Abstract
Objective
To report participant characteristics relevant to identifying health inequities in systemic lupus erythematosus (SLE) randomized controlled trials conducted in Canada.
Methods
We conducted a scoping review by searching MEDLINE (Ovid) and Embase (1990 to June 2023), and CENTRAL (inception to June 2023). Eligible studies: used an RCT design; evaluated interventions (pharmacologic and non-pharmacologic) among SLE patients aged ≥18 years; and were conducted in Canada. Data extraction was guided by the Campbell and Cochrane Equity Methods Group’s PROGRESS-Plus framework on 11 factors leading to health inequities (
Results
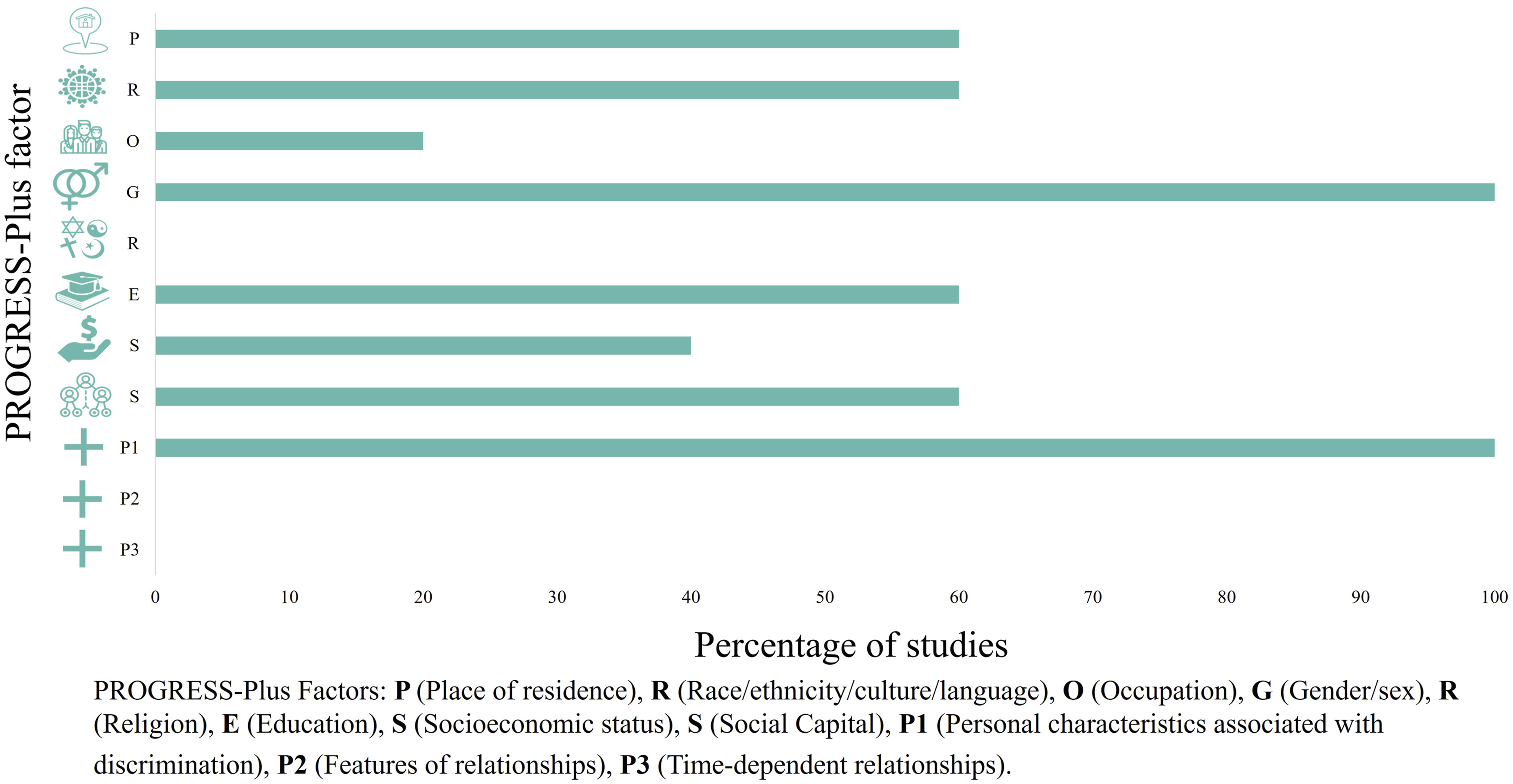
Of 1901 unique records, 6 met the inclusion criteria. Sex and age were the only PROGRESS factors that were reported in all studies. The majority of participants were female (84.4% to 100%), and mean ages of participants ranged from 42 to 52.3 years. Place of residence, race, education, and social capital were reported in three studies. Socioeconomic status was reported in two studies, and occupation was reported in one study. Religion, features of relationships, and time-dependent relationships were not reported in any included studies.
Conclusion
Limited reporting of determinants of health inequities in RCTs for SLE in Canada suggests the need for reporting standards to support equity, diversity, and inclusion practices in research.
Introduction
There is an urgent need to address inequities in health created by structural and social disparities. Race, sex and/or gender identity, sexual orientation, socioeconomic status (SES), and religion among others have been documented to result in unequal allocations of power and resources.1,2 This unequal allocation can impact health in various ways, such as through one’s ability to access health services. This is particularly important for complex chronic conditions such as systemic lupus erythematosus (SLE), an autoimmune condition with multiple organ targets, as patients require longitudinal access to care to manage disease activity and symptoms and preserve their health, and disease burden is not equally distributed.
The Campbell and Cochrane Equity Methods Group’s PROGRESS-Plus
3
framework outlines (risk) factors (‘PROGRESS factors’) that lead to inequities in health (i.e.
Efforts to address health inequities for SLE care and research in Canada have identified that access to care is negatively affected by racism and geography. 10 To add to these efforts, it is also important to examine how determinants of health inequities have been considered in SLE clinical research in Canada, particularly because research is how effective therapies for SLE are identified. Missing these determinants of health inequities may limit opportunities to intervene or design interventions, understand treatment, or could inadvertently exacerbate disparities by not including all population groups. Though specific determinants of health inequities such as race and ethnicity are often reported in certain countries (e.g. the US11,12), to our knowledge, there has been no similar investigation of the reporting of a broad definition of determinants of health inequities in SLE trials conducted in Canada. As such, our objective was to conduct a scoping review of Canadian randomized controlled trials (RCTs) of interventions among people with SLE to characterize participants and identify which, if any, of a broad range of determinants of health inequities are reported, and how, guided by the PROGRESS-Plus framework.
Methods
Inclusion criteria
We followed the Arksey and O’Malley framework for conducting scoping reviews. We included studies that: 1) used an RCT design; 2) evaluated interventions, defined as either pharmacological or non-pharmacological treatments or services; 3) included participants with SLE; 4) conducted in Canada; 5) published in English; and 6) published between 1990 and July 2022. These criteria were selected based on our prior scoping reviews of other rheumatic diseases.13,14
Information sources and search
We conducted a search in MEDLINE (Ovid 1990 to June 2023), Embase (Ovid 1990 to June 2023), and CENTRAL (inception–June 2023) (Supplemental Materials). The search strategy was adapted from the sensitivity-maximizing Cochrane Highly Sensitive Search Strategy for identifying randomized trials in MEDLINE (2008 revision). Following best practices, we consulted with a librarian to develop and refine our search.
Study selection
All retrieved publications were independently screened for eligibility, first by title and abstract (VV) and then by full text (MT and VV). Any records judged eligible for inclusion proceeded to full text review. Discussion between all authors occurred to resolve uncertainty and to achieve consensus about inclusions.
Data extraction and synthesis of results
We extracted the following data from included studies: authors, title, journal, year, purpose of the study, and study intervention. Of particular interest was the reporting of eleven PROGRESS-Plus factors: 1) Place of residence; 2) Race, culture, ethnicity, and language; 3) Occupation; 4) Gender and sex; 5) Religion; 6) Education; 7) Socioeconomic status; 8) Social capital; 9) Personal characteristics associated with discrimination; 10) Features of relationships; and 11) Time-dependent relationships. For studies that reported PROGRESS-Plus factors, we extracted further details such as how these factors were defined and operationalized, and the distribution of study participants. Since this was a review, we did not recruit participants, and ethics approval was not required.
Results
Included studies
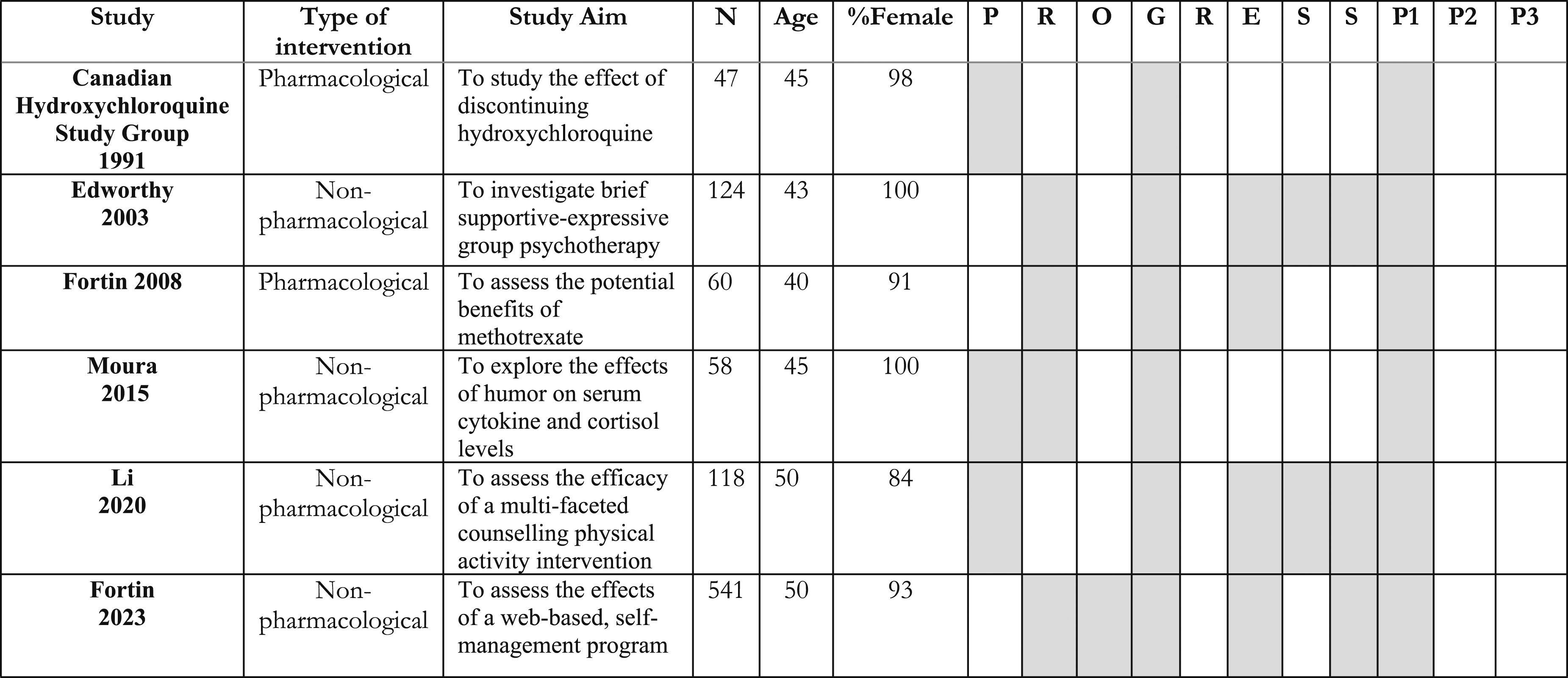
After screening 1901 unique records, 6 studies were included (Figure 1), with the earliest study being published in 1991, and the most recent in 2023. Of the included studies, 2 evaluated pharmacological interventions15,16 and 4 evaluated non-pharmacological interventions17–20 (Table 1). PRISMA. Characteristics of included studies and reporting of PROGRESS-Plus factors. Shaded cells denote factor reported in the study and blank cells denote factor not reported; PROGRESS-Plus Factors: 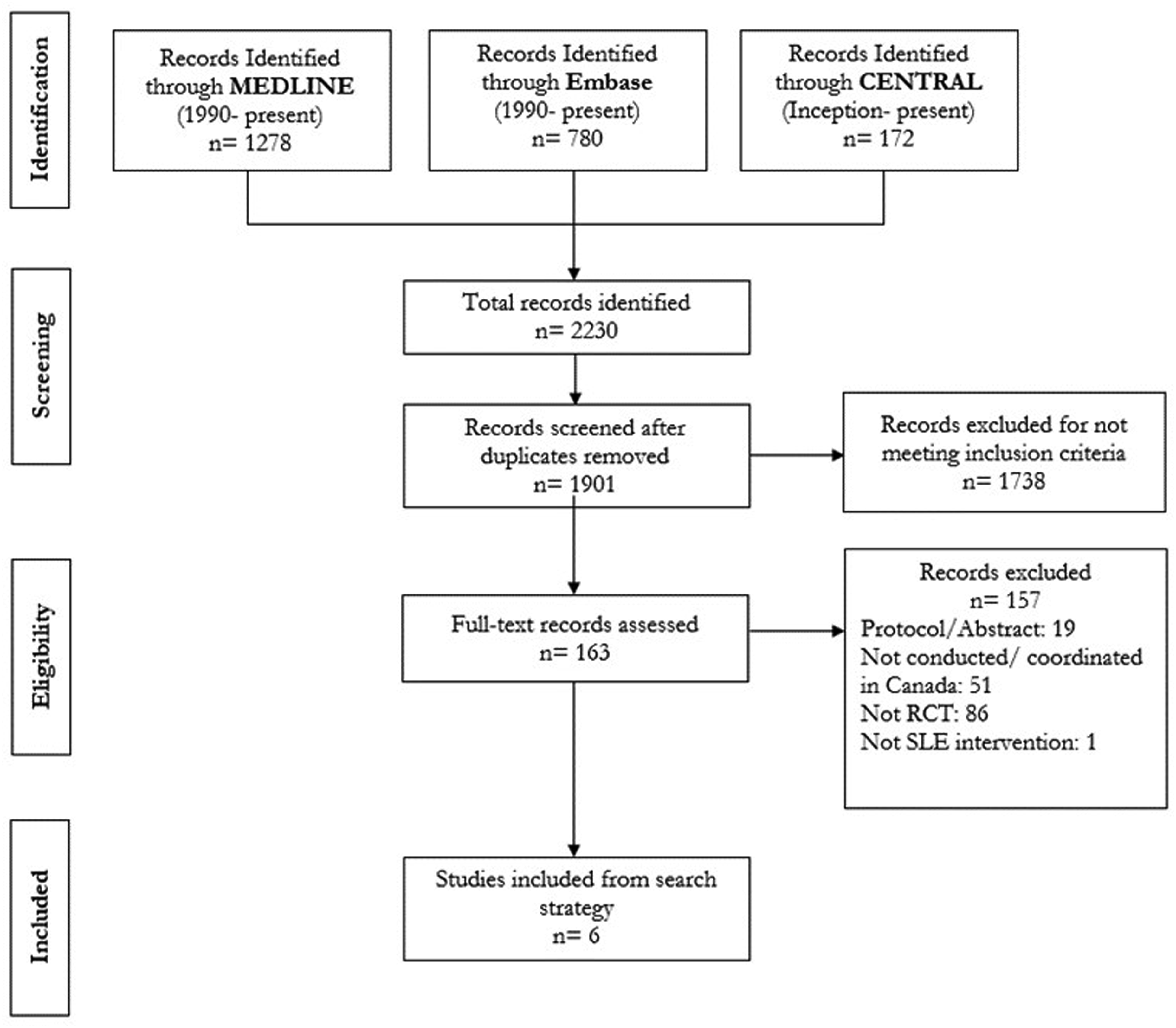
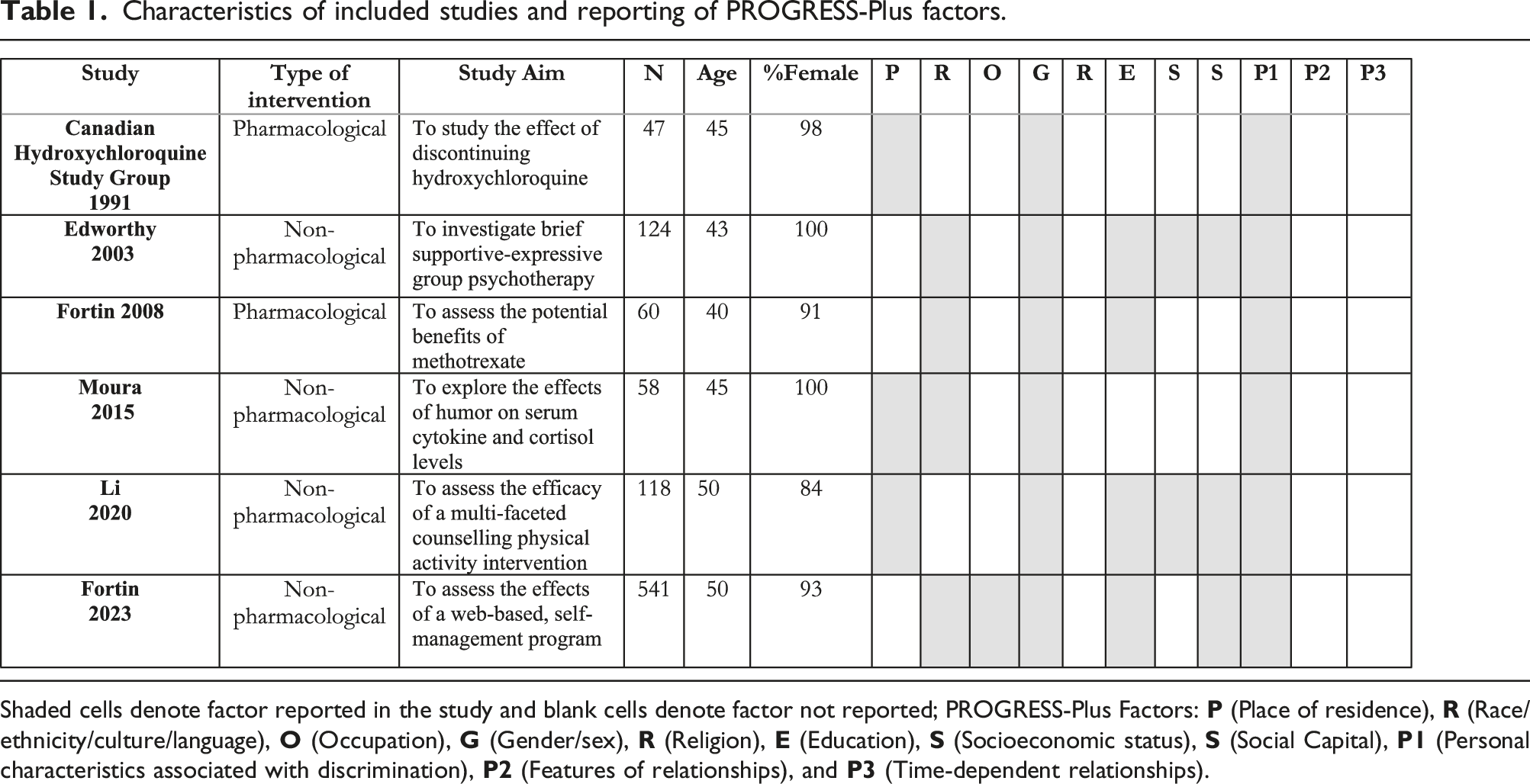
Characteristics of participants
All included studies reported sex and age (i.e. personal characteristics associated with discrimination). Participants in included RCTs were predominantly female (84% to 100% of participants in included studies). Reported mean ages of participants ranged from 40.2 (IQR 34.0–48.2) years to 52.3 (SD 14.1) years, which suggests individuals were largely middle-aged. Participants were predominantly White (72.4% to 84% of participants when reported).
Reporting of progress-plus factors
We summarize the reporting of PROGRESS-Plus factors in the identified studies here and in Figure 2, in the order of the framework. (8) (9) PROGRESS-Plus factors collected and reported in studies.
Discussion
We conducted a scoping review on intervention trials for people with SLE conducted in Canada to better understand how (or whether) determinants of health inequities are reported. Of 6 studies identified, all reported the sex and age distribution of the participants. The participants were generally middle-aged, female, and White. The next most reported determinants were place of residence, race, education, and social capital.
Our findings highlight three key issues. First, there is limited reporting of determinants of health inequities in RCTs of SLE in Canada to date. As aforementioned, sex and age were the only PROGRESS-Plus factors consistently reported across all 6 studies; other factors were either reported to a limited degree or not reported at all. This is problematic as SLE is a chronic disease, estimated to affect one in 2000 Canadians, 22 requiring significant resources (financial and healthcare supports) in its treatment. For example, one study conducted out of Alberta, Canada found that the mean direct cost incurred by an individual with SLE is about $8000 CAD per year. 23 This is about $2500 CAD more than mean cost estimates for an individual with rheumatoid arthritis ($5500 CAD) per year. 24 This cost is in part due to medications, and coverage varies based on individual private insurance and provincial coverage as Canada does not have a national Pharmacare plan. Coverage is important to consider because in settings where there is no universal coverage for much of the population, the impact on healthcare access is heightened. In a cohort study in California, over 25% of participants reported medication cost concerns, and these concerns were associated with worse patient-reported outcomes. 25 Treatment approaches in SLE are therefore significantly influenced by income level (which is linked to education and occupation), and these factors must be considered when evaluating interventions. Other relevant considerations are the amount of social support patients have, as chronic conditions present a significant amount of stress. A Canadian study found that females diagnosed with SLE before the age of 30 were less likely to be married/living common-law than expected based on general population rates, suggesting that sex and age can put patients more at risk of experiencing negative social effects (i.e. family relationships) of SLE. 26 Furthermore, several studies have shown that SLE disproportionally affects minorities, with studies in the US citing that Black patients have earlier onset and more severe SLE.27,28 In Canada, research has shown that First Nations females have twice the prevalence of SLE compared to non–First Nations females. 7 Due to this unequal distribution of SLE, it is therefore imperative that the diversity of the population is well represented in research cohorts to ensure that research outcomes are relevant to the typical population affected by these conditions. Without adequate reporting of these factors however, it is difficult to assure this.
The second issue is the lack of standardization in categorization. For example, when collecting race data, the reporting options differed in each study (e.g. White vs Caucasian); White refers to a race category while Caucasian refers to ethnicity. A distinction should be made in the reporting of the two, as an individual may be considered White racially but not Caucasian (or vice versa). This is an issue because without standardization it is challenging to compare and contrast different studies with respect to inclusion of different population groups. Having clearly defined race and/or ethnicity categories can help ensure that different racial and ethnic groups are well represented across research trials.
The final, and perhaps most significant, issue is that our findings demonstrate a lack of representation in clinical trials within Canada. As aforementioned, those who are middle-aged, female, and White are more likely to be represented. Given that research shows that minorities are disproportionately affected by SLE, this lack of representation is concerning. Issues with representation in SLE trials have also been recognized in other countries, such as the US, where there remain challenges enrolling participants from different populations.6,11 Sheikh et al. identified 5 key barriers to enrolment of diverse patients in SLE trials, including access, opportunity, health literacy, culture, and mistrust. 6 These barriers should be considered when trying to improve representation and enrolment of diverse participants for SLE trials in Canada. Furthermore, researchers have suggested that guidance, transparency, and consensus with regulatory agencies are necessary to ensure future SLE studies reflect the true population living with the disease. 29 A key example of this was the lack of racial and ethnic representation in belimumab clinical trials in the US, which led to mixed findings for Black patients, as there were insufficient numbers to conclude safety. As a result, the FDA initiated the Efficacy and Safety of Belimumab in Black Race Patients with SLE (EMBRACE) trial, where they were able to conclude that belimumab was safe for Black patients. 12 Ultimately, without adequate representation in clinical trials, we are unable to determine whether treatments are effective for different populations, and thus appropriate measures should be taken to improve representation in SLE trials within Canada.
Limitations
There are limitations associated with this scoping review. Studies were limited to RCTs as they are instrumental for identifying and evaluating effective therapies; however, this may have led to either underestimating or overestimating the true extent of health inequities in Canada, as we did not include other study designs. Further, RCTs may be limited in their capacity to measure determinants of inequity (e.g. have comprehensive questionnaires) and time for participants to complete such measures. Future studies to replicate our findings in reviews of observational studies and point-of-care trials to address determinants of equity would be beneficial. Finally, we used the PROGRESS-Plus framework to guide extraction of reporting of determinants of inequities; however, this framework may not be exhaustive, as other factors may be relevant that may not be captured within PROGRESS-Plus (e.g. social exclusion/inclusion, income distribution, neighbourhood safety, and infrastructure). However, PROGRESS-Plus is a leading framework for reporting health inequities and allowed us to formally consider how Canadian RCTs for SLE collect and report these factors.
Conclusion
Determinants of inequities are not commonly reported or measured in Canadian RCTs for SLE interventions, which may present significant problems in the translation of findings to improved and personalized care. More importantly, where we could determine based on the available reporting, there remains a lack of representation in participants of SLE trials. Ongoing and future SLE research in Canada should carefully reconsider engagement practices, design and reporting to address health inequities introduced by standard practices.
Supplemental Material
Supplemental Material - Reporting of determinants of health inequities and participant characteristics in randomized controlled trials of systemic lupus erythematosus in Canada: A scoping review
Supplemental Material for Reporting of determinants of health inequities and participant characteristics in randomized controlled trials of systemic lupus erythematosus in Canada: A scoping review by Megan Thomas, Vanay Verma, Niloofar Gheshlagi, John Esdaile, Antonio Avina-Zubieta, Cheryl Barnabe, Mark Harrison, and Mary A De Vera in Lupus
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a Planning Grant from the Canadian Institutes of Health Research, ‘Towards addressing equity considerations in research and care of patients with inflammatory arthritis (ACCESS)’ (Funding reference number: #PCS-183488). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.