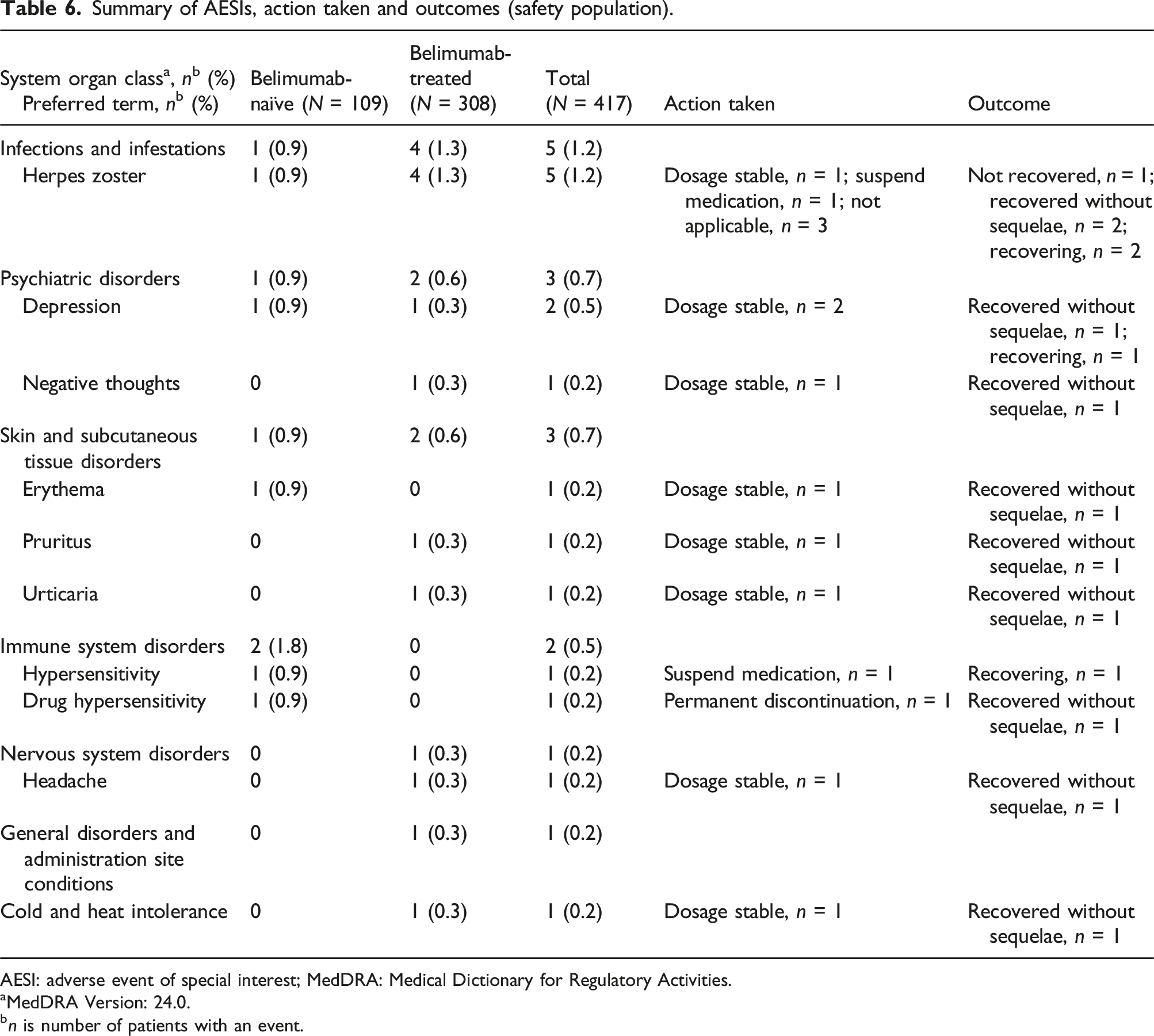
AESI: adverse event of special interest; MedDRA: Medical Dictionary for Regulatory Activities.
aMedDRA Version: 24.0.
bn is number of patients with an event.
Patient safety subgroup analysis
Overall, 14 paediatric patients (<18 years of age; 3.4% of the total safety population) and five older adult patients (≥65 years of age; 1.2% of the total safety population) were included in the respective subgroup analyses. Six paediatric patients (1.4% of the total safety population) experienced 14 AEs, two paediatric patients (0.5% of the total safety population) experienced three SAEs (cerebral thrombosis, oedema and rash, for which outcomes were ‘recovering’ at the end of the study) and one paediatric patient (0.2% of the total safety population) experienced one AESI. There were no ADRs in this subgroup.
Among older adult patients, four patients (1.0% of the total safety population) experienced 24 AEs, one patient (0.2% of the total safety population) experienced one ADR, two (0.5% of the total safety population) experienced five SAEs and one (0.2% of the total safety population) patient experienced two AESIs.
Tolerability in the total safety population
A total of 136 patients discontinued treatment (n = 34 belimumab-naïve patients [31.2%]; n = 102 belimumab-treated patients [33.1%]). Among patients who discontinued treatment, the reason for discontinuation was an AE or SAE for 11.8% (n = 4) of belimumab-naïve patients and 2.9% (n = 3) of belimumab-treated patients, while ‘other’ was the reason for 73.5% (n = 25) of belimumab-naïve patients and 95.1% (n = 97) of belimumab-treated patients. ‘Other’ reasons included the COVID-19 pandemic (at least seven patients discontinued due to COVID-19 management/concern, none of whom tested positive for COVID-19 infection), consideration of pregnancy, personal reasons, unwilling to continue, financial reasons, poor efficacy and improvement in health. There was no reason recorded for 14.7% (n = 5) of belimumab-naïve patients and 2.0% (n = 2) of belimumab-treated patients.
Discussion
This Phase 4, prospective, observational study provides new real-world safety and tolerability data for belimumab when used as part of standard medical care in Chinese patients with SLE. Of more than 400 enrolled patients, 37.9% experienced an AE, the majority of which were mild or moderate in severity, and few experienced an SAE (5.3%) or an ADR (6.0%). No new safety concerns were identified for the use of belimumab in clinical practice in China.
The most common types of AEs or SAEs were infections and infestations, which is consistent with pivotal clinical trials of belimumab,13–15 as well as trials in Asia and China.16,17 This study adds to limited existing real-world safety evidence for belimumab in the treatment of SLE in China.21,28 Previous observational studies in this population identified upper respiratory tract infection, herpes zoster, pneumonia and urinary tract infection as the most common AEs, which is consistent with the present study. AEs, SAEs and ADRs were mutually exclusive categories in the present study; therefore, direct comparison of the incidence of events between this and other studies may be limited.
Serious infections are listed under Warnings and Precautions in the belimumab label, along with hypersensitivity reactions and psychiatric events.29,30 As such, these events were captured as AESIs in the current study and have also been studied extensively in a large, international Phase 4 trial (the BASE trial). 20 There were only two hypersensitivity reactions and three psychiatric disorders in the present study that were categorised as AESIs (including one case of negative thoughts), and in all cases patients either recovered without sequelae or were recovering at the end of the study. Rates of serious infusion and hypersensitivity reactions and serious depression were slightly more common with belimumab versus placebo in the BASE study, but the proportion of patients experiencing these events were low (serious infusion and hypersensitivity reactions: n = 8/2002 vs n = 2/2001; serious depression: n = 7/2002 vs 1/2001). 20 The current study did not include a comparator arm; however, the number of infections categorised as AESIs in the present study was also low (five patients, all herpes zoster). Data from the BASE study suggested that while belimumab does not increase the chances of a serious infection, it may increase the risk of a fatal infection if a serious infection occurs. 20 This highlights the need to monitor patients and manage the risk of infection.
As well as adding to evidence in adults with SLE, the current study also reports the first paediatric real-world safety data in China. Although the number of paediatric patients was small, no new safety signals were identified in this subgroup. Previously, in the first clinical trial of paediatric patients receiving belimumab, which was carried out in North, Central and South America, Europe and Japan, the safety profile was consistent with observations in adults with SLE and there were no new safety concerns. 31 In addition, this study also reports the safety of belimumab in the older-adult population, which similarly raised no new safety concerns.
Approximately 75% of the population in this study had been receiving belimumab prior to study enrolment. Prior to and over the course of the study, belimumab-treated patients had a total mean exposure of 298 days compared with 155 days of exposure during the study for those who were belimumab-naïve prior to study entry. Although both groups were followed for the same length of time, and the overall number of ADRs was low and similar between the two groups, there were some small numerical differences in rates of certain ADRs between the belimumab-treated and belimumab-naïve patients. For example, infections and infestations classed as ADRs occurred in 3.2% of belimumab-treated patients versus 0.9% of belimumab-naïve patients. It is possible that the difference in exposure duration contributed to this difference, as effects on the immune system may take some time to develop. In the present study, three patients (1.0%) in the belimumab-treated group had decreased white blood cells versus none in the belimumab-naïve group. However, in a continuation study of belimumab with data up to 13 years, infection rates each year remained stable over time despite an increasing proportion of patients having serum immunoglobulin levels below the lower limit of normal. 32 It should be noted that patients who tolerated belimumab in that study may have been more likely to remain on-treatment, thus, findings may not be representative of all patients with SLE. It is possible that the belimumab-treated population was enriched with patients who tolerated treatment in the present study, given their previous belimumab exposure for SLE; however, rates of treatment discontinuation were similar between the two groups in the follow-up period, suggesting that this was unlikely.
Limitations of the current study include the short follow-up period of 24 weeks; longer-term assessment is needed to provide more robust evidence of the safety profile in this population. However, approximately three-quarters of patients received belimumab before the study commenced, and these patients had almost double the total length of exposure compared with those who were belimumab-naïve. Although safety data were not collected in the exposure period before study start, this additional exposure represents longer-term use. The belimumab-treated group did not include patients who may have received belimumab and then discontinued prior to the study start date. However, the similar incidence of ADRs between belimumab-naïve and belimumab-treated patients observed in this study suggests that the tolerability of belimumab remains consistent over time. Logistical aspects may also have limited the findings. The study was carried out in hospitals within large cities, and patients in rural areas may not have returned for repeat doses of belimumab and thus may have contributed to the ‘other’ population of patients who discontinued treatment. The inclusion of similar types of study sites may also limit the generalisability of the results. The study was conducted during the COVID pandemic period, which may have affected belimumab treatment schedules and follow-up due to national quarantining and public distancing measures to prevent the spread of the virus; exposure rates to infections may have been affected in addition, as patients may have been isolating and/or following national quarantining and public distancing procedures.
The safety profile of belimumab demonstrated in this real-world study is aligned with global belimumab studies and the known safety profile, with no new safety signals identified. The findings support the use of belimumab for Chinese patients with SLE and will inform clinical decision-making, following belimumab’s recent addition to the therapeutic armamentarium for patients with SLE in China.
Supplemental Material
Supplemental Material - Belimumab safety in adult and paediatric Chinese patients with systemic lupus erythematosus: A Phase 4, multicentre, observational study
Supplemental Material for Belimumab safety in adult and paediatric Chinese patients with systemic lupus erythematosus: A Phase 4, multicentre, observational study by Zhuoli Zhang, Zhizhong Ye, Shanzhi He, Lin Tang, Chuanmei Xie, Songlou Yin, Shuhong Chi, Jing Yang, Qinghong Yu, Min Yang, Xuefeng Zhao, Yifan He, Jingwen Hu, Weibo Wang and Annie Tung in Lupus.
Footnotes
Acknowledgments
The authors would like to thank the participating patients and their families, clinicians, and study investigators. Medical writing and submission support was provided by Tasmin Long, PgCert, Fishawack Indicia Ltd., UK, part of Avalere Health, and was funded by GSK. Authors have authorised the manuscript submission via third party and approved all statements and declarations.
Author contributions
S.C., S.H., L.T., C.X., S.Y., Q.Y., M.Y., X.Z., J.H., Z.Z., Z.Y. and J.Y. contributed to the acquisition of the data. X.Z., S.Y., Y.H., W.W., Z.Z. and A.T. contributed to the conception or design of the study. Y.H., J.H., W.W., Z.Z. and A.T. contributed to the analysis or interpretation of the data. Z.Z. also revised the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Z.Z. has received honoraria from/was a speaker for GSK, Eli Lilly, Novartis, and Pfizer. S.C. received honoraria from/was a speaker for GSK. S.H. has received speaker/honoraria from GSK. X.Z., Y.H., J.H. and A.T. are employees of GSK. W.W. is an employee of GSK and holds stocks and shares in GSK. Z.Y., L.T., C.X., S.Y., J.Y., Q.Y. and M.Y. have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study, including medical writing support, was funded by GSK (GSK Study 213210).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
