Abstract
Systemic lupus erythematosus (SLE) is an autoimmune disease that affects multiple organs. SLE can affect the lung, the pulmonary vasculature, and the pleura. A 38-year-old female with limb pain and ecchymosis who later developed pulmonary thromboembolism and alveolar hemorrhage is presented here. Clinical, imaging, laboratory, and histopathological evidence is presented. The patient met the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) criteria for SLE. Furthermore, the patient had a Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) score of 35; thus, indicating severe disease. This case is an example of concomitant venous and arterial lung complications in an SLE patient.
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease that affects multiple organs. It is characterized by the production of different antibodies (e.g., anti-nuclear antibodies, ANA, and/or anti-dsDNA antibodies, anti-Ro/La, anti-RNP, and anti-Sm antibodies), 1 as well as having different clinical presentations (e.g., glomerulonephritis, cytopenia, arthritis, and photosensitive rash). 2 The prevalence of SLE has been estimated to be 30–50 per 100,000, which is equivalent to 500,000 patients in Europe and 250,000 in the United States of North America. 3 The respiratory system can be involved in up to 50–70% of SLE patients and be the initial manifestation in 4–5% of the cases. 4 SLE can affect various structures of the respiratory system: 1) the lung (i.e., interstitial pneumonia, bronchiolitis obliterans with pneumonia, acute lupus pneumonitis); 2) the vasculature (i.e., pulmonary hypertension, alveolar hemorrhage, reversible acute hypoxemia, and antiphospholipid syndrome); and 3) the pleura.5,6 Pleurisy can be present in up to 60% of SLE, while chronic interstitial pneumonia is 3–13%, and alveolar hemorrhage is less than 2%. 7 The risk of complications and mortality depend on the type and extent of respiratory involvement and the presence of comorbidities. 4
In the case presented below, a patient with SLE debuted with a clinical pulmonary involvement; ultimately, suffering from pulmonary thromboembolism and alveolar hemorrhage. In the discussion, we provide the reader with the relevant literature to put this case into clinical context. This case report should provide the clinician with an example of a rare presentation of SLE involving concomitant venous and arterial pulmonary complications.
Case presentation
A 38-year-old female arrived at the Emergency Department with oppressive pain in the right pelvic limb with a 6/10 intensity on the analogous visual scale, with increased volume and ecchymosis at the posterior tibial region. The patient did not allude to any exacerbating or mitigating causes. The patient’s family history included a mother with type 2 diabetes and a father with arterial hypertension (AH); other relevant aspects of family history were questioned and denied. Among the patient’s personal history, tobacco consumption is reported for more than 10 years at a rate of three cigarettes per day (i.e., 30 pack-years), but currently, the patient refers to be a non-smoker. The patient denied a history of allergies, blood transfusions, recent travel, tattoos, and piercings. The patient had a 2-year history of AH, treated with calcium channel blockers, angiotensin II receptor antagonists, and beta-blockers. The patient also referred a personal history of deep vein thrombosis 2-years prior treated with anticoagulant (i.e., acenocoumarin, weaned off for over 12 months). The patient had no history of lung disease, asthma during childhood, and chronic or degenerative disease. Obstetric history includes six gestations, one miscarriage, one cesarean delivery, and four normal deliveries.
Upon admission, the patient had the following vital signs: blood pressure 110/80 mmHg; heart rate 110 bpm; respiratory rate 20 rpm; oxygen saturation of 88% with noninvasive ventilation at a rate of 10 L/min; body temperature 37°C. We found a patient with a Glasgow coma score of 15 points, somnolent, without focal neurologic deficits nor meningeal signs, aware of her environment, with reference to place, time, and people. Upon inspection, abdominal dissociation and accessory muscles use were noted. The thorax had decreased expansion without vibrations or fremitus during palpation. No asymmetries or abnormal findings in tone intensity, pitch, duration, and quality through direct percussion. Precordial auscultation reveals tachycardia 110 bpm, heart sounds of good intensity without extra heart sounds. The lower right extremity with edema, hyperemia, increased local temperature with ecchymosis, and fovea ++/+++. The Homan’s sign and Pratt test were positive for deep vein thrombosis; Lowenberg’s and Bancroft’s signs were not present. Wells score of 4 (i.e., 1), Calf swelling >3 cm compared to the other leg, 2) localized tenderness along the deep venous system, 3) pitting edema, confined to symptomatic leg, and 4) previously documented DVT was reported for deep vein thrombosis (i.e., high risk).
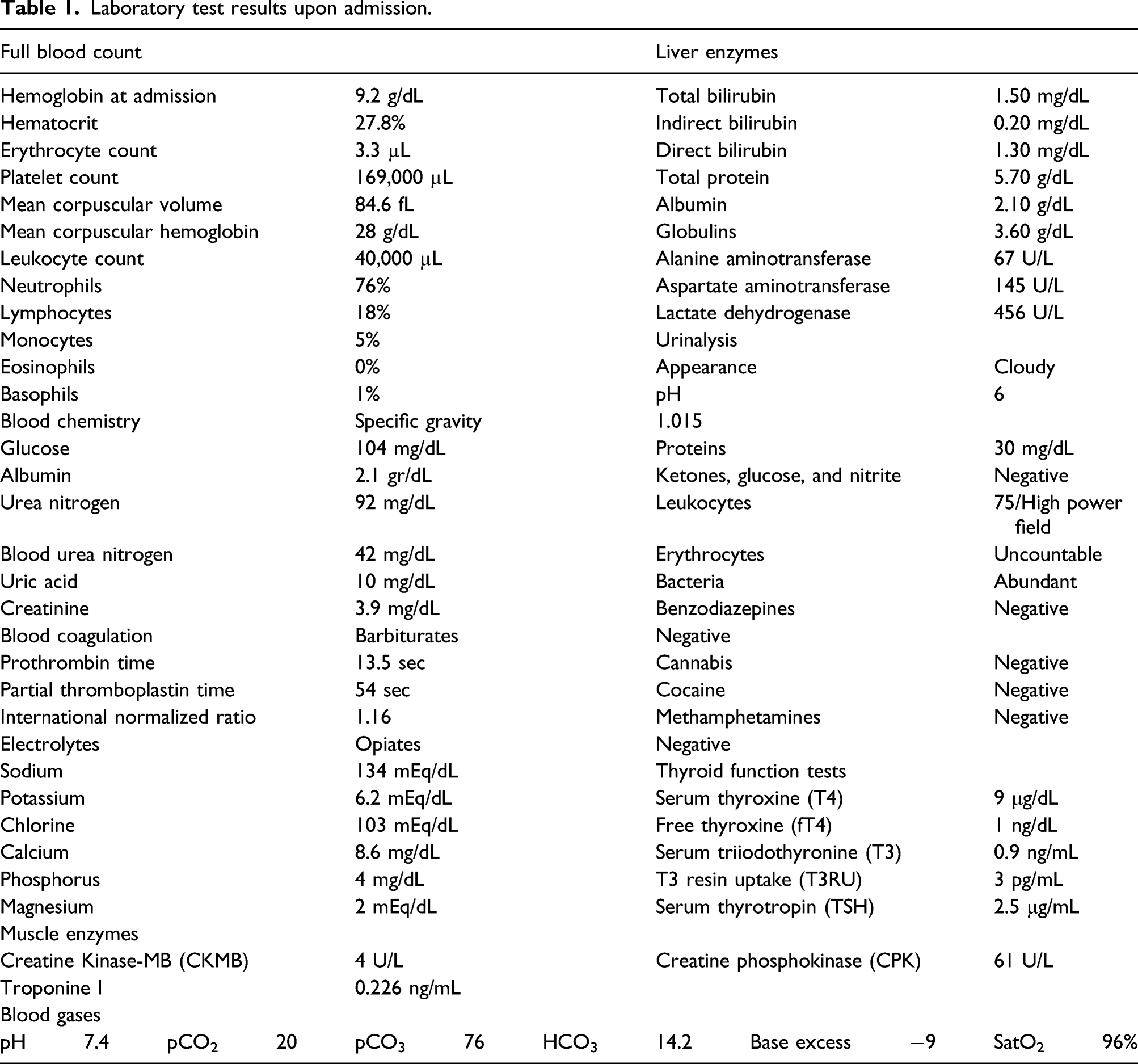
Laboratory test results upon admission.
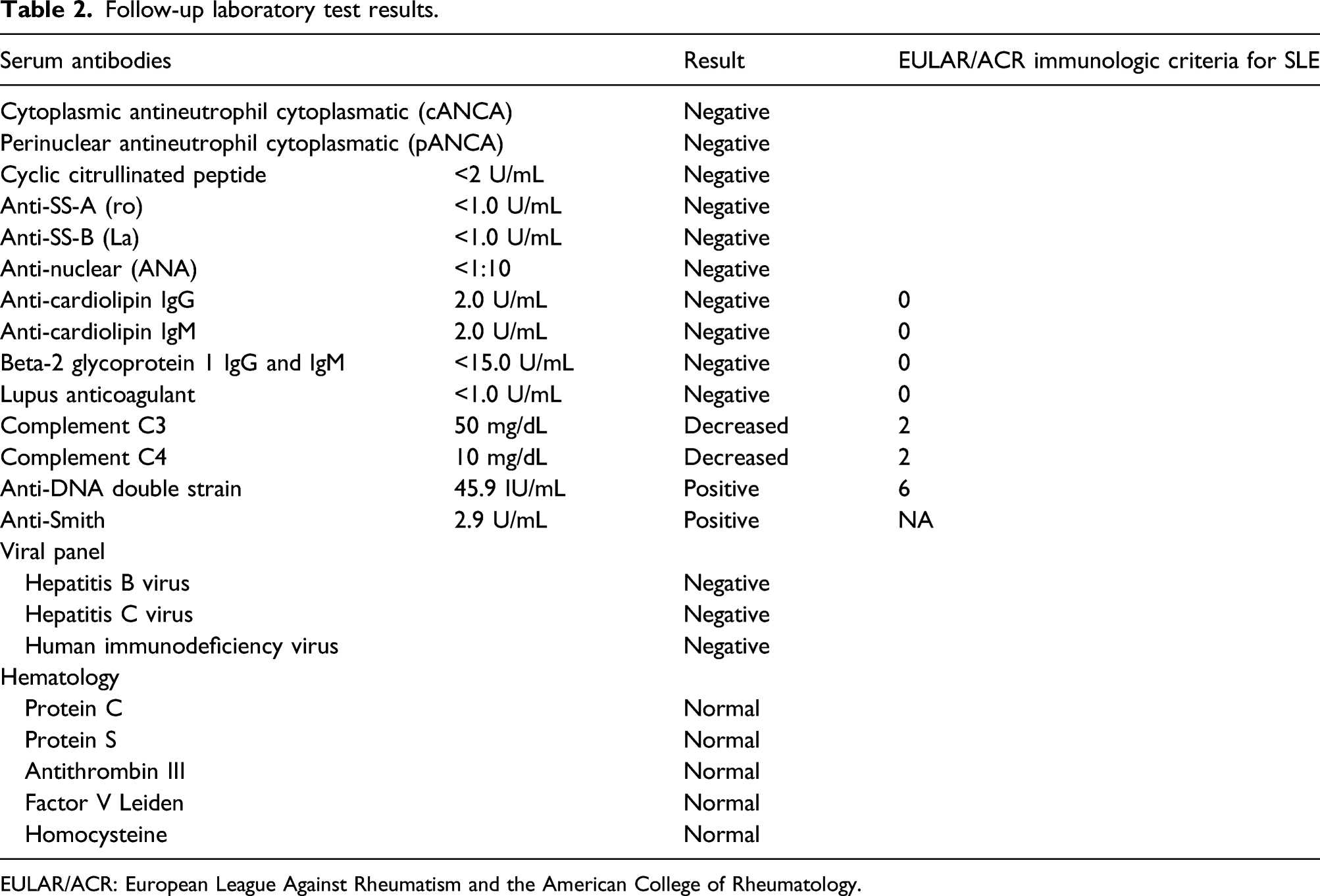
Follow-up laboratory test results.
EULAR/ACR: European League Against Rheumatism and the American College of Rheumatology.
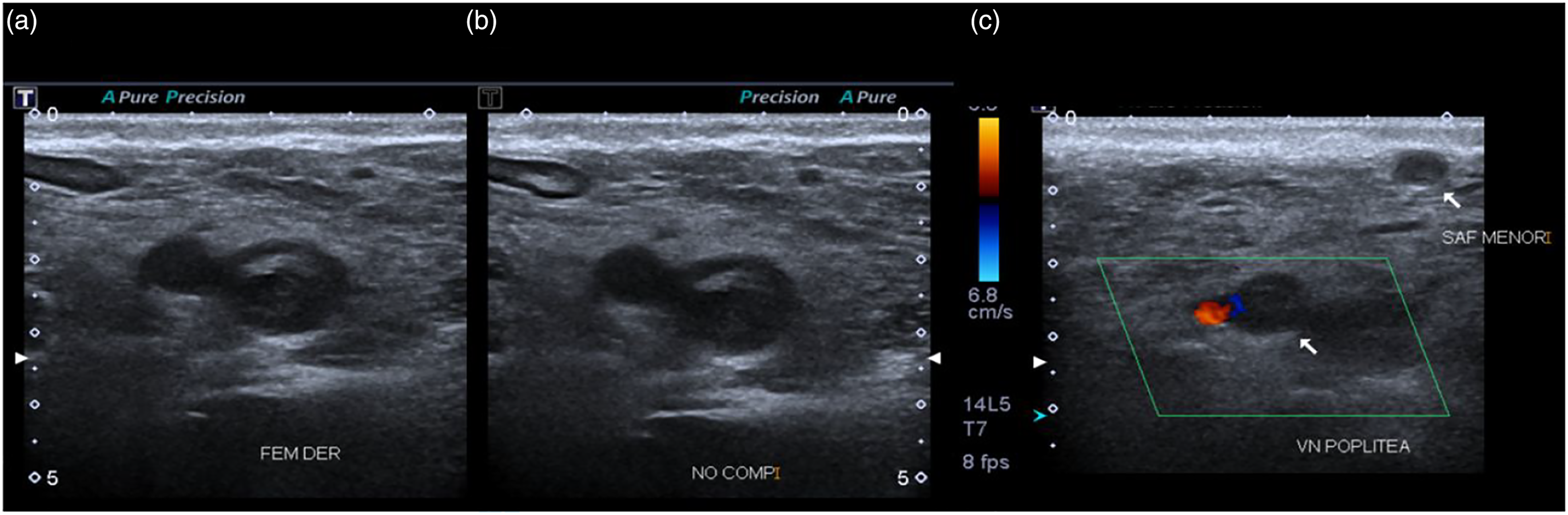
Venous ultrasonography showing thrombi.
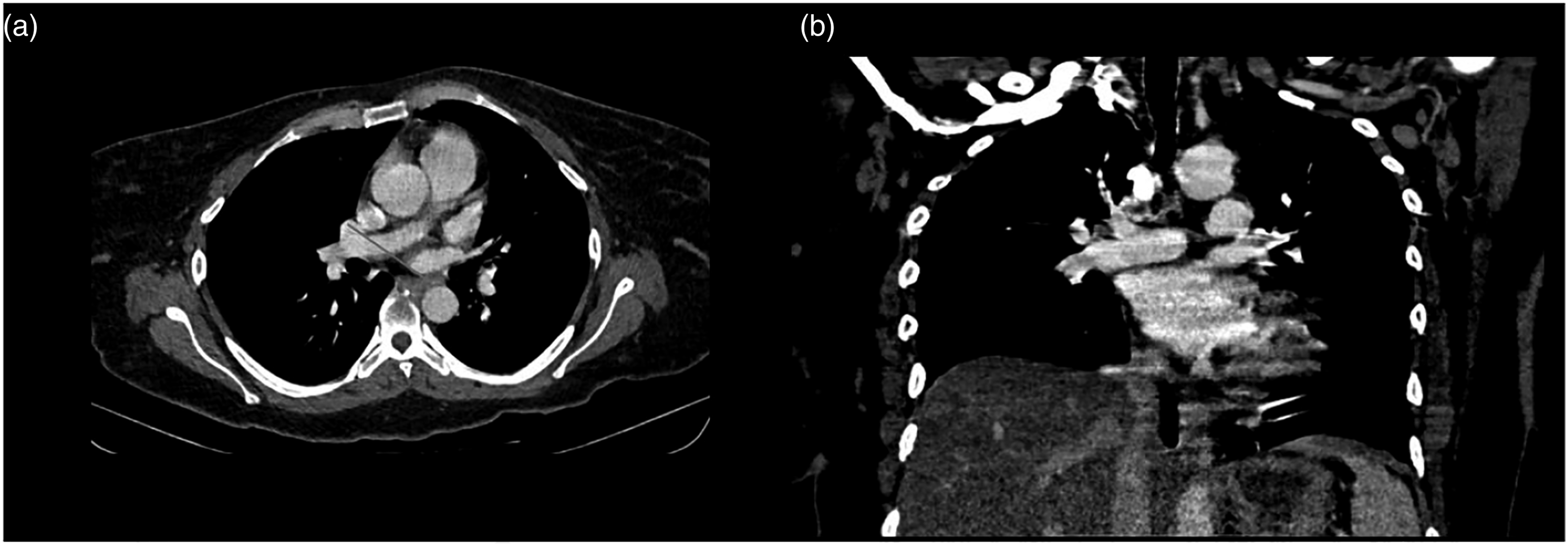
Computed tomography angiography of pulmonary vasculature showing embolism. Computed tomography angiography of the pulmonary vasculature.
Clinical evolution
After the diagnosis of PE was established, anticoagulant therapy was initiated with low molecular weight heparin. The patient presented hemodynamic instability 24 h after being admitted to the hospital. The patient had a blood pressure of 70/36 mmHg and a mean arterial pressure of 48 mmHg, as well as decreased awareness. Advanced airway management was initiated with endotracheal intubation and mechanical ventilation. A transthoracic echocardiogram (TTE) was performed at the request of the cardiology department, reporting middle segment right ventricular hypokinesia, pulmonary artery pressure of 32 mmHg, tricuspid annular plane systolic excursion 18 mm, right and left ventricle relationship 1.0, left ventricular ejection fraction of 35%, and cardiac output of 1.8 L/min.
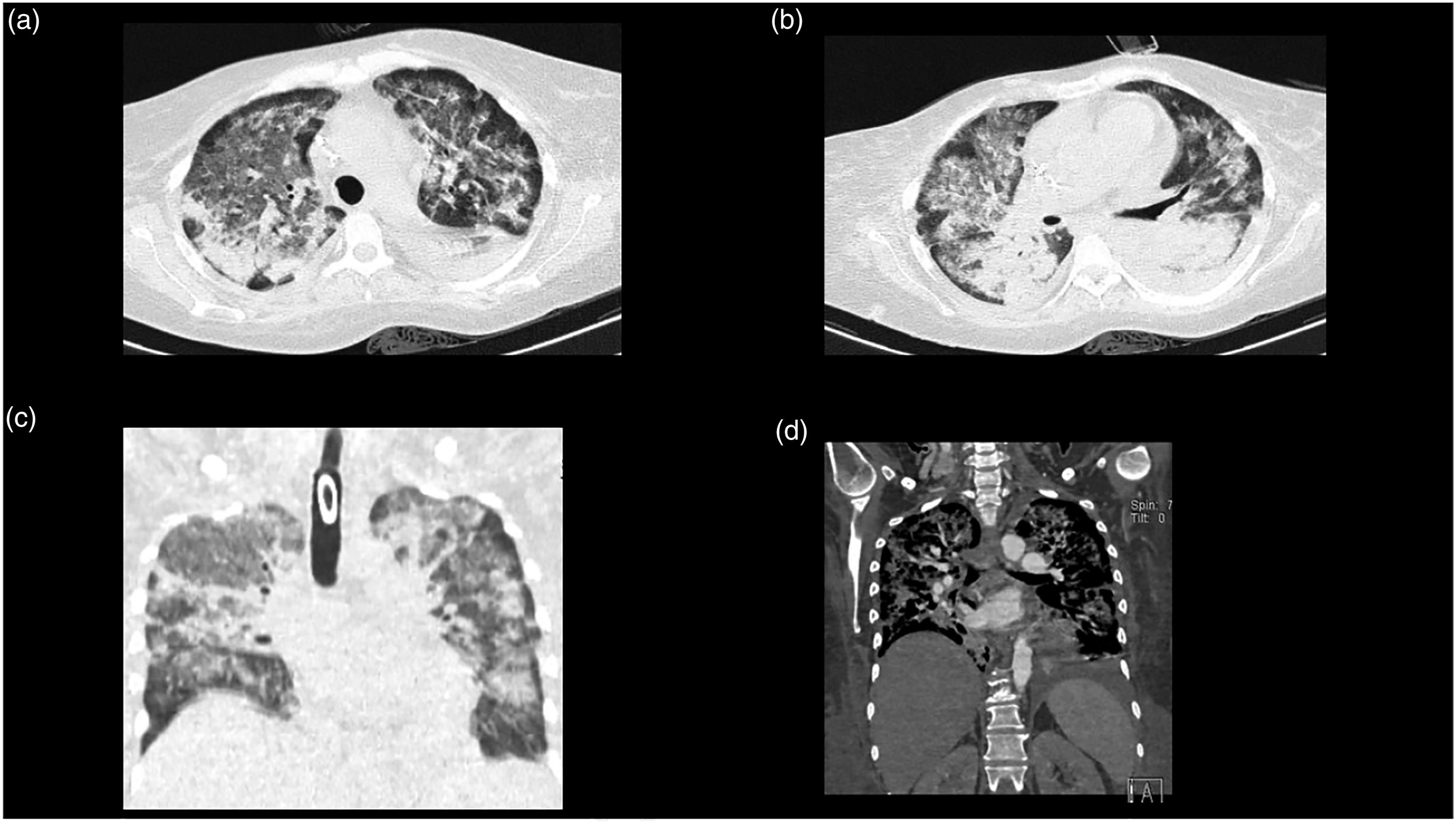
Vasopressors were initiated with norepinephrine (0.5 mcg/kg/min) and dobutamine (0.2 mcg/kg/min). Thrombolysis with alteplase 15 mg IV bolus was initiated, followed by a 50 mg IV 30-minute infusion and a 35 mg 60-minute infusion. After 72 h, the patient had anuria with a creatinine of 2.8 mg/dL, urea nitrogen of 146 mg/dL, and blood urea nitrogen of 68 mg/dL. Continuous renal replacement therapy (CRRT) was initiated with six sessions. On the ninth day of CRRT, the follow-up laboratory workup was creatinine of 1 mg/dL, urea nitrogen of 58 mg/dL, and blood urea nitrogen of 27 mg/dL; furthermore, the BUN/creatinine urinary output ratio was 1 mL/kg/hr. On day 11 of hospitalization, the patient had hemoptysis with a hemoglobin drop greater than 2 g/dL. A chest X-ray revealed increased radiodensity at both hemithorax (Figure 3(a)). Alveolar hemorrhage was suspected and two blood units were transfused. Table 3 reports the follow-up hemograms throughout the patient’s hospitalization. Blood cultures and bronchial secretion cultures were performed and reported negative. The patient did not accept to undergo bronchoscopy with bronchioalveolar lavage. A new chest CT scan was performed, revealing small hypodense regions with air bronchogram that merge at the posterior lobules (Figures 3(b), 4(a)(d)a–d4(a(d)), as well as bilateral pleural effusion with predominance in the left side (Figures 3(c)–(d)). Follow-up chest X-ray and computed tomography. Follow-up hemograms at different time points. Follow-up lung computed tomography. Computed tomography (CT). 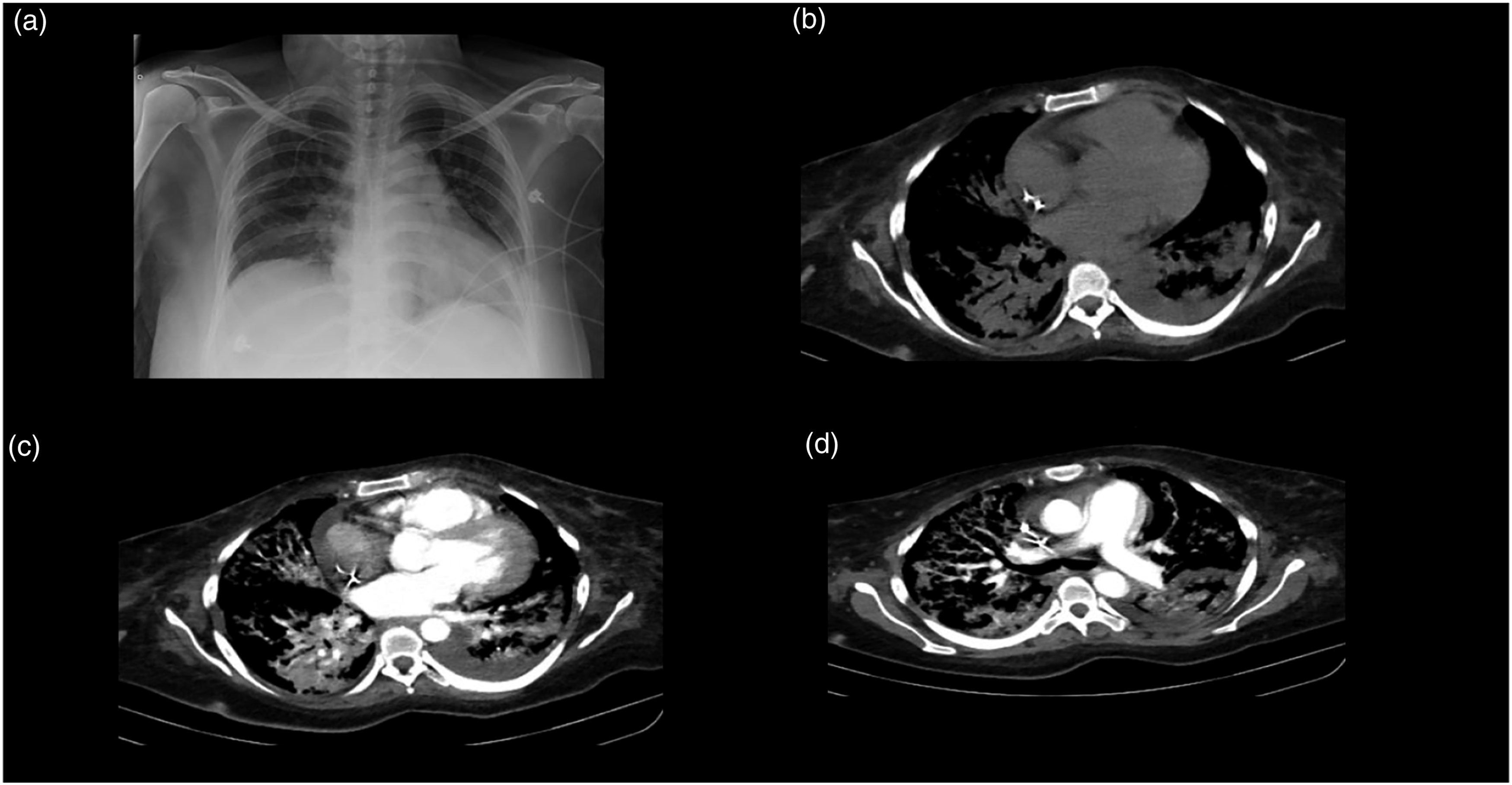
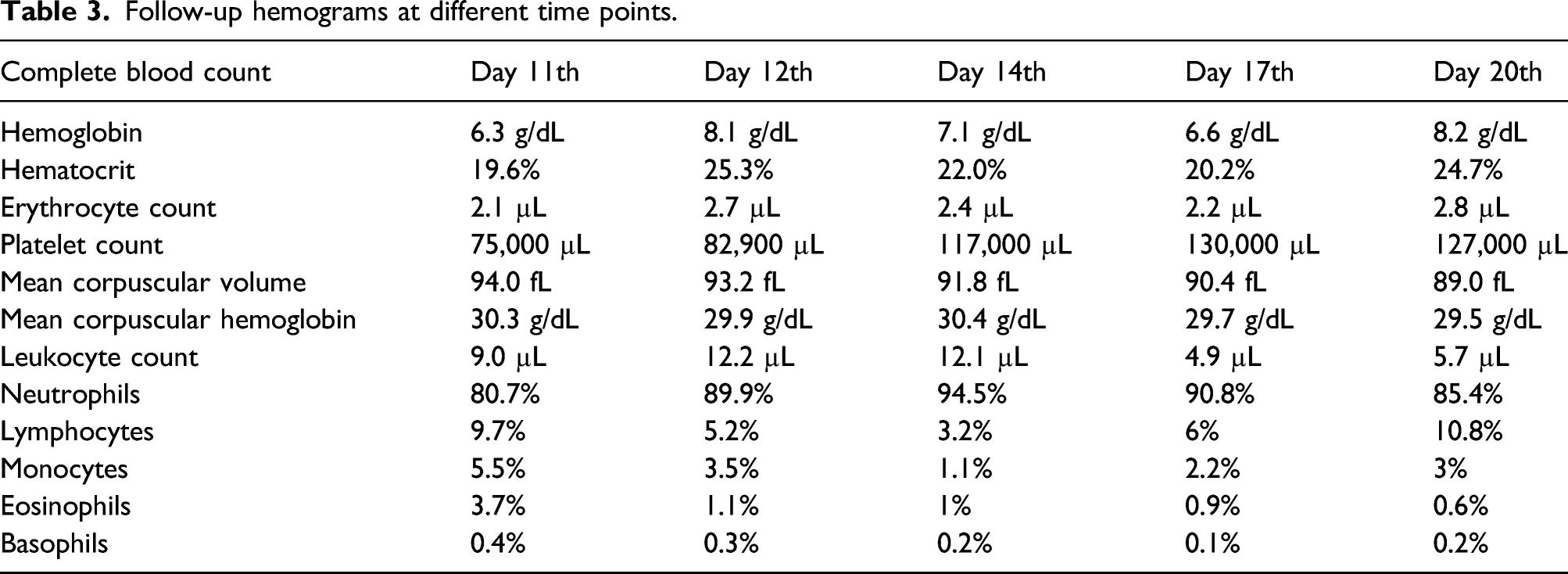
In search of an autoimmune etiology, the following tests were requested: antineutrophil cytoplasmatic antibodies (ANCA), cytoplasmatic (cANCA), perinuclear (pANCA), cyclic citrullinated peptide, anti-SS-B (La), anti-SS-A (Ro), and anti-nuclear antibodies were all negative (Table 2). In search of SLE anti-cardiolipin IgM and IgG antibodies, Beta-2 glycoprotein 1 IgG and IgM antibodies, and lupus anticoagulant were negative; however, anti-double-stranded deoxyribonucleic acid anti-Smith antibodies were positive, as well as Complement C3 and C4 were decreased (Table 2). Thus, the diagnosis of SLE was integrated as per the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) criteria (Aringer et al., 2020); the patient had at least one clinical criteria (i.e., pleura effusion) and score ≥10 points (Table 2). After SLE diagnosis was integrated, treatment with three IV boluses of methylprednisolone 1 g was administered; followed by prednisone 0.5 mg/kg q24 h. Meanwhile, protein, S protein, antithrombin III, factor V Leiden, and homocysteine levels were reported normal. The patient continued with a torpid evolution secondary to active hemorrhage, which required transfusion of two more blood bags, raising hemoglobin levels from 6.3 g/dL to 8.2 g/dL (Table 3). After 20 days of hospitalization, the patient died after a cardiac arrest. A percutaneous pulmonary biopsy was performed postmortem, reporting septal and interstitial space thickening, lymphocyte and plasma B cell presence compatible with chronic interstitial inflammation (Figure 5). Pulmonary biopsy. Histopathological sections of the lung. Top row: hematoxylin and eosin staining. Bottom row: Masson’s trichrome staining. Thickening of the interstitial spaces (e.g., collagen seen in blue), chronic inflammation with lymphocytes and plasma cells can be observed.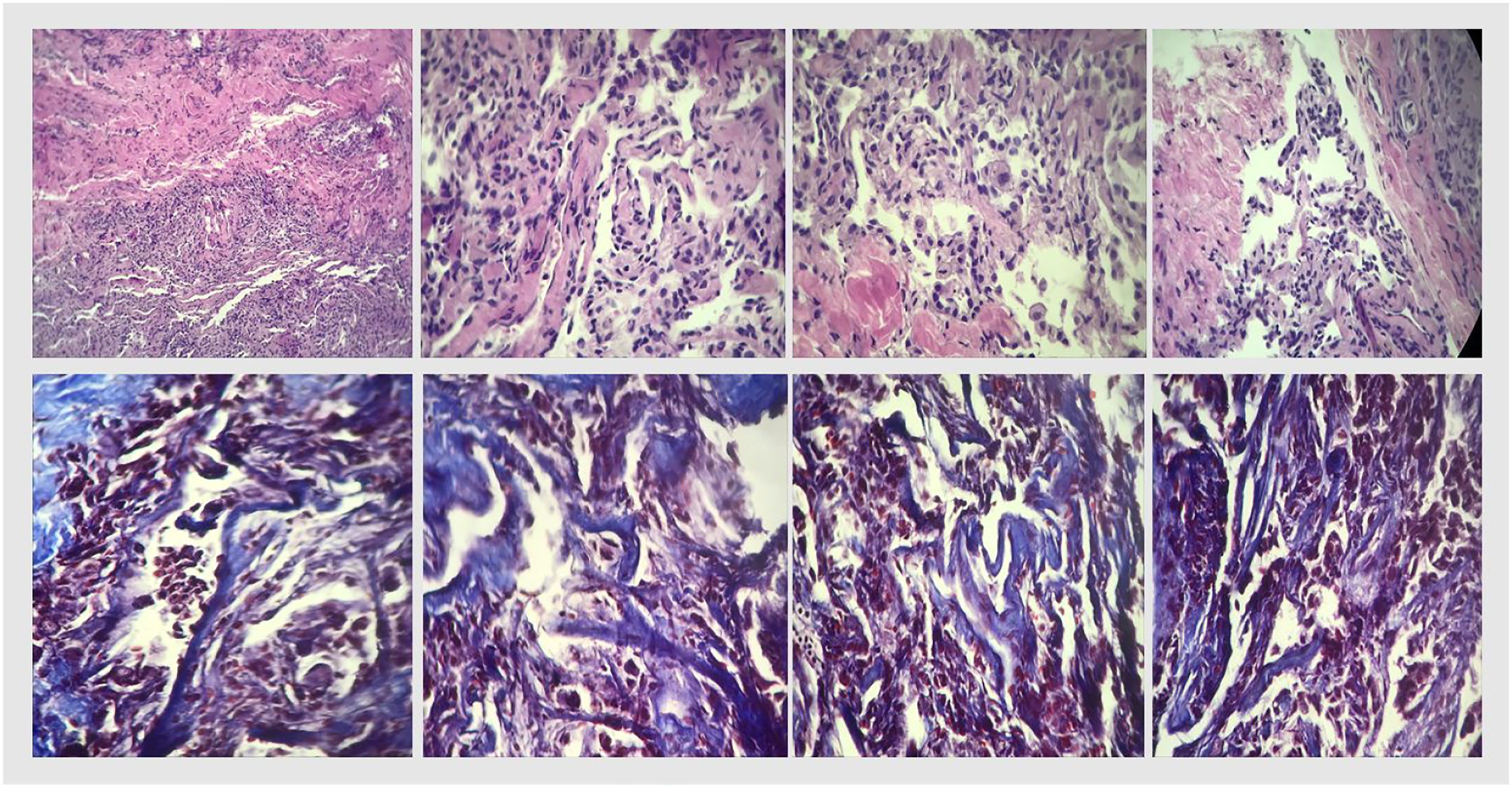
Discussion
We report the first recorded case of concomitant thromboembolism and alveolar hemorrhage associated with SLE. The patient met the SLE classification criteria 8 and had a Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) score of 35; thus, indicating a severe disease. 1 Deep vein thrombosis and pulmonary embolism occur in approximately 9% of SLE cases. 9 Antiphospholipid antibodies (IgG and IgM) are associated with an increase in thromboembolic events up to 35–42%. 10 However, the patient in the case presented above had negative antinuclear antibodies which is a very rare finding in SLE, as only approximately 6.2% of cases can be negative. 11 Factors related to negative ANA antibodies are disease activity (i.e., systemic lupus erythematosus disease activity index score ≥10), lupus nephritis, and immunosuppressant use. 12 The patient had an SLEDAI-2K score of 35; thus, a severe disease activity could be associated with the negative ANA antibodies laboratory result. Among other clinical manifestations associated with SLE are 1) pulmonary embolism, 2) lung infarction, 3) pulmonary hypertension, 4) pulmonary artery thrombosis, 5) pulmonary micro-thromboses, 6) acute respiratory distress syndrome, 7) diffuse alveolar hemorrhage, and 8) postpartum hemolytic uremic syndrome. 13 In the subsequent paragraphs, we will put the case presented above into clinical context.
Alveolar hemorrhage was suspected in this patient due to the presence of hemoptysis, persistently decreased hemoglobin (>2 g/dL), as well as the presence of pulmonary infiltrates in the imaging studies.14,15 Early manifestations of SLE can be diverse and nonspecific (e.g., fatigue, fever, and malaise).2,16 Alveolar hemorrhage is a medical emergency that produces acute respiratory failure, requiring an early diagnosis, as well as aggressive treatment [17]. The radiological patterns suggestive of alveolar hemorrhage can be present 48–72 h after the clinical onset. 4 SLE patients who present alveolar hemorrhage have approximately a 50% mortality rate. 18 Advanced age, presence of massive hemoptysis, need for mechanical ventilation, previous plasmapheresis treatment, presence of thrombocytopenia, as well as infections have been associated with higher mortality risk. 4 Alveolar hemorrhage secondary to SLE is more common in young women, 19 as it was in this case. The following etiologies must be considered in the presence of alveolar hemorrhage: primary capillaritis (e.g., microscopic polyangiitis, ANCA-associated vasculitis, granulomatosis with polyangiitis, eosinophilic granulomatosis with polyangiitis, isolated pauci-immune pulmonary capillaritis, and Henoch–Schönlein purpura) and without capillaritis (e.g., SLE, scleroderma, primary anti-phospholipid syndrome, polymyositis, rheumatoid arthritis, mixed connective tissue disease, drug-induced vasculitis, IgA nephropathy, and anti-glomerular basement membrane antibody disease).15,20,21 Thrombocytopenia and low C3 levels are predictive factors for alveolar hemorrhage in SLE 14 ; this case presented with reduced complement levels but not thrombocytopenia. Furthermore, although SLE patients have leucopenia, the patient in the case presented above had leukocytosis due to a septic process. Meanwhile, lupus nephritis has been linked with alveolar hemorrhage in SLE patients in 64–100%. 14 Additionally, imaging studies can assist to diagnose alveolar hemorrhage in SLE patients; in chest X-ray, opacities can be seen in the central and basal airways, while in a chest CT, ground glass opacifications without significant interlobular septal thickening. 22
Systemic lupus erythematosus has a three times greater risk of pulmonary embolism, a four times greater risk of deep vein thrombosis, and three times greater risk of venous thromboembolism than in the general population, a year after the diagnosis. 23 Although the patient was not positive for the lupus anticoagulant, the patient had deep vein thrombosis. However, fifty percent of patients with SLE and lupus anticoagulants will present venous thrombosis within 20 years. 24 Furthermore, lupus anticoagulants are a risk factor for the first event of venous thrombosis, as well as thrombi recurrence. 25 Patients with SLE are at increased risk of venous thromboembolism events due to inflammation and/or the presence of antiphospholipid antibodies. 2 In the case presented above, antiphospholipid antibodies were negative; however, SLE was further explored with a second antiphospholipid antibody determination. 25 Other etiologies of thrombotic events, in this case, were excluded: protein C and S deficiency, antithrombin III and Factor V Leiden alterations, use of estrogens, among others. 26
Management of alveolar hemorrhage in SLE has been previously successfully treated with corticosteroids and cyclophosphamide, 27 as well as with pulse methylprednisolone, cyclophosphamide, and plasma exchanges. 28 However, in the case presented above, the patient had a torpid evolution even after employing similar management. Among the treatment options available for alveolar hemorrhage in SLE are high corticosteroid doses (e.g., methylprednisolone), high immune-system suppressants (e.g., azathioprine, methotrexate, and mycophenolate), plasma exchanges, rituximab, belimumab.3,16,21 In the case presented above, the patient had alveolar hemorrhage 11 days after alteplase administration; while previous studies reported a similar complication after 4–5 days after the use of thrombolytic medication.29,30 Furthermore, a recent case series reported that alveolar hemorrhage secondary to thrombolytic administration was observed during the first 4–12 h posterior administration 31 ; thus, the alveolar hemorrhage might have occurred irrespective of the thrombolysis. Among the multiple risk factors for alveolar hemorrhage that the patient did have were development thrombocytopenia and leukopenia, low C3 levels, high (ds)DNA levels, severe disease activity, and lupus nephritis.4,14 Finally, heart failure, in this case, could be associated with the pulmonary vasculature occlusion severity, which could have led to a right ventricular failure and a subsequent acute circulatory collapse. Furthermore, the patient had sepsis, which has been associated with cardiomyopathy (i.e., 20–50% of the cases), particularly, diastolic dysfunction, which can contribute to decreased cardiac output secondary to right ventricular failure, leading to biventricular failure, thus hemodynamic instability and death.32, 33
Limitations
The patient refused to undergo a bronchial lavage to exclude an infectious alveolar hemorrhage origin. This is an important limitation as the patient had leukocytosis upon admission to the Internal Medicine department. Furthermore, the patient did not accept the management of his condition with rituximab or plasma exchanges, which could have positively impacted the patient’s prognosis.
Conclusion
The case presented above is the first report in the literature that consists of concomitant thromboembolism and alveolar hemorrhage secondary to SLE. We present clinical, imaging, laboratory, and histopathological evidence. The patient met the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) criteria for SLE. Furthermore, the patient had a Systemic Lupus Erythematosus Disease Activity Index 2000 (SLEDAI-2K) score of 35; thus, indicating severe disease.
Final Diagnosis
Thromboembolism and alveolar hemorrhage secondary to systemic lupus erythematosus disease.
Footnotes
Acknowledgments
This study was supported by CONACyT (Consejo Nacional de Ciencia y Tecnología) Grant #440591. This research did not receive any specific grant from funding agencies in the commercial sector. We would like to commend the work of the medical staff (i.e., specialists, medical residents, and nursing staff) of the Internal Medicine Department at Hospital General Regional No. 21 IMSS de León, Guanajuato, México.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
The clinical data supporting the conclusions of this article is included in the article.
Author Contributions
Jiménez-Zarazúa, O: original idea, manuscript preparation, data analysis, and interpretation.
Vélez-Ramírez, LN: data acquisition, data interpretation, and manuscript correction.
Ramírez-Casillas CA: data acquisition, data interpretation, and manuscript preparation.
Mondragón, JD: data analysis and interpretation, manuscript preparation, and manuscript correction.
