Abstract
Aim
To compare frequency, incidence rates (IR), risk factors and outcomes of a first venous thromboembolic event (VTE) between patients with systemic lupus erythematosus (SLE) and controls.
Methods
Using state-wide longitudinal hospital data from Western Australia (WA), we recorded venous thrombosis (VT) and pulmonary embolism (PE) in patients with SLE (n = 1854, median age 40, 86% female) and matched hospitalised controls (n = 12,107, median age 40 years, females 88.6%) in the period 1985–2015. Results presented are medians, frequency, IR per 1000 person years (PY) and odds, rate, or adjusted hazard ratios (OR/RR/a-HR) with 95% confidence intervals (CI).
Results
Patients with SLE had significantly higher odds (12.8 vs 3.3%; OR 4.26, CI 3.60–5.05) and IR for a first VTE (10.09 vs 1.52; RR 6.64; CI 5.56–7.79). Over the three study decades, the IR for PE declined in patients with SLE from 7.74 to 3.75/1000 PY (p < .01) with no changes observed for VT or in controls. VTE recurred more frequently in patients with SLE (24.1% vs 10.2 %) (p < .01). Antiphospholipid antibodies (aPL) (a-HR 4.24, CI 2.50–7.19), serositis (a-HR 2.70, CI 1.86–3.91), lupus nephritis (a-HR 1.75 CI 1.25–2.33) and thrombocytopenia (a-HR 1.65 (1.10–2.49) were the strongest disease risk factors for VTE only in patients with SLE, while arterial hypertension, smoking and obesity were independent VTE risk factors for both groups. VTE was not associated with an increased risk for arterial events, but PE increased the risk for pulmonary hypertension (PH) in both patients with SLE (a-HR 6.47, CI 3.73–11.23) and controls (a-HR 9.09, CI 3.50–23.63). VTE increased the risk of death in both patients with SLE (a-HR 2.02, CI 1.50–2.70) and controls (a-HR 6.63, CI 5.21–8.42) after 10 years of follow-up.
Conclusions
VTE affected 12.8% of patients with SLE at six times the VTE rate in controls with aPL as the strongest, but not the only risk factor in SLE. The risk of PH was increased in both groups following PE, but VTE did not associate with an increased risk of arterial events.
Keywords
Introduction
Patients with systemic lupus erythematosus (SLE) are at increased risk of thrombotic events due to the combined effects of chronic inflammation, vascular damage and acquired coagulation disorders such as antiphospholipid antibodies (aPL).1,2 Venous thromboembolism (VTE) in patients with SLE manifests in various blood vessels such as lower and upper limb deep vein thrombosis, thrombophlebitis, cerebral venous sinus thrombosis and splanchnic vein thrombosis, subsequently referred to as VT, and pulmonary embolism (PE). VTE is the third most frequent thrombotic event in the population, 3 while in SLE, VTE frequency eclipses myocardial infarction and stroke and associates with significant morbidity and mortality.4–6 A recent meta-analysis of 44 cohort studies estimated the pooled absolute VTE risk for patients with SLE at 5% with an incidence rate 8/1000 person years (PY) and a hazard ratio of 4.38 compared to general population. 7 However, the meta-analysis also reported significant data heterogeneity as most studies were from small cohorts, only four studies included a control group and few studies reported more than one VTE subtype. VTE risk in general depends on several variables such as age, lifestyle factors and comorbid conditions with disease specific features providing the additional risk for patients with SLE.1,2 With few population-based studies and scarce data from this region, we compared the incidence, odds and consequences of multiple VTE types in an observational long-term study comprising Australian patients with SLE and controls.
Methods
This observational study was a retrospective analysis of prospectively collected health data. Data were sourced from the Western Australia Rheumatic Disease Epidemiological Registry (WARDER) that contains routinely collected health data over the period January 1st, 1985 to December 31st, 2014, from public and private health care organisations in Western Australia (WA). . WARDER data are drawn from the Hospital Morbidity Data Collection (HDMC), Emergency Department Data Collection (EDDC), WA Cancer Registry and Death Registrations, for which there is mandatory reporting by state-wide health care institutions. These registries are maintained by WA Health and data across all datasets have been effectively linked through a validated process of probabilistic matching and clerical review to provide deidentified individual longitudinal health data over the whole study period.8,9
Case selection
WARDER contains health data for patients with rheumatic diseases and similar data for a control cohort free of inflammatory rheumatic disease.10,11 SLE patients were identified among WARDER participants by a first discharge diagnosis code (ICD9-CM: 710.0 or ICD-10-AM: M32.0-9). A single diagnostic discharge code for SLE has an 80%–96% positive predictive value for correctly identifying SLE patients in administrative health data. 11 As most SLE patients are hospitalised at least once during their disease course with an average of two admission over 15 years for either SLE or unrelated complications, 12 these figures suggest that our dataset represented the great majority of patients with SLE in WA over the 30-year study period. For each SLE patient, we defined the index admission as the first hospital contact with a diagnostic code for SLE, and subsequently, propensity matched each SLE patients on age, gender and index admission year with up to seven non-exposed controls who were free of inflammatory rheumatic disease over the study period. Controls reflect the general population well 13 and presented with a large variety of baseline conditions at index admission (Suppl Figure 1). The final dataset for this study contained sociodemographic data and all principal and secondary diagnoses and procedures registered during all visits (hospital and emergency) since the index admission date for each participant. This longitudinal case record for each participant was subsequently linked to death registry data.
Definitions
VTE manifestations were lower and upper limb deep vein thrombosis, thrombophlebitis, cerebral venous sinus thrombosis and splanchnic vein thrombosis, subsequently referred to as VT, and pulmonary embolism (PE). VTE coding followed standardised procedures from the Australian Classification Index pathways with separate coding algorithms employed for pulmonary embolism (PE) and venous thrombosis (VT) (Suppl table 1). The use of ICD-codes has been deemed valid for the identification of VTE in administrative databases with PPV of 80–90%.14–17 When PE was registered alongside a VT code, only the PE code was included in analyses as it is the more severe manifestation on the VTE spectrum. Recurrence of VTE were defined as any new VTE event more than 90 days after the first VTE event. Antiphospholipid syndrome (APS) was defined as VTE with positive aPL antibody testing while thrombophilia was defined as presence of APS and/or other coagulation disorders such as activated protein C resistance (Suppl table 1). aPL antibodies are only recorded in the databases in the context of a clinical syndrome compatible with SLE/APS with isolated finding of aPL antibodies not recorded. In addition, we registered comorbidities and lifestyle factors (Suppl table 1) and accrual of overall and organ‐system‐specific damage according to the validated and prognostically important updated Charlson comorbidity index (CCI) at index admission and last observation. 18 Given our patient selection process we modified the CCI (m-CCI) by omitting the rheumatic disease score.
Primary study outcomes were VTE incidence rates (IR) per 1000 person years (PY) and secondary outcomes were recurrence of VTE defined as any new VTE event more than 90 days after the first VTE event, pulmonary hypertension (PH) (as defined in Suppl table 1) and death.
The Western Australia Department of Health Ethics Committee provided approval of deidentified data for this project (project no. 2016.24, extension approval in 2021), that was performed in accordance with the Helsinki Declaration of 1964 and its later amendment.
Statistical analysis
Descriptive statistics include median and interquartile range (IQR) for continuous variables with Kruskal–Wallis test for comparison, categorical data described as frequency and proportion and group comparison tested with Odds Ratios (OR) with 95% CI and Fisher’s exact test. Primary outcomes (VTE IR per 1000 PY) are presented with 95% CI derived from Poisson distribution and changes in VTE IR over time were assessed by linear least squares regression analysis using the coefficient of determination (R-squared, R2) as the goodness-of-fit measure where higher coefficients (range 0 to 1) indicating a better fit for increasing or decreasing incidence rates over time with p-values derived from analysis of variance. We applied logistic regression for the analysis of VTE risk factors that were significant in univariate analysis and included these in age and gender adjusted Wald backward multivariate models. Survival data were based on Kaplan–Meier estimates with logrank testing for subgroup comparisons. All statistical analyses were performed using SPSS software v29.0 (IBM, USA) and OpenEpi software with two-sided p-values (p) < 0.05 considered to be statistically significant.
Results
Patient characteristics at time of index admission for patients with SLE and controls and incidence rates of VTE. Values represent median (interquartile range, IQR), number of patients (%) and hazard ratio (HR), incidence rate/1000 person years or rate ratio (RR) all with 95% confidence interval (CI).
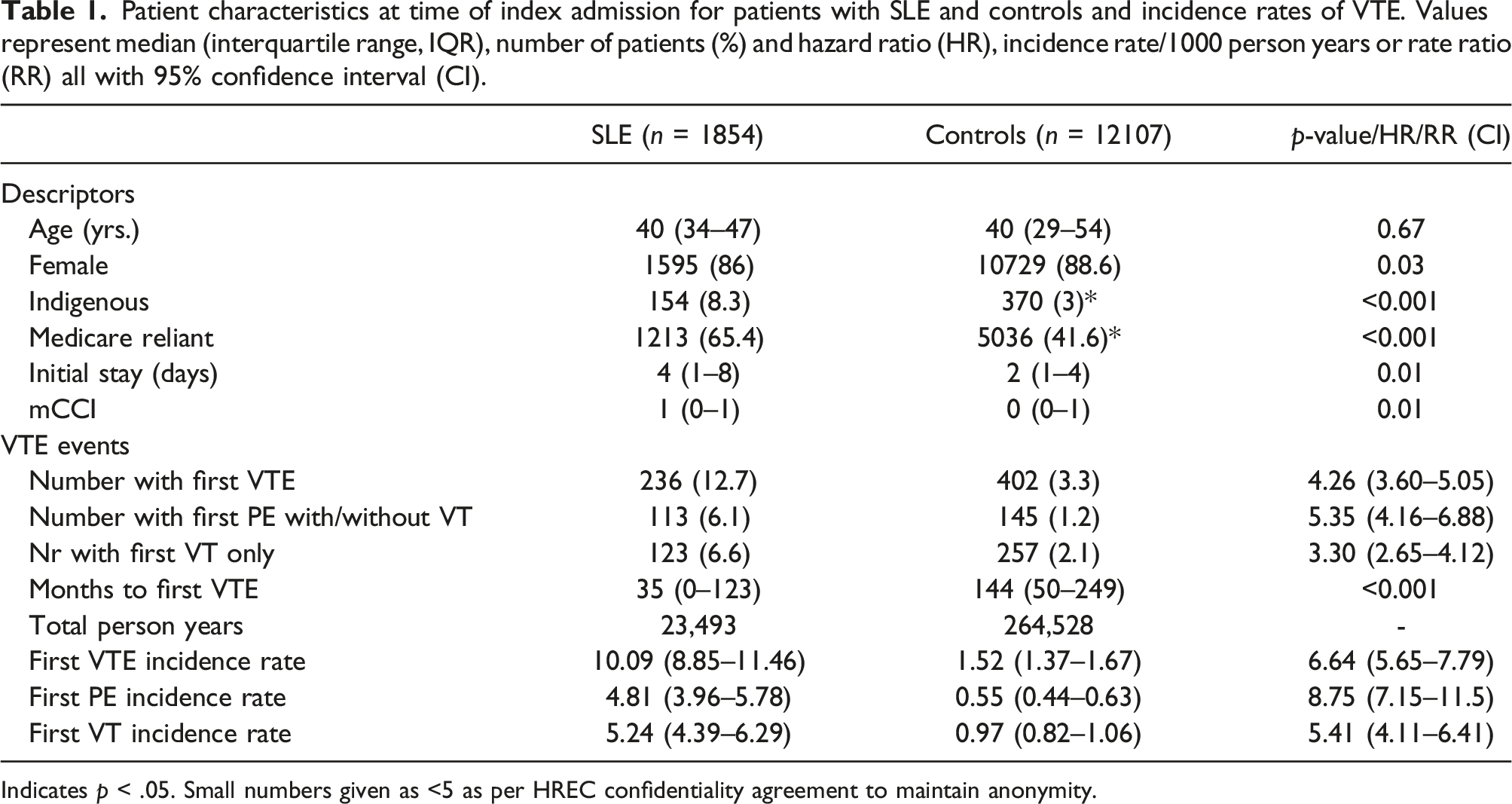
Indicates p < .05. Small numbers given as <5 as per HREC confidentiality agreement to maintain anonymity.
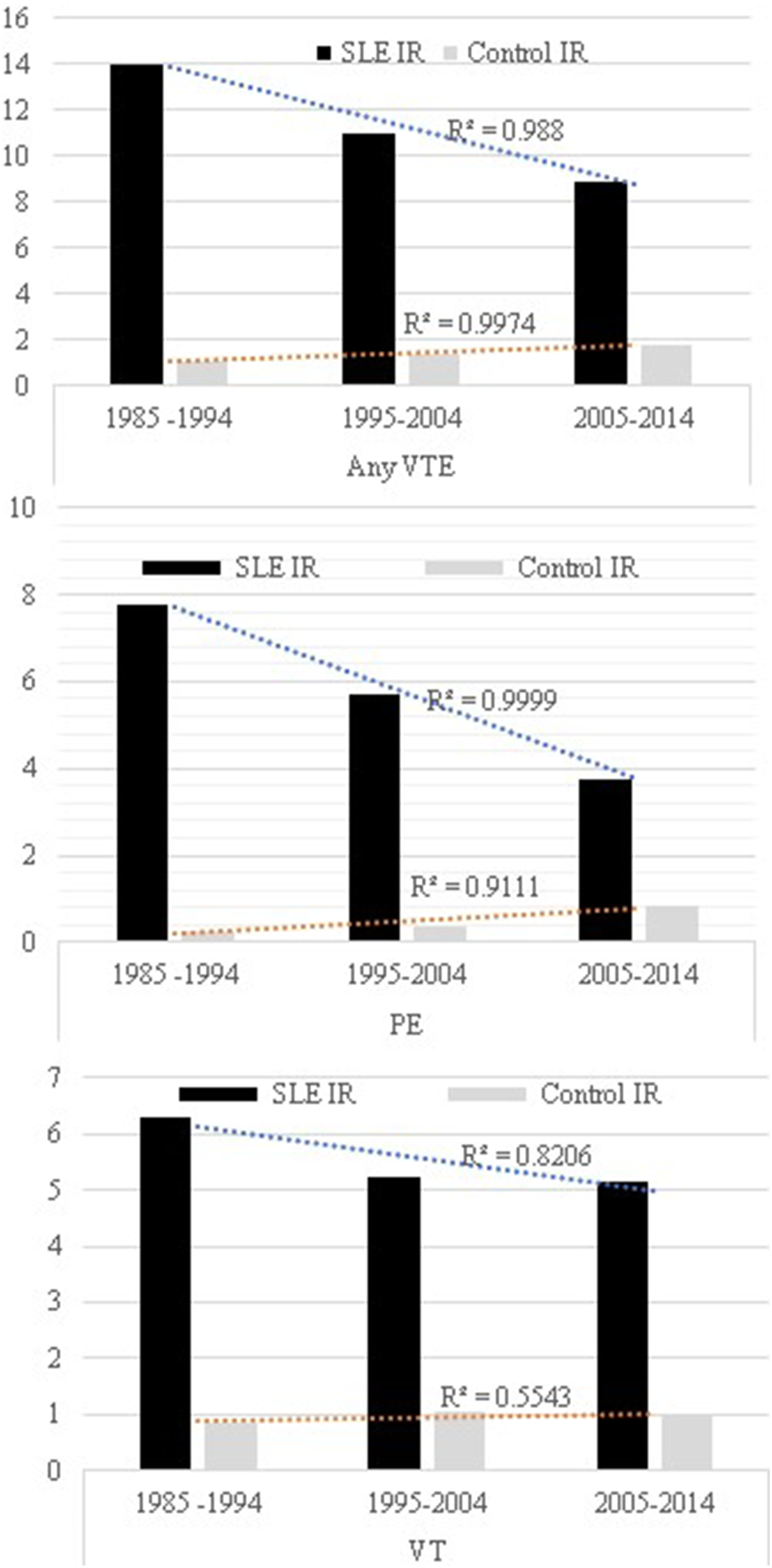
Temporal trend of incidence rates (IR) ) per 1000 person years for all venous thrombotic events (anyVTE), pulmonary embolism (PE) and venous thrombosis (VT) in patients with systemic lupus erythematosus (SLE) and controls. R2 denotes the coefficient of determination.
Frequency, crude hazard ratio (HR) and adjusted hazard ratio (a-HR) for the association between risk factors and a first venous thrombotic event (VTE) in patients with Systemic Lupus Erythematosus (SLE) and controls. Values represent number of patients (%) and hazard ratio (HR) with 95% confidence interval.
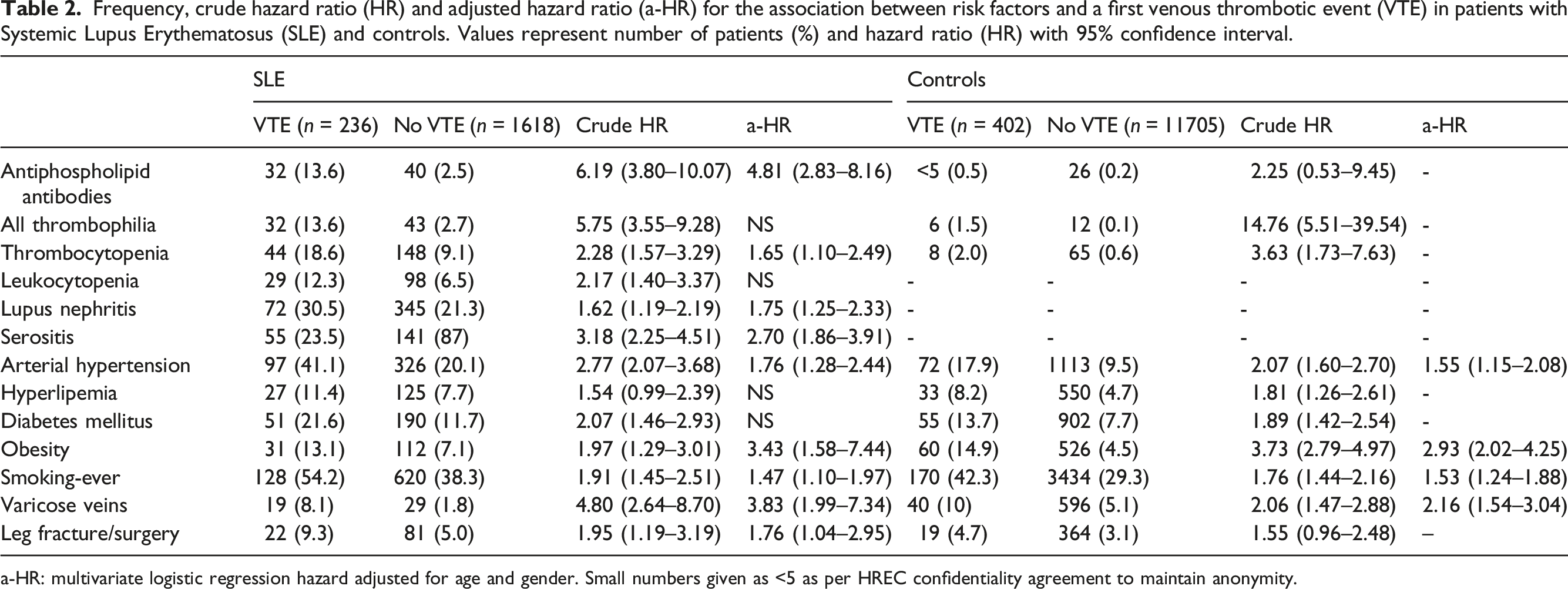
a-HR: multivariate logistic regression hazard adjusted for age and gender. Small numbers given as <5 as per HREC confidentiality agreement to maintain anonymity.
Frequency, crude hazard ratio (HR) and adjusted hazard ratio (a-HR) for death and arterial events following first venous thrombotic event (VTE) in patients with Systemic Lupus Erythematosus (SLE) and controls. Values represent number of patients (%) and hazard ratio (HR) with 95% confidence interval.
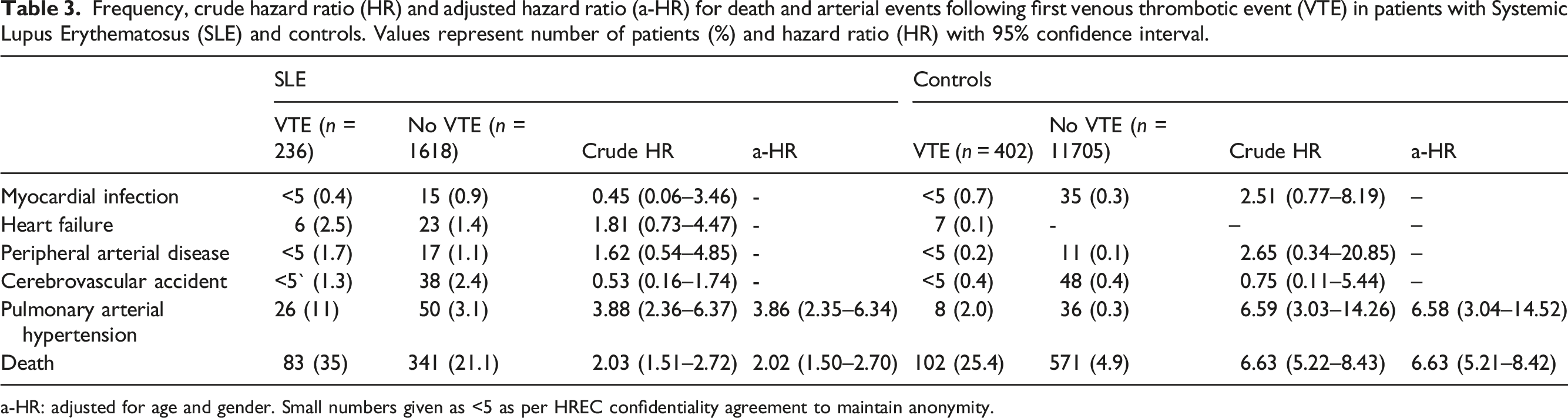
a-HR: adjusted for age and gender. Small numbers given as <5 as per HREC confidentiality agreement to maintain anonymity.
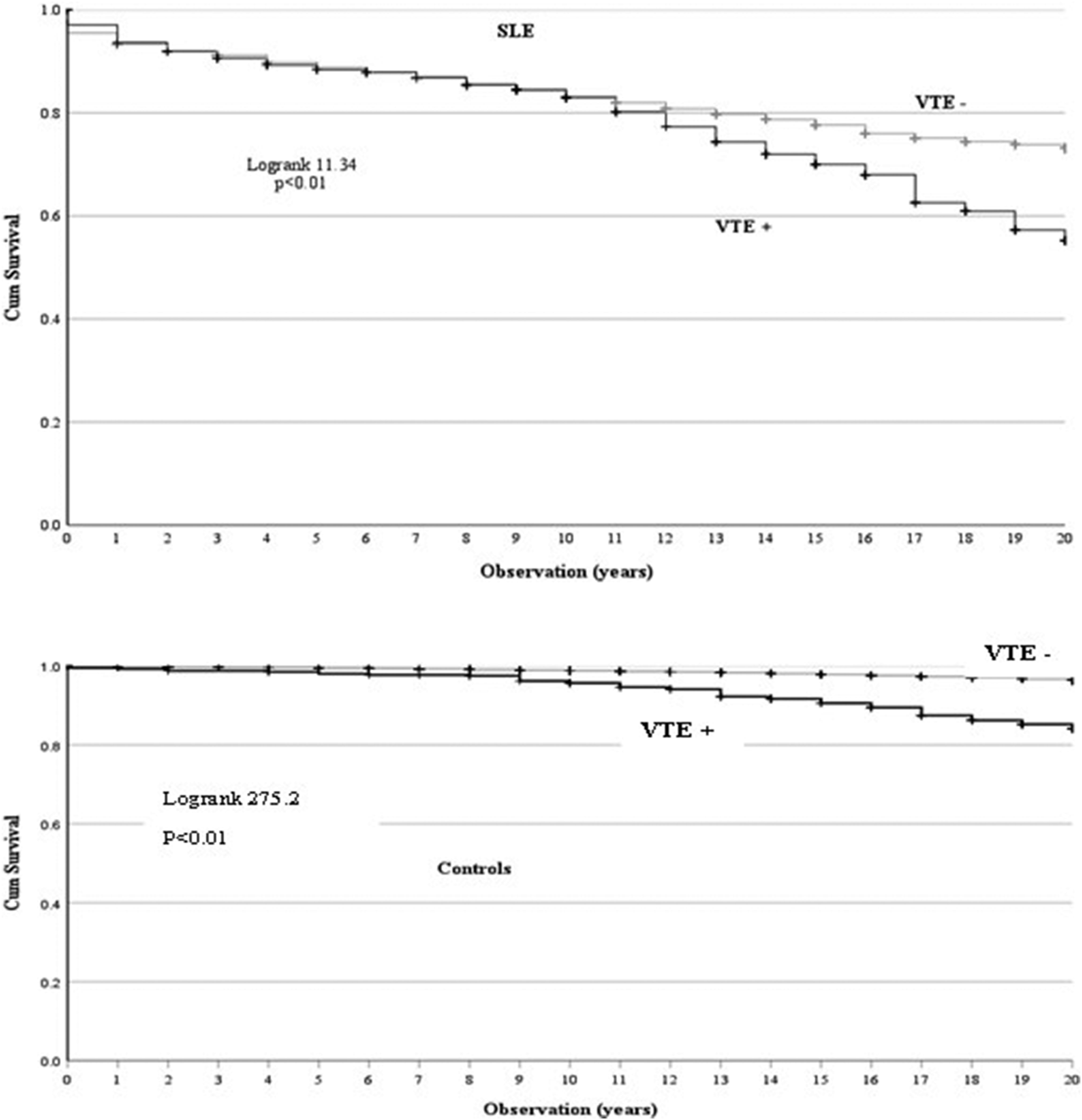
Kaplan–Meier survival curves for A) patients with systemic lupus erythematosus (SLE) and B) controls based on occurrence of a first venous thrombotic event (VTE+/VTE−).
Discussion
This long-term study on absolute and relative risk and rates for VTE found that 236/1854 (12.7 %) of patients with SLE developed VTE over a median observation time of 12 years compared to 402/12,107 (3.3%) of controls (OR 4.26, CI 3.6–5.1). The corresponding IRR was 6.6 times higher in SLE than in controls. These differences were more pronounced for PE than VT, even though PE incidence declined over time. The main VTE risk factor was aPL presence in SLE and other forms of thrombophilia for controls, with traditional risk factors evenly distributed. In both groups, VTE did not associate with an increased risk of arterial thrombotic disease, except for the association between PE and PH prevalence.
The absolute VTE risk in patients with SLE was 12.7% which higher than 5–8% reported in the meta-analysis but aligns with a number of previous studies,5,19,20 while the increase in odds of 4.26 compared to controls is a close fit with the estimated 4.38 times pooled higher risk in the meta-analysis. 7 Correcting VTE risk for observation time, we found the IR for a first VTE was 10.1/1000 PY which lies well within the reported range of 5.5–11.1 (7) and was 6.6 times higher than in controls, not dissimilar to the 4.8 increase in VTE incidence observed in one of the largest cohort studies among 4863 individuals with SLE in Canada. 21 Despite differences in the level of detail for defining VTE, the absolute and time adjusted relative risk of VTE in this SLE cohort and controls was thus largely in line with current literature.
As in most reports the IR for VT (5.2/1000 PY) was higher than for PE (4.8/1000 PY), but the difference was not as large as in other studies, likely because we excluded VT when it was concurrent with PE. The higher rate ratio for PE than VT suggests a stronger propensity to PE development in SLE than controls. Whether this is based on other lupus manifestations such as endothelial dysfunction or proteinuria or solely on aPL presence is not clear. The long observation period allowed analysis of trends, where we found a significant reduction in the IR for PE over time. Given the time frame, this is likely the result of increasing recognition of SLE associated thrombophilia and the development of various anticoagulative measures for primary and secondary prevention over time.22,23 However, almost a quarter of patients with SLE experienced a recurrence of VTE over the study period, which is similar to the 25% recurrence observed in a recent multicentre study that applied the same time frame for VTE recurrence and found that aPL were the main predictor for recurrent VTE after adjusting for other provoking factors. 24
Many studies have reported the presence of one or more aPL as a major risk factor for VTE in patients with SLE25–28 and our study confirms that aPL antibodies increase the hazard of VTE by a factor of 4 in patients with SLE with lower hazards for the association with thrombocytopenia and lupus nephritis. Other thrombophilia factors such a factor V Leiden or protein C or S deficiency (recorded in did not further increase these odds in patients with SLE, but they were the main risk factor for VTE in controls, where aPL were not a risk factor. However, it should be noted that aPL testing may not have been done as frequently in non-SLE patients with thrombosis as in SLE patients and thus underreported. Surgery and cancer have traditionally been considered main risk factors for VTE, but there is increasing recognition that traditional cardiovascular risk factors also contribute to VTE risk. 3 Our findings confirm that in addition to disease related risk factors these traditional factors contribute to VTE risk in patients with SLE in a fashion similar to controls. This opens potential new avenues to reduce VTE risk in patients with SLE, especially those with persistent aPL who have not (yet) developed VTE.25,29
The occurrence of PE confers a risk of immediate and potentially life-threatening haemodynamic complications and entails the risk of developing into chronic PH due to organisation of thromboembolic material and vascular remodelling. 30 There is relatively little data on the long-term consequences of PE in general, but PH development has been observed in up to 5% of patients with PE. 30 Although the proportion of patients developing PH was much higher in SLE than controls (4.1% vs 0.3%), the occurrence of VTE increased the hazard ratio of PH significantly in both groups. While the use of ICD coding limits the ability to distinguish between the various mechanisms contributing to secondary PH, these figures suggest increased attention should be paid to the possibility of clinically silent chronic and/or recurrent PE by follow-up imaging of the pulmonary circulation in patients with SLE especially. 30
We found a notable absence of an association between VTE and arterial thrombotic events in patients with SLE, which confirms the idea that these two vascular manifestations have different pathogenic mechanisms with a larger contribution of aPL to VTE than arterial thrombosis.1,27 While the occurrence of VTE increased the hazard of death in both groups, this was not an immediate effect as the survival curves only started to differ after many years. The effect was most pronounced in patients presenting with PE confirming a long-term mortality risk seen with PE in cohort studies, 31 while VT alone did not increase mortality in patients with SLE in contrast to controls.
The limitations of this study include the lack of classifying and prognostic markers such as composite measures of SLE disease activity, confirmatory repeat testing and subtyping for aPL and other autoantibodies and type and duration of anticoagulant treatment. As a result, several confounding variables remain unadjusted for. The use of ICD coding only allows identification of patients with a positive result of aPL antibody testing in the context of APS manifestations. Thus, we cannot tabulate how many patients and controls were tested for aPL antibodies overall while underreporting may have confounded the data on other types of thrombophilia. Also, while aPL testing likely was done in the context of a thrombotic event, the frequency of APS in controls may be an underestimate. Our control group cannot be considered completely healthy controls, but we have earlier demonstrated that our controls reflect the general population well in terms of overall prognosis.11,13 Pregnancy as a risk factor for VTE was as frequent in controls (25%) and SLE patients (24.5%). Our data are based on physician diagnoses at discharge of care and while the deidentified nature of the dataset did not allow validation of VTE, this approach has been shown to have high positive predictive value and is also supported by the fact that VTE IR in our controls was similar to the population IR of 1.5/1000PY in the USA.15,32 The index admission used to identify SLE patients did not necessarily coincide with the time of clinical diagnosis, and we cannot exclude having missed some SLE patients with a mild phenotype never requiring hospital care. We recorded data based on hospital attendance (admission or ED. visit) and may have missed patients with VTE who were not referred for specialist care, but current guidelines in Australia suggest hospital referral with suspected VTE. 33 Importantly also, novel oral anticoagulant therapy for VTE was not approved in Australia until after the end of our observation period. Our analyses used the first event to classify VTE subgroups and we did not study the effect of a crossing over of subtypes. The strength of this study lies in its ability to study longitudinal validated data on VTE frequency and outcomes in a large population-based group of patients with SLE and controls.
In conclusion, the development of VTE decreased over study period but was overall six times higher in patients with SLE than controls. APL antibodies, thrombocytopenia and lupus nephritis were the main risk factor in SLE patients. We also found arterial hypertension, smoking and obesity as modifiable risk factors in both groups. The occurrence of PE in patients with SLE associated with a four times higher prevalence of PH.
Supplemental Material
Supplemental Material - Long-term incidence, risk factors and complications for venous thromboembolism in patients with systemic lupus erythematosus
Supplemental Material for Long-term incidence, risk factors and complications for venous thromboembolism in patients with systemic lupus erythematosus by Johannes Cornelis Nossent, Helen Isobel Keen, David Brian Preen and Charles Anoukpar Inderjeeth in Lupus.
Footnotes
Acknowledegments
The authors acknowledge the Data Custodians of the Hospital Morbidity Data Collection (HMDC), Emergency Department Data Collection (EDDC), the State Registry of Births, Deaths and Marriages and the WA Electoral Commission for data provision. We thank the staff at Data Linkage Branch at the Western Australian Department of Health for their assistance in collating the data and at the Western Australian Clinical Coding Authority for continued support.
Author contributions
Johannes Nossent, Helen Keen, David Preen and Charles Inderjeeth are members of the steering committee, that procured and supervised the WARDER dataset. Johannes Nossent designed the current study and was responsible for extraction and analysis of the data. Johannes Nossent, Helen Keen, David Preen and Charles Inderjeeth contributed to interpretation of the data and engaged in drafting the manuscript and revising the manuscript critically.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Arthritis Foundation of Western Australia with an unrestricted grant to JCN.
Ethical statement
Contributorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
