Abstract
Background
Systemic lupus erythematosus (SLE) is an autoimmune disease that typically affects women aged 16–55 years. Cardiovascular disease (CVD) is a well-recognized complication of SLE. This systematic literature review and meta-analysis evaluated the relative risk (RR; compared with non-SLE controls), absolute risk (AR; as incidence proportion, n/N), and incidence rate (IR) of CVD events (including stroke, myocardial infarction [MI], and CVD [composite or undefined]) in adult patients with SLE. The RR of CV risk factors (including hypertension, diabetes, and metabolic syndrome [MetS]) was also examined.
Methods
PubMed and Embase were searched on September 10, 2020. Observational studies published between January 2010 and September 2020 that reported RR, AR, and/or IR of CVD events, or RR of CV risk factors, were eligible. Pooled risk estimates were calculated using a random-effects model.
Results
Forty-six studies (16 cross-sectional, 15 retrospective cohort, 14 prospective cohort, and 1 case–control) were included in meta-analyses. Most studies were considered high quality (Critical Appraisal Skills Programme checklists). Compared with adults without SLE, patients with SLE had statistically significantly higher RRs (95% CIs) of stroke (2.51 [2.03–3.10]; 12 studies), MI (2.92 [2.45–3.48]; 11 studies), CVD (2.24 [1.94–2.59]; 8 studies), and hypertension (2.70 [1.48–4.92]; 7 studies). RRs of diabetes (1.24 [0.78–1.96]; 3 studies) and MetS (1.49 [0.95–2.33]; 7 studies) were elevated but not significant. RRs of stroke and MI were generally higher in younger versus older patients with SLE. In patients with SLE, the pooled estimate of AR (95% CI) was 0.03 (0.02–0.05), 0.01 (0.00–0.02), and 0.06 (0.03–0.10) for stroke (7 studies), MI (6 studies), and CVD (8 studies), respectively. The pooled estimate of IR per 1000 person-years (95% CI) was 4.72 (3.35–6.32), 2.81 (1.61–4.32), and 11.21 (8.48–14.32) for stroke (10 studies), MI (6 studies), and CVD (8 studies), respectively. Although heterogeneity (based on I2 value) was high in most analyses, sensitivity analyses confirmed the robustness of the pooled estimates.
Conclusions
This meta-analysis found an increased risk of stroke, MI, CVD, and hypertension in patients with SLE compared with the general population, despite substantial heterogeneity across the included studies.
Introduction
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease characterized by a wide spectrum of clinical manifestations and an unpredictable relapsing-remitting course. SLE typically affects women aged between 16 and 55 years and is characterized by multiorgan damage mediated by tissue-binding autoantibodies and immune complexes, leading to acute and chronic inflammation of various organs and tissues.1–3
Cardiovascular disease (CVD) is a well-recognized complication of SLE, and there is strong evidence to suggest that patients with SLE have at least a 2- to 3-fold elevated risk of myocardial infarction (MI), congestive heart failure, cerebrovascular disease, and overall CVD mortality than the general population.4,5 Patients with SLE have increased mortality, with accumulation of organ damage being the major determinant. A bimodal mortality pattern has been described showing that early mortality is associated with disease activity and infections, whereas late mortality is associated with atherosclerotic complications. 6
There have been significant improvements in long-term survival in patients with SLE. Morbidity and mortality due to comorbid causes are declining 7 ; however, the decline is similar to that of the general population, and mortality remains disproportionately high in the SLE population, particularly in younger patients with SLE. 8 The relative risk (RR) of CVD is higher in younger women (aged 19–39 years) with SLE compared with the general population, 9 and the absolute risk (AR) of CVD increases with age in both men and women with SLE. 1
Understanding the magnitude of CVD risk, including stroke, MI, and cardiovascular (CV) death, as well as traditional CV risk factors, in patients with SLE would support the identification of high-risk patients in clinical practice and allow for implementation of tailored prevention and treatment strategies in addition to SLE management. The objective of this meta-analysis was to evaluate the RR (compared with the general population or suitable proxies), AR, and incidence rate (IR) of CV events, including stroke, MI, and CVD, in adult patients with SLE. An additional objective was to evaluate the ratio of risk of CV risk factors, including hypertension, diabetes, and metabolic syndrome (MetS), in patients with SLE compared with the general population.
Methods
Search strategy
This study was conducted using the “Preferred Reporting Items for Systematic Reviews and Meta-analysis” guidelines 10 and followed a prespecified protocol (not registered). For the systematic literature review (SLR) and meta-analysis (MA), the available literature was retrieved from the electronic medical databases MEDLINE (using the PubMed platform) and Embase (using the Elsevier platform) on September 10, 2020. The search was limited to observational studies published between January 2010 and September 2020 that reported on CV events and CV risk factors in adult patients with SLE. The keywords used in the search focused on SLE, CV events, CV risk factors, and the risk or incidence of CV events (Supplementary Table 1, Supplementary Table 2). The CV events of interest included MI, angina, heart failure, stroke, peripheral arterial disease, transient ischemic attack (TIA), major adverse CV events, and CVD. The CV risk factors included hypertension, obesity, smoking, diabetes, dyslipidemia, MetS, and chronic kidney disease.
Selection criteria
Although there were no geographic or language limitations on the searches, only English-language articles were included during screening. Observational studies (cohort, cross-sectional, case–control, registry studies, and other observational) were eligible for inclusion; randomized and nonrandomized clinical trials, case reports, editorials, etc., were excluded. SLRs and MAs were hand-searched to identify additional relevant studies. Studies that included patients with diseases other than SLE or pediatric (aged <18 years) patients with SLE were not eligible, unless results for adults with SLE were reported separately. The outcomes of interest included the RR of CV events in adults with SLE compared with adults without SLE; the AR of CV events in patients with SLE; and the IR of CV events in patients with SLE. In addition, to evaluate the ratio of risk of CV risk factors, studies reporting on CV risk factors in patients with SLE compared with the general population were also included in the SLR. Subgroups of interest included patients differentiated by age, sex, ethnicity, geographical location, disease severity, disease duration, prior treatment received, and antiphospholipid syndrome/antiphospholipid antibodies. The CV events reported in this publication were stroke, MI, and general CVD (e.g., composite outcomes or CVD not otherwise defined), and the CV risk factors were hypertension, diabetes, and MetS. Other events included in the SLR were not prioritized for the MA because of insufficient data.
Data extraction and quality assessment
Data were extracted from full-text publications. References to other publications within a study were traced to original sources, where appropriate. When multiple publications reporting on the same study were identified, the primary publication was extracted first; data from secondary publications were extracted if not reported in the primary publication. All extracted data were quality-checked and verified against the source document by a second researcher not involved with the extraction. A quality assessment recommended by the Critical Appraisal Skills Programme (CASP; https://casp-uk.net/) was conducted for each individual observational study. Individual CASP checklists were used to evaluate case–control studies and cohort studies. A modified version of the CASP cohort study checklist was used to evaluate cross-sectional studies. Studies were classified as low, medium, or high quality, or as unclear; all studies were included in primary analyses regardless of quality.
Statistical analysis
General meta-analysis methods
Meta-analyses for RR, AR (incidence proportion), and IR were conducted for each outcome when there were ≥3 studies reporting the respective measure for the respective outcome. The restricted maximum likelihood random-effects model was used to calculate the pooled risk estimates and 95% confidence intervals (CIs) for all outcomes. Fixed-effects models were also fit for completeness. Higgins’s I2 was used to estimate the percentage of variance explained by heterogeneity. 11 Sensitivity analyses were performed to assess the robustness of the primary pooled estimates as well as the impact of study design or risk measures on the effect size; these included leave-one-out analysis (i.e., MA in which exactly one study is omitted; this method demonstrates how each individual study affects the overall estimate), limiting analysis to studies with comparable CV event definitions, and limiting analysis to high-quality studies. Metaprop or metafor in R (R Foundation for Statistical Computing, Vienna, Austria) was used for analyses. 12 Forest plots were visually inspected to assess the consistency of the study data.
Meta-analysis methods for relative effects
Measures such as odds ratio, hazard ratio (HR), RR, IR ratio, or standardized IR were used for comparative measures of the ratio of risk of stroke, MI, CVD, hypertension, diabetes, and MetS for patients with SLE versus the general population. Because data were scarce for most endpoints, all comparable studies were included in the primary MA. These measures were pooled in the MA and are hereafter referred to collectively as RRs. Because of differences in adjustment factors used across the studies, unadjusted measures were used in preference to adjusted measures where both were available. Studies were included in the analysis regardless of the length of follow-up under the assumption of proportional hazards over time. Subgroup analyses were based on age and sex compared with matched non-SLE controls.
Meta-analysis methods for absolute risk
For the analysis of AR of stroke, MI, and CVD in patients with SLE, the AR was defined as a proportion. The contributing studies provided information on the number of events (n/N), and the double-arcsine transformation was used for this analysis.
Meta-analysis methods for incidence rates
For the analysis of IR of stroke, MI, and CVD in patients with SLE, results from studies were adjusted to be in the consistent form of per 1000 person-years (PY). The metafor package was used with the metarate command, with the events based on the IR per 1000 PY and the PY of follow-up used as inputs.
Results
Search results
The electronic literature search identified 1591 articles for title and abstract screening, of which 278 articles were retrieved for Level 2 screening of the full text; 7 additional articles were identified from published SLRs (Figure 1). A total of 87 articles met the inclusion criteria for the SLR. After review, 46 of these were included in ≥1 MA (Figure 1), and 41 were excluded for various reasons (Supplementary Table 3). Flow diagram of the study selection process. MA: meta-analysis; SLR: systematic literature review.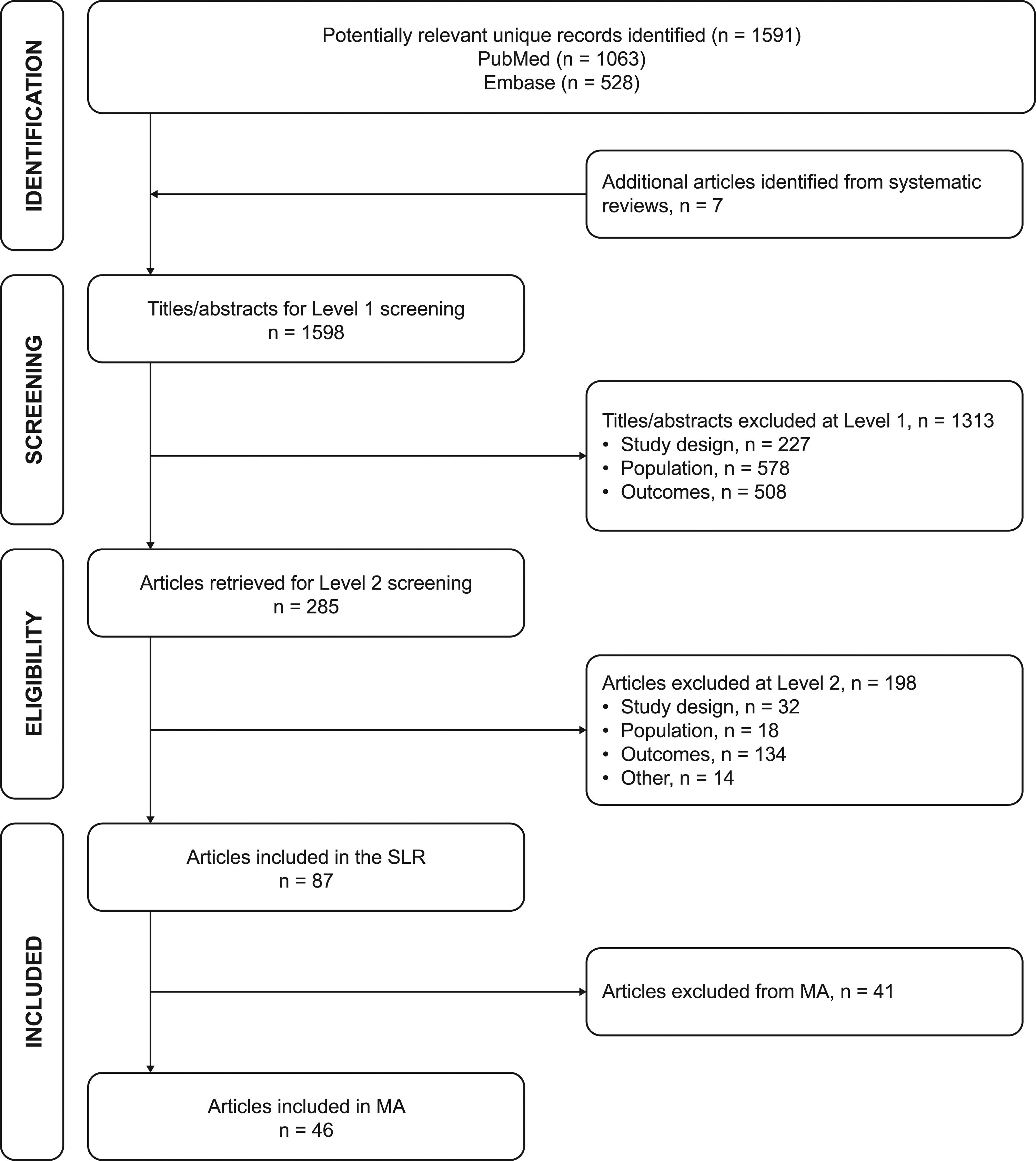
Characteristics of included studies
Baseline characteristics of the included studies and participants.
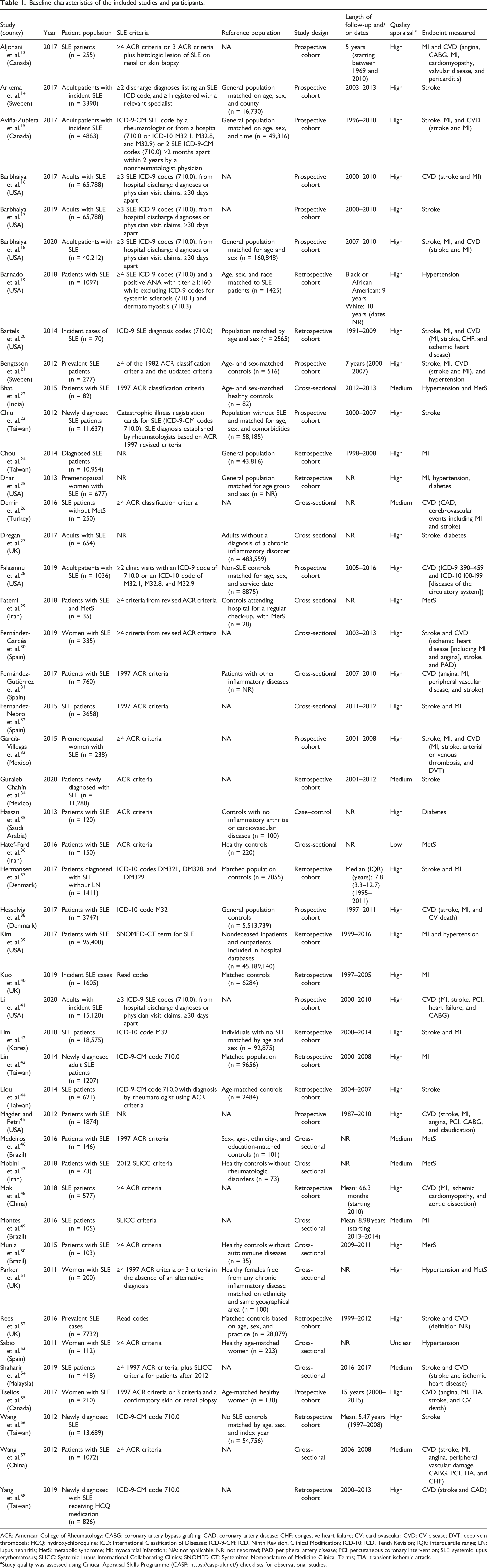
ACR: American College of Rheumatology; CABG: coronary artery bypass grafting; CAD: coronary artery disease; CHF: congestive heart failure; CV: cardiovascular; CVD: CV disease; DVT: deep vein thrombosis; HCQ: hydroxychloroquine; ICD: International Classification of Diseases; ICD-9-CM: ICD, Ninth Revision, Clinical Modification; ICD-10: ICD, Tenth Revision; IQR: interquartile range; LN: lupus nephritis; MetS: metabolic syndrome; MI: myocardial infarction; NA: not applicable; NR: not reported; PAD: peripheral artery disease; PCI: percutaneous coronary intervention; SLE: systemic lupus erythematosus; SLICC: Systemic Lupus International Collaborating Clinics; SNOMED-CT: Systemized Nomenclature of Medicine-Clinical Terms; TIA: transient ischemic attack.
aStudy quality was assessed using Critical Appraisal Skills Programme (CASP; https://casp-uk.net/) checklists for observational studies.
The CV events of stroke, MI, and CVD were reported as endpoints in 18, 15, and 20 studies, respectively (Table 1). The definition of CVD varied across studies, although most studies included key events such as MI and stroke within a composite CVD outcome. The CV risk factors of hypertension, diabetes, and MetS were reported as endpoints in 7, 3, and 7 studies, respectively. Given the limited number of studies reporting other CV events and CV risk factors (e.g., smoking and dyslipidemia), the MA focused on the CV events of stroke, MI, and CVD, together with the CV risk factors of hypertension, diabetes, and MetS.
Relative risk of stroke, MI, and CVD in patients with SLE compared with the general population
Twelve studies were included in the analysis of the RR of stroke in patients with SLE compared with the general population.14,15,18,20,21,23,27,37,42,44,52 A statistically significantly higher risk of stroke was observed in those with SLE than in the general population (Figure 2(a)). The RR estimates across the studies ranged from 1.61 to 5.34, and the pooled estimate was 2.51 (95% CI: 2.03–3.10). Substantial heterogeneity was observed in the RR estimates across the studies (I2 = 89.6%). As there were differences in the measures reported by most studies, a sensitivity analysis was conducted including only those studies reporting an HR (8 studies).14,15,18,23,37,42,44,56 The pooled estimate was similar to the primary analysis (2.39; 95% CI: 1.96–2.92), although substantial heterogeneity remained (I2 = 90.5%; Supplementary Figure 1). Leave-one-out sensitivity analyses of studies reporting HR resulted in estimates for the RR of stroke ranging from 2.37 to 2.83 (Supplementary Table 4). RR of (a) stroke, (b) MI, and (c) CVD in patients with SLE versus the general population. CI: confidence interval; CVD: cardiovascular disease; df: degrees of freedom; FE: fixed-effects; MI: myocardial infarction; RE: random-effects; RR: relative risk; SLE: systemic lupus erythematosus.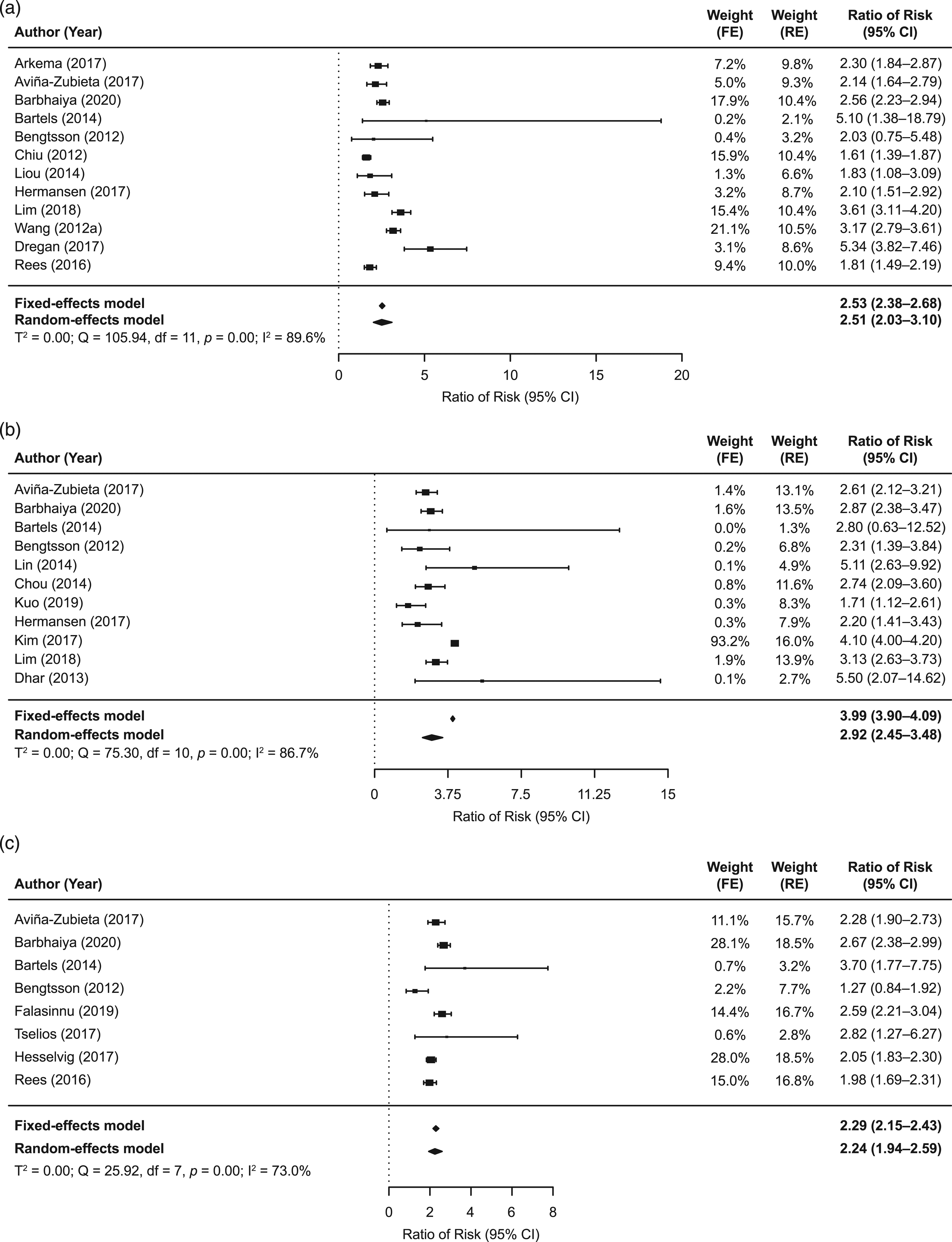
In total, 11 studies were included in the primary analysis of the RR of MI in patients with SLE compared with the general population (Figure 2(b)).15,18,20,21,24,25,39,40,42,43 A statistically significant, 2.9-fold higher risk of MI was seen in those with SLE compared with the general population (RR = 2.92 [95% CI: 2.45–3.48]), with RRs from individual studies ranging from 1.71 to 5.50. Substantial heterogeneity was observed (I2 = 86.7%), possibly related to differences in the measures of RR used in the included studies. To investigate this, a sensitivity analysis including only those studies reporting an HR (n = 7) was conducted,15,18,24,37,40,42,43 resulting in a pooled estimate of 2.76 (95% CI: 2.46–3.10), similar to the primary analysis, but with reduced heterogeneity (I2 = 48.3%) (Supplementary Figure 2).
Eight studies were included in the primary analysis of the RR of CVD in patients with SLE compared with the general population (Figure 2(c)).15,18,20,21,28,38,52,55 A statistically significant, 2.2-fold higher risk of CVD was observed in those with SLE compared with the general population (RR = 2.24 [95% CI: 1.94–2.59]). Substantial heterogeneity (I2 = 73.0%) was observed in the RR estimates across the studies, which ranged from 1.27 to 3.70. A sensitivity analysis using only those studies with CVD definitions of stroke and/or MI (n = 4)15,18,21,38 found a pooled estimate of 2.10 (95% CI: 1.63–2.70), although substantial heterogeneity remained (I2 = 83.7%) (Supplementary Figure 3). To evaluate whether differing measures of risk contributed to heterogeneity, a sensitivity analysis using only those studies that reported HR (n = 4)15,18,38,55 found a pooled estimate of 2.34 (95% CI: 2.00–2.73), but substantial heterogeneity remained (I2 = 71.6%) (Supplementary Figure 4).
Relative risk of hypertension, diabetes, and MetS in patients with SLE compared with the general population
The RR of hypertension was statistically significantly higher in patients with SLE compared with the general population (2.70 [95% CI: 1.48–4.92]; 7 studies; Supplementary Figure 5(a)).19,21,22,25,39,51,53 Heterogeneity was high (I2 = 99.7%); leave-one-out sensitivity analyses confirmed the estimated RR (range: 2.00–3.13), but substantial heterogeneity remained (I2 = 90.6–99.8%) (Supplementary Table 5). Although not statistically significant, the RRs of diabetes (1.24 [95% CI: 0.78–1.96]; 3 studies; Supplementary Figure 5(b))25,27,35 and MetS (1.49 [95% CI: 0.95–2.33]; 7 studies; Supplementary Figure 5(c))22,29,36,46,47,50,51 also indicated an elevated risk of these factors in patients with SLE compared with non-SLE controls. Heterogeneity was moderate in the diabetes analysis (I2 = 30.4%) and substantial in the MetS analysis (I2 = 69.6%); leave-one-out sensitivity analysis that excluded 1 study 36 with a notably lower RR resulted in an estimated MetS RR of 1.74 (95% CI: 1.32–2.30), with I2 = 0%.
AR of stroke, MI, and CVD in patients with SLE
In total, 7 studies were included in the primary analysis of the AR (i.e., incidence proportion) of stroke in patients with SLE (Figure 3(a)).18,30,32,33,42,44,54 The AR of stroke in patients with SLE ranged from 0.01 to 0.06, and the pooled estimate was 0.03 (95% CI: 0.02–0.05). Substantial heterogeneity was observed in the AR estimates across the studies (I2 = 98%). The results of the leave-one-out sensitivity analysis resulted in estimates ranging from 0.03 to 0.04 (Supplementary Table 6). Because the heterogeneity could be due to differences in the definition of stroke, a sensitivity analysis was conducted using only those studies where stroke definitions were reported and were not likely to include TIAs (n = 3).18,30,33 This estimate resulted in a pooled AR of 0.03 (95% CI: 0.01–0.06), although substantial heterogeneity remained (I2 = 94%) (Supplementary Figure 6). A sensitivity analysis was also conducted to evaluate the impact of disease duration on the AR of stroke, which included 4 studies reporting disease duration.30,32,44,54 The pooled estimate for this sensitivity analysis was 0.05 (95% CI: 0.04–0.06), with moderate heterogeneity (I2 = 57%) (Supplementary Figure 7). Visual assessment suggests a possible trend for higher risk with longer disease duration, but the small number of studies limits interpretation. AR of (a) stroke, (b) MI, and (c) CVD in patients with SLE. AR: absolute risk; CI: confidence interval; CVD: cardiovascular disease; FE: fixed-effects; FU: follow-up; MI: myocardial infarction; RE: random-effects; SLE: systemic lupus erythematosus.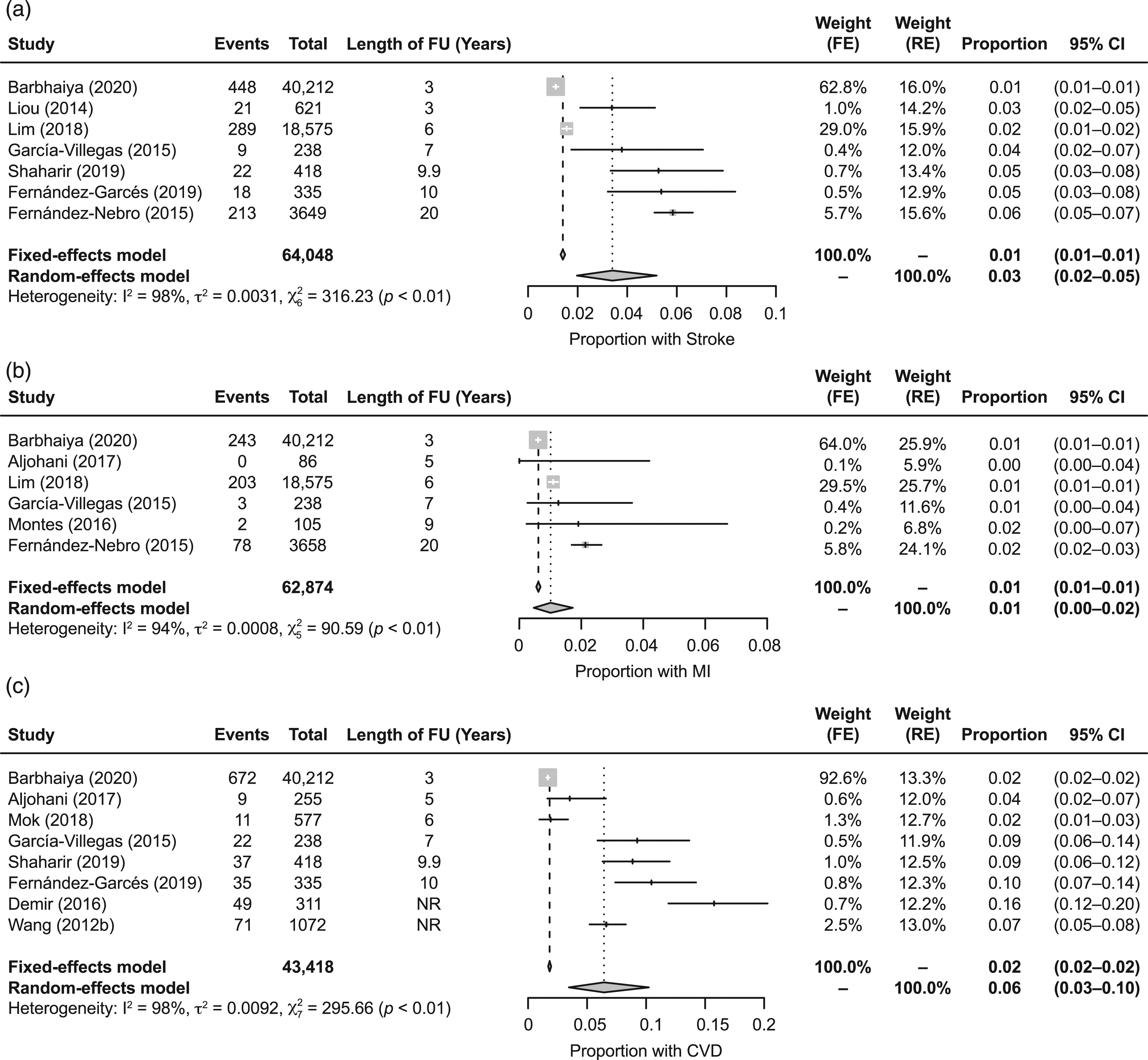
In total, 6 studies were included in the primary analysis for the AR of MI in patients with SLE (Figure 3(b)).13,18,32,33,42,49 The AR of MI ranged from 0.00 to 0.02, with a pooled estimate of 0.01 (95% CI: 0.00–0.02). Substantial heterogeneity was observed in the AR estimates across the studies (I2 = 94%). A leave-one-out sensitivity analysis resulted in estimates of 0.01 for all analyses (Supplementary Table 7).
In total, 8 studies were included in the primary analysis of the AR of CVD in patients with SLE (Figure 3(c)).13,18,26,30,33,48,54,57 The AR of CVD ranged from 0.02 to 0.16, with a pooled estimate of 0.06 (95% CI: 0.03–0.10). Substantial heterogeneity was observed in the AR estimates across the studies (I2 = 98%). To evaluate whether the heterogeneity could be due to differences in how CVD was defined, a sensitivity analysis was conducted with only those studies whose CVD definitions were stroke and/or MI (n = 4).18,26,33,54 The pooled estimate for this sensitivity analysis was 0.07 (95% CI: 0.02–0.14), although substantial heterogeneity remained (I2 = 98%) (Supplementary Figure 8). To further evaluate the heterogeneity and robustness of the pooled estimates, a sensitivity analysis was also conducted with only those studies assessed as high quality (n = 5).13,18,30,33,48 The pooled AR estimate for this sensitivity analysis was 0.05 (95% CI: 0.02–0.09), with substantial heterogeneity remaining (I2 = 95%) (Supplementary Figure 9).
Incidence rate of stroke, MI, and CVD in patients with SLE
Ten studies were included in the primary analysis of IR of stroke in patients with SLE (Figure 4(a)).14,15,17,18,34,37,42,44,52,56 The IR of stroke ranged from 1.25 to 8.18 per 1000 PY, with a pooled estimate of 4.72 (95% CI: 3.35–6.32). Substantial heterogeneity was observed (I2 = 99%). Pooled estimates from the leave-one-out sensitivity analysis ranged from 4.43 to 5.22 (Supplementary Table 8). Further sensitivity analysis was conducted using only those studies where stroke definitions were reported and were not likely to include TIAs (n = 6)14,15,17,18,34,37; in this analysis, the pooled estimate for the IR of stroke was 4.94 (95% CI: 3.05–7.27), but substantial heterogeneity remained (I2 = 99%) (Supplementary Figure 10). IR of (a) stroke, (b) MI, and (c) CVD in patients with SLE. CI: confidence interval; CVD: cardiovascular disease; FE: fixed-effects; IR: incidence rate; MI: myocardial infarction; PY: person-years; RE: random-effects; SLE: systemic lupus erythematosus.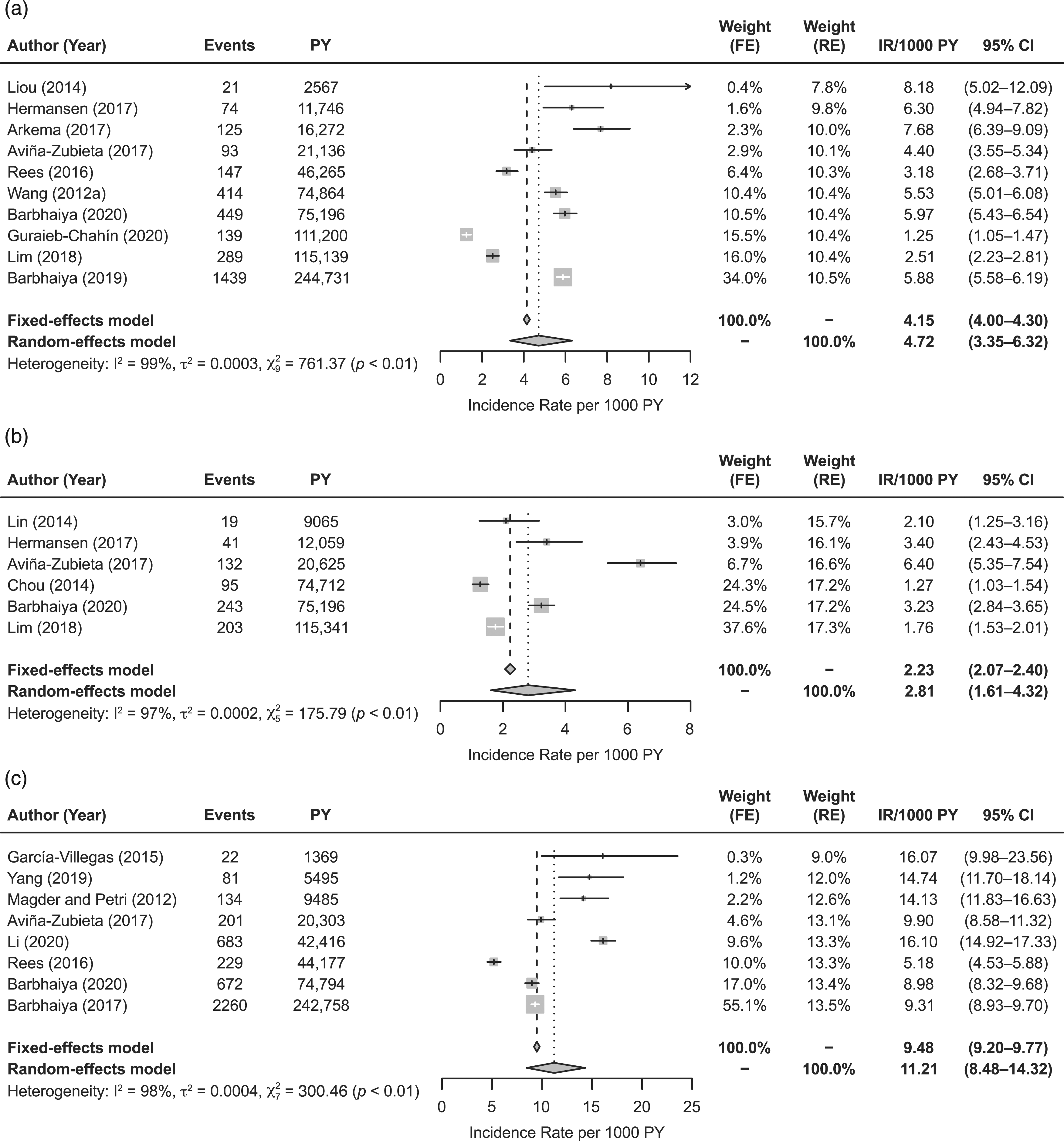
Six studies were included in the primary analysis of the IR of MI in patients with SLE (Figure 4(b)).15,18,24,37,42,43 The IR of MI ranged from 1.27 to 6.40 per 1000 PY, with a pooled estimate of 2.81 (95% CI: 1.61–4.32). Substantial heterogeneity was observed (I2 = 97%). The leave-one-out sensitivity analysis resulted in estimates ranging from 2.25 to 3.20 and with substantial heterogeneity remaining (I2 ranging from 95.1% to 97.7%) (Supplementary Table 9).
In total, 8 studies were included in the primary analysis of the IR of CVD in patients with SLE (Figure 4(c)).15,16,18,33,41,45,52,58 The IR of CVD ranged from 5.18 to 16.10 per 1000 PY, with a pooled estimate of 11.21 (95% CI: 8.48–14.32). Substantial heterogeneity was observed (I2 = 98%). A sensitivity analysis including only those studies with CVD definitions of stroke and/or MI found an estimate of 9.26 (95% CI: 8.92–9.61) with no heterogeneity (I2 = 0%) (Supplementary Figure 11). The leave-one-out sensitivity analysis resulted in estimates ranging from 10.51 to 12.21 (Supplementary Table 10).
Subgroup analyses of RR of stroke, MI, and CVD by age and sex
Meta-analyses were not conducted for CV events by age and sex because of limited data. As an alternative, results from individual studies are shown. Although studies used different age group definitions, the RRs of stroke (5 studies; Figure 5(a)),14,15,18,37,56 MI (5 studies; Figure 5(b)),15,18,21,24,37 and CVD (3 studies; Figure 5(c))15,18,38 were generally higher in younger patients with SLE compared with older patients with SLE, especially in patients younger than 40 years, when compared with age-matched non-SLE controls. RR of (a) stroke by age, (b) MI by age, (c) CVD by age, (d) stroke by sex, and (e) MI by sex. CI: confidence interval; CVD: cardiovascular disease; MI: myocardial infarction; RR: relative risk.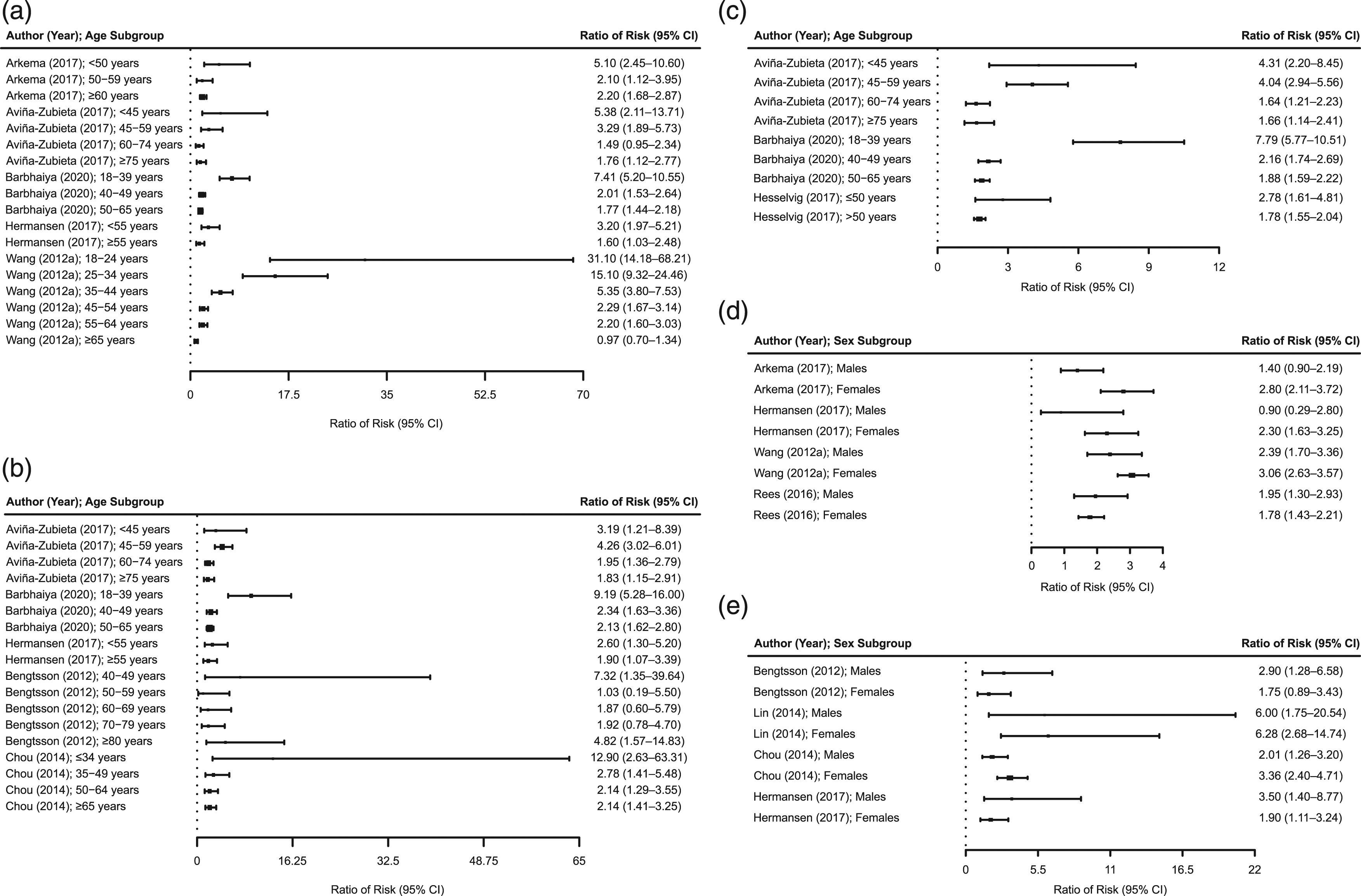
For sex, the RR of stroke in patients with SLE compared with the general population appeared higher in women than in men in 3 of 4 included studies (Figure 5(d)).14,37,52,56 In contrast, there was no consistent pattern in the RR of MI by sex among 4 included studies (Figure 5(e)).21,24,37,43 Only 2 studies reported CVD by sex;31,58 therefore, these studies are not displayed.
Discussion
The main objective of this MA was to provide pooled estimates of the RR of CV events in patients with SLE compared with the general population or suitable proxies. Despite substantial heterogeneity, there was a 2- to 3-fold increased risk of stroke, MI, and CVD with SLE compared with the general population, consistent with previous MAs.4,5,59,60 Yazdany et al. reported that the RRs of stroke and MI were significantly higher in those with SLE compared with the general population (2.13 [95% CI: 1.73–2.61] and 2.99 [95% CI: 2.34–3.82], respectively). 5 Similarly, Gu et al. reported an RR of 1.96 (95% CI: 1.52–2.53) for stroke and an RR of 3.04 (95% CI: 1.81–5.11) for MI. 59 Lu et al. reported an RR of 2.30 (95% CI: 1.52–3.50) for stroke, 2.66 (95% CI: 1.97–3.59) for MI, and 2.35 (95% CI: 1.95–2.84) for CVD, 4 whereas Restivo et al., who only included prospective cohort studies published between 2015 and 2019, reported that patients with SLE had an elevated risk of symptomatic CV events (RR = 1.98 [95% CI: 1.18–3.31]). 60 Interestingly, these previous MAs found a similarly high level of heterogeneity as in the current MA. Despite this heterogeneity, the consistency of RR estimates from the primary and sensitivity analyses, and similar findings across the published studies, supports the elevated risk of CV events in patients with SLE. Moreover, visual assessment of the forest plots of subgroup analyses by age suggested that the RR of stroke, MI, and CVD compared with an age-matched non-SLE population was greater in younger patients with SLE compared with older patients with SLE, consistent with the findings of Yazdany et al. for stroke. 5 Data on the RR of stroke, MI, and CVD in patients with SLE versus sex-matched non-SLE cohorts were limited.
Unlike previous MAs, the current analysis also estimated the AR and IR of CV events in patients with SLE. The estimated pooled ARs for stroke, MI, and CVD were 0.03, 0.01, and 0.06, respectively, which are generally higher than observed in the general population without SLE.18,42,44 However, differences were seen in the AR across the studies, especially for CVD, which may relate to the diversity of outcomes included in this category. There was a visual trend for increased AR of CVD with longer follow-up, although visual assessment of the data did not indicate this relationship for stroke or MI (data not shown). The pooled estimates for the IR of stroke and MI in this MA (4.72 per 1000 PY and 2.81 per 1000 PY, respectively) were similar to those reported by Gu et al. (4.4 per 1000 PY and 2.0 per 1000 PY, respectively). 59 As with AR, the IR of these CV events, as well as for CVD (11.21 per 1000 PY in this MA), was higher in patients with SLE than in the general population.14,15,18,24,37,42,43,44,52,56
The RR of the CV risk factors of hypertension, diabetes, and MetS was also elevated in patients with SLE compared with non-SLE controls (2.70 [1.48–4.92], 1.24 [0.78–1.96], and 1.49 [0.95–2.33], respectively), although statistical significance was only observed for hypertension. Our MA results are similar to those of Lu et al., who reported a significantly increased RR for hypertension but not diabetes (2.31 [1.62–3.29] and 1.19 [0.90–1.58], respectively). 4 Although traditional CV risk factors are likely to contribute to increased risk of CV events in patients with SLE, the underlying mechanisms are complex and are believed to also include inflammation caused by SLE, the presence of antiphospholipid antibodies, and SLE-specific risk factors (e.g., SLE disease activity and duration, renal disease, organ damage [as measured by the Systemic Lupus International Collaborating Clinics/American College of Rheumatology Damage Index], and corticosteroid therapy).1,61,62
To our knowledge, this is the first SLR and MA to assess the risk of not only stroke and MI but also general CVD, including subgroups based on age and sex, as well as the risk of CVD risk factors such as hypertension, diabetes, and MetS across multiple observational study types in patients with SLE compared with the general population. As noted above, this is also the first MA to report pooled estimates of AR and IR for CV events in patients with SLE. Moreover, this MA applied a single protocol to the investigation of multiple endpoints and epidemiologic measures. An additional strength of this MA is that it uses evidence from multiple observational study types to investigate the risk of CV events in adults with SLE compared with the general population or other control groups. By using multiple study types, the evidence base is widened and more comprehensive. Although most included studies were considered high quality, the potential for bias is a limitation of all observational studies. Moreover, the inclusion of multiple study types likely contributed to the heterogeneity observed for most endpoints. This heterogeneity may have resulted not only from differences in study design but also from differences in patient characteristics, study quality, length of follow-up, and control group selection and matching across the studies, as well as the use of different definitions of events, especially for CVD and, to a lesser extent, stroke. However, although the results from sensitivity analyses of CVD and stroke using more comparable definitions were similar to the primary analysis results, heterogeneity remained, and caution should be used in the interpretation of the findings. Finally, this MA was unable to examine some factors that potentially affect CVD risk in patients with SLE, such as smoking and dyslipidemia, because of a limited number of studies on these factors.
Conclusions
In summary, evidence from this MA suggests that there is an increased risk and incidence of stroke, MI, and CVD in patients with SLE compared with the general population. Despite study differences and heterogeneity in the pooled estimates, the results of sensitivity analyses were similar to those of the primary analyses, supporting the robustness of the estimates of risk. Given the high risk of CV mortality and morbidity in patients with SLE, especially in younger patients, it is imperative that risk factors are routinely assessed and appropriate preventive measures are integrated into the management plan. Additionally, more rigorous and comparable prospective cohort studies should be conducted to further evaluate CV risk and the impact of disease- and patient-related risk factors.
Supplemental Material
Supplemental material - Cardiovascular events and risk in patients with systemic lupus erythematosus: Systematic literature review and meta-analysis
Supplemental material for Cardiovascular events and risk in patients with systemic lupus erythematosus: Systematic literature review and meta-analysis by Natalia Bello, Kristin J Meyers, Jennifer Workman, Louise Hartley, and Maureen McMahon in Lupus.
Footnotes
Acknowledgments
The authors would like to thank Emma Hawe of RTI Health Solutions, who was the project lead on the analyses. Medical writing assistance was provided by Roisin McCarthy of Eli Lilly and Company and Rebecca Lew, PhD, CMPP, of ProScribe – Envision Pharma Group, and was funded by Eli Lilly and Company. ProScribe’s services complied with international guidelines for Good Publication Practice.
Author contributions
All authors participated in the interpretation of study results and in the drafting, critical revision, and approval of the final version of the manuscript. LH, JW, KJM, and NB developed the study design, and LH was involved in the concept, design, and conduct of the meta-analyses.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NB, KJM, and JW are employees and minor shareholders of Eli Lilly and Company. LH is an employee of RTI Health Solutions, which was contracted by Eli Lilly and Company to conduct the meta-analyses. MM reports receiving funding from Eli Lilly and Company for medical writing and statistical support for this study and consulting and/or speaker fees from AstraZeneca, Aurinia Pharmaceuticals, Bristol Myers Squibb, Eli Lilly and Company, and GlaxoSmithKline outside of this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Eli Lilly and Company, manufacturer/licensee of baricitinib.
Role of the sponsor
Eli Lilly and Company was involved in the study design, data collection, data analysis, and preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
