Abstract
Anifrolumab is a new therapeutic approach for individuals with systemic lupus erythematosus (SLE) directed at blocking the type 1 interferon pathway. Despite the expanding body of literature on Anifrolumab, an essential aspect remains absent: the subjective patient experience of treatment effects and implications on patients’ health-related quality of life (HRQoL). The present study aimed to fill this void by elucidating the nuanced perspectives of SLE patients receiving Anifrolumab treatment by conducting qualitative in-depth interviews (IDIs). SLE patients at Aarhus University Hospital who had received at least three infusions of Anifrolumab were approached for inclusion in the study, which comprised two main elements: (1) qualitative IDIs and (2) collection of patient data from electronic medical records (EMRs). The IDIs were semi-structured and based on a discussion guide that included open-ended and close-ended questions. Verbatim transcripts were coded and analysed using qualitative software to understand concepts important to patients and to understand patients’ own experiences before and after Anifrolumab therapy. A clinical chart review was conducted using EMR data at baseline, 3 months, and 6 months after Anifrolumab initiation. IDIs were completed with 14 patients, and EMR data was collected from 16 patients (treatment days range: 62-474). Of the 23 symptoms spontaneously reported by patients prior to Anifrolumab treatment, fatigue, joint pain, sun sensitivity, joint stiffness, skin rashes, and hair loss were the most common. Most symptoms improved, and none worsened during treatment. Patients reported significant impacts of disease on daily life before treatment: day-to-day activities, social life, emotional aspects, physical activity, concentration/memory, work/employment, and family/romantic relationships. Patients reported improvements in all aspects after treatment but were still impacted. From the EMR data, we observed a fall in disease activity after treatment initiation with a concomitant reduction in the use of corticosteroids. This study provides valuable insights into the subjective experiences of SLE patients treated with Anifrolumab, and the findings collectively contribute to a comprehensive understanding of the treatment’s efficacy from the patients’ perspective and its tangible effects on both subjective and objective parameters in SLE patients.
Keywords
Introduction
Systemic Lupus Erythematosus (SLE) imposes a substantial and variable clinical burden marked by debilitating disease manifestations, irreversible organ damage, and concurrent comorbidities and complications.1,2 In Denmark, the prevalence of SLE is approximately 2000 individuals, and around 90% of cases are women.3–5 These women are typically diagnosed earlier on in life, especially around childbearing age.4,5
SLE is characterized by periods of high disease activity and variable organ system involvement.1–3 Patients with SLE commonly experience symptoms across the whole body, such as rash, alopecia, oral ulcers, arthritis, headaches, seizures, hematologic abnormalities, lupus nephritis, as well as cardiovascular and gastrointestinal symptoms.1,2,4 According to a 2020 survey conducted by Lupus Europe, SLE patients reported a median of nine symptoms out of a total of 21, with fatigue being the most prevalent (85.3%) and most bothersome. 6 The top three symptoms that respondents expressed a desire to alleviate were fatigue and weakness, joint pain and swelling, and muscle pain and weakness. 6
Anifrolumab is a novel therapeutic approach for individuals with SLE that inhibits the type 1 interferon pathway. 7 By binding to subunit 1 of the type 1 interferon receptor (IFNAR1) with high specificity and affinity, Anifrolumab inhibits type 1 IFN signaling and causes internalization of IFNAR1, which inhibits IFN response gene expression and downstream inflammatory and immunological processes. 7 Anifrolumab was approved by EMA in 2022 for clinical use based on its efficacy as an add-on therapy to the standard of care in moderate to severe SLE. 8 A post-hoc analysis of phase 3 clinical trials demonstrated that responders had lower flare rates, higher rates of sustained glucocorticoid reduction to ≤7.5 mg/day, and greater improvements in patient-reported fatigue, as measured by the Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) scale, and health-related quality of life (HRQoL), as measured by the Short Form 36 (SF-36) Health Survey. 9
Despite the expanding body of literature on Anifrolumab, an essential aspect remains absent: the subjective patient experience of treatment effects and its implications for daily life. Understanding the patient experience is crucial for developing effective treatments and improving overall HRQoL. Patients’ unique experiences with the disease, its symptoms, and the various treatment modalities may not align with the parameters typically measured in clinical trials. Symptoms such as fatigue, pain, and the psychological toll of chronic illness are often underrepresented or insufficiently addressed. The patient perspective is instrumental in providing a more comprehensive understanding of the disease burden, treatment efficacy, and the trade-offs between benefits and potential adverse effects.
The present study aimed to fill this knowledge gap by elucidating the nuanced perspectives of SLE patients receiving Anifrolumab treatment by conducting qualitative, in-depth interviews (IDIs) with the patients. The study’s primary objective was to enhance our understanding of the treatment’s perceived efficacy from the standpoint of those directly affected and its impact on their lives.
Methods
Design
This was a cross-sectional, non-interventional qualitative interview study employing a hybrid approach, combining (1) primary data from one-on-one qualitative IDIs and (2) secondary data from a collection of patient data from electronic medical records (EMRs). All SLE patients at the Department of Rheumatology, Aarhus University Hospital, Denmark, who had received at least three infusions of Anifrolumab were approached for inclusion between May and August 2023. Patients provided written consent to participate in the study following oral and written study information.
Inclusion criteria consisted of signed informed consent, consent to be audio recorded during IDIs, age of at least 18 years, a clinical diagnosis of SLE confirmed by the treating physician, receiving at least three infusions of Anifrolumab, EMR recorded at baseline, and 3 months after starting Anifrolumab, prescription of Anifrolumab for the first time according to the country-specific label, residence in Denmark, fluent communication in Danish, and physical and cognitive ability to participate in a 60-minute IDI in one sitting.
Discussion guide
The 60-minute IDIs were conducted virtually based on a study-specific discussion guide that included open-ended and close-ended questions and were moderated by a trained interviewer in Danish. The interviews were conducted by an independent company (IQVIA). Interviewers were without relation to patients or treating physicians and had no knowledge of the patient’s disease state or manifestations.
The discussion guide comprised sections about SLE and treatment experience before starting Anifrolumab, in particular focusing on their experiences from 12 months before being on Anifrolumab (including perceived disease severity, symptoms and impacts, and periods of high and low disease activity [disease flares]), and SLE and treatment experience since being on Anifrolumab (including perceived disease severity, symptoms and impacts, and disease flares). The patients were first asked to speak openly about the symptoms and impacts they had due to their SLE before initiating Anifrolumab treatment. The discussion guide contained a symptom list and an impact list from which the moderator could probe for symptoms and impacts not spontaneously mentioned by the patients. If the patients reported having the symptom or impact before being explicitly probed on it, this was defined as a spontaneous reporting of the symptom/impact. For symptoms and impacts experienced by patients, bothersomeness ratings were collected using 11-point numerical rating scales (NRS) where 0 = not at all bothersome and 10 = extremely bothersome. The saliency of symptoms and impacts was tracked and determined by plotting the level of bothersomeness against the number of patients spontaneously reporting it. Concept saliency was defined as ≥50% of patients experiencing the concept with a bothersomeness rating of ≥5. We used a similar NRS when asking patients whether they were satisfied with the Anifrolumab treatment (0 = not at all satisfied and 10 = extremely satisfied), a three-point scale asking for patients’ perceived disease severity (mild/moderate/severe) before and after treatment, and dichotomous questions (yes/no) of whether they had experienced disease flares before and after treatment and whether they would recommend Anifrolumab to other patients.
In preparation for the interviews, the interviewer reviewed the discussion guide and participated in mock interview sessions with an experienced qualitative researcher to test the discussion guide and discuss the flow and structure of the questions and associated probes.
Data analysis
The IDIs were audio-recorded, and verbatim transcripts of the interviews were translated into English and then coded and analysed using MaxQDA qualitative software 10 to understand concepts (symptoms and impacts) relevant and important to SLE patients as well as to understand patients’ own experiences before and after Anifrolumab therapy. Given the broad study objective of describing patients’ experiences of their disease and treatment, an inductive thematic analysis approach was used where the goal of coding was to enable identification of the concepts (signs, symptoms, and HRQoL impacts) most important and relevant to patients for their experiences of their SLE and treatment. Coders reviewed each transcript to identify text that included concept expressions and tagged selected text with a code. The codes were organized within a coding framework established at the start and refined/expanded during the coding process. Two coders were involved in coding the aggregate of transcripts, and inter-rater agreement was evaluated between coders to ensure coding consistency. Once the coding process was completed, outputs listing the number of occurrences of the codes were generated and served as the basis for the results. 11
The treating physicians conducted a clinical chart review using EMR data at baseline, 3 months, and 6 months (where available) after Anifrolumab initiation. Data were collected on SLE manifestations, biochemical data, and treatments at baseline and follow-up.
The study was performed using ethical principles consistent with the Declaration of Helsinki, International Conference on Harmonisation – Good Clinical Practice, Good Pharmacoepidemiology Practice, and the applicable legislation on Non-Interventional Studies and/or Observational Studies. Since no biological material was included in the study, the study was exempted from formal Danish ethical approval.
Results
Patient characteristics and enrolment
At the time of study enrolment, 18 patients had undergone Anifrolumab treatment since its availability in the Department of Rheumatology, Aarhus University Hospital, Denmark, starting April 2022. Two patients were no longer receiving the treatment; one ceased due to pregnancy, and the other discontinued after experiencing 12 months of remission followed by tapering. Of the remaining 16 patients, two were only included in the EMR data collection but excluded from the IDIs; one was due to mental illness, and the other was due to unwillingness to be interviewed. For transparency reasons, EMR data for all 16 patients is presented.
Demographics, clinical characteristics and medication use at baseline.

SLEDAI = Systemic Lupus Erythematosus Disease Activity Index; SLICC(ACR) = Systemic Lupus International Collaborating Clinics of American College of Rheumatology; SD = Standard Deviation.
Qualitative in-depth interviews
Symptoms and impacts on daily living
Symptoms and impacts mentioned before and after Anifrolumab treatment.
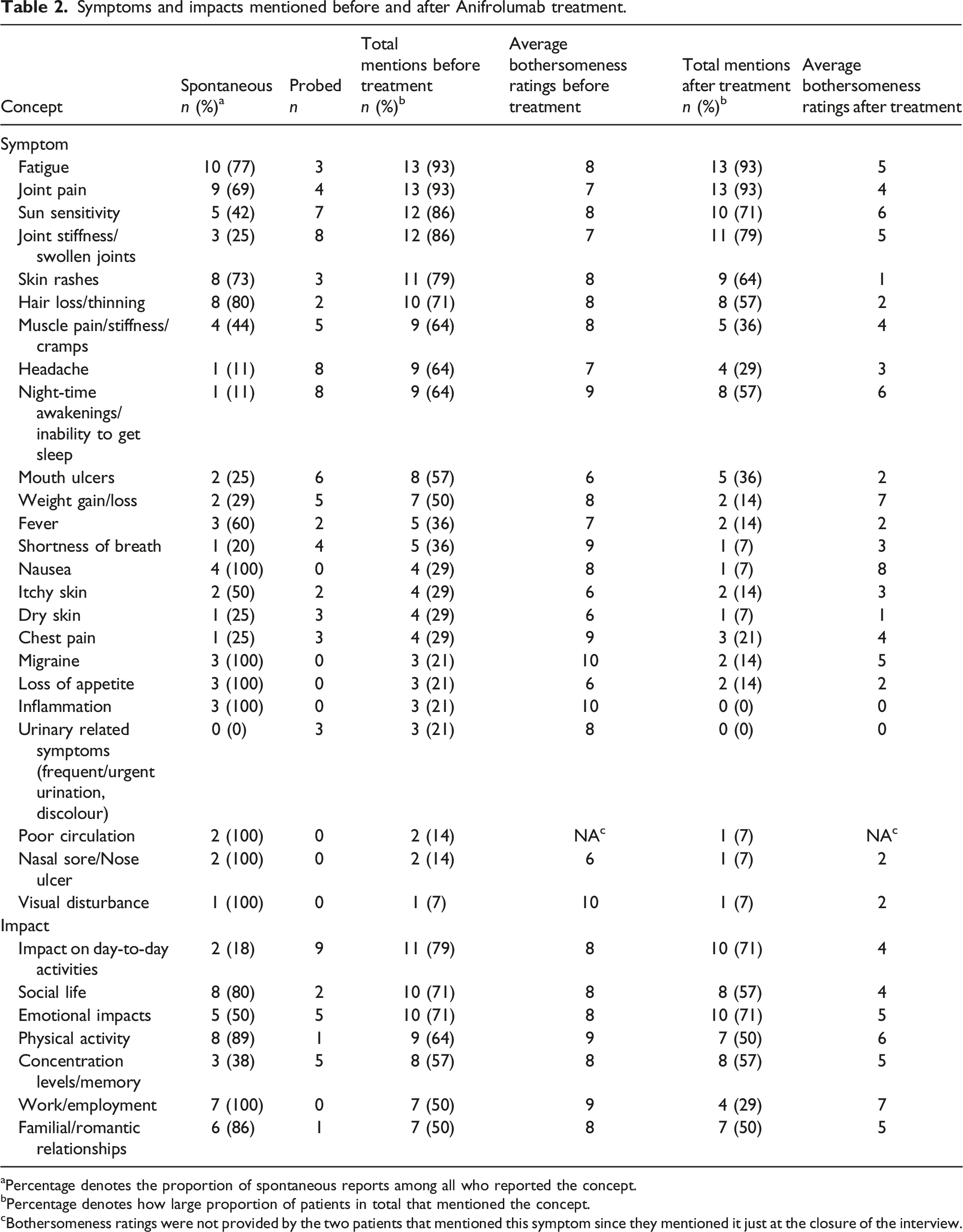
aPercentage denotes the proportion of spontaneous reports among all who reported the concept.
bPercentage denotes how large proportion of patients in total that mentioned the concept.
cBothersomeness ratings were not provided by the two patients that mentioned this symptom since they mentioned it just at the closure of the interview.
Table 2 shows that seven separate impacts were spontaneously reported before treatment, where impact on day-to-day activities was the most frequently reported (79%). All impacts were mentioned by fewer or equally many patients after treatment compared to before treatment.
In sum, no new symptoms or impacts emerged following treatment, and fewer patients, or equally as many patients, mentioned all symptoms and impacts after treatment compared to before treatment.
The NRS bothersomeness ratings on all symptoms and impacts reported by each patient, combined with the frequency by which each symptom/impact was mentioned before and after treatment, contribute to an increased understanding of the treatment’s efficacy from the patients’ perspective. Figure 1(a) and (b) and 2(a) and (b) illustrate the saliency of symptoms and the disease’s impact on daily life before and after Anifrolumab treatment. Prior to Anifrolumab treatment, we identified 11 salient symptoms (Figure 1(a)). Thus, the symptoms reported most prior to Anifrolumab treatment, together with severe bothersomeness ratings, were fatigue, joint pain, joint stiffness, sun sensitivity, skin rashes, hair loss, muscle pain, headache, night-time awakenings/inability to sleep, mouth ulcers and weight gain/loss. Post-treatment, salient symptoms were reduced to four (fatigue, joint stiffness, sun sensitivity, night-time awakening/inability to sleep) (Figure 1(b)). Thus, the bothersomeness of all symptoms improved but were still present, and none worsened during the treatment. (a) Symptom saliency before Anifrolumab treatment, (b) Symptom saliency after Anifrolumab treatment. (a) Impact saliency before Anifrolumab treatment, (b) Impact saliency after Anifrolumab treatment.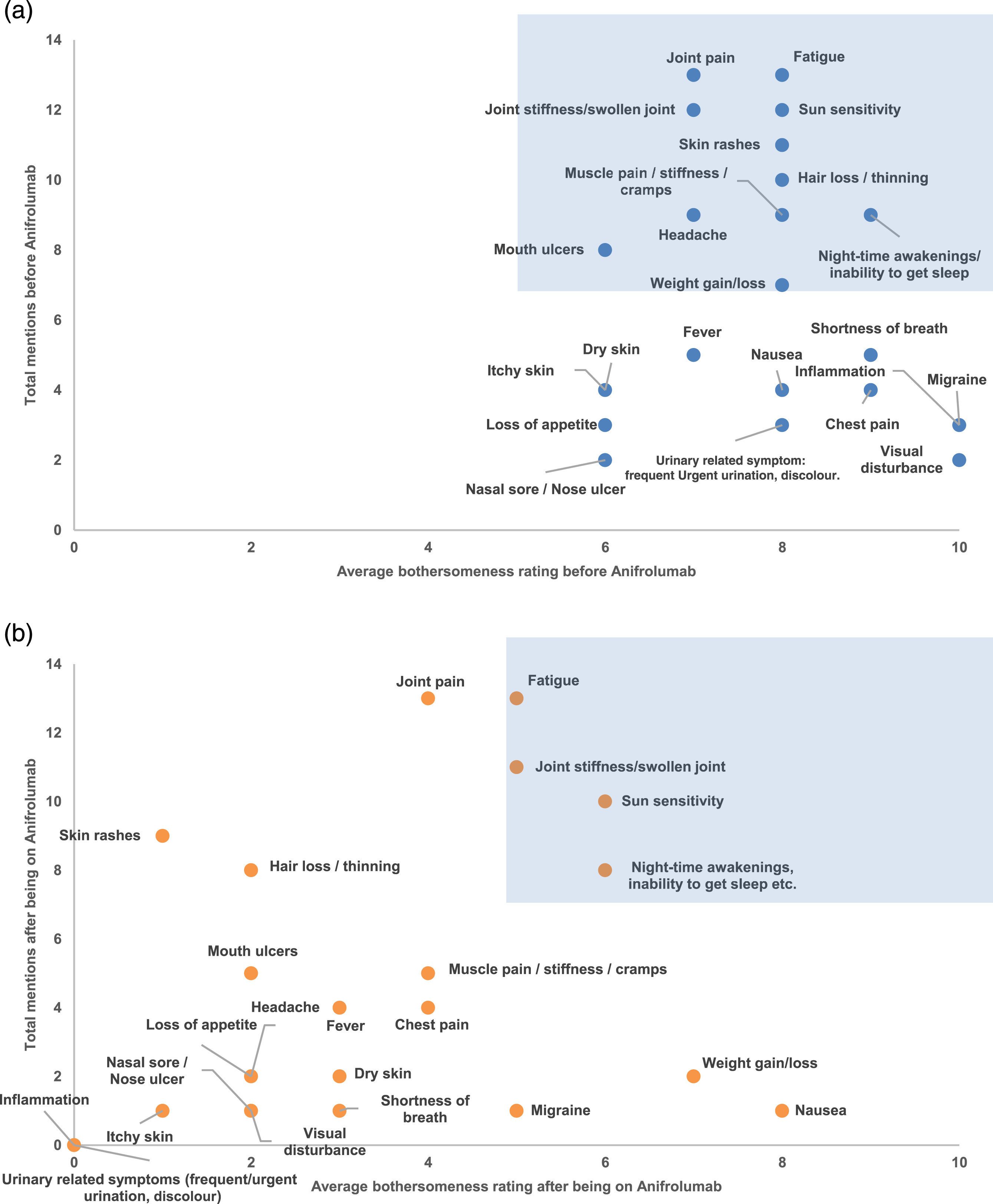
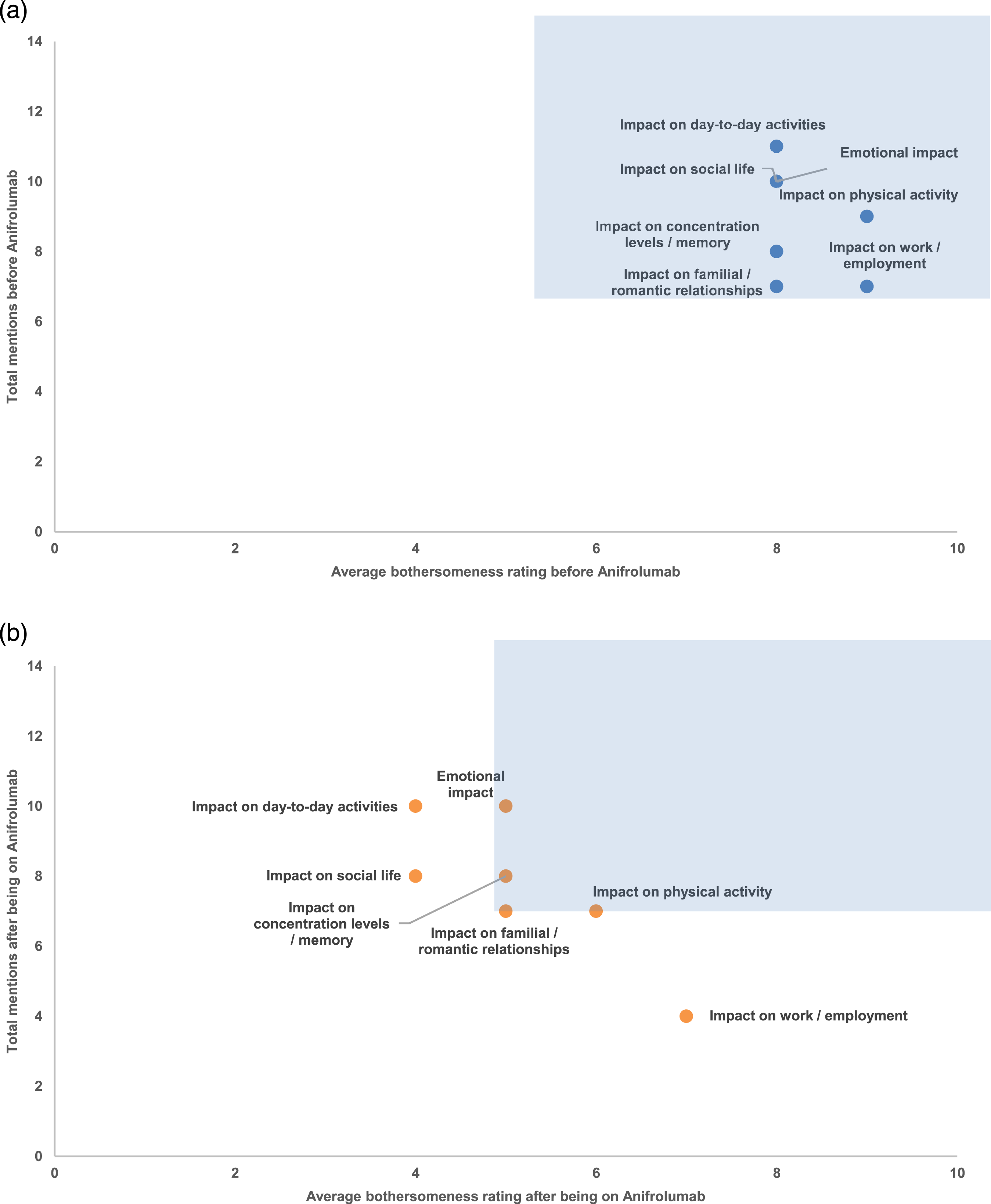
Patients reported significant impacts of disease on daily life before treatment: day-to-day activities, emotional aspects, social life, physical activity, concentration/memory, work/employment, and family/romantic relationships (Figure 2(a)). Whereas all these seven impacts were salient before treatment, four impacts remained salient following treatment, encompassing emotional impacts, effects on concentration/memory, impacts on familial/romantic relationships, and effects on physical activity (Figure 2(b)).
Perceived disease severity, disease flares, and treatment satisfaction
Before Anifrolumab treatment, zero patients (0%), seven patients (50%), and seven (50%) perceived their disease as mild, moderate and severe, respectively. After treatment, the corresponding figures were six (43%) for mild, eight (57%) for moderate, and zero (0%) for severe. Eleven patients (79%) reported that they had experienced disease flares (periods of high and low disease severity) during the 12 months before starting Anifrolumab treatment. The average time for these patients between treatment initiation and the interview date was 286 days. Two (14%) patients reported that they had experienced disease flares since Anifrolumab treatment initiation. Patients were generally satisfied with Anifrolumab treatment (mean score 8 [SD = 1.5 and range 6-10] on the 0-10 NRS), and all patients (100%) said they would recommend the treatment to other patients. The main reason given for these ratings was that the treatment helped patients manage their SLE symptoms, as reported by 12 (86%) of the patients. One patient had a herpes outbreak during treatment, and one patient mentioned that travelling a long distance for treatment was a reason for being less satisfied.
Clinical chart review
Disease activity changes during treatment with Anifrolumab
SLEDAI-2K changes on Anifrolumab.
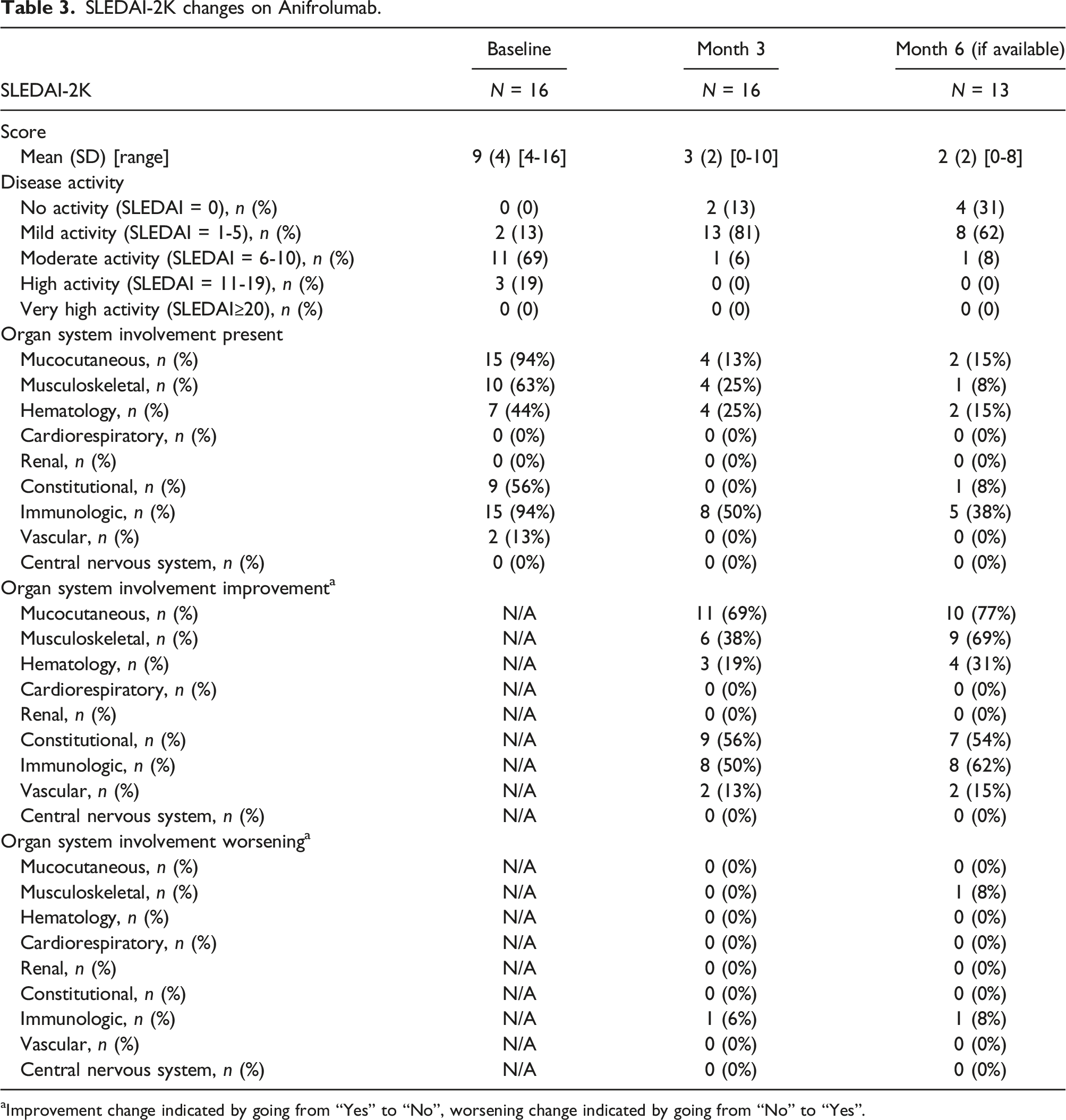
aImprovement change indicated by going from “Yes” to “No”, worsening change indicated by going from “No” to “Yes”.
Regarding the categorization of SLEDAI-2K disease activity at baseline, the majority of patients (N = 11, 69%) were classified as moderate. This shifted to mild at month three (N = 13, 81%) and month six (N = 8, 62%). Changes were also observed in organ system involvement between baseline, month three, and month six. The most significant change was observed in mucocutaneous organ involvement, which decreased from N = 15 (94%) at baseline to N = 4 (13%) at month three and N = 2 (15%) at month six. Additionally, immunologic organ system involvement was N = 15 (94%) at baseline, N = 8 (50%) at month three, and N = 5 (38%) at month six (Table 3).
Glucocorticoid use before and after Anifrolumab treatment
More than half (56%) of the patients received glucocorticoids upon commencing Anifrolumab treatment, as illustrated in Figure 3. An average reduction in dosage was observed at both 3 months and 6 months following the initiation of Anifrolumab therapy. Forty-four percent did not receive glucocorticoids when Anifrolumab was initiated, but were on other standard therapies. Oral corticosteroid usage before (at baseline) and at 3 and 6 months after Anifrolumab treatment. Note: Percentage calculated as proportion of sample (baseline: N = 16, month 3: N = 16, month 6: N = 13).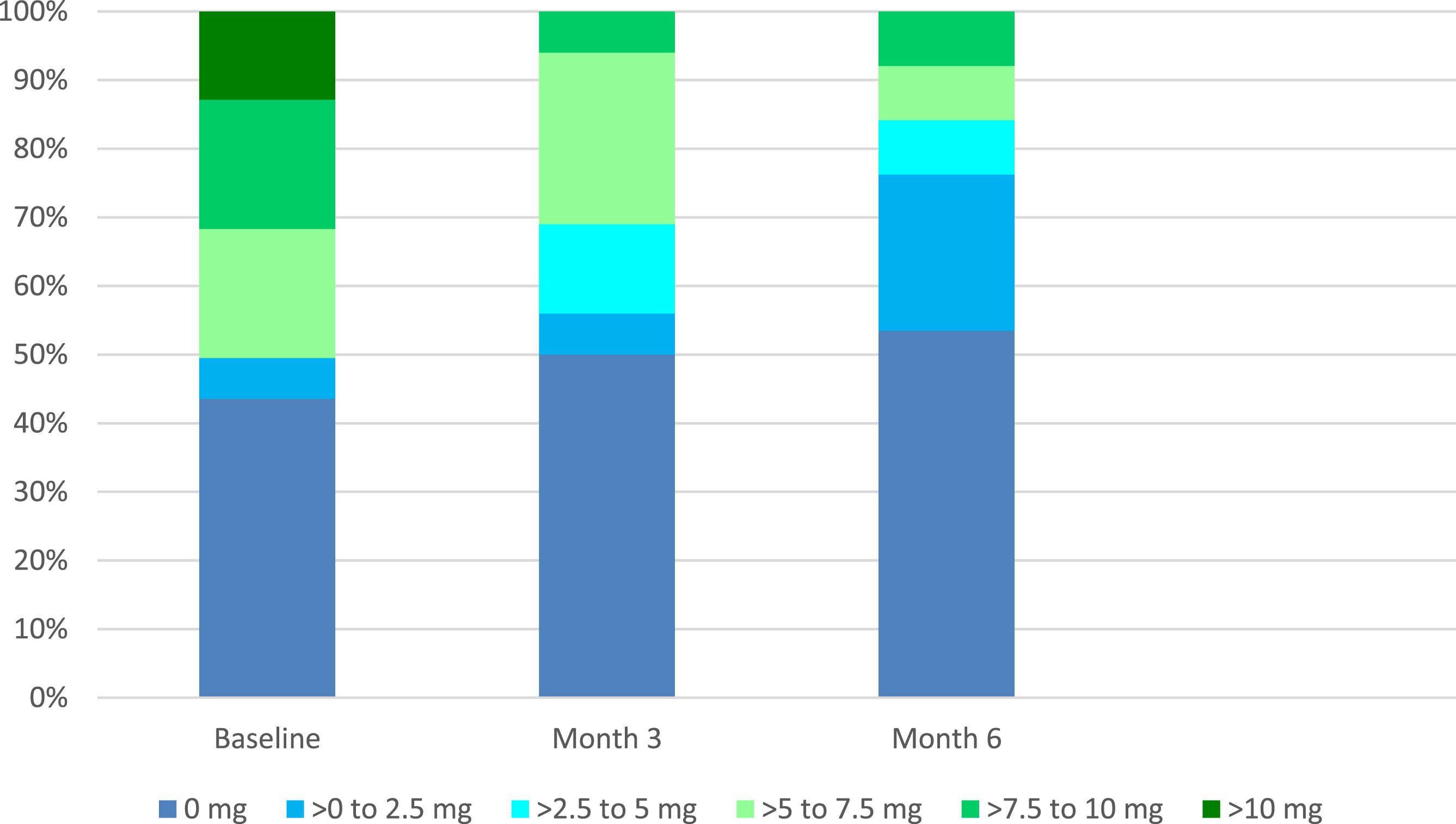
Laboratory results before and after Anifrolumab
Laboratory results showed changes which included a low C3 level (<0.9) at baseline in N = 11 (69%) of patients, N = 9 (56%) at month three, and N = 5 (38%) at month six. A low lymphocyte count (<1.3) was recorded for N = 13 (81%) at baseline, N = 9 (56%) at month three, and N = 6 (46%) at month six (Supplementary Table 1). No patients were treated for a herpes zoster infection during the study, and 2/16 (13%) patients were treated for a respiratory tract infection within the first three months of treatment (Supplementary Table 2).
Discussion
With this study, we aimed to elucidate the patient experience of treatment with Anifrolumab, both on symptoms related to SLE and the impact the disease has on daily life before and after initiating treatment. The absence of a robust representation of the patient perspective in clinical trials significantly limits our understanding of the efficacy of therapeutic interventions. It hampers the complete comprehension of the impact of the disease on patients’ daily lives.
IDIs are a robust and unbiased research method with the purpose to highlight the patients’ disease and treatment experiences, as they allow patients to speak openly about their perspective, unguided by fixed questions and response options. In contrast to traditional PROs for symptom and HRQoL assessments that most often are included in clinical trials, in IDIs, only the relevant symptoms and impacts that patients mention are scored on the NRS scales.
The impact of SLE disease on daily life experienced by patients included in the current study was high and was scored high in all areas by the patients. This is in congruence with previous research findings indicating that individuals with SLE exhibit a diminished functional status compared to the general population. 12 Moreover, distinct manifestations of SLE, including disease activity, specific disease manifestations, and the presence of, for example, fibromyalgia, have been identified as factors influencing HRQoL. SLE disease significantly impacts HRQoL in patients living with the condition, 13 and several studies have demonstrated significant discordance between the assessments of SLE disease activity and global health as perceived by physicians and patients.13,14 This paradox underscores the importance of incorporating measures of HRQoL alongside assessments of disease activity and damage in clinical trials to capture the multifaceted impact of SLE comprehensively. In the current study, we observed an apparent reduction in the impact SLE disease had on daily life after initiating Anifrolumab. Although the disease still impacted daily life, it was reduced in all seven areas that patients spontaneously reported.
Cutaneous involvement significantly impacts the HRQoL of SLE patients. 15 It is well described that visible dermatologic disease can affect work life, personal relationships, and leisure activities. 16 Numerous cutaneous manifestations and their sequels, which may include scarring and depigmentation, possess the potential for disfigurement, thereby instigating heightened self-consciousness among patients concerning their physical appearance in both domestic and professional settings and may contribute to social isolation. 17 Skin rash and hair loss were two of the most mentioned symptoms by patients in the current study, with a high average bothersome score. Cutaneous symptoms exhibited the most substantial alteration in perceived bothersomeness, decreasing from an average score of 8 prior to treatment to 1 post-treatment. Notably, 91% of patients observed an amelioration in skin-related concerns, with no cases of reported deterioration. This aligns with findings from a post hoc analysis of the TULIP trials by Morand et al. 18 highlighting the pronounced efficacy of Anifrolumab on mucocutaneous manifestations during treatment. Reducing skin involvement in SLE will have a major impact on the patient’s social life, as also demonstrated in the current study.
Another crucial aspect often inadequately addressed is the patient perspective on treatment tolerability. Treatment-related side effects in the current study were infrequent. Specifically, only two patients sought treatment for a respiratory tract infection within the initial three months of therapy, and no patients were treated for herpes zoster infection. A majority of patients (9/16; 56%) were vaccinated against herpes zoster prior to initiating treatment. Importantly, this is a short-time study of the lifespan of a SLE disease. The annualized rate of herpes zoster and bronchitis was 7.2% and 12.2%, respectively, on Anifrolumab treatment in the pivotal TULIP-2 clinical trial. 7
When queried about their satisfaction with the treatment, respondents provided an average score of 8 on a scale ranging from 0 to 10. All patients (100%) would recommend the treatment to others. These nuanced facets of the treatment experience can only be comprehensively understood through direct insights from those undergoing the interventions.
In our investigation, we observed a notable correlation between the patients’ self-reported symptom reduction and the decrease in the disease activity score, which reduced from an average of 9 to 2. Previous research has demonstrated the predictive capacity of patient self-assessment using the EuroQol-5-domain (EQ-5D) for organ damage accrual in SLE. 19 Consequently, in the era of treat-to-target strategies, leveraging the patient perspectives on HRQoL as a clinically pertinent treatment target in SLE, gauged directly by the patients, becomes a relevant focal point.
Current treatments for SLE include the long-term use of immunosuppressants and corticosteroids. Low dose prednisolone results in a good long-term control of the disease activity. 20 However, prolonged use of glucocorticoids can result in increased morbidity and mortality. 21 Many individuals with SLE can sustain prolonged periods of remission by combinations of non-biologic drugs, with a key emphasis on maintaining a prednisone dosage of 5 mg or less per day, as outlined in the DORIS remission criteria. 22 DORIS remission has demonstrated efficacy in mitigating the long-term accrual of disease-related damage. 23 It is important to notice, that patients achieving remission through non-biologic interventions do not align with the intended target population for Anifrolumab utilization. Anifrolumab was approved by the European Medicines Agency in February 2022 for the treatment of moderate to severe SLE and demonstrated a reduced need for corticosteroid use in patients with SLE in the TULIP-trials. 24 To hinder long-term side effects inflicted by systemic glucocorticoid treatment, there is an imperative need for new treatment options that allow us to taper glucocorticoids in SLE where current treatment options do not assure remission. This is because even low doses of systemic glucocorticoid treatment are associated with an increased risk of infections, osteoporosis, and increased mortality. 25 In the current study, we observed a reduction in steroid use across patients, fulfilling one of the purposes of adding Anifrolumab to the standard of care in SLE treatment. Further, in 44% of cases, we could avoid reintroducing steroids during the patient’s flare by adding Anifrolumab to standard of care. Patients were initiated on Anifrolumab due to a flare in their disease. In our clinical practice, we aim to taper prednisolone to zero after initiating both csDMARD and bDMARD therapies, and should patients experience a flare during the tapering process, we introduce supplementary therapies. Based on our observations of Anifrolumab’s rapid efficacy, we try, when possible, to add Anifrolumab without reinitiating prednisolone. In several cases, this has been possible.
The limitations of this study include, firstly, the relatively small number of patients included, which may impact the generalizability of the findings. Secondly, the study’s patient selection process introduces a potential bias, as individuals were chosen for the specific treatment based on their symptoms. While this aligns with real-world clinical practices, it introduces the possibility of selection bias, as not all patients were uniformly chosen, potentially influencing the generalizability of the results. Thirdly, the retrospective nature of the study introduces the risk of recall bias, as participants may not accurately recollect past events or experiences. This could influence the reliability of the reported symptoms and treatment outcomes. Still, during the interviews, no patients revealed any problems of remembering how it was for them before they started on Anifrolumab treatment.
A noteworthy strength of this study is its status as the largest investigation to date, focusing on the patient perspective of Anifrolumab treatment in a real-life clinical setting. Another strength lies in the unbiased approach to patient-reported symptoms. By directly capturing patients’ perspectives, the study minimizes potential biases introduced by intermediary assessments, providing a more authentic reflection of the patient experience with Anifrolumab treatment. The study also benefits from an extensive dataset, encompassing clinical and treatment data for nearly all patients who underwent the specified treatment at this specific centre. This comprehensive approach contributes to a more thorough, unselected understanding of the treatment’s real-world impact.
In conclusion, our study provides valuable insights into the subjective experiences of SLE patients treated with Anifrolumab. The symptoms commonly reported by patients before treatment demonstrated notable improvement, with no exacerbation observed during the treatment course. Furthermore, the significant impact of SLE on various aspects of daily life was acknowledged by patients, and while these impacts persisted post-treatment, significant enhancements were reported. Additionally, objective clinical data revealed a decrease in disease activity and a reduction in corticosteroids after Anifrolumab initiation.
These findings collectively contribute to a comprehensive understanding of the treatment’s efficacy and its tangible effects on both subjective and objective parameters in SLE patients.
Supplemental Material
Supplemental Material - Anifrolumab treatment improves patient-reported quality of life and decreases disease activity and corticosteroid use in patients with systemic lupus erythematosus: A qualitative study in Denmark
Supplemental Material for Anifrolumab treatment improves patient-reported quality of life and decreases disease activity and corticosteroid use in patients with systemic lupus erythematosus: A qualitative study in Denmark by Anne Troldborg, Lauren Remkus, Daniel Eek and Bent Deleuran in Lupus
Footnotes
Acknowledgments
We would like to acknowledge all the patients who took the time to participate in this study. We are also grateful to Åsa Hanna Lindelöf, Emma Hjort and Bine Kjøller Bjerregaard at IQVIA for performing the interviews; to Neeti Prasad and Rachael Kendrew at IQVIA for coding the interview data; to Alexandra Colon at IQVIA for leading the coding of the qualitative data team; and to Owen Cooper at IQVIA for conducting the analysis and synthesis of these data. The study was sponsored by AstraZeneca.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AT has received speaker fees from AstraZeneca and Boehringer Ingelheim. LR is an employee of AstraZeneca. DE is an employee of AstraZeneca and owns stock in the company. BD has received speaker fees from AstraZeneca, Galapagos, and Eli Lilly.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the AstraZeneca.
Data availability statement
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
