Abstract
Objective
During the COVID-19 pandemic, many research studies were adapted, including our longitudinal study examining cognitive impairment (CI) in systemic lupus erythematosus (SLE). Cognitive testing was switched from in-person to virtual. This analysis aimed to determine if the administration method (in-person vs. virtual) of the ACR-neuropsychological battery (ACR-NB) affected participant cognitive performance and classification.
Methods
Data from our multi-visit, SLE CI study included demographic, clinical, and psychiatric characteristics, and the modified ACR-NB. Three analyses were undertaken for cognitive performance: (1) all visits, (2) non-CI group visits only and (3) intra-individual comparisons. A retrospective preferences questionnaire was given to participants who completed the ACR-NB both in-person and virtually.
Results
We analysed 328 SLE participants who had 801 visits (696 in-person and 105 virtual). Demographic, clinical, and psychiatric characteristics were comparable except for ethnicity, anxiety and disease-related damage. Across all three comparisons, six tests were consistently statistically significantly different. CI classification changed in 11/71 (15%) participants. 45% of participants preferred the virtual administration method and 33% preferred in-person.
Conclusions
Of the 19 tests in the ACR-NB, we identified one or more problems with eight (42%) tests when moving from in-person to virtual administration. As the use of virtual cognitive testing will likely increase, these issues need to be addressed – potentially by validating a virtual version of the ACR-NB. Until then, caution must be taken when directly comparing virtual to in-person test results. If future studies use a mixed administration approach, this should be accounted for during analysis.
Introduction
One of the effects of the COVID-19 pandemic was the suspension of many health research projects as face-to-face contact needed to be limited. As such, researchers tried to adapt their studies to use a virtual environment where possible. 1 Virtually testing cognitive function is possible, and prior to the pandemic, there was already a shift to online assessment, especially using computerised cognitive tests. 2
CANTAB® and ANAM are two established systems that provide validated normalised computerised cognitive batteries. 3 Administration of these systems requires minimal training, and reporting is much easier as it automatically calculated. CANTAB® has an established online version of their tasks 4 and ANAM has recently released an online version. CANTAB® studies have found many of the tests to be comparable in both in-person and online versions. However, caution must be used when assessing response time measures. These can be affected by both internet speed and hardware use, for example, mouse versus touch screen. 5
The use of ANAM and CANTAB® to test cognitive impairment (CI) in systemic lupus erythematosus (SLE) is increasing but the American College of Rheumatology neuropsychological battery (ACR-NB) of tests remains the gold standard. 6 This battery is primarily a face-to-face paper assessment. It includes 19 tests examining six different cognitive domains. Understanding how performance may be affected when this non-computerised assessment is administered in a virtual manner is currently unknown.
Prior to the COVID-19 pandemic, we had started a longitudinal project examining CI in SLE, with participant data collected at 0, 6, 12 and 24 months. Our protocol included the ACR-neuropsychological battery (ACR-NB) and ANAM, and all study visits were conducted face-to-face in-person. The study was suspended at the start of the pandemic in 2020 but parts were reopened including the use of an adapted version of the ACR-NB, which allowed virtual administration to start in February 2021.
The purpose of this study was to assess the comparability of the ACR-NB when administered in-person versus virtually. In addition, we explored which administration method participants preferred.
Methods
Data from participants enrolled in our longitudinal CI in SLE study was used. This data included demographic, clinical, and psychiatric characteristics, and the modified American College of Rheumatology neuropsychological battery (ACR-NB), full details are published elsewhere.3,7 Participants in this study have up to four research visits at 0, 6, 12 and 24 months. Inclusion criteria required all participants to meet 2019 EULAR/ACR classification criteria for SLE, 8 to be aged between 18 and 65 years old and to have an adequate level of English to enable completion of the cognitive tasks. Consecutive participants were approached from the Toronto Lupus Clinic at the University Health Network (UHN) Toronto Western Hospital. Patients provided written informed consent in accordance with the Helsinki Declaration and the study was reviewed and approved by the UHN Research Ethics Board (CAPCR ID: 15-9582).
In-person data was collected between July 2016 and 16th March 2020. The study was then paused due to COVID-19 restrictions. Web-based virtual cognitive assessments began 8th February 2021 and are still ongoing.
Cognitive tests
In-person
Cognitive tests administered and the differences between the in-person and virtual tests.

BOLD indicates significant changes to tests administered. COWAT: controlled oral word association test, RCFT: Rey complex figure test, HVLT-R: Hopkins verbal learning test-revised, WAIS: Wechsler adult intelligence scale, SDMT: symbol digit modalities test.
Virtual
The administration of the ACR-NB in a virtual environment was conducted in the same way as the in-person administration except that it was done through a video call. This also meant changes were required to five of the tests; two had to be completely removed and three were changed from written to verbal tests, see Table 1.
All cognitive test scores were converted into z-scores.
After completion of the battery, participants’ level of cognitive function was determined. Participants were categorised as cognitively impaired if they met the following criteria (CI algorithm):
Participants must have impaired performance in two or more domains. Domains 1–4 were considered impaired if one or more tests within the domains had z-score ≤ −1.5. Domains 5 and required two or more tests to have z-score ≤ −1.5. 11
Participants were further categorised into one of three groups based on their cognitive functioning over time. These groups were defined as follows: 1. No CI, participant did not meet the definition for CI at any study visit. 2. Fluctuating CI, participant was impaired at some visits and not at others. 3. Persistent CI, participant was impaired at all study visits.
An additional questionnaire was added retrospectively to ask participants about their experiences of in-person versus virtual visits. This questionnaire was given to all participants that undertook both virtual and in-person cognitive assessments (Supplementary Figure 1).
Analysis
All data available was assessed for completeness. Visits with ≥5 cognitive test results or ≥2 cognitive domains missing were removed from the analysis. Data was split into two groups, in-person versus virtual visits. Characteristics were compared between in-person and virtual visit data using Mann–Whitney U or chi-square.
Three different analyses were conducted on the cognitive data to examine differences between in-person and virtual assessment performance, using Mann–Whitney U or Wilcoxon Signed Ranks test: 1. Analysis of all available visit data. 2. Analysis of visit data from participants who remained non-cognitively impaired over all visits. 3. Intra-individual longitudinal analysis of those who had both an in-person and virtual visit at different time points. An additional delta analysis was also performed. This examined change in performance from two in-person visits compared to change in performance from an in-person visit and virtual visit.
The purpose of the three analyses enabled us to control for potentially confounding factors. Analysis 1 provided an overview of all data available. Analysis 2 allowed us to control for participants who may have naturally fluctuating CI levels over time. Analysis 3 enabled us to control for other factors that may affect cognition, such as medication, depression and disease damage, but may remain more stable within participants.
Results
Results from 816 visits were available. After removing visits with missing data, 801 visits were included. 328 participants contributed to these visits. In-person visits accounted for 696 visits and virtual 105 visits. Baseline characteristics, split by administration method can be seen in Supplementary Table 1. No significant differences were seen in visit characteristics except for ethnicity, disease damage and anxiety, where higher level of damage was noted amongst the in-person visits and higher level of anxiety amongst the virtual visits. In terms of ethnicity, there were more Black and Caucasian in-person visits compared to more Asian and Other ethnicities for the virtual visits.
Across both visit types, disease activity (as assessed by the SLEDAI) was seen in the following systems: central nervous n = 15 (1.8%), vascular n = 14 (1.7%), musculoskeletal n = 54 (6.7%), renal n = 96 (12%), dermatologic n = 103 (13%), serosal n = 10 (1.2%), immunologic n = 467 (58%), constitutional n = 10 (1.2%) and haematologic n = 63 (7.8%). Also, across both visits types, 300 visits had patients with antiphospholipid syndrome and ACR criteria categories were split as follows: malar rash n = 543 (68%), discoid rash n = 111 (14%), photosensitivity n = 467 (58%), oral ulcers n = 432 (54%), arthritis n = 658 (82%), serositis n = 263 (33%), renal disorder n = 345 (43%), neurologic disorder n = 75 (9%), haematologic disorder n = 707 (88%), immunologic disorder n = 726 (91%) and antinuclear antibody positive n = 795 (99%).
All visits
Comparisons between in-person and virtual visits cognitive test results for all visits.
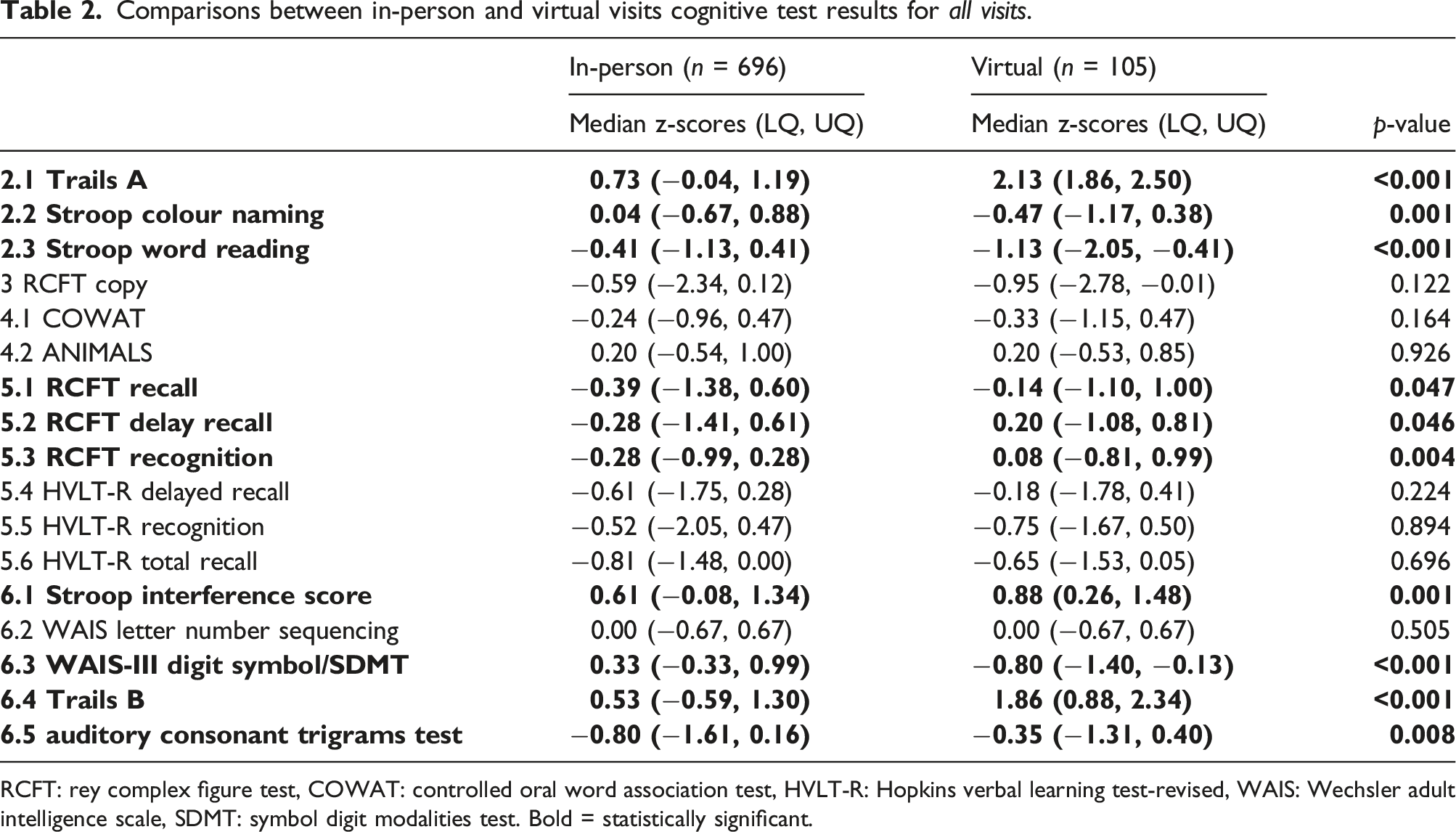
RCFT: rey complex figure test, COWAT: controlled oral word association test, HVLT-R: Hopkins verbal learning test-revised, WAIS: Wechsler adult intelligence scale, SDMT: symbol digit modalities test. Bold = statistically significant.
Visits of those with no CI
A total of 394 visits were classified as not cognitively impaired, comprised of 346 in-person visits and 48 virtual visits.
Comparisons between in-person and virtual visits cognitive test results for non-CI visits only.
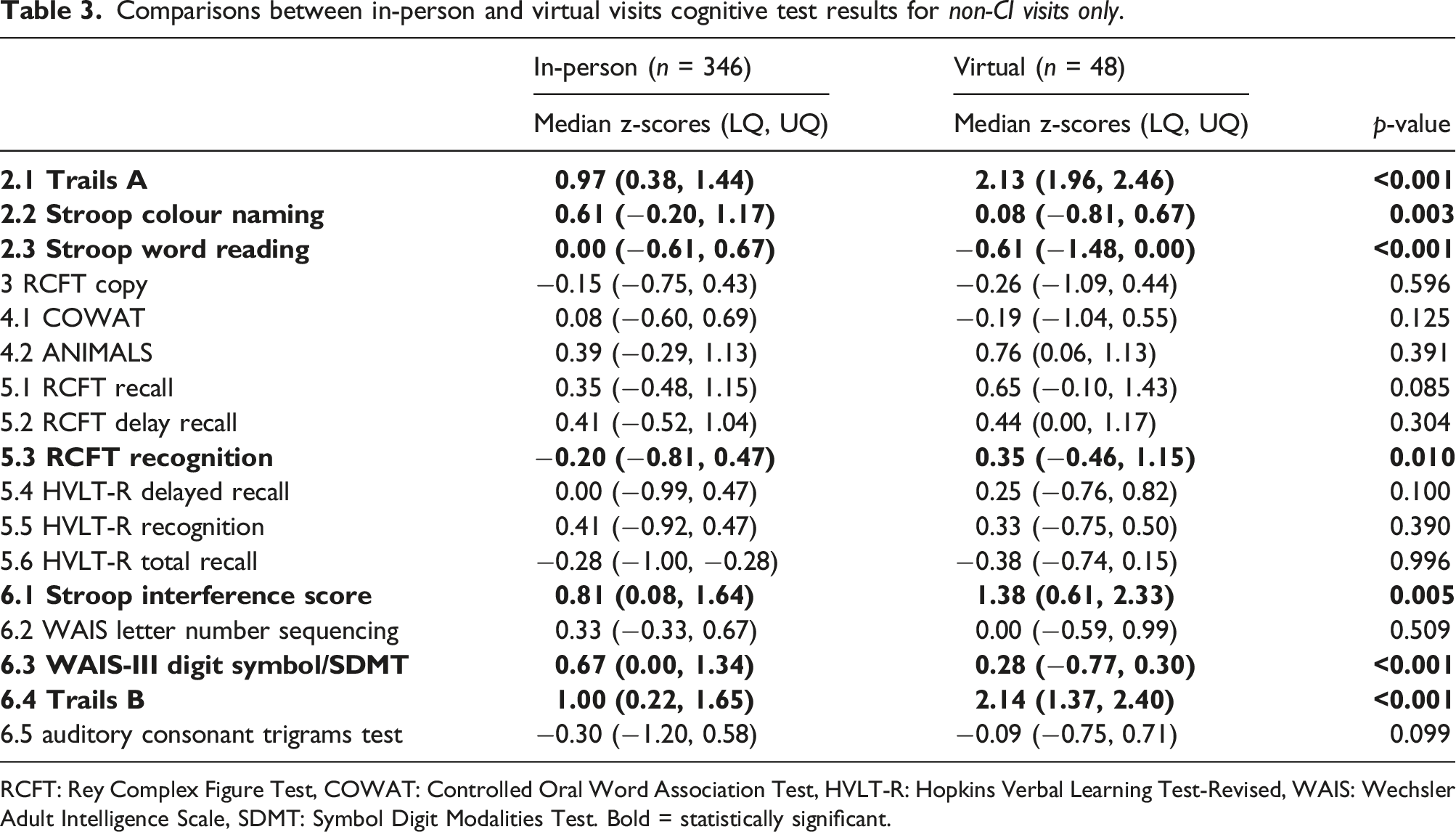
RCFT: Rey Complex Figure Test, COWAT: Controlled Oral Word Association Test, HVLT-R: Hopkins Verbal Learning Test-Revised, WAIS: Wechsler Adult Intelligence Scale, SDMT: Symbol Digit Modalities Test. Bold = statistically significant.
Intra-individual longitudinal comparisons
Intra-individual participant comparisons between in-person and virtual visits cognitive test results.
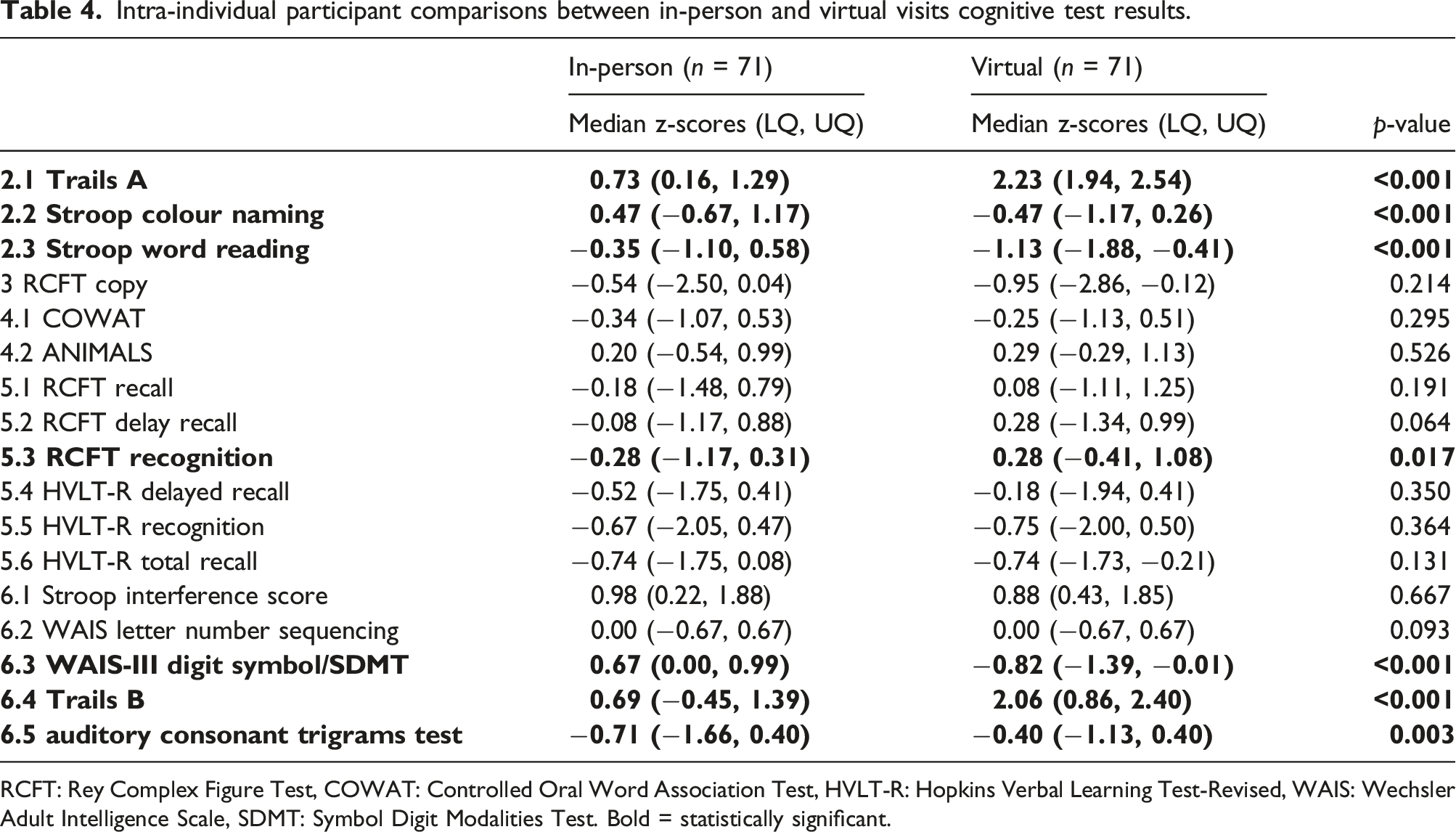
RCFT: Rey Complex Figure Test, COWAT: Controlled Oral Word Association Test, HVLT-R: Hopkins Verbal Learning Test-Revised, WAIS: Wechsler Adult Intelligence Scale, SDMT: Symbol Digit Modalities Test. Bold = statistically significant.
A summary of which cognitive tasks were significantly different for the three analyses.

Changes in CI status
The intra-individual comparisons found that performance on seven of the cognitive tests were significantly different between virtual and in-person assessments. Further examination of how these differences may have affected participant CI status stratification, based on our CI algorithm, found that 11/71 (15%) participants had a change in CI status. Three participants were moved from CI into the not cognitively impaired group and eight from non-impaired into the impaired group. Figure 1 illustrates which participants’ test scores cross the −1.5 CI cut-off. If participant in-person and virtual scores are separated by the cut-off, this may have changed the participants CI status. Spider diagrams of individual participant z-scores for the seven cognitive tests affected by administration method in the intra-individual analysis. These diagrams show results from the 71 participants who had both an in-person and virtual visit. Where only one result (in-person or virtual) crosses the red line this may indicate a change in CI status for that individual.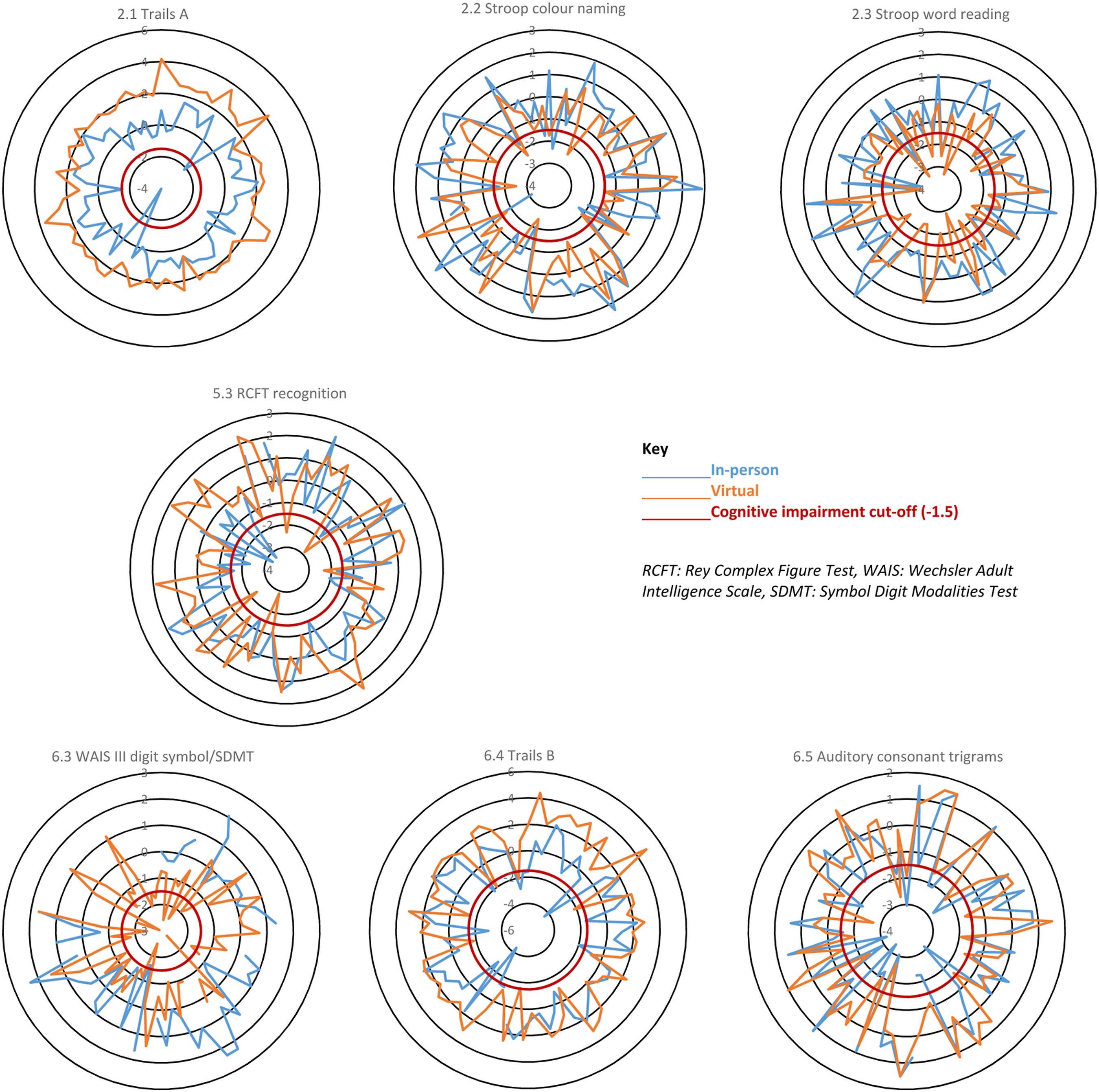
Preference questionnaire
Sixty-six out of 71 participants completed the preference questionnaire. The results showed a preference for the virtual assessment (n = 30, 45%), compared to n = 22, 33% for in-person and n = 14, 21% who had no preference. The main reason people preferred the virtual visit was that they did not need to travel; this was followed by ‘it is more convenient’ and ‘reduces in-person contact during COVID-19’. With regards to how the test is run, the majority of participants (n = 50, 76%) felt that there was no difference in difficulty between virtual and in-person testing. When asked if anything could make the virtual experience better, of the 61 who commented, the majority (n = 52) said they had no suggestions, five participants expressed a wish for more technical support and two participants suggested more information was needed regarding what additional equipment (e.g. pencils) they would need during the assessment.
Discussion
Of the 19 tests in the ACR-NB, we identified one or more problems with eight (42%) tests as we moved from in-person to virtual administration including tests that cannot be administered virtually (n = 2), tests that required modification (n = 3), and tests (n = 6) that showed significant and consistent performance differences according to mode of administration. The tests primarily affected were those that had been changed from a written to verbal response and had a timed element as well as those that required a visual presentation. Three tasks, Trails A and B and WAIS-III digit symbol/SDMT, were changed from a written to verbal response. This change made both Trails A and B easier. Trails A is particularly affected when converted to verbal and other researchers have suggested that a verbal version of Trails A is not appropriate. 12 In a similar remote study, in patients with multiple sclerosis, the Trail Making Tests were also used but to avoid issues with verbal versus written response, paper copies of the tests were posted out prior to the assessment to ensure that the test could be done in written form. The results were more comparable but participants still performed better on Trails A when administered remotely. 13 Using a written version remotely may be a better method for ensuring comparability to in-person testing but is not ideal for the long-term switch to virtual assessment. If testing is to move to a remote setting long-term, then better computerised adaptations of the Trails Making Test are required.
A verbal version of the WAIS-III digit symbol test was also required for our study. As such, the WAIS-III digit symbol was replaced by the SDMT. These tests have been previously shown to be comparable, 14 with the exception that the SDMT can be responded to verbally and not just in written form. 15 For these tests, the participant is scored based on how many correct answers they can give within a set timeframe. The WAIS-III digit symbol test is assessed over 120 s, whereas the SDMT is shorter and assessed over 90 s, as such accounting for the speed advantage from a verbal response. However, our cohort performed worse on the SDMT in the virtual environment compared to the in-person administration of the WAIS-III digit symbol. This suggests additional adaptations are required to ensure that these tests are comparable in an SLE cohort; however, it is also worth noting that this task had some missing data.
The remaining three tests that were consistently significantly different were the Stroop colour naming and word reading and the RCFT recognition. For both Stroop results, participants performed worse during the virtual administration of this test compared to in-person. The reasoning for this altered performance is believed to be linked to the presentation of the stimuli. As with the in-person administration, the participant is presented with one sheet showing all the words they are required to read. In-person, they are handed a letter size sheet of paper. Virtually, all the words are presented onscreen. This latter version means the words are smaller and harder to read and therefore negatively affects performance. Other studies adapting cognitive tests from in-person to virtual have found similar issues due to display restrictions. 13 Contrary to this, participants performed better on the RCFT recognition, this may be due to environment. For example, studies have shown cognitive performance is affected by stress and anxiety,16,17 states that can be affected by environment such as a home setting versus a laboratory setting.
The intra-individual longitudinal comparison found that seven tests were potentially affected by administration method. Further exploration of performance on these tests found that only 15% of the participants changed CI status (based on our CI algorithm) due to performance on one or more of these seven tests. This suggests that while there were statistically different results from virtual versus in-person cognitive testing, this may not result in a difference in CI classification.
This work was needed in response to adaptations made to our CI in SLE study because of the COVID-19 pandemic. As such, the analysis for this study was planned retrospectively and the ideal comparison of administering both methods (virtual and in-person) on the same day to the same participant in a counterbalanced way was not possible and must be considered a limitation of this work. Instead, we undertook three analyses to try and control for as many confounding factors as possible. Also, we were unable to counterbalance the administration method. All in-person visits were undertaken prior to the virtual visits, so we cannot rule out learned effects. It is also worth noting that the anxiety levels in the virtual administration method were higher than in-person. This difference is likely connected with the COVID-19 pandemic where global levels of anxiety increased. 18 Finally, using computer technology in a study may have health inequality implications as it requires access to computers and the internet as well as computer literacy 19 and therefore may have biased our study cohort or affected our results.
Assessing cognitive function virtually, especially for research, is likely to become more common even as we see COVID-19 restrictions lifted. Our research found that virtual administration was preferred over in-person testing by participants and there are many benefits for researchers too, mainly regarding time and flexibility to deliver the testing. As such, research needs to ensure that the newly adapted virtual tests are validated in the same way the in-person versions were. New cut-offs may need to be established to ensure comparability between the administration methods.20–22 Our study has shown that it is possible to adapt the ACR-NB but that caution needs to be taken if comparing in-person to virtual administration. Future research may find results more comparable if established computerised cognitive batteries, such as the CANTAB® and ANAM, are used instead. 23 Ultimately, the new virtual way of working is likely to remain 24 and future cognitive research needs to ensure accurate testing is available and validated.
Supplemental Material
Supplemental Material - In-person versus virtual administration of the American College of Rheumatology gold standard cognitive battery in systemic lupus erythematosus: Are they interchangeable?
Supplemental Material for In-person versus virtual administration of the American College of Radiology gold standard cognitive battery in systemic lupus erythematosus: Are they interchangeable? by ML Barraclough, JP Diaz-Martinez, A Knight, K Bingham, J Su, M Kakvan, C Muñoz Grajales, MC Tartaglia, L Ruttan, J Wither, MY Choi, D Bonilla, N Anderson, S Appenzeller, B Parker, P Katz, D Beaton, R Green, IN Bruce and Z Touma in Lupus
Footnotes
Declaration of Conflicting Interests
MB reports financial support by Arthritis Society of Canada, Canadian Institutes of Health Research, Physician’s Services Incorporated, The Province of Ontario Early Research Award, The Lupus Research Alliance, Lupus Canada, Lupus UK, and has honoraria from UHN International conference on Arthritis; AK reports grants from Lupus Research Alliance & US Department of Defense; CIHR Long Covid study grant; KB reports financial support from Labatt Family Network and University of Toronto Dept of Psychiatry’s Excellence Fund; CM receives consulting fees from MitogenDx, Mallinckrodt Pharmaceuticals, AstraZeneca, GSK; JW reports indirect support from Lupus 21st Century – indirect support from multiple drug companies, participation on Astra Zeneca Advisory Board, indirect salary support from unrestricted funds given by Pfizer for a research chair in the Division of Rheumatology; MYC reports consulting fees from MitogenDx, Mallinckrodt Pharmaceuticals, AstraZeneca, GSK; BP reports consulting fees from Fresenius Kabi and Vifor Pharma, honoraria from Astra Zeneca, UCB and Roche Chugai, meeting attendance support from Abbvie and Eli Lilly; DB is executive director & chair of OMERACT; zettatesla is supported by grants from Schroeder Arthritis Institute, Lupus Ontario, Arthritis Society of Canada, Canadian Institutes of Health Research, Physicians’ Services Incorporated, The Province of Ontario (Early Research Award), The Lupus Research Alliance and reports a relationship with consulting fees from AstraZeneca AB, Merck KGaA, GlaxoSmithKline Inc, and UCB Biopharma SRL, and Sarkana Pharma, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the This project is funded by grants from the Arthritis Society of Canada, Canadian Institutes of Health Research, Physician’s Services Incorporated, the Province of Ontario Early Research Award, and the Lupus Research Alliance. Dr Michelle Barraclough is supported by a fellowship from the Arthritis Society. Dr Touma is supported by the Department of Medicine, University of Toronto. Dr Touma’s laboratory is supported by Lupus ON, the Schroeder Arthritis Institute and donations from the Kathi and Peter Kaiser family, the Lou and Marissa Rocca family, the Bozzo family, and the Stacey and Mark Krembil family. Dr Wither is supported by a Pfizer Chair Research Award.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
