Abstract
Fatigue is a frequently reported and disabling symptom in patients with systemic lupus erythematosus (SLE). The management of Lupus-associated fatigue (LAF) is complex and requires the exclusion of disease activity and comorbidities as potentially treatable causes. Standard of care recommendations includes psychological counselling and regular physical activity. However, many SLE patients still report the persistence of LAF despite these measures. Therefore, pharmacological management may be required, which has been insufficiently investigated in clinical trials. Here, we report two patients who improved with pharmacological treatment with modafinil (MODA), a central nervous system stimulant. Both patients had an overall low disease activity (SLEDAI-2K score of 0). Their FACIT fatigue scores were 15 and 20, respectively (with a maximum score of 52, where 52 indicates the best quality of life). With MODA treatment, the first patient’s FACIT fatigue score improved from 15 to 42, the second patient’s score from 20 to 37. In the latter patient, it returned to 21 after stopping the drug and increased back again to 37 after re-treatment.
In conclusion, our report demonstrates, for the first time, that MODA treatment is a potential pharmacological treatment option in selected patients with LAF. Clinical trials in SLE are required to confirm our observations.
Keywords
Background
Fatigue is a frequent and disabling symptom in patients with systemic lupus erythematosus (SLE). As recently reviewed by Mertz et al., the diagnostic approach and treatment of this condition represent a major challenge in clinical practice. 1 A comprehensive assessment of treatable causes and associated comorbidities is required. Different clusters of patients with lupus-associated fatigue (LAF) have recently been identified in a transnational registry study. 2 Briefly, three clusters were identified: the largest cluster showed low disease activity, moderate fatigue, and low anxiety or depression. 2 This population reflects the majority of patients also seen in our clinics. While a comprehensive assessment and targeted approach, including physical activity and psychological intervention, are useful for many patients, some patients may, nevertheless, benefit from pharmacological therapy. Reports of medical treatments for LAF are limited. Here, we report the effect of modafinil (MODA) for LAF in two patients, representing a novel and, as of yet, not reported approach to LAF.
Pharmacology of modafinil
MODA is a central nervous system stimulant with wake-promoting actions. The racemic compound MODA and its active enantiomer armodafinil, which has a longer duration of action, have been demonstrated to improve wakefulness in conditions with excessive daytime sleepiness (e.g. narcolepsy, obstructive sleep apnea, shift work sleep disorder, and idiopathic hypersomnia). 3
In Europe, MODA is currently only indicated for the treatment of narcolepsy at a daily dose of 200 mg (up to 400 mg). The lowest effective starting dose is recommended to minimize the risk of drug-related adverse effects. 4 In addition to neuropsychiatric complications (see below), the development of skin and hypersensitivity reactions have rarely been reported with armodafinil. 5 Cardiovascular adverse events have also been documented in association with MODA because of its presumed effect in rising catecholamine levels, mainly attributable to adrenomedullary discharge. 6 Despite the fact that clinical studies gave conflicting results in this regard,6–9 MODA should not be used in patients with uncontrolled hypertension or cardiac arrhythmias as per recommendation of the European Medicines Agency (EMA). 4 Wake-promoting is related to monoaminergic effects, increasing cortical activity in the frontal lobe, enhancing the actions of histamine, norepinephrine, serotonin, dopamine, and orexin systems. 10 The precise mechanisms of action are unknown, but they have been shown to increase dopaminergic neurotransmission by blocking dopamine transporters. 3 Potential side-effects include alterations of mood, perception, thinking, and feeling. 3 However, increases in dopamine levels are milder and longer-lasting than other psychostimulants, making them well-tolerated with attenuated withdrawal symptoms and low addictive potential. 10 Despite having the same half-life (∼13 h), MODA and the R-isomer armodafinil differ in their elimination profiles. 11 Conflicting results on the efficacy of MODA in multiple sclerosis-related fatigue have been reported. 12 On the other hand, armodafinil was associated with improved sarcoidosis-associated fatigue compared with placebo. 13
Case reports
Assessment of fatigue and disease activity
We assessed LAF with the FACIT fatigue score, a self-completed 13-item questionnaire designed and validated to evaluate fatigue in chronic diseases. Each question refers to the patient’s experience during the last seven days, expressed in a 5-point Likert scale format. The items are pooled to generate a total score in which higher values represent better quality of life (range 0–52). As a measure of disease activity, the SLEDAI-2K score was reported as previously published. 14 Both patients provided written informed consent.
Case description patient #1
The first patient is a 50-year-old Caucasian female with a 10-year history of SLE. The diagnosis was based on arthritis, photosensitivity, discoid rash, Raynaud’s syndrome, positive antinuclear antibodies (ANA), and anti-histone antibodies. There was no evidence of neuropsychiatric lupus or lupus nephritis. Her current treatment included 2.5 mg of prednisolone, hydroxychloroquine (HCQ), azathioprine (AZA), and vitamin D. There was no evidence of serological activity. Overall, her SLEDAI-2K score was zero. Her main symptom was significant fatigue, interfering with social life and her ability to work. Other concurrent disorders, such as secondary Sjögren’s syndrome, diabetes mellitus or thyroid disease, as causes of fatigue, were ruled out with physical examination and basic laboratory examinations. After supportive treatment and counselling (physical activity and psychological support), a shared decision was made to add MODA with a starting dose of 100 mg/day (increasing to 200 mg/day after one week) to her pharmacological management. Before the initiation of treatment, depressive symptoms were assessed during the routine clinical visit but were not reported by the patient. The fatigue levels were recorded using the FACIT fatigue score before and after five months of treatment (Figure 1). She improved from a baseline score of 15 to 42 after 5 months, which is stable at 42 after 10 months of follow-up. She continues treatment without side effects. Of note, no change in physical activity occurred during the treatment period.
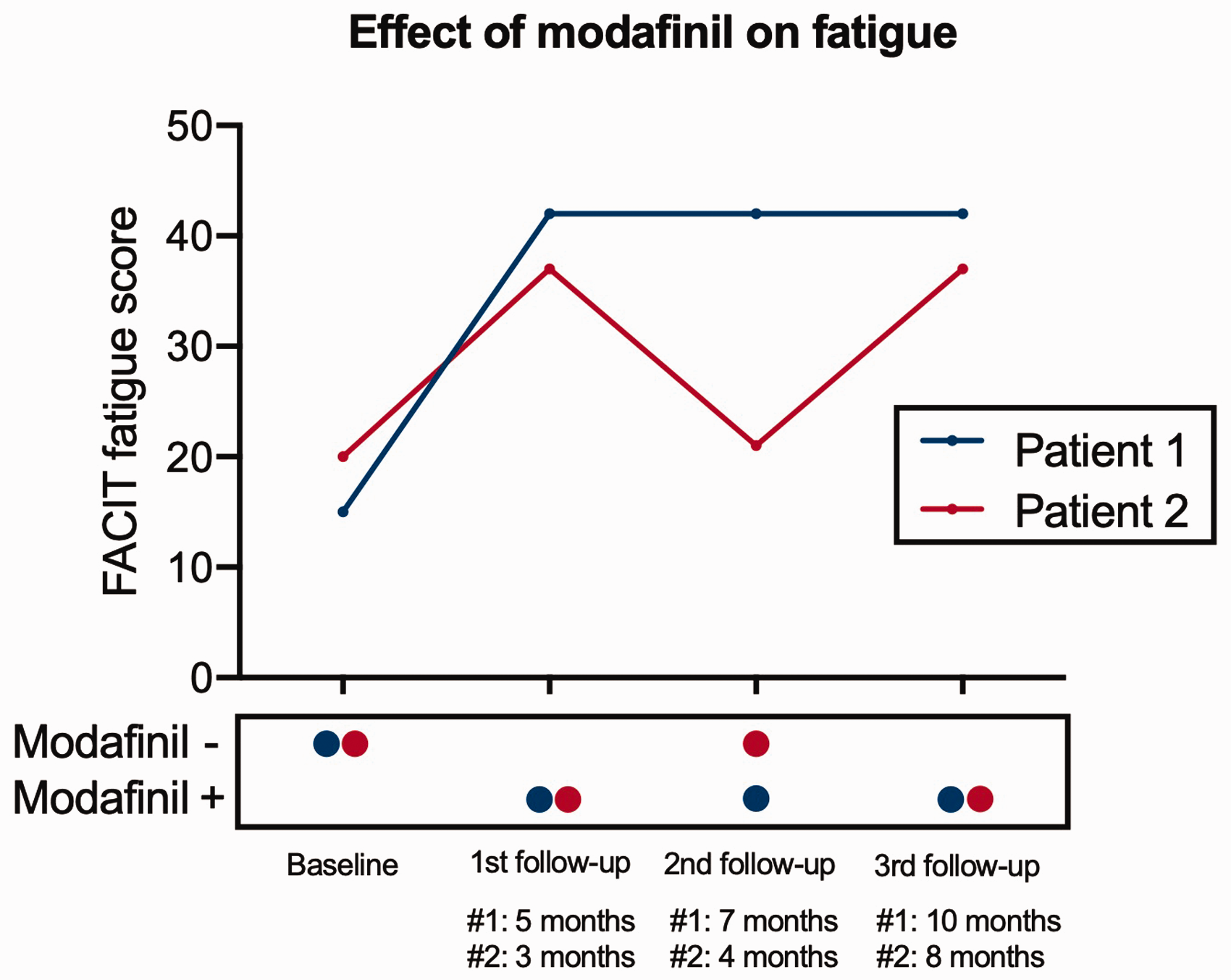
FACIT fatigue scores of two patients before and after treatment initiation with modafinil. In patient #1 (blue line/dots), scores improved from 15 to 42 and stayed stable with continued drug intake. In patient #2 (red line/dots), scores improved from 20 to 37, but almost returned to baseline (21) after stopping the drug. After continuing modafinil, scores again improved to 37 after 8 months of follow up.
Case description patient #2
The second patient is a 49-year-old Caucasian female with an 11-year history of SLE. She had very similar clinical manifestations as patient #1 (arthritis, photosensitivity, discoid rash, positive ANA, and anti-double-stranded DNA antibodies). Her disease was well-controlled with HCQ, AZA, 5 mg/day of prednisolone, and vitamin D. Her concurrent treatment included duloxetine for mild depression; she reported stable symptoms with duloxetine treatment. The SLEDAI-2K was zero at her most recent follow-up visit to the clinic before the initiation of MODA. Other concurrent disorders potentially contributing to fatigue were ruled out. She had been participating in a structured exercise program for several years without any relevant effect on fatigue. After considering all therapeutic options, MODA was initiated at a dose of 100 mg per day. After three months, she stopped taking the drug due to a lack of continued prescription. Her FACIT fatigue scores improved from 20 at baseline to 37 at the three-month assessment but returned almost to baseline (21) one month after stopping the drug (Figure 1
Discussion
We here report two cases of patients with significant LAF severe enough to interfere with activities of daily living. Overall, our two cases improved with MODA treatment. In SLE, the minimal clinically important difference for FACIT fatigue has been estimated at a change of 5.9 points; 15 our patients showed an improvement of 27 and 17 points, respectively.
For the management of LAF, one crucial question must be addressed: is LAF a result of persisting disease activity or accompanying symptom of SLE itself? As recently shown by Arnaud et al., 2 most patients (about two-thirds in their study) with fatigue as the predominant symptom have a low SLE disease activity and low rates of depression and anxiety. According to this ana-lysis, the patients described in our report would fit in this cluster.
MODA has been described as useful in several conditions, such as multiple sclerosis and sarcoidosis-associated fatigue. In a meta-analysis of the efficacy of MODA for multiple sclerosis-associated fatigue, as measured by the Modified Fatigue Impact Scale score, MODA was superior to placebo. 12 In sarcoidosis-associated fatigue, armodafinil, which is pharmacologically similar to MODA, has been shown to improve the Fatigue Assessment Scale compared to placebo. 13 Surprisingly, we have not identified any reports on MODA or armodafinil in SLE patients by a scoping literature search.
Overall, our case reports supports the use of MODA for severe LAF in selected patients. Nevertheless, there are important limitations: first, our findings in two patients are not generalizable until they are confirmed in larger, placebo-controlled trials. Second, we did not formally assess anxiety or depression as comorbidities but interrogated the patients before treatment initiation for suggestive symptoms. One patient already received duloxetine for mild depression but depressive symptoms had been stable throughout the treatment and follow-up.
In conclusion, based on our experience, MODA is a potential candidate drug to be investigated in clinical trials for disabling LAF, especially in the absence of associated and treatable comorbidities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
