Abstract
Purpose
To investigate whether low complement levels can predict worse outcomes in patients hospitalized with positive anti-phospholipid antibodies.
Methods
This was a retrospective cohort study. We obtained demographics, laboratory, and prognostic data of all consecutive patients hospitalized between 2007 and 2021, for whatever reason, with at least one positively abnormal anti-phospholipid antibody, who were also tested for complement levels (C3 or C4). We then compared the rates of long-term mortality, 1-year mortality, deep vein thrombosis, and pulmonary emboli between groups of low complement and normal complement levels. Multivariate analysis was used to control for levels of clinical and laboratory confounders.
Results
We identified 32,286 patients tested for anti-phospholipid antibodies. Of those patients, 6800 tested positive for at least one anti-phospholipid antibody and had a documented complement level. Significant higher mortality rates were found in the low complement group, with an odds ratio for mortality (OR 1.93 CI 1.63–2.27 p < .001). Deep vein thrombosis and pulmonary emboli rates were similar. Multivariate analysis confirmed that low complement was an independent predictor for mortality after controlling for age, sex, dyslipidemia, chronic heart failure (CHF), chronic kidney disease (CKD), and anemia.
Conclusions
Our study results indicate that low complement is associated with significantly higher mortality rates in admitted patients with elevated levels of anti-phospholipid antibodies. This finding correlates with recent literature suggesting a vital role for complement activation in anti-phospholipid syndrome.
Introduction
Antiphospholipid Syndrome (APS) is an autoimmune disease characterized by increased risk for vascular (arterial or venous) thrombosis or pregnancy morbidity in the presence of anti-phospholipid antibodies. However, it is known that in healthy persons without APS, elevated anti-phospholipid antibodies are relatively frequent, occurring in 1%–5% of the population, increasing to 50% among elderly patients with chronic diseases1,2. The presence of those antibodies in healthy patients, those with Infection, and malignancies, do not correlate necessarily with an increased risk of thrombosis or other manifestations of the anti-phospholipid syndrome.3–5
The mechanisms by which aPLs induce thrombosis in the Antiphospholipid Syndrome are unclear; several have been suggested, among them complement activation.6–9 The complement system is a system of enzymes and regulatory proteins of the innate immune system that play a crucial role in the inflammatory response to various pathogenic stimuli. Complement activation can be initiated via three pathways—the classical, the lectin, and the alternate. Complement activation plays a significant role in host defense mechanisms against infection and inflammation but is also implicated in the pathogenesis of the anti-phospholipid syndrome. The complement and coagulation pathways are interconnected, and expanding evidence indicates that complement may be activated in patients with anti-phospholipid syndrome10–12. Recent studies found that complement inhibition in murine models of anti-phospholipid syndrome reverses some anti-phospholipid antibodies-associated complications13,14. Furthermore, with the emerging role of complement in anti-phospholipid syndrome, an anti-C5 monoclonal antibody was recently suggested as a treatment for catastrophic anti-phospholipid syndrome15,16.
Our study was designed in an attempt to translate animal model findings to observations in humans and specifically to try and see if complement activation translates into deleterious clinical findings among hospitalized patients who tested positive for high anti-phospholipid antibodies.
Methods
Study design
We performed a retrospective analysis of all consecutive patients admitted to the Chaim Sheba Medical Center, the largest tertiary hospital in Israel, between 1/3/2007 and 05/09/022. We then pulled out all the cases tested for anti-phospholipid antibodies, for whatever reason, whether positive or negative. Of all those cases, we pulled out only the cases who were found to have at least one positive anti-phospholipid antibody. We than pulled out all the relevant clinical and laboratory data on those cases. We further divided this groups of cases with at least one positive antibody into two groups one with normal complement levels and the other with low complement levels. We than compared all the variables between the groups of low complement and normal complement levels. Inclusion criteria mandated age above 18, at least one positive anti-phospholipid blood test, and a measurement of complement level on the same admission. We did not use the discharge diagnosis or the background diagnoses for inclusion or exclusion from the study.
Laboratory measurements
Patients with positive anti-phospholipid antibodies were defined as those who tested positive for one or more of the following anti-phospholipid antibodies: lupus anticoagulant, IgG/IgM anti-cardiolipin antibodies, or IgG/IgM anti-β2-glycoprotein I antibodies (β2GPI).
Anti-β2-glycoprotein I antibodies (isotypes IgG and IgM) and anti-cardiolipin antibodies were considered positive above 20 U/mL, which is according to the manufacturer’s recommendations (BioRad BioPlexTM 2200 system [USA]) and the international consensus guidelines. Three-step testing with the diluted Russell’s viper venom time (dRVVT) screen and dRVVT confirm assays were used to assess the presence of LAC as recommended by the Scientific Standardization Subcommittee on Lupus Anticoagulant/Phospholipid Antibodies. In addition to the dRVVT method, we also used silica clotting time (SCT), an activated partial thromboplastin time (APTT)-based test, which is also recommended by the Scientific and Standardization Committee for lupus anticoagulant/anti-phospholipid antibodies of the International Society on Thrombosis and Haemostasis for lupus anticoagulant testing. All elevated results were confirmed by a second test plus a screen and confirmatory test on a 1:1 mix of the sample with normal plasma.17–19
Exposure variables
The primary exposure variable was complement levels. ELISA assays measuring complement C3 and C4 were performed on EDTA plasma samples according to the manufacturer’s protocol. Low c3 was considered below 90 mg/dL, and low c4 was considered below 10 mg/dL. The complement cut offs were measured according to the manufacturer recommendation (BN™ II System, Siemens Healthineers).
Outcome variables
Mortality data were extracted from the Israeli National Population Register and were available for all cases. The primary outcome of the current study was all-cause mortality. Survival data were available for all subjects from the Israeli Population Registry. We analyzed both 1-year mortality following admission and long-term mortality (defined as-time from admission till the date of the extraction of the data for the study, namely, 05/09/2022). Secondary outcomes were death from sepsis or an incidence of Deep Vein Thrombosis or Pulmonary Emboli.
After study approval by the local institutional review board, we retrieved patients’ data from their electronic medical records.
Statistical analysis
The patient’s baseline demographic and clinical data were retrieved from the Electronic Medical Record. Diagnoses were based on computerized hospitalization records (International Classification of Diseases, Ninth Revision [ICD‐9] codes), laboratory tests, medications, physiological signals (e.g., ECGs), radiological images (e.g., echocardiograms and angiograms), and procedures' reports. Continuous variables were expressed as mean ± SD if normally distributed or median with interquartile range if skewed. Normality was determined using Anderson-Darling and Shapiro-Wilk tests for normality. Categorical variables were presented as frequency (%). Continuous data were compared with the student’s t-test, and categorical data were compared using the chi-square test or Fisher exact test. Using the log-rank test, survival analysis was depicted using Kaplan–Meier curves, comparing cumulative survival across strata. Univariate regression modeling was used to determine the unadjusted Odds ratio (OR) for the primary and secondary outcomes. A general logistic regression multivariate model was constructed to investigate the correlation and control for important confounders. We then conducted several sensitivity analyses. When choosing comorbidities to include in the multivariate models we aimed to include the most common diseases that are statistically significantly different between groups, are of a large proportion in the population and are known to impact mortality. Several other analyses are included in the supplementary Appendix. In addition, we conducted multivariate analysis to correct for Anti-β2-glycoprotein I antibodies and anti-cardiolipin antibodies levels. The rationale for this analysis was to test whether higher antibody levels in the low complement group, and not the low complement itself affects the mortality. Another sub analysis was done to control for the effect of having systemic lupus erythematosus (SLE) on the results
Al analyses were performed using R software version 4.1.0 (R Foundation for Statistical Computing). An association was considered statistically significant for a two-sided p value of less than 0.05.
Results
During the research period, 2007–2022, we identified 32,286 patients who were tested for positive anti-phospholipid antibodies. Of these patients, 6800 were also tested for complement levels and were found to be eligible for study enrollment. In 25,486 patients, complement levels were not tested, and they were not enrolled.
Basic demographics and laboratory data of patients with at least one positive anti-phospholipid antibody.
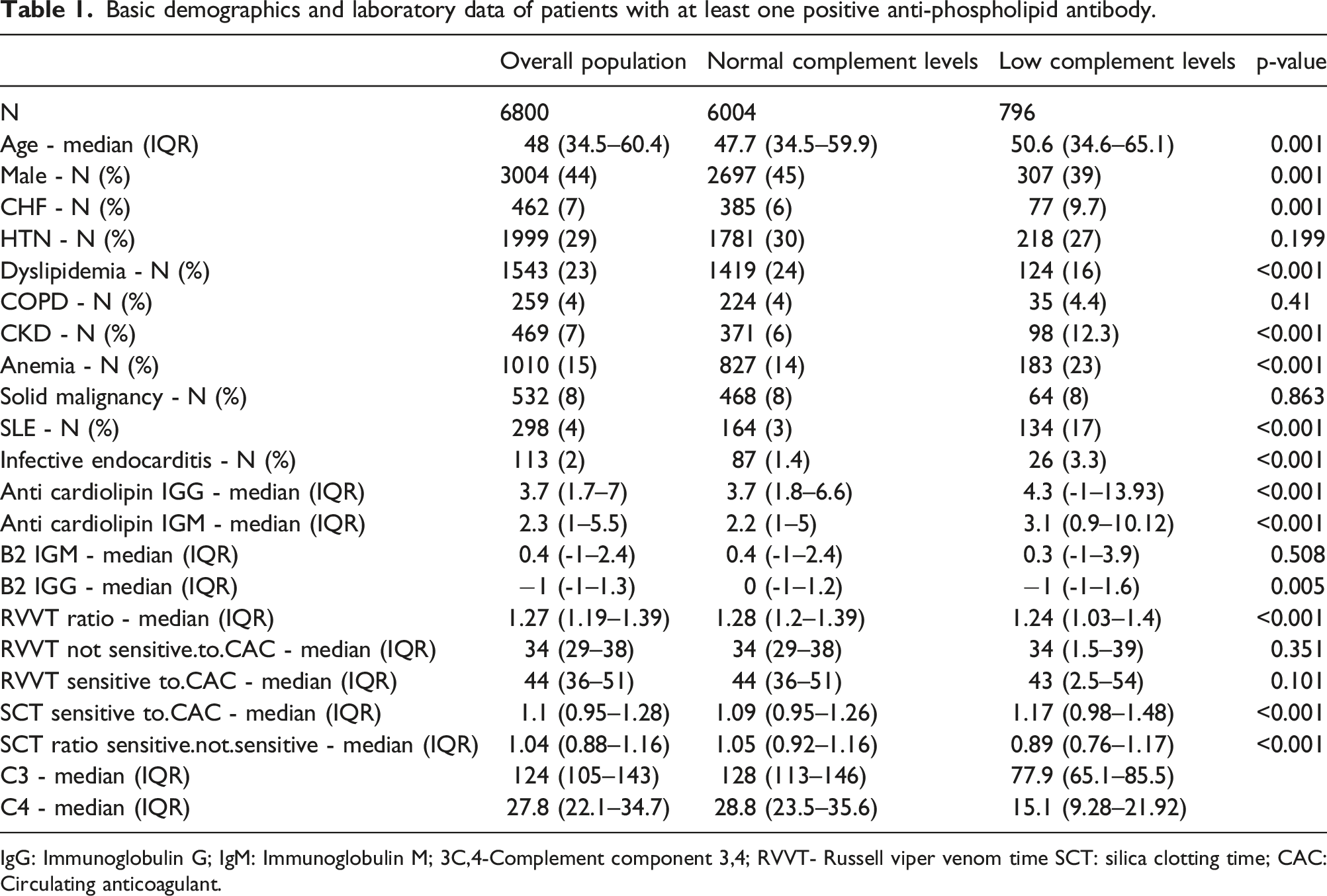
IgG: Immunoglobulin G; IgM: Immunoglobulin M; 3C,4-Complement component 3,4; RVVT- Russell viper venom time SCT: silica clotting time; CAC: Circulating anticoagulant.
Six thousand and four (88%) cases were found to have at least one positive anti-phospholipid antibody and normal complement levels, while 796 (12%) cases were found to have at least one positive anti-phospholipid antibody but had low complement levels (either c3 or C4). The median C3 level in the standard complement levels group was 128 (IQR 113–146), and the median level of C3 in the low complement levels group was 77.9 (IQR 65.1–85.5). The median level of C4 in the standard complement levels group was 28.8 (IQR 23.5–35.6), and in the low complement levels group was 15.1 (IQR 9.3–21.9).
As detailed in Table 1, the median levels of β2GPI IgG, β2GPI IgM (anti-β2-glycoprotein I antibodies), anti-cardiolipin antibodies IgG, and anti-cardiolipin antibodies IgM were found to be significantly higher among the low complement levels group of patients.
Univariate analysis
Mortality and morbidity rates based on complement levels.
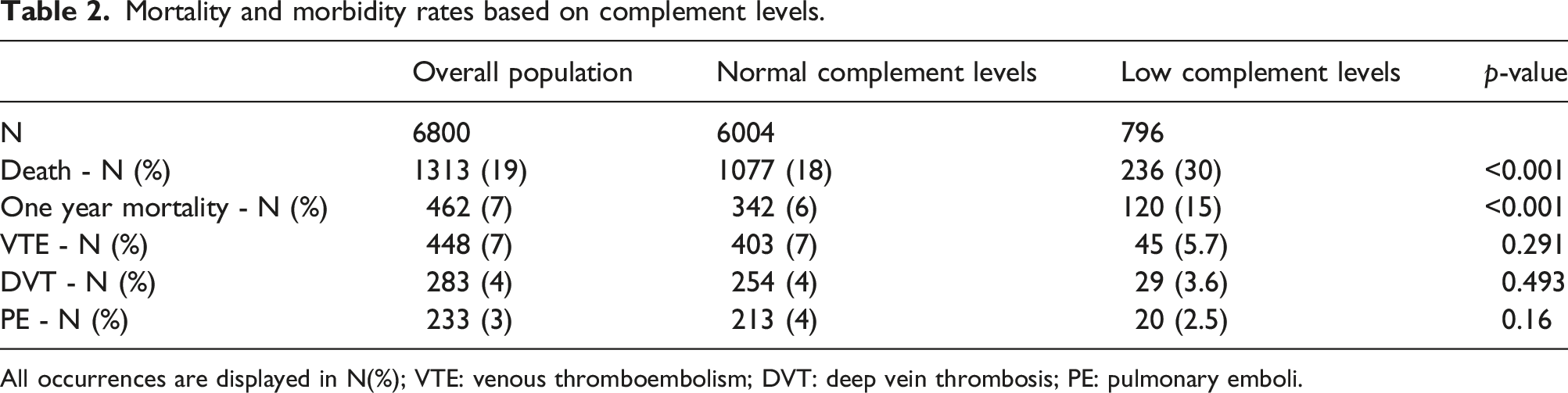
All occurrences are displayed in N(%); VTE: venous thromboembolism; DVT: deep vein thrombosis; PE: pulmonary emboli.
We analyzed both short-term and long-term mortality. Time-based survival analysis showed that 1-year and 10-year survival rates were lower in the low complement levels group (84.7% vs 94.3% and 69.7% vs 81.3%, respectively). Log-Rank for comparison p < .001. Figure 1 depicts the cumulative 10-year survival analysis using a Kaplan–Meier curve. When both C3 and C4 were low, that resulted in a higher hazard ratio for mortality (HR 2.38 CI 1.60–3.53 p < .001) Cumulative 10-year survival of low versus normal complement population.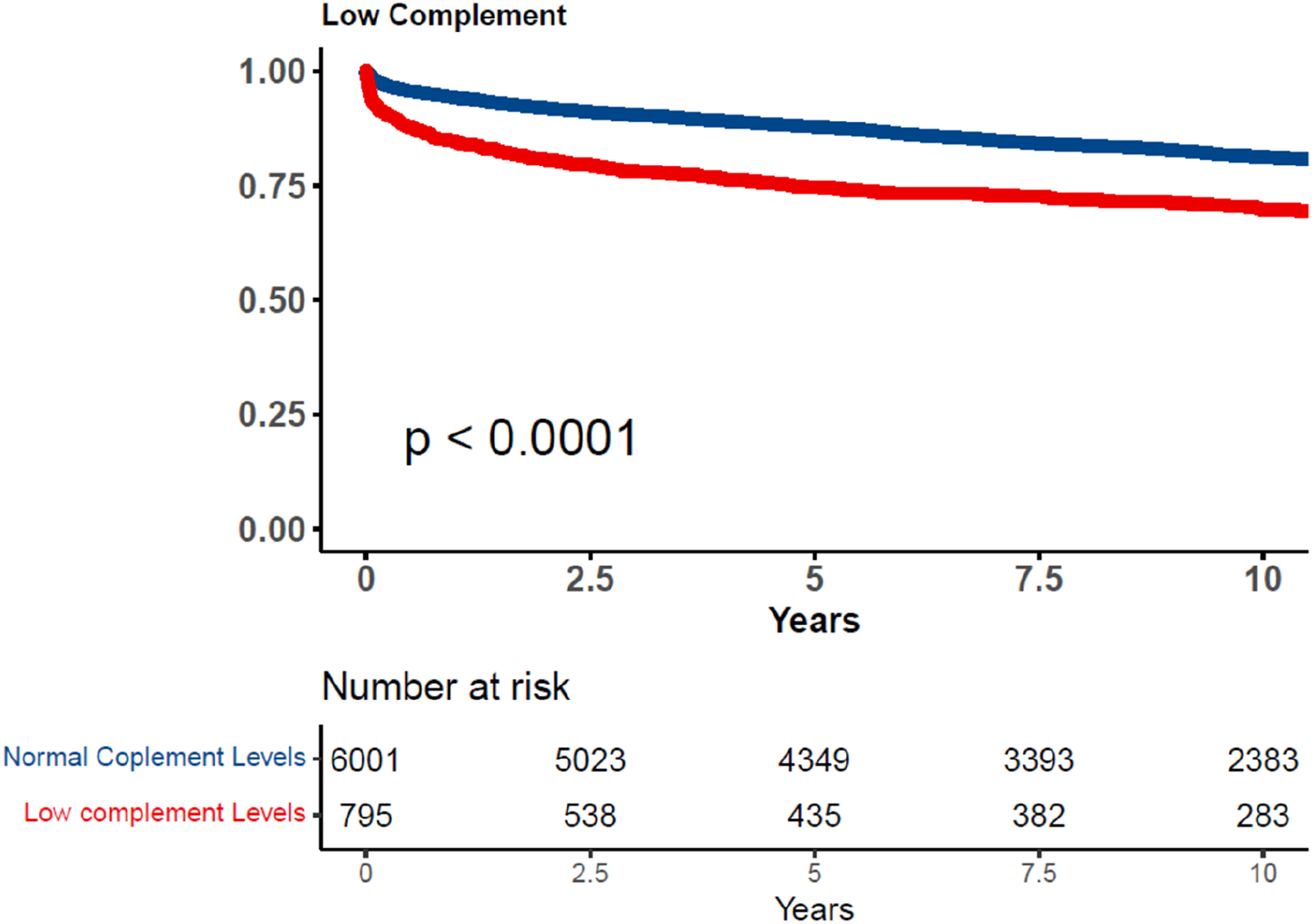
Multivariate analysis
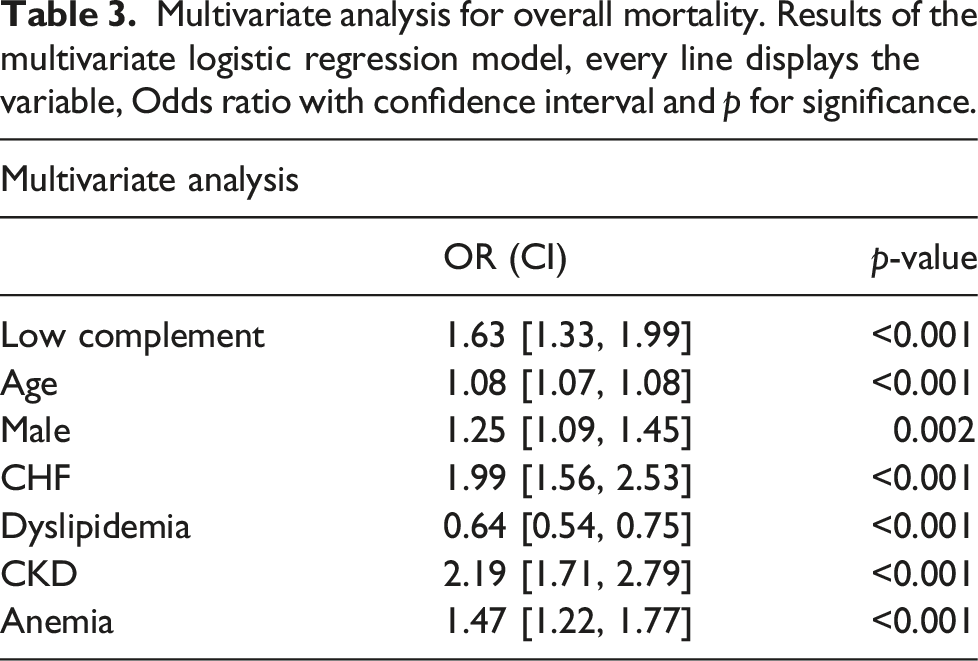
Multivariate analysis for overall mortality. Results of the multivariate logistic regression model, every line displays the variable, Odds ratio with confidence interval and p for significance.
Discussion
In this study of a large cohort of hospitalized patients with high anti-phospholipid antibodies levels, we found that low complement activity is associated with significantly higher mortality rates. We believe this is the first description of this correlation. This finding was validated using multivariate analysis controlling for age, sex, and multiple comorbidities.
Although anti-phospholipid antibodies profile helps in risk stratification of patients with anti-phospholipid syndrome, the absence of a reliable biomarker to distinguish clinically relevant from clinically irrelevant anti-phospholipid antibodies levels (and to predict which patients are at risk of experiencing first or recurrent thrombosis), is a significant gap in the routine clinical care of patients tested positive for anti-phospholipid antibodies without evidence of the anti-phospholipid syndrome. Using C3 and C4 levels as an additional prognostication tool to better stratify patients with positive anti-phospholipid antibodies is relatively easy and accessible. An appearance of anti-phospholipid antibodies characterizes a myriad of infectious diseases; however, their presence does not necessarily lead to the development of thrombotic events and, consequently, anti-phospholipid syndrome.
Interestingly, in our cohort, low C3 and C4, though associated with mortality, did not increase the risk for pulmonary emboli or deep vein thrombosis. This may support the previous suggestion that anti-phospholipid antibodies not in the context of clinical APS may often not represent a true cause of venous hypercoagulability.20–23 Our cohort lacked data to examine if arterial thrombosis events were the source for the increased mortality in the low complement group. The groups had similar rates of solid malignancies therefore the increased mortality was not attributed to the malignancies. In order to look whether the higher mortality rates were indeed related to the lower complement levels and not to comorbidities, we conducted a multivariate analysis which found low complement to be associated with higher mortality rates even after correction for age, CKD, CHF, anemia, and other comorbidities.
Only 4% of our large cohort were diagnosed with systemic lupus erythematosus; and 2% had a diagnosis of infective endocarditis This finding is in line with previous studies that found positive anti-phospholipid antibodies to be widespread among hospitalized patients with infectious diseases, malignancies, and other conditions.1,2,23 Our findings of the increased mortality rates amongst the low complement group remained valid regardless of the inclusion or exclusion of the SLE and infective endocarditis patients.
To our knowledge, low complement as a marker for increased mortality in the setup of patients with positive anti-phospholipid antibodies has not been well described. In a previous study by Saraiva et al. 24 in patients with primary anti-phospholipid syndrome, lower-than-normal levels of C3 were independently associated with triple positivity. Triple positive anti-phospholipid antibodies are known14,25 to be associated with higher thrombotic risk in patients with anti-phospholipid syndrome, but no data was given about mortality rates.
Other authors have proposed a pathogenic model in which anti-phospholipid antibodies are the first hit and can induce complement activation and consequent thrombosis.6,13 In contrast, patients who also have a pathogenic complement regulatory gene mutation (second hit) are predisposed to uncontrolled complement activation, leading to the catastrophic anti-phospholipid syndrome in the setting of a complement-amplifying trigger such as infection, surgery, pregnancy, or autoimmune disease6,13. The recent positive experience using eculizumab, an anti-C5 monoclonal antibody in catastrophic anti-phospholipid syndrome, further strengthens our findings 15 . An Italian group found that low circulating levels of complement components were associated with poor pregnancy outcomes in terms of the gestational week at delivery, neonatal birth weight, neonatal birth weight percentile, fetal loss, preterm delivery, and birth weight < 2500 g 26 .
Limitations
This study has several limitations. First, this is a retrospective cohort study based on electronic medical records (EMR) data. This was mitigated by not using data logged by medical personnel and is error-prone while keeping data from laboratory machinery and the national population registry.
The second limitation was the use of all-cause mortality; while it is evident that low complement impacts mortality, based on this study alone, we cannot assert the cause for the mortality and whether there are any specific preventive measures to be taken. Also, the results of the study could have been skewed by selection bias, as we do not know why the physicians performed the complement level test only in 6800 patients but did not do the same in the 25,486 patients which were not enrolled. Last but not least, our study conclusions are relevant to patients with positive anti-phospholipid antibodies in general, and the conclusions shouldn’t be directly drawn regarding patients with the anti-phospholipid syndrome.
Conclusion
In hospitalized patients tested positive for high anti-phospholipid antibodies levels, low complement levels are associated with significantly higher mortality rates. These findings are consistent with recent literature, suggesting that complement activation plays a vital role in the anti-phospholipid syndrome.
Supplemental Material
Supplemental Material - Hospitalized patients with positive antiphospholipid antibodies who have low complement levels are at increased risk for death—a retrospective cohort study
Supplemental Material for Hospitalized patients with positive antiphospholipid antibodies who have low complement levels are at increased risk for death—a retrospective cohort study by Edward Itelman, Maxim Perelman, Natali Bivar, Daniella Kent, Adva Vaisman, Gad Segal, Liat Negru, Amir Dagan in Lupus
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s note
All authors had access to the data and a role in writing the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
