Abstract
Introduction
Systemic lupus erythematosus (SLE) predominantly affects females of reproductive age but can affect patients later in life with variable disease presentations. Late-onset (LO)-SLE is defined as SLE diagnosed at age 50 years or later, which includes 20% of all SLE patients. 1 Studies comparing LO-SLE to non-LO-SLE (NLO-SLE) have been conflicting.2–6 Some studies show a higher prevalence of autoantibodies such as SSA and SSB in LO than NLO-SLE patients5,6 with other studies not demonstrating a difference in prevalence of autoantibodies.2,7 Studies have examined the clinical presentation of LO and NLO-SLE patients.2,4 While LO-SLE patients were less likely to present with renal involvement,2–4 they had higher rates of serositis,4,5 more comorbid conditions such as cardiovascular disease,3,5,7 and higher mortality rates.5,7 As patients with SLE face diagnostic delays, understanding disease presentation in SLE is important to reducing diagnostic delays. 8 With this clinical dilemma and lack of US studies, we examined the differences between the clinical phenotypes of LO and NLO-SLE patients. In addition to small and conflicting studies on disease presentation in LO-SLE, few studies have examined if prescribing patterns differ in LO-SLE compared to NLO-SLE patients. Further, few studies exist that include a large sample of diverse LO-SLE patients in the US. Using a large, EHR-based cohort of SLE individuals, we compared demographics, disease characteristics, SLE-specific antibodies, and medication prescribing practices in LO vs. NLO-SLE individuals.
Methods
The Synthetic Derivative
After receiving approval from the Institutional Review Board of Vanderbilt University Medical Center (VUMC), we used a large, de-identified, mirror image of our EHR called the Synthetic Derivative. The Synthetic Derivative has been previously described and currently contains over 3.6 million individuals from 1989 to present. 9 Data includes demographics, billing codes, inpatient and outpatient notes, medications, laboratory data, radiology, and pathology reports. Health data from outside VUMC is not available.
Identifying systemic lupus erythematosus individuals
Potential SLE individuals were identified using a previously published and validated algorithm that requires ≥4 counts of the SLE International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9) code of 710.0 and a positive anti-nuclear antibody (ANA), defined as a titer of ≥1:160, while excluding for subjects with ICD-9 codes for systemic sclerosis (710.1) and dermatomyositis (710.3). The algorithm has a positive predictive value of 89% and a sensitivity of 86% and has been externally validated.10,11 We performed chart review on all potential SLE individuals identified by the algorithm to ensure they were diagnosed with SLE by a specialist (dermatology, rheumatology, or nephrology) (Supplemental Figure 1). As SLE classification criteria are not systematically collected in rheumatology notes, 10 we used specialist diagnosis to define SLE case status. As our primary analysis examines prevalence of SLE autoantibodies at time of SLE diagnosis, we excluded SLE individuals who did not have autoantibodies at diagnosis at our institution.
Outcomes and variables assessed
We assessed demographics including sex, race, ethnicity, and age at SLE diagnosis. Race and ethnicity are collected as a mixture of self-report and administrative entry. Prior studies have validated that these race assignments align with genetic ancestry. 12 Age at SLE diagnosis was defined as age at first SLE billing code or age at SLE diagnosis documented in notes, whichever came first. We conducted sensitivity analyses excluding individuals without documented dates of diagnosis. We performed chart review to assess for autoantibodies including dsDNA, SSA (Ro), SSB (La), Smith, RNP, lupus anticoagulant, anticardiolipin, and beta-2-glycoprotein I antibodies at SLE diagnosis. Autoantibodies were measured using enzyme-linked immunosorbent assays with manufacturer values to determine positivity. We only counted autoantibody testing performed at VUMC, as outside labs could not be confirmed. We calculated the percentage of individuals with positive autoantibodies by decade of SLE diagnosis. SLE nephritis was defined by renal pathology or clinical diagnosis by a rheumatologist or nephrologist.
We performed chart review for ever use of commonly prescribed medications in SLE including antimalarials, conventional disease modifying antirheumatic drugs (DMARDs), biologics, and renal transplant regimens (Supplemental Table 1). Medications are compiled from inpatient and outpatient prescribing systems and a validated software program called MedEx. 13 MedEx mines medications from free text in clinical documents.
Statistical analyses
We evaluated for differences in demographics, autoantibodies, presence of SLE nephritis, and medications ever prescribed in LO-SLE vs. NLO-SLE individuals using the Mann–Whitney U test for continuous variables and Chi-square test for categorical variables, as a priori, we anticipated non-normal distributions in the data. Categorical variables were expressed as relative frequencies. Continuous variables were expressed as median values with interquartile ranges. SLE individuals with missing demographic and clinical data were excluded. We performed logistic regression to estimate the association of LO-SLE with sex, race, nephritis, positive dsDNA, and medications prescribed. For logistic regression models, odds ratios (ORs) and 95% confidence intervals (95% CIs) are reported. Analyses were conducted using R version 4.3.2.
Results
Systemic lupus erythematosus cohort
Characteristics in late-onset SLE versus non-late-onset SLE.
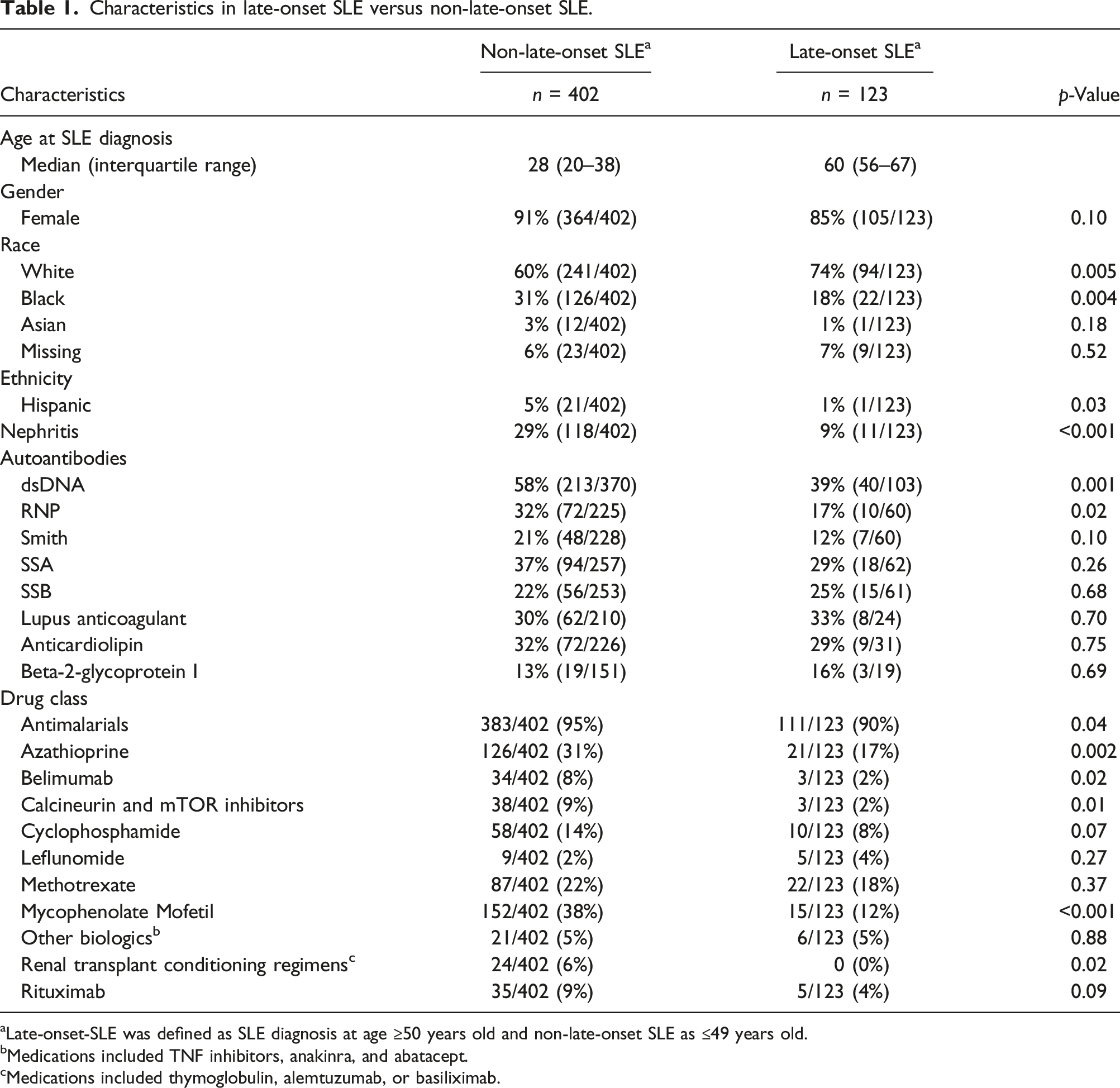
aLate-onset-SLE was defined as SLE diagnosis at age ≥50 years old and non-late-onset SLE as ≤49 years old.
bMedications included TNF inhibitors, anakinra, and abatacept.
cMedications included thymoglobulin, alemtuzumab, or basiliximab.
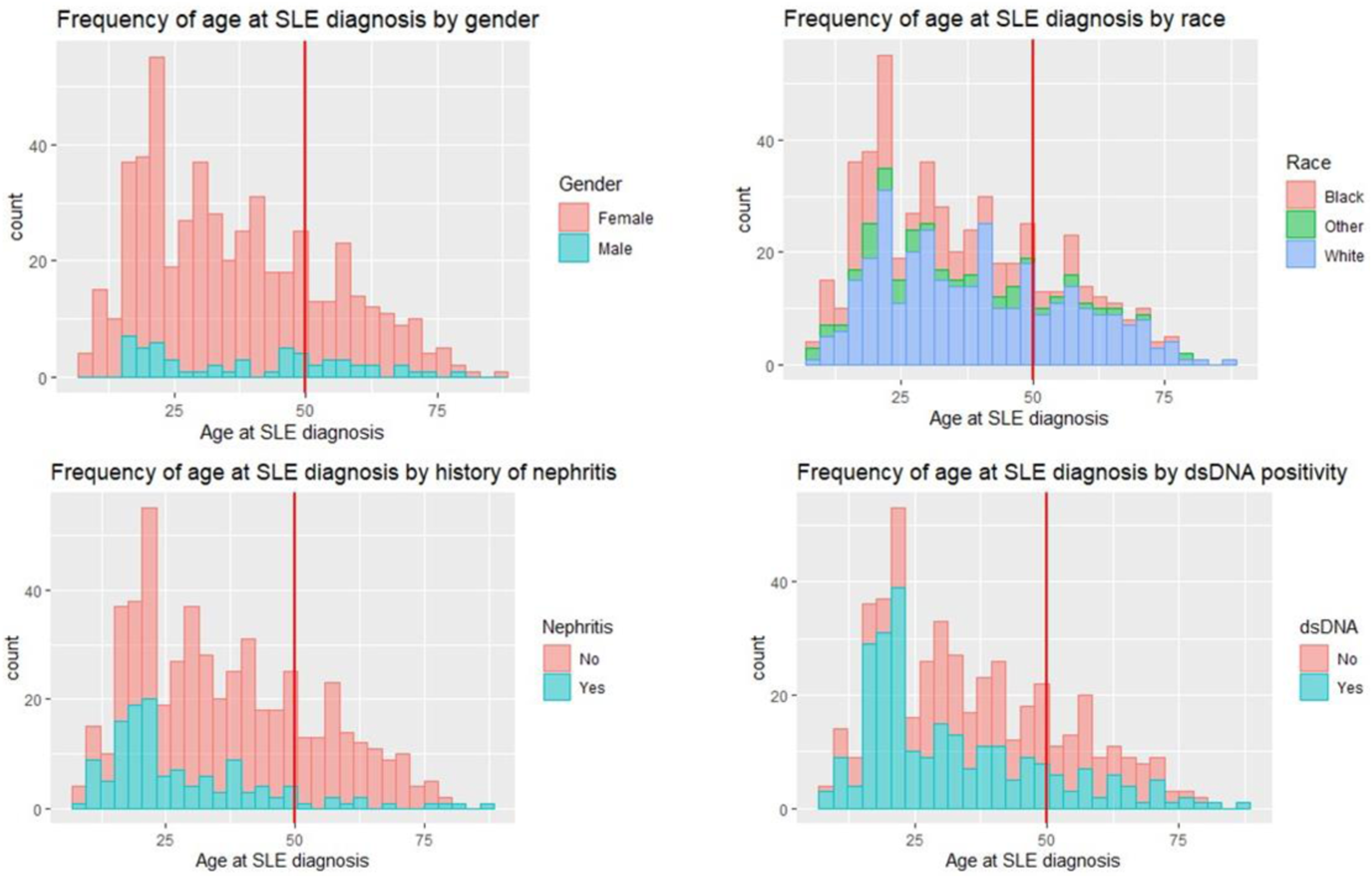
Frequency of age at SLE diagnosis by demographic and clinical characteristics. (a) Frequency of age at SLE diagnosis by gender. Red line at age 50 years old distinguishes late-onset-SLE (LO-SLE) (SLE diagnosed at ≥50 years old) from non-late-onset-SLE (NLO-SLE) (SLE diagnosed <50 years old). Both LO-SLE and NLO-SLE individuals were predominantly female. (b) Frequency of age at SLE diagnosis by race. Other indicates Asian and mixed race groups. LO-SLE individuals were more likely to be White vs. NLO-SLE individuals. (c) Frequency of age at SLE diagnosis by history of SLE nephritis. SLE nephritis defined as either diagnosis by biopsy or clinical diagnosis by a rheumatologist or nephrologist. LO-SLE individuals were less likely to develop SLE nephritis. (d) Frequency of age at SLE diagnosis by positive dsDNA antibody. DsDNA positivity was determined at time of SLE diagnosis with manufacturer values to determine positivity. LO-SLE individuals were less likely to have positive dsDNA antibodies.
Autoantibodies
We compared autoantibody positivity in LO vs. NLO-SLE individuals. LO-SLE individuals were less likely to have positive dsDNA (39% vs. 58%, p = 0.001) (Figure 1(d)) and RNP (17% vs. 32%, p = 0.02) with no differences in Smith, SSA, SSB, lupus anticoagulant, anticardiolipin, and beta-2-glycoprotein I antibodies compared to NLO-SLE individuals (Table 1). We then analyzed autoantibody positivity by decade of age at SLE diagnosis. We observed that autoantibody positivity declined with increasing age (Figure 2). Specifically, individuals diagnosed with SLE in the first two decades of life had dsDNA positivity rates of 75% with positivity declining to 33% in the fifth and sixth decades (Figure 1(d)). Similar patterns were observed for RNP and Smith. SLE autoantibody positivity by decade of SLE diagnosis. The x-axis shows the age at SLE diagnosis in years and organized by decade. The y-axis shows the percent of patients diagnosed with SLE in that decade who had a positive autoantibody test at time of SLE diagnosis. Late-onset-SLE individuals were less likely to have positive dsDNA and RNP antibodies compared to non-late-onset SLE individuals with no differences in Smith, SSA, and SSB. Rates of dsDNA and RNP positivity declined with increasing age.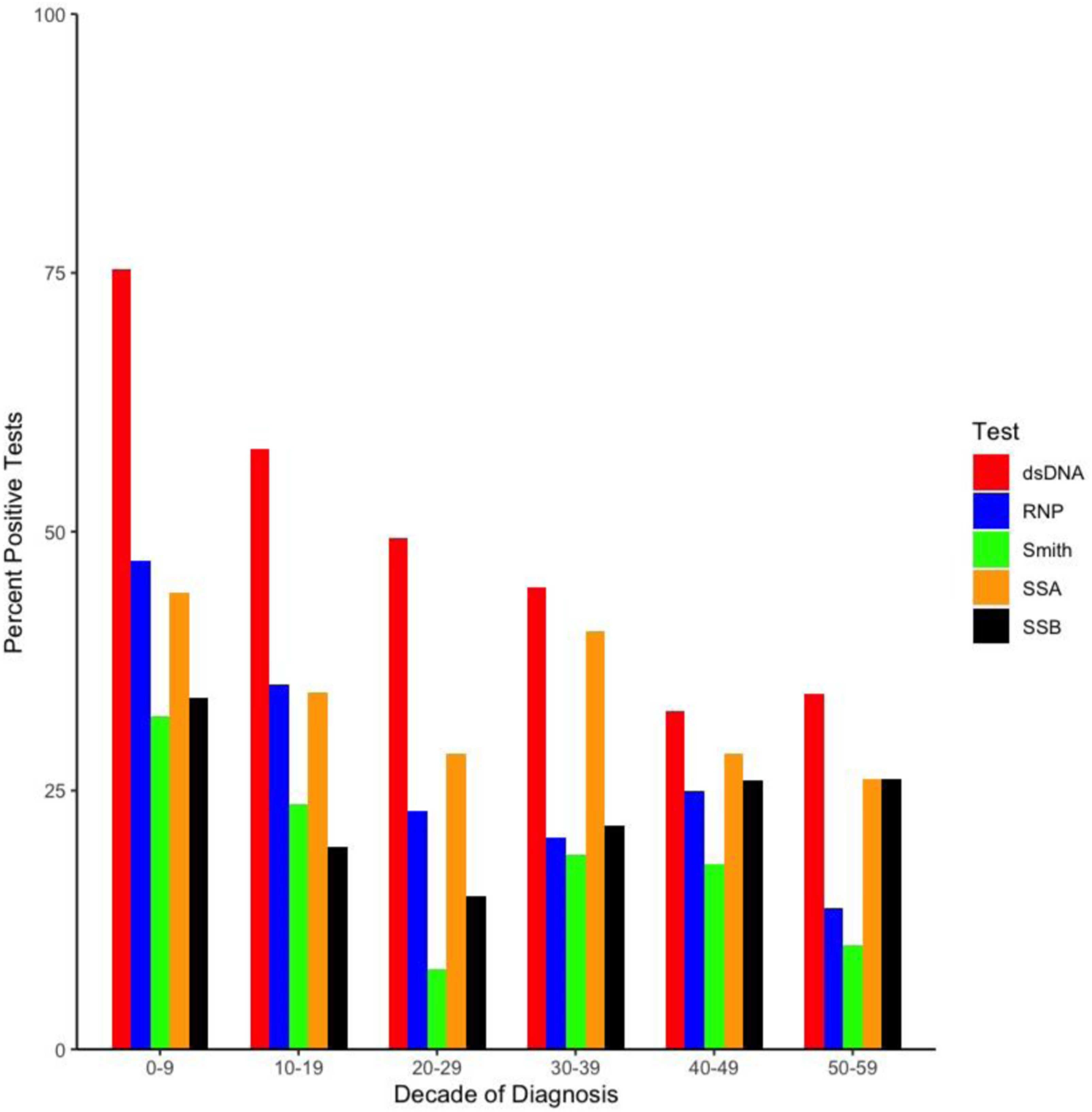
Medications
We compared SLE medications ever prescribed in LO vs. NLO-SLE (Table 1). LO-SLE individuals were less likely to be prescribed multiple classes of SLE medications including antimalarials (90% vs. 95%, p = 0.04), azathioprine (17% vs. 31%, p = 0.002), mycophenolate mofetil (12% vs. 38%, p < 0.001), and belimumab (2% vs. 8%, p = 0.02). LO-SLE individuals were also less likely to be on calcineurin inhibitors (2% vs. 9%, p = 0.01) and renal transplant conditioning regimens (0% vs. 6%, p = 0.02). There were no significant differences in rates of methotrexate, leflunomide, or cyclophosphamide.
Sensitivity analyses
For LO-SLE individuals, we were more likely to rely on first SLE billing code vs. documented date in clinic note for SLE diagnosis (32% vs. 15%, p < 0.05). Sensitivity analyses were conducted for all LO-SLE vs. NLO-SLE comparisons above excluding individuals with missing documented dates of SLE diagnosis. Findings from these comparisons did not differ from the above results.
Logistic regression models
LO-SLE individuals were less likely to have nephritis (OR = 0.24, 95% CI 0.11–0.47, p < 0.001) after adjusting for race and sex (Supplemental Table 2). LO-SLE individuals were also less likely to have positive dsDNA (OR = 0.55, 95% CI 0.34–0.87, p = 0.01) after adjusting for race and sex. There was no significant difference in hydroxychloroquine prescribing (OR = 0.53, 95% CI 0.24–1.21, p = 0.12) after adjusting for race and sex.
Discussion
We compared LO and NLO-SLE individuals in a large, real-world, EHR-based cohort. Nephritis and autoantibody positivity was less frequent in LO-SLE individuals compared to NLO-SLE individuals, even after adjusting for sex and race. LO-SLE individuals were less likely to receive antimalarials and multiple classes of immunosuppressant medications. Our findings are important as they show that age impacts SLE presentation and management of SLE.
Similar to some but not all studies, we found a lower prevalence of SLE-specific autoantibodies in LO-SLE individuals. In one Spanish study of 59 LO-SLE individuals, prevalence of dsDNA and Smith positivity were significantly lower in LO-SLE vs. NLO-SLE individuals. 7 In contrast, a Canadian study that included 161 LO-SLE individuals, who were predominantly White, showed equal prevalence of dsDNA in LO-SLE vs. NLO-SLE individuals. 6 Lastly, a Turkish study with 20 LO-SLE individuals found no statistical difference in autoantibody positivity between LO-SLE and NLO-SLE. 14 These studies may have different findings due to differences in study design, differences in race and ethnicity of the study population, and low power to detect differences. These studies reviewed above are prospective SLE cohorts with patients meeting SLE classification criteria, which contrasts with our real-world EHR-based study that requires an individual be diagnosed with SLE by a specialist. Our study provides data on autoantibody positivity in a larger and more diverse population of LO-SLE individuals.
We observed less frequent rates of nephritis in LO-SLE vs. NLO-SLE individuals. A milder disease course with less frequent nephritis has also been observed in other cohort studies.3,6,7,15 Lower rates of positive dsDNA and nephritis were still observed in LO vs. NLO-SLE even after adjusting for race. Thus, differences in race in LO vs. NLO-SLE individuals were not solely driving clinical differences. We also observed antimalarials and multiple immunosuppressants were prescribed less frequently in LO-SLE individuals, which likely reflects and confirms the lower severity of disease in LO-SLE. Few studies focus on the impact of age of SLE diagnosis on prescribing patterns in SLE. Two studies agree with our findings and found that antimalarials and immunosuppressants were less frequently prescribed in LO-SLE vs. NLO-SLE individuals.3,7,15 With less frequent nephritis in LO-SLE individuals, it was expected that LO-SLE individuals would be less frequently prescribed medications used for renal transplant as well as mycophenolate mofetil and azathioprine. We also found less frequent prescribing of antimalarials in LO-SLE, which agrees with one of our prior studies 16 and a Danish study. 17 We hypothesize that LO-SLE individuals may not have been prescribed antimalarials due to less active disease compared to NLO-SLE individuals. Further, since age is a risk factor for antimalarial ocular toxicity, this concern may have influenced prescribing patterns. 16
While we have a relatively large and diverse study of LO-SLE individuals in the EHR, there are several limitations. First, we have a single center based in the Southeastern US that may not be generalizable to other SLE populations. With using the EHR, we have missingness in our autoantibody data. Some individuals may have autoantibody data from outside the institution and these autoantibodies may not be rechecked and available in our dataset. There was also missingness for 19% of SLE individuals in documentation of SLE diagnosis dates with LO-SLE patients having more diagnosis dates missing. This could potentially introduce bias. We re-ran our analyses excluding individuals with missing documented diagnosis dates in clinic notes and found no differences in the overall conclusions, suggesting this differential missingness did not introduce systemic bias into our results. As SLE classification criteria are not systematically collected in rheumatology notes, 10 we could not assess for differences in SLE classification criteria in LO vs. NLO-SLE individuals. As our data source is a de-identified EHR, death data is not available, so we could not examine for differences in mortality in LO vs. NLO-SLE individuals. Lastly, we assessed for prescribing patterns for medications but could not assess medication use or adherence, as pharmacy fill data is not available in our de-identified dataset.
We observed that LO-SLE may be less likely to fit an expected SLE presentation with less frequent positive autoantibodies at diagnosis and less severe disease with lower rates of nephritis, even after adjusting for race. As SLE is a heterogeneous disease with highly variable disease presentations, understanding how age impacts SLE disease presentation is important to help reduce diagnostic delays in SLE.
Supplemental Material
Supplemental Material - Comparison of late-onset and non-late-onset systemic lupus erythematosus individuals in a real-world electronic health record cohort
Supplemental Material for Comparison of late-onset and non-late-onset systemic lupus erythematosus individuals in a real-world electronic health record cohort by Ganiat Adeogun, Alex Camai, Ashley Suh, Lee Wheless, and April Barnado in Lupus.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases (1K08 AR072757-01, R01 AR080629, Barnado); National Institutes of Health/National Center for Research Resources (UL1 RR024975, VUMC); and National Institutes of Health/National Center for Advancing Translational Sciences (ULTR000445, VUMC); VACSR&D (CX-002452, Wheless). This publication does not represent the views of the Department of Veteran Affairs or the United States Government.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
