Abstract
Background
Lupus nephritis (LN) is associated with poor outcomes and a significant risk of progression to end-stage renal disease (ESRD). Some patients with resistant LN do not respond adequately to current treatment options and need alternative strategies or therapies.
Objective
The objective is to evaluate the efficacy and safety of rituximab as a re-induction therapy (Re-RTX) followed by maintenance therapy for patients with resistant LN.
Methods
Twenty-four patients with resistant LN (failed initial induction therapy or severe relapse after remission) were analyzed. Re-RTX was co-administered with other immunosuppressants. The primary KDIGO criteria outcomes included renal response (complete and partial), disease progression, relapses, and infections.
Results
The median age was 28 years (IQR 24.5–42), and the female-to-male ratio was 11:1. All patients had active LN, and 91.3% had proliferative LN. Baseline creatinine was 1.075 mg% (IQR 0.7–1.38), and mean urine protein-to-creatinine ratio (UPCR) was 4.9 (IQR 2.8–6.65). Of the patients receiving RTX as re-induction therapy, 66.6% (16/24) had failed initial induction therapy with other immunosuppressants, whereas 33.3% (8/24) had severe relapse during maintenance therapy.
Re-RTX had a favorable renal response at 6 months, with 91.7% of the patients responding (20.8% complete response and 70.8% partial response). At 12 months, 58.3% of the patients maintained a renal response (25% complete response and 33.3% partial response). Approximately one-third of patients relapsed within a year.
Fourteen patients (58.3%) continued RTX maintenance therapy with two different treatment regimens. At 6 months, Regimen-1 (500 mg every 6 months) resulted in a partial response in 43% (3/7) and relapse in 57% (4/7) of patients. Regimen 2 (1 g dose per year) achieved a complete response in 28.5% (2/7) and a partial response in 71.5% (5/7) with no relapses at 6 months.
At a median follow-up of 29 months, adverse renal outcomes were observed in 29.16% of the patients with progression to advanced chronic kidney disease (CKD) or end-stage renal disease (ESRD). The overall use of Re-RTX was considered safe, with a reported infection prevalence of 16%, which is comparable to the existing data.
Conclusion
Re-RTX demonstrated efficacy and safety as an induction therapy for resistant LN. However, the response waned after 1 year, underscoring the need for optimized maintenance therapy.
Introduction
Systemic lupus erythematosus (SLE) is a complex multisystem autoimmune disease with various genetic, hormonal, and environmental factors that influence the course and severity of the disease.
Renal involvement in SLE is common and, despite adequate therapy, is often associated with unsatisfactory outcomes, with 10%–30% of patients progressing to end-stage renal disease (ESRD) over 15 years 1 Additionally, even with treatment, the outcome of lupus nephritis (LN) varies significantly, with a relapsing-remitting course being the most common. This results in accrual damage and a poor long-term prognosis. The reported incidence of flares in LN varies from 27% to 66%. 2 Numerous studies have found that nephritic flares significantly negatively predict long-term renal outcomes. 3
Despite various therapeutic choices for these patients, a distinct subgroup of patients with LN demonstrate an inadequate response to current treatment options or recurrent relapses. The initial response to treatment determines the long-term outcome in patients with LN nephritis; thus, patients with resistant LN have a poor prognosis. 4 These patients with “resistant LN” require alternate or more effective therapies to manage their LN and improve their quality of life. Therefore, there is a need to develop novel therapies for this subset of patients.
Over the years, randomized controlled trials on effective B cell-directed immunosuppressive drug therapy have yielded mixed results in LN. Although drugs such as belimumab are effective as add-on therapy in LN, 5 anti-CD20 directed antibody-like rituximab (RTX) failed to demonstrate statistically significant results compared to placebo.6,7 However, multiple open-label trials and registry studies have shown the efficacy of RTX in LN,8–13 especially in patients with resistant LN.14–16 Furthermore, a meta-analysis on the efficacy of RTX in treating LN revealed significant variations in response to treatment between different racial groups. Complete remission rates (CRRs) for Caucasian, East Asian, and Hispanic patients were found to be 77%, 38%, and 28%, respectively. 17 These findings underscore the importance of studying the efficacy of RTX and other treatments for LN in diverse populations to address potential racial disparities. 18 In this context, we describe our experience with the use of Re-RTX along with other immunosuppressants in South Asian ethnic patients with resistant LN.
Aims and objectives
To evaluate the efficacy and safety of rituximab as a re-induction therapy (Re-RTX) followed by maintenance therapy for patients with resistant LN.
Materials and methods
Study design
This single-center retrospective observational study was conducted at an LN clinic at a tertiary care hospital in Mumbai. The long-term renal outcomes of this cohort of patients have been previously published. 19 All patients attending this clinic from 2013 to 2022 were screened for resistant LN.
Patients
Resistant LN patients with complete records with at least 1 year of follow-up and treated with RTX were included in the analysis.
Definition of resistant LN
Failed induction regime
Patients who did not achieve at least a partial response (PR) or experienced worsening of proteinuria after the initial induction regimen (as per KDIGO guidelines) despite receiving adequate therapy for 6–12 months. The dosing of drugs was according to KDIGO criteria.
Failed maintenance regime
Patients with LN who achieved a complete response (CR) or PR to the initial induction regimen (consisting of six cycles of CYC and 2–3 g/day of MMF) for at least 6 months but experienced severe relapse (defined as an increase in creatinine of >1.5 mg/dL and/or an absolute increase in the urinary protein-to-creatinine ratio >5000 mg/g) despite having good maintenance doses of MMF (1–2 g/day), AZA (50–100 mg/day), or CYC (50–100 mg/day) in addition to prednisolone (5–10 mg/day).
Exclusion criteria
• Patients already on dialysis or with stage 5 chronic kidney disease (CKD) or ESRD. • Patients who received RTX therapy for non-renal disease activity. • Less than 1 year of follow-up after RTX.
Data collection
The following data were collected: sex, age of LN onset, serum creatinine, C3, urine protein-creatinine or albumin-creatinine ratios, 24-h urinary protein (in selected cases), and renal histology. All kidney biopsies were reported by an experienced nephropathologist according to the International Society of Nephrology/Renal Pathology Society (ISN/RPS) 2003 classification.
General treatment principles in all Re-rituximab LN patients
For induction, all patients were treated with either intravenous pulse methylprednisolone for 3 days, followed by an oral prednisolone taper or increased oral prednisolone (1 mg/kg), followed by tapering. In most patients, the maintenance treatment included low-dose steroids, MMF (1–2 g), or AZA (1–2 mg/kg). Hydroxychloroquine was administered in all cases if there were no contraindications. Hypertension was treated to achieve a target blood pressure of 140/90.
Re-rituximab induction therapy for resistant LN
All patients received intravenous RTX as an induction regimen, with two doses of 1 g each 2 weeks apart, or 500 mg every week for 4 weeks.
Re-rituximab maintenance therapy for resistant LN
Owing to the lack of specific treatment guidelines, the selection of patients, RTX dosage, and duration between RTX infusions for maintenance were variable. In most cases, RTX was added to a conventional agent for maintenance.
Patients who showed a good response to Re-RTX at 6 months after failing other induction regimens and those who relapsed after 1 year of induction therapy after a good response at 6 months were administered RTX as a maintenance regimen.
Two maintenance regimes were used. - 6 monthly RTX – dose 500 mg. - 12 monthly RTX – dose 1 gm.
Outcome measures
CR, PR, progression to CKD and ESRD, mortality, relapses, and infections were studied.
Renal response—defined as per KDIGO 2012 criteria 20
• CR: Return of serum creatinine to the previous baseline, plus a decrease in the urine protein/creatinine ratio to less than 500 mg/g (UPCR <0.5). • PR: Stabilization (±25%) or improvement of serum creatinine, but not normal, plus a ≥50% decrease in the urine protein/creatinine ratio (UPCR). If there was nephrotic range proteinuria (UPCR ≥3000 mg/g), improvement requires a 50% reduction in UPCR and a UPCR <3000 mg/g. • No response: Patient failing to achieve partial or complete remission within 6 to 12 months of therapy • Chronic kidney disease (CKD): Persistent doubling of serum creatinine level lasting at least 6 months with a creatinine of >1.5 mg%. • End-stage renal disease (ESRD): The need for dialysis therapy for greater than 3 months/renal transplant • Renal flares were defined as mild, moderate, and severe.
Statistics
For continuous variables, we used the median with the interquartile range for most variables rather than the mean and standard deviation to account for the reduced sample size, eliminate the impact of outliers, and provide a better sense of the data distribution in our study. The results for categorical variables are presented as percentages and proportions.
Results
Twenty-four patients with resistant LN were treated with Re-RTX. The patient selection method is illustrated in Figure 1. Showing the method of patient selection for resistant lupus nephritis.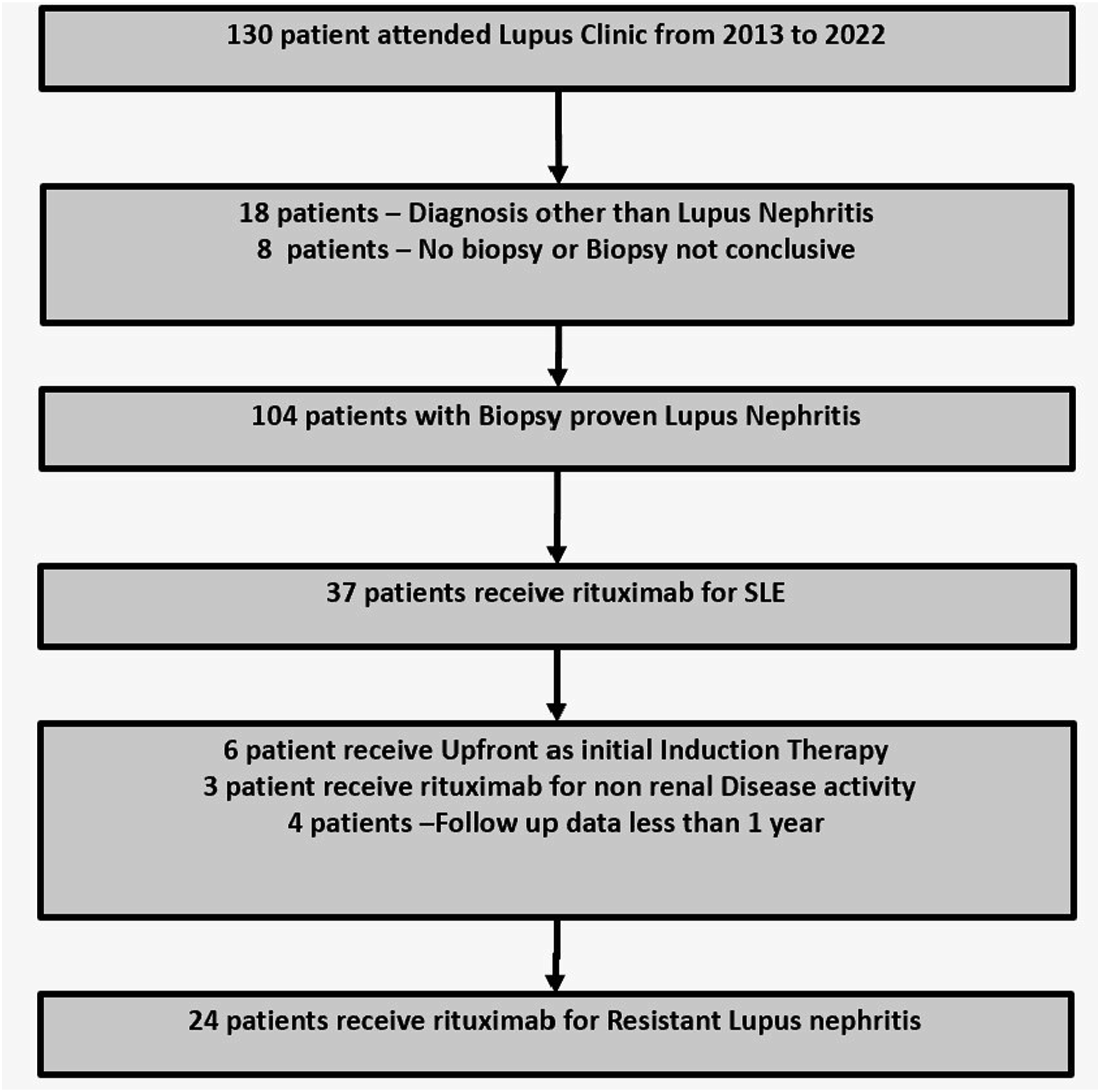
Baseline characteristics of resistant LN patients treated with rituximab.
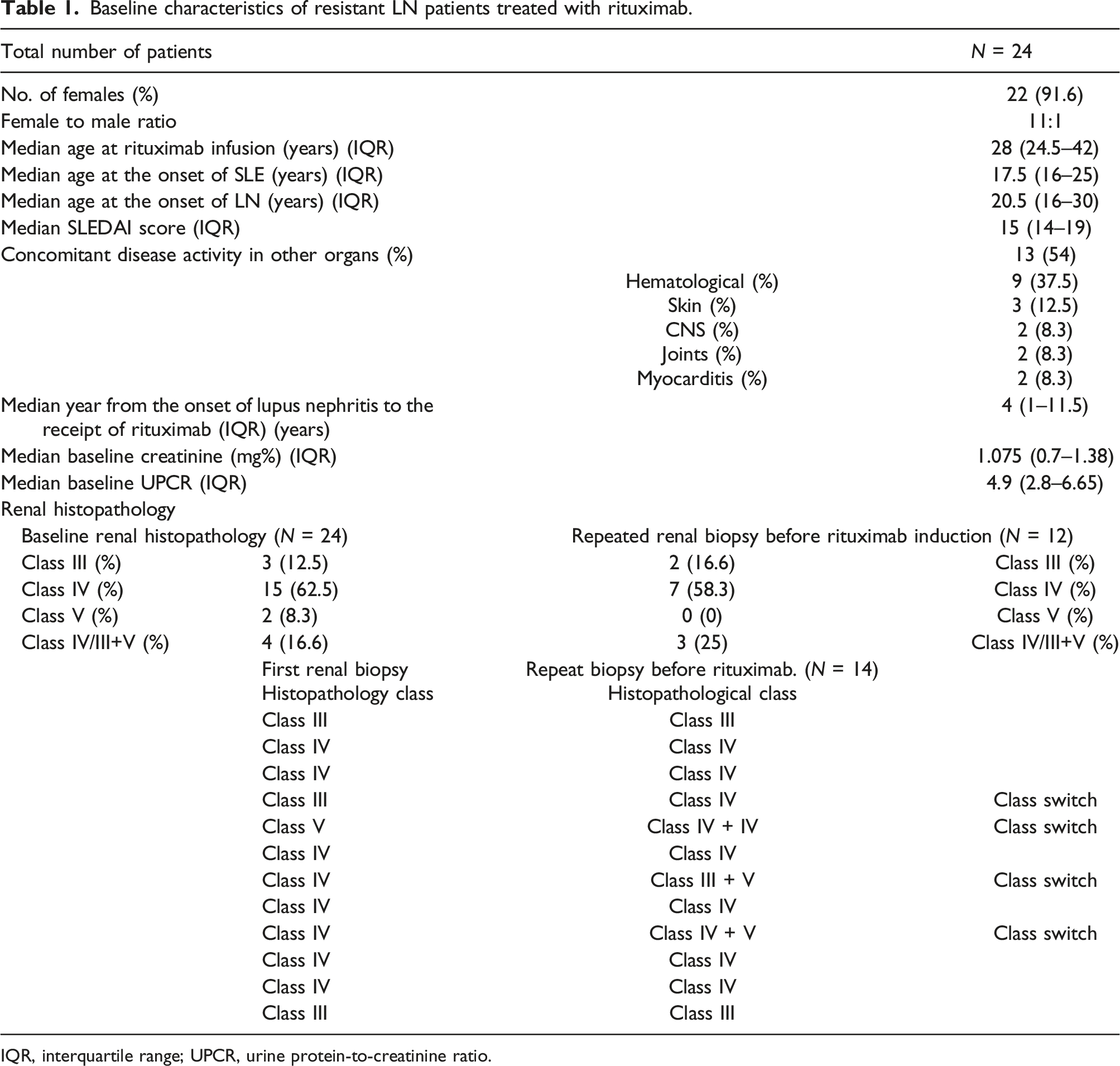
IQR, interquartile range; UPCR, urine protein-to-creatinine ratio.
All patients with resistant LN had high disease activity at inclusion, with a median SLEDAI score of 15 (IQR: 14–19). Thirteen (54%) patients had disease activity in other organs, with hematological involvement being the most common (37.5%), followed by the skin (12.5%), CNS (8.3%), joints (8.3%), and myocarditis (8.3%).
Repeat renal biopsy performed in 12 (50%) patients showed the following WHO classes: 7/12 (58.3%) WHO Class IV, 2/24 (16.6%) Class III, and 3/12 (25%) had Class (IV/III) + V. Four patients underwent class switching, with three of them from class IV to Class (IV/III) + V. The median baseline creatinine was 1.075 mg% (IQR: 0.7–1.38), and the median baseline urine protein to creatinine ratio (UPCR) was 4.9 (IQR: 2.8–6.65).
Indication for rituximab re-induction therapy
Sixteen out of 24 patients who received Re-RTX had failed initial induction therapy, while 8 had a severe relapse on maintenance therapy (Supplemental S1). Re-RTX was used after a single drug initial induction regimen failed in 13 patients, and in two patients, it was used after two sequential initial induction regimes failed. All patients received concomitant steroid therapy, with 8 patients receiving three IV pulses of 500 mg methylprednisolone followed by a 40 mg/day prednisolone taper, and 8 patients received a prednisolone taper of 30 mg/day (Supplemental S2). In all patients, the steroid dose was tapered to achieve maintenance doses of 5–10 mg/day after 6–12 weeks of induction therapy.
Short-term response to induction therapy (table)
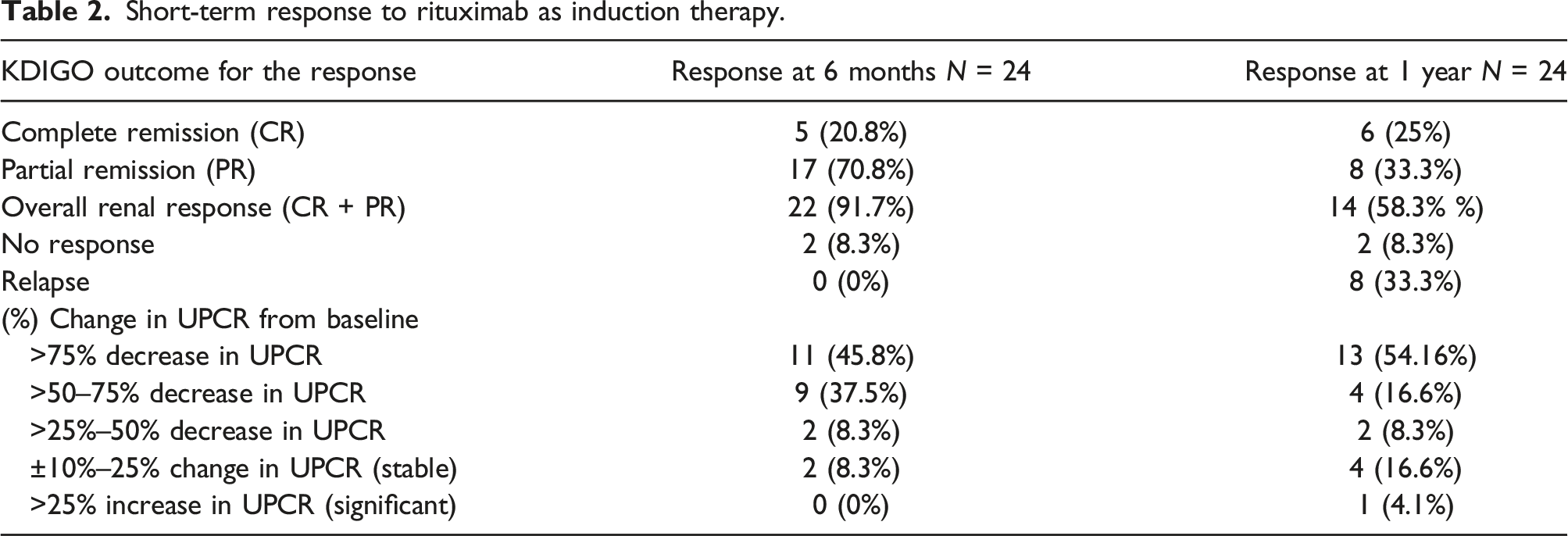
At 6 months, 22/24 (91.7%) patients responded to Re-RTX therapy, 5/24 (20.8%) had CR, 17/24 (70.8%) had PR, and 2/24 (8.3%) had no response. Similarly, at 6 months, 22/24 (91.7%) patients showed an improvement in proteinuria, whereas it was stable in 2/24 (8.3%) patients.
At 12 months, 14 of 24 patients (58.3%) responded to Re-RTX therapy, with 6/24 (25%) achieving CR, 8/24 (33.3%) achieving PR, and 2/24 (8.3%) showing no response. According to the KDIGO criteria, 8/24 (33.3%) patients relapsed after 1 year. At 12 months, 19/24 (79%) patients showed a significant improvement in proteinuria, while it was stable in 4/24 (16.6%) patients and increased significantly in the other 1/24 (4%) patients. Seven patients (29.1%) received an additional dose of RTX 500 mg at 6 months.
Short-term response to rituximab as induction therapy.
Maintenance therapy
After completing 6 months of Re-RTX induction therapy, all patients were assigned to receive maintenance immunosuppression therapy. Of the 24 patients, 14 (58.3%) continued RTX as maintenance therapy, while 10 (41.6%) received conventional maintenance therapy using medications such as MMF, CYA, and AZA (Supplemental S3). The decision to use maintenance RTX therapy and its dosage and duration varied owing to the absence of definitive guidelines.
Response to rituximab as maintenance after repeat induction for resistant LN.

Regimen 2 involved administering RTX at a higher 1 gm dose every year. Before initiating maintenance treatment, one patient was in PR, one patient was in CR, and five patients had already experienced relapse 12 months after the induction. Consequently, a higher dose of RTX was used for maintenance therapy in these seven patients. After 6 months of maintenance RTX, of 2/7 patients (28.5%) achieved CR, while five patients (71.5%) showed PR with no reported relapses. At 1-year follow-up from RTX maintenance, data from 5 patients showed that 2 (40%) had a CR, 2 achieved PR (40%), and only 1 (20%) patient experienced a relapse (Supplemental S4).
Overall outcome
After a median follow-up of 29 months (IQR: 18–54 months), 12/24 (50%) patients had a CR and 5/24 (20.8%) had a PR. Adverse renal outcomes were observed in of 7/24 (29.16%) patients who progressed to advanced CKD or ESRD. Three patients developed CKD stage 3–4; four progressed to ESRD, of which two patients remained on hemodialysis, and two underwent living donor renal transplantation.
Adverse events
One patient developed active pulmonary tuberculosis, two had herpes zoster, and one had a gluteal abscess and cytopenia during the study. Posterior reversible encephalopathy syndrome (PRES) was observed in two patients who received RTX: one during the first infusion and the other during the third infusion. In addition, two (8%) patients had infusion-related allergic reactions, while none had hypogammaglobulinemia.
Discussion
This study focused on Re-RTX therapy in patients with resistant LN, a subset that is often challenging to treat. Notably, the cohort predominantly comprised females with active proliferative LN with high disease activity and nephrotic-range protein urea but relatively well-preserved renal function, emphasizing the importance of selecting the right subset of patients for this therapy.
The study demonstrated a favorable renal response to Re-RTX in resistant LN patients, with 91.7% responding at 6 months, 20.8% achieving CR, and 70.8% achieving PR. However, responses declined over time, with approximately one-third of the patients experiencing relapses at 12 months. This decline in the response emphasizes the need for optimized maintenance therapy.
In a meta-analysis, RTX showed good therapeutic efficacy in patients with resistant LN, with 73% showing a global renal response rate, 51% achieving CR, and 34% achieving PR, however. 21 Our study had lower CR rates, possibly due to variations in refractory LN definitions, different CR and PR criteria, varying doses of concomitant medications, and the aggressive nature of SLE in Indian patients. 22 However, consistent results were observed in large registry studies from the United Kingdom, 10 Italy 23 and multi-centric studies from East Asians, 24 showing an overall response rate of 50% to 65%, with a higher prevalence of PR over CR, similar to our findings. A comprehensive comparative review of the relevant literature is presented in Supplemental S5.12,23,25–34
Optimizing maintenance therapy for LN is crucial for preventing relapse and organ damage. However, lacking specific guidelines, decisions on continuing RTX as maintenance therapy were individualized in our study, used in 58% of patients often as an add-on after successful induction. Two regimens were tested: Regimen 1 (RTX 500 mg every 6 months) had a 57% relapse rate at 6 months, whereas Regimen 2 (RTX 1 g yearly) showed no relapse. Although promising, determining optimal dosing intervals is vital when considering B cell repopulation dynamics. Notably, the rarity of utilizing RTX for maintenance suggests a potential area for further research.
The overall use of RTX was safe. Large multi-centric studies and registry studies have reported the prevalence of infection to be between 10 and 16%. We reported the prevalence of infection in 16% of patients, which is comparable to the reported data. There were a few manageable infusion-related allergic reactions. Additionally, we noted PRES in 2 patients post-RTX, a rare occurrence in SLE. Despite potential confounding factors, similar cases have been reported by other authors.10,12
Our study has several limitations. First, the sample size was relatively small, which may limit the generalizability of the findings. Second, the retrospective nature of the study design and the absence of a control group restrict the ability to establish definitive conclusions regarding the comparative effectiveness of RTX compared to other treatments. Furthermore, our study did not measure serology for a response, including CD20 positive B cell count and anti-dsDNA levels, which could have provided further insights into treatment responses.
Notably, the strengths of the study included clear criteria for resistant LN, adherence to standard definitions, and reduced corticosteroid confounding. Furthermore, our study was done in a pragmatic setting, addressing practical issues in LN management, ensuring racial homogeneity, and reducing biases associated with variable LN management by nephrologists and rheumatologists. 35
In conclusion, our study demonstrated the effectiveness and safety of Re-RTX as an induction and maintenance therapy for patients with resistant LN. The renal response rates and proteinuria improvement observed in our study were modest, but acceptable for resistant LN. However, the response tended to decline after 1 year, highlighting the need for optimum maintenance therapy, which may include low-dose RTX, the dosing regimen of which may need further refinement. Further research, including larger prospective studies and RCTs, is needed to establish RTX's long-term efficacy and safety of RTX in the treatment of resistant LN, including optimization of the RTX dosing schedule for maintenance treatment.
Supplemental Material
Supplemental Material - Rituximab as add-on therapy in patients with resistant lupus nephritis who have failed induction or maintenance therapy with other agents: A real-world experience from a single center in Mumbai
Supplemental Material for Rituximab as add-on therapy in patients with resistant lupus nephritis who have failed induction or maintenance therapy with other agents: A real-world experience from a single center in Mumbai by Sandeep Yadav, C Balakrishnan, Gurmeet Mangat and Jatin Kothari in Lupus.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
