Abstract
Background
Systemic lupus erythematosus (SLE) patients experience a premature and more severe presentation of coronary artery disease. The underlying mechanisms of accelerated coronary artery disease in SLE patients remain to be elucidated.
Methods
By using atherosclerosis combining a SLE murine model, we proved that the onset of SLE aggravates atherosclerosis. Although the onset of SLE reduced blood lipids slightly, immune deviation contributed to aggravated atherosclerosis in lupus mice. Lupus atheroma were characterized by inflammatory cell infiltration, such as gathered dendritic cells, macrophages, and IgG deposition.
Results
Decreased lymphocytes and magnified dendritic cells in the spleen were also observed in lupus mice. Hydroxychloroquine prevented atherosclerosis progression mainly by reversing immune status abnormality caused by SLE. Serum interferon alfa levels were not changed in lupus mice.
Conclusion
These findings strongly suggested that anti-inflammatory therapies and hydroxychloroquine provide a new possible strategy for treating SLE patients with atherosclerosis.
Introduction
Coronary artery disease (CAD) is the deadliest disease with the highest morbidity and mortality, both in developed and developing countries. Atherosclerosis is the underlying pathological change in CAD. Atherosclerosis is initiated with endothelial cell injury and oxidized low-density lipoprotein (oxLDL) deposition in the arterial wall. Monocytes from the circulation are attracted to the subendothelial space and activated, named macrophages. oxLDL is then internalized by macrophages from the circulation and arterial smooth muscle cells. These lipid-rich cells are termed ‘foam cells’. Macrophages produce a variety of cytokines and accelerate smooth muscle cell and fibroblast migration. Finally, a plaque inside the vascular wall, or an atheroma, is formed.
Systemic lupus erythematous (SLE) is the most common systemic autoimmune disease, characterized by elevated dsDNA antibodies and autoantibody to nuclear antigen. Due to improved diagnosis and effective management, atherosclerosis-related diseases have emerged as a major cause of long-term mortality in SLE patients. The risk of myocardial infarction in SLE patients is at least five times greater than that of the general population. 1 In a previous study, we found that CAD patients with SLE were younger and had multivessel lesions. 2 We assumed that SLE may aggravate CAD in some SLE-specific ways. In a follow-up study, we found that SLE patients tended to have more traditional cardiovascular risks, such as hypertension, diabetes mellitus and dyslipidemia. 3 These observations can be explained by the fact that lupus nephritis and steroid medicines may lead to hypertension, diabetes and dyslipidemia. However, patients with both CAD and SLE have less traditional cardiovascular risk factors. This phenomenon prompts the theory that the abnormal immune status of SLE patients may accelerate atherosclerosis. Following studies have confirmed this assumption. Arterial stiffness, an indicator of atherosclerosis, was associated with SLE-specific factors, such as longer disease duration and intravenous cyclophosphamide use. 4 Although in previous clinical studies we have observed the association between SLE and arterial stiffness, the reliability of the conclusion is hampered by the fact that arterial stiffness is just a surrogate marker of atherosclerosis. In this study, we used a murine model to observe atherosclerosis combined with SLE directly.
Apolipoprotein E (ApoE) is an essential component of chylomicrons, very low-density lipoprotein (LDL) and high-density lipoprotein (HDL). ApoE plays a central role in lipid metabolism. Dysfunction of ApoE leads to elevated blood lipids and atherosclerosis.5,6 Mice lacking ApoE (ApoE–/– mice) have been used widely as a stable and practical model of hyperlipidemia and atherosclerosis. The plaque composition of ApoE–/– mice is similar to human lesions and this makes it an excellent atherosclerosis model. In addition, a high fat diet even accelerates atheroma formation. 7
Pristane, also known as tetramethylpentadecane, has been proved to induce lupus by intraperitoneal injection in several mice strains, including BALB/c mice and C57BL/6 mice.8,9 Pristane-induced SLE is characterized by elevated dsDNA antibodies and nephritis. 10 However, reports on the injection of pristane in ApoE–/– mice are limited. The overexpression of interferon-stimulated genes (ISGs) is considered a mechanism of SLE. The pristine–lupus model reproduces the overexpression of ISGs in mice. In this study, we induced lupus in athrosclerosis-prone ApoE–/– mice by the intraperitoneal injection of pristane.
Hydroxychloroquine has long been employed in the management of SLE. The efficiency of hydroxychloroquine in disease activity control and flare prevention is well documented. Furthermore, the side effects of hydroxychloroquine are relatively mild and rare. Rheumatologists even recommend using hydroxychloroquine as a ‘background medication’, which means the vast majority of SLE patients should be prescribed with hydroxychloroquine. Hydroxychloroquine mainly acts through accumulating within the lysosomes to increase pH, by which it interferes with phagocytosis as well as self-antigen presentation. 11 Hydroxychloroquine has been proved to attenuate risk factors of CVD. In NZB/W mice, a lupus model, HCQ improved endothelial function.12 A case control study reported that the use of hydroxychloroquine was inversely correlated to carotid atheroma formation detected by ultrasonography, independent of lipid modulation effects. 13 However, as yet there has been no study on the effect of hydroxychloroquine on atherosclerosis.
In this study, we use a murine model to investigate directly the severity and content of lupus atherosclerosis, as well as the effect of hydroxychloroquine treatment on atherosclerosis.
Materials and methods
Mice grouping and modelling
The study used 8-week-old female wild type C57BL/6 (B6) and ApoE–/– mice with B6 background (both bought from Vital River Laboratory Animal Technology Co. Ltd.). All mice were housed and maintained in individual ventilated cages in specific pathogen-free conditions at the Institute of Laboratory Animal Sciences (ILAS) at the Chinese Academy of Medical Sciences and Peking Union Medical College, research licence no. SYXK (Beijing) 2015-0035. All procedures were carried out in compliance with the guidelines of institutional animal care and use committees of the ILAS (ILAS-PG-2015-016). Mice were randomly divided into different groups. Mice in the lupus model groups (n = 30) received an intraperitoneal injection of 0.5 ml pristane (Sigma Aldrich) at the age of 8 weeks, while mice in the control group (n = 32) received an intraperitoneal injection of 0.5 ml phosphate-buffered saline (PBS). Mice in the hydroxychloroquine treatment group (n = 15) received 0.2 mg/ml hydroxychloroquine in the drinking water, while other mice received drinking water without hydroxychloroquine. Six months later, mice were killed after overnight fasting. Aortas, serum and heart were harvested.
dsDNA antibody analyses
Concentrations of anti-dsDNA antibodies were measured by ELISA according to the manufacturer’s instructions (Haisian Biotech).
Oil Red O staining of aortic valve atherosclerosis plaques
Tissue around the aortic sinus and aortic valve was harvested and embedded in optimal cutting temperature compound medium. Frozen sections (7 µm) of aortic sinus were obtained. Frozen sections were fixed in 10% formalin for 10 minutes and dehydrated in 100% propylene glycol for 2 minutes at room temperature. Slices were incubated in Oil Red O solution for one hour at room temperature. Then slices were developed in 85% propylene glycol for 1 minute and rinsed in distilled water twice. Nuclei were stained in haematoxylin for 2 minutes and washed in tap water for 2 minutes.
Oil Red O staining of whole aortas for en face analysis
Adventitial fat of the aortas was thoroughly trimmed away. Aortas were fixed in 10% formalin and washed in PBS on a shaker overnight. Aortas were then dehydrated in 100% propylene glycol for 2 minutes at room temperature and were incubated in Oil Red O solution for 3 hours at room temperature. Aortas were then developed in 85% propylene glycol three times and washed overnight in PBS on a shaker. Aortas were pinned out using minutien pins (Finescience tools).
Immunofluorescence staining of aortic sinus atherosclerosis plaques
Antigen blocking was performed with 10% rat serum. Macrophages were detected by rat anti-CD68-Alexa Fluor 647 (BD Biosciences). Dendritic cells (DCs) were detected by Armenian hamster monoclonal to CD11c-Biotin and TSA Kit (HRP-Streptavidin and Alexa Fluor 488 Tyramide). IgG deposition was detected by goat anti-mouse IgG H&L-FITC (BD Biosciences). All slides were mounted with prolonged gold antifade reagent with DAPI (Invitrogen). No antibody controls revealed any immunofluorescence.
Image capture and analysis
Images were captured using a digital camera connected to a microscope and analyzed by Image-Pro Plus 6.0 software. The staining positive area and total area of sections and aortas were calculated.
Flow cytometry analysis of aorta cells
Single cell suspensions were prepared from aortas. Aortas were cut into pieces and each aorta was digested with 900 U collagenase I, 250 U collagenase XI, 1200 U hyaluronidase and 120 U DNase in 2 ml D-PBS for 1.5 hours at 37℃. The antibodies used for aorta cell staining were anti-CD11c-APC (1:100), anti-CD68-Alexa Fluor 488(1:100) and anti-CD45-PE (1:100) from BD Biosciences. All cells were incubated with antibodies for 30 minutes at 4℃. Cells were washed with fluorescence-activated cell sorter (FACS) buffer twice and immediately analyzed on a BD FACS Calibur flow cytometer (Aria I). Data were analyzed using FlowJo software.
Serum biochemical indexes analysis
Total cholesterol (TC), LDL-cholesterol and total IgG in serum were tested using the Chemistry System (Beckman Coulter AU5800).
Statistical analysis
Data were reported as means ± standard errors. Data among multiple groups were compared with multivariate analysis of variance, and data between two groups were analyzed by Student’s t test. P < 0.05 was considered significant. Relationships were analyzed by partial correlation analysis. |r| > 0.8 indicates highly correlated and 0.5 < |r| < 0.8 indicates moderately correlated. Graphs were made by GraphPad Prism 6. SPSS software (version 19.0) was used for all statistical analysis.
Results
Pristane injection induced lupus-like manifestation in both C57BL/6 and ApoE–/– mice, hydroxychloroquine partly reversed lupus demonstration
Elevation of serum autoantibodies concentration, especially dsDNA antibody, is specific for lupus disease and critical for the diagnosis of lupus. We tested the serum dsDNA concentration 6 months after pristane injection. DsDNA antibodies in both pristane injected C57BL/6 and ApoE–/– mice were elevated remarkably. Furthermore, hydroxychloroquine-treated mice presented with decreased dsDNA antibodies (P < 0.001) (Figure 1(a)). Some pristine-injected mice presented with alopecia and skin haemorrhage (Figure 1(c)), which were similar to human lupus clinical features. PBS-injected mice had no skin lesions (Figure 1(b)). These phenomena suggest that the lupus disease model was successfully set up in both C57BL/6 and ApoE–/– mice.
dsDNA antibodies and skin lesions. (a) dsDNA antibodies in mice sera 6 months after pristane or phosphate-buffered saline (PBS) injection. (b) PBS-injected mice had no skin lesions. (c) Alopecia and skin haemorrhage of lupus mice. HCQ: hydroxychloroquine. *P < 0.05, **P < 0.01.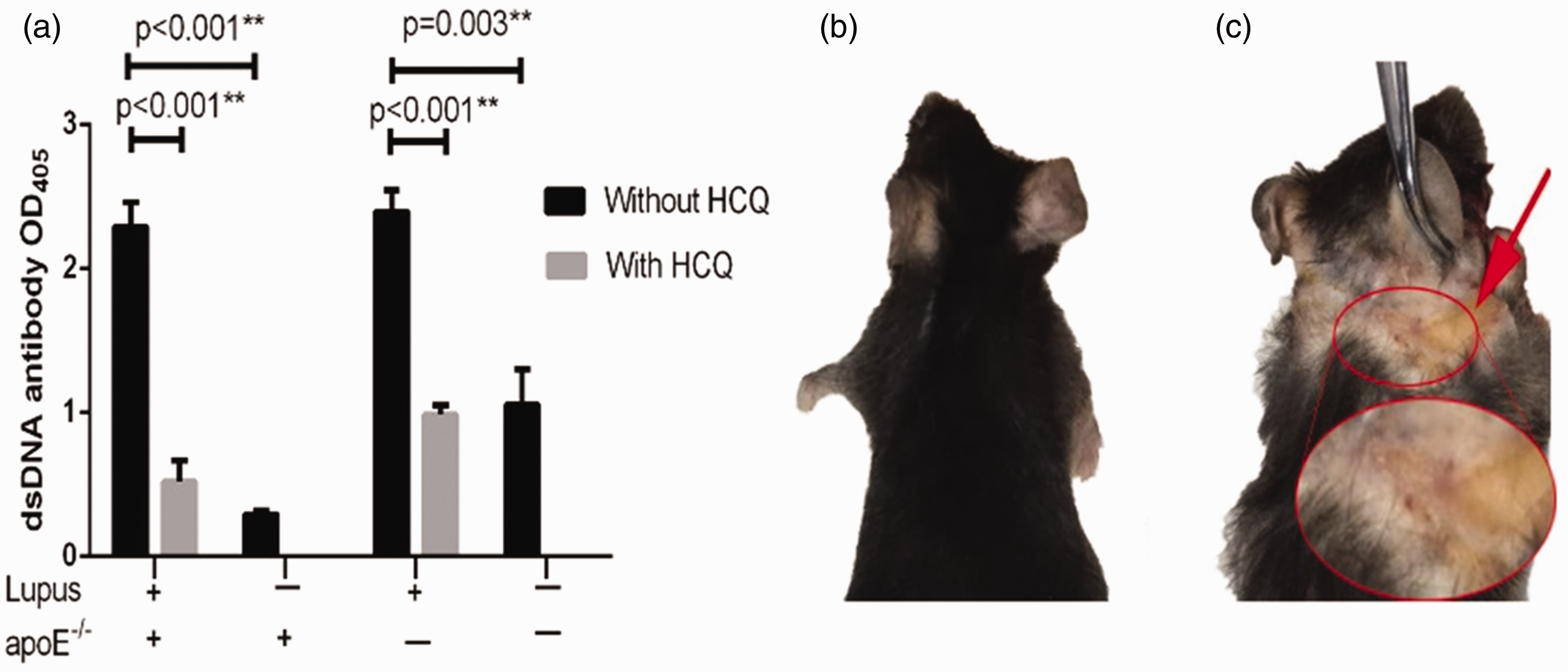
Onset of lupus led to more severe atherosclerosis, while hydroxychloroquine alleviated atherosclerosis
Oil Red O is a dye specific for staining lipids. When used for vessel staining, Oil Red O marks atheroma. By calculating the percentage of area stained by Oil Red O, the severity of atherosclerosis can be evaluated. There are two common methods to access atherosclerosis development in mice. One is by staining a frozen section of aortic sinus, and the other one is by staining of the aorta en face. In order to improve reliability, we used both methods in this research to assess the severity of atherosclerosis.
Only ApoE–/– mice developed atherosclerosis when killed at the age of 8 months. No atherosclerosis was observed in C57BL/6 mice (Figure 2(a)). For this reason, only ApoE–/– mice were used for further analysis. From multivariate analysis, we found that pristane treatment also worsened the extent of atherosclerotic plaque in both the aorta (Figure 2(a) and (b)) and aortic sinus in ApoE–/– mice (Figure 2(c) and (d)). Furthermore, mice fed a high fat diet suffered from significantly more severe atherosclerosis (data not shown).
Aorta Oil Red O staining. (a) Oil Red O staining of aorta. (b) Percentage of Oil Red O staining positive area in aorta. (c) Oil Red O staining of aortic sinus section. (d) Percentage of Oil Red O staining positive area in aortic sinus section. (e)–(h) Effect of HCQ on aorta Oil Red O staining. (e) Oil Red O staining of aorta. (f) Percentage of Oil Red O staining positive area in aorta. (g) Oil Red O staining of aortic sinus section. (h) Percentage of Oil Red O staining positive area in aortic sinus section. HFD: high fat diet fed; CD: chow diet fed; HCQ: hydroxychloroquine.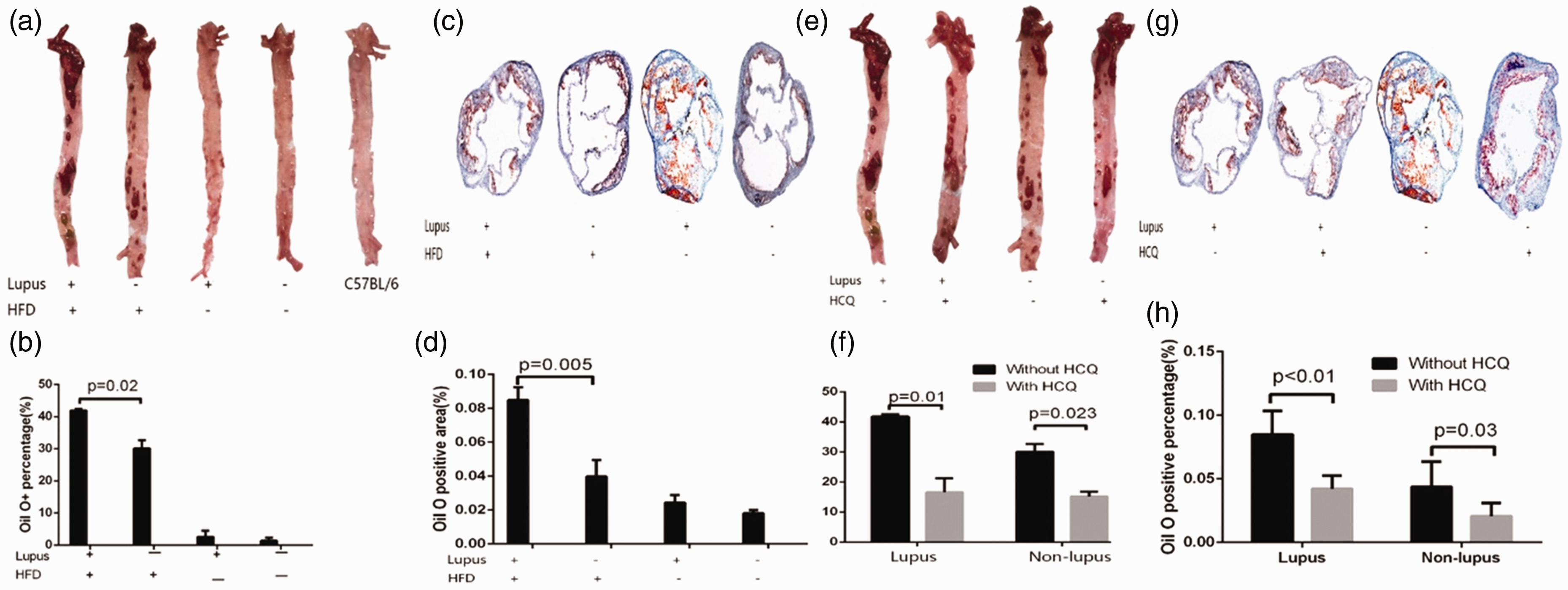
Multivariate analysis also showed that in ApoE–/– mice, hydroxychloroquine treatment ameliorated the severity of atherosclerosis in both the pristane-treated and control treated mice (Figure 2(e)–(h)).
Serum TC and LDL-cholesterol level decreased in lupus mice
As described above, the onset of lupus worsened the severity of atherosclerotic lesions in ApoE–/– mice. Then we sought to find the reason for accelerated atherosclerosis. Dyslipidemia has long been branded the most critical risk factor for atherosclerosis. Elevated TC and LDL-cholesterol, accompanied by decreased HDL-cholesterol, contribute to more significant atheroma. Therefore, we first tested the serum lipid level.
In ApoE–/– deficient mice that received a high fat diet, pristane treatment resulted in lower serum TC (P = 0.017) and LDL-cholesterol (P = 0.014) levels compared to control treated (i.e. non-lupus) mice. As we expected, ApoE–/– mice had higher LDL-cholesterol and TC levels compared to C57BL/6 mice (P < 0.001), and mice fed with a high fat diet also had higher LDL-cholesterol and TC levels (Figure 3(a)) and (Figure 3(b)).
Serum lipid level. (a) Serum total cholesterol (TC) level. (b) Serum low-density lipoprotein (LDL) cholesterol. Lupus mice had lower serum TC level and LDL-cholesterol level compared to non-lupus mice. *P < 0.05.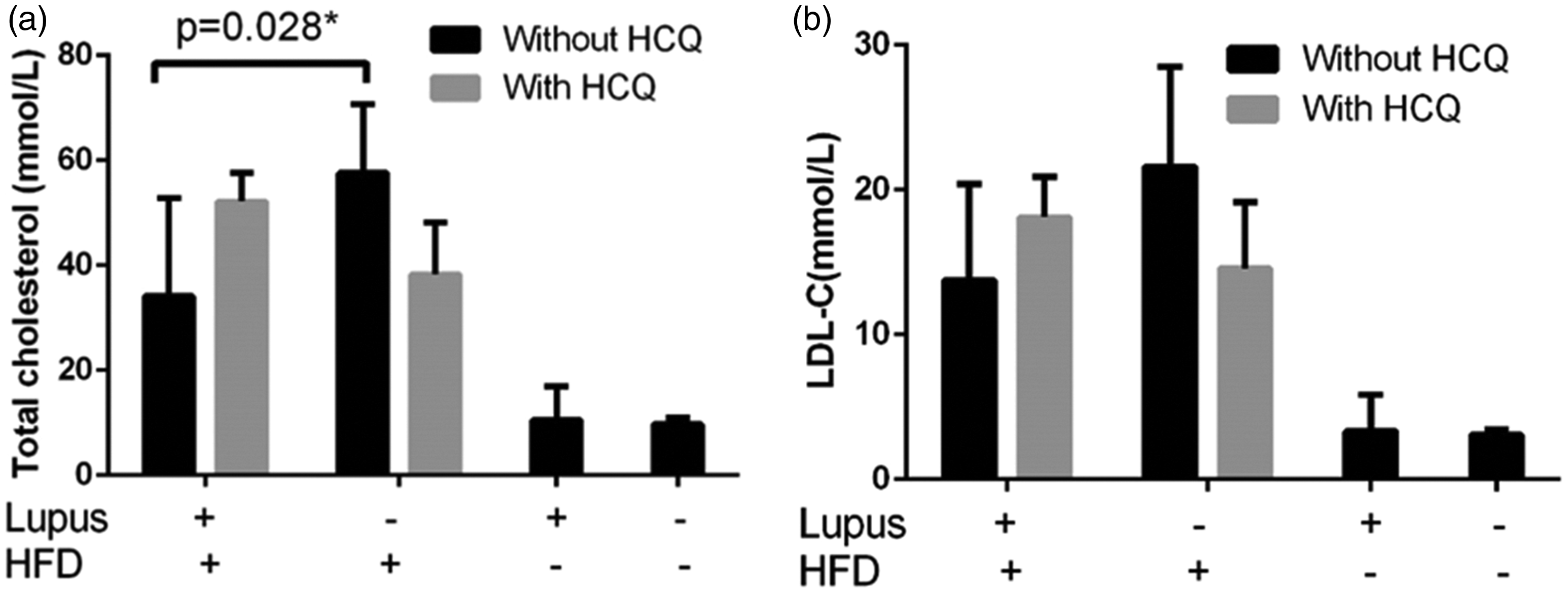
However, the HDL-cholesterol level was not influenced by the atherosclerosis-prone genotype (P = 0.986), cholesterol content of feed (P = 0.458), lupus disease onset (P = 0.632) or hydroxychloroquine treatment (P = 0.861) (data not shown).
Although we found that hydroxychloroquine treatment led to atheroma shrinkage, hydroxychloroquine treatment had no significant effect on the cholesterol content of the chow in serum.
Onset of lupus increased inflammatory cells in aortas, while hydroxychloroquine administration reduced aortic inflammatory cells
Although the onset of lupus accelerated atherosclerosis, serum lipids, the most significant traditional risk factor for atherosclerosis, was alleviated. Thus, we hypothesized that certain lupus-specific factors contribute to atherosclerosis in ApoE–/– background lupus mice. On the other hand, in spite of the fact that hydroxychloroquine administration alleviated atherosclerosis, the blood lipid level was not affected by hydroxychloroquine. We assumed that hydroxychloroquine may also act directly on lupus disease to alleviate atherosclerosis. To test this, plaque immunofluorescence staining and aortic cell flow cytometry were applied.
The aorta leukocyte number may reflect the activity of inflammatory reactions. We used CD45 as a marker for leukocytes in aortic cell flow cytometry. Aortic leukocytes increased significantly in lupus mice (P < 0.001). Mice fed with a high fat diet also had significantly greater leukocyte numbers (P = 0.006, data not shown). In pristine-injected lupus mice, there was a numerical trend towards lower levels of aortic leukocyte infiltration in the hydroxychloroquine-treated group (P = 0.066). However, hydroxychloroquine had no impact on aortic leukocytes in non-lupus mice (P = 0.219) (Figure 4(a)).
Inflammatory cells in aortas. (a) Aortic leukocytes increased significantly in lupus mice. Aortic leukocytes decreased in lupus mice when hydroxychloroquine was administered. (b)–(d) Onset of lupus increased inflammatory cells in aortas, while hydroxychloroquine administration reduced aortic inflammatory cells. (b) Flow cytometry analysis of CD68+ and CD11c+ cells. (c) Aortic CD45+CD68+ cell numbers. (d) Aortic CD45+CD11c+ cell numbers. (e)–(f) Macrophages and dendritic cells are highly correlated. (e) Correlation analysis of CD68+ cell number and CD11c+ cell number. (f) Immunofluorescence staining of CD68+ and CD11c+ cells.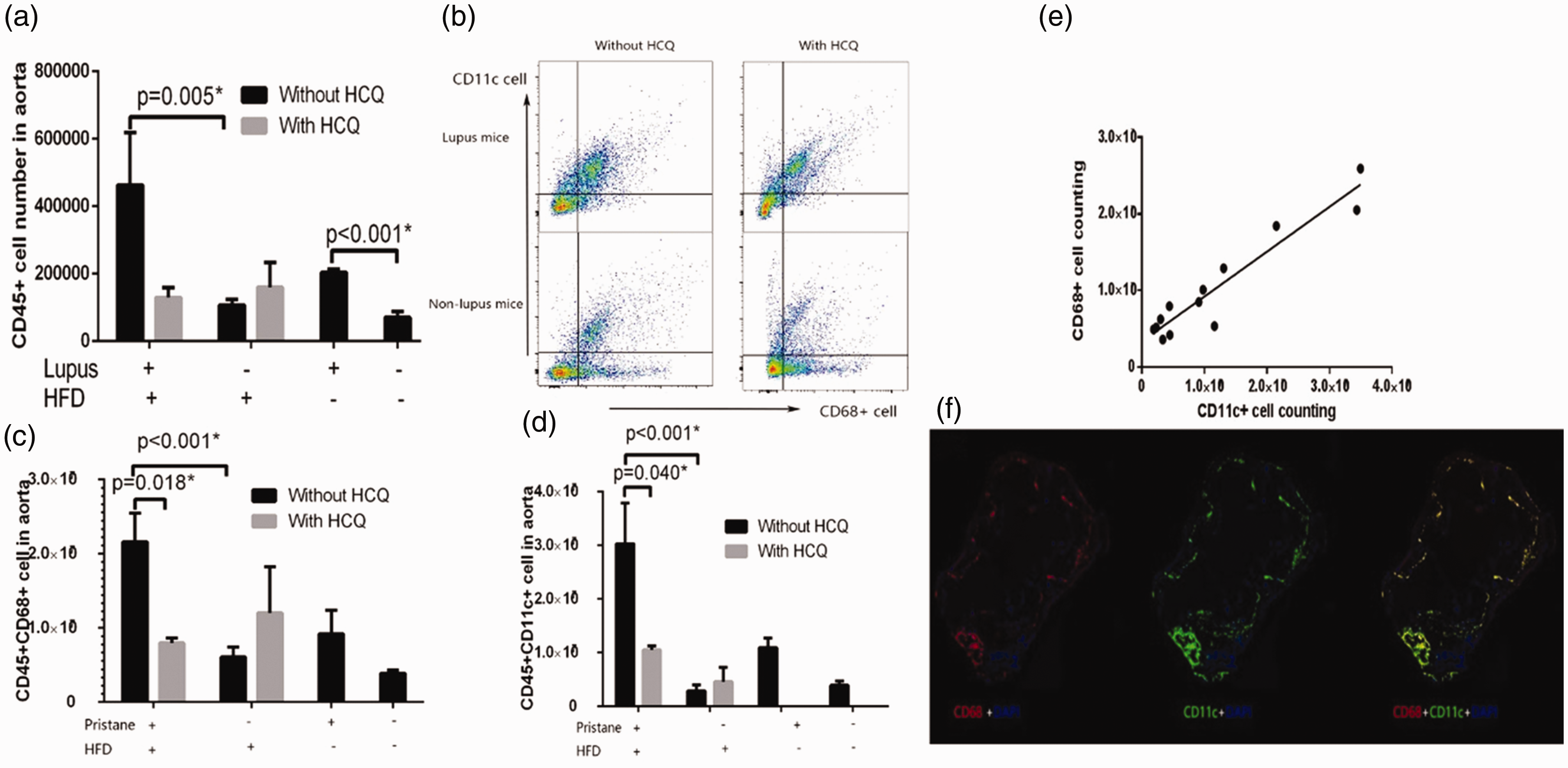
We further analyzed which group of leukocyte numbers changed significantly. Macrophages are critical for oxLDL phagocytosis and DCs play a key role in activation downstream immune reactions. Both of these cells are crucial for atheroma development. We marked macrophages with CD68 antibodies, and marked DCs with CD11c antibodies. Lupus mice had more macrophages (P < 0.001) and DCs (P < 0.001) in the aorta. High fat diet fed mice also had more macrophages (P = 0.002) and DCs (P = 0.006) in the aorta (data not shown). Hydroxychloroquine administration reduced aortic macrophage numbers (P = 0.018) and DC numbers (P = 0.040) in lupus mice. However, hydroxychloroquine had no effect on macrophage numbers (P = 0.114) or DC numbers (P = 0.293) in non-lupus mice (Figure 4(b)–(d)).
We further counted cells expressing both CD68 and CD11c. These cells may well be able to devour antigens as well as present antigens. CD68+CD11c+ cells increased in aortas of lupus mice (P < 0.001). Mice fed with a high fat diet also had more CD68+CD11c+ cells in aortas (P = 0.026). Hydroxychloroquine reduced CD68+CD11c+ cell numbers in lupus mice (P = 0.030), while it had no effect in non-lupus mice (P = 0.229).
We further analyzed the relationship between macrophage and DC numbers. Partial correlation analysis revealed that aortic macrophages and DCs were highly correlated (r = 0.873, P = 0.001) (Figure 4(e)). By means of immunofluorescence staining, we found that CD68+ and CD11c+ cells were almost totally co-localized; 97.2% CD68+ cells and 99.6% CD11c+ cells were co-located (Figure 4(f)).
Higher levels of IgG deposition within plaques were observed in lupus mice, which was ameliorated by hydroxychloroquine treatment
SLE is characterized by excessive IgG deposition in connective tissues. We assumed that IgG deposition also contributed to enhanced atheroma in lupus mice and wondered whether hydroxychloroquine treatment might affect the level of IgG deposition. Immunofluorescence staining of IgG revealed that the area of IgG deposited was similar to fat deposited, both mainly inside the aortic sinus. However, lipid deposition was demonstrated predominantly adjacent to the vessel lumen while IgG was instead deposited towards the adventitia. IgG deposition in plaques was more obvious in lupus mice (P = 0.046). High fat diet feeding also led to more IgG deposition (P = 0.004). Hydroxychloroquine treatment significantly suppressed IgG deposition (P < 0.001) (Figure 5(a)–(d)). IgG deposition was barely seen in C57BL/6 mice.
Extra IgG deposition within plaques was observed in lupus mice, while hydroxychloroquine treatment reduced IgG deposition in plaques. (a) and (c) Immunofluorescence staining of IgG in aortic sinus in ApoE–/– mice. (b) Oil Red O staining compared to IgG staining. (d) IgG deposition area in aortic sinus cross-section. (e) Serum IgG elevated in lupus mice. (f) Serum IgG and IgG deposition were not correlated.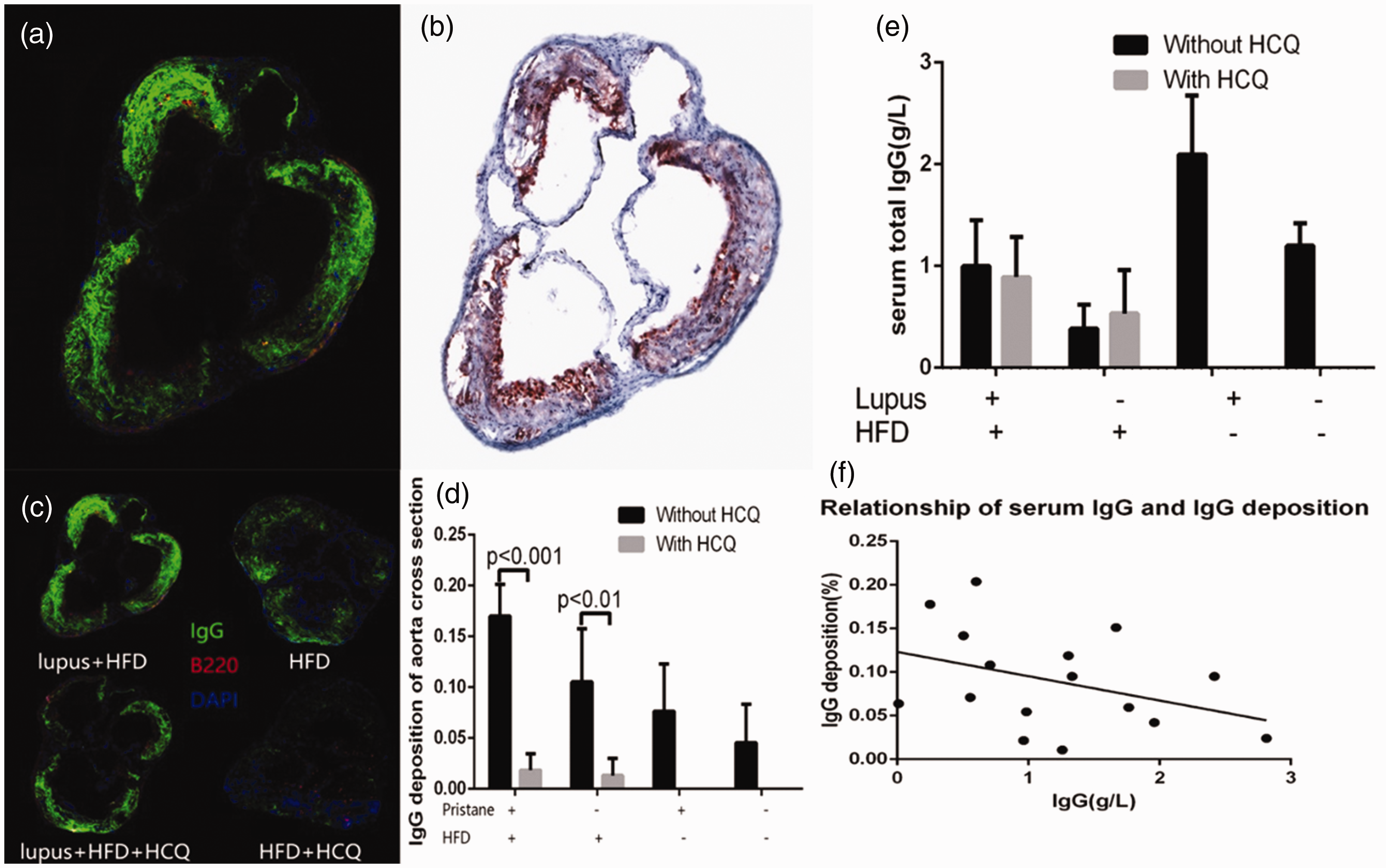
Serum IgG elevated in lupus mice
Many SLE patients display elevated serum IgG levels. In our studies, we similarly found that serum IgG levels were elevated in lupus mice (P < 0.001). Hydroxychloroquine treatment had no effect on IgG levels (P = 0.442) (Figure 5(e)). Partial correlation analysis revealed that serum IgG and IgG deposition were not correlated (r = –0.360, P = 0.207) (Figure 5(f)).
No significant serum IFN-α level change was detected in lupus mice
Previous studies have found that the interferon alfa (IFN-α) signal is correlated with the pathogenesis of SLE as well as atherosclerosis risk factors. Herein we measured the mice serum IFN-α level. Neither the onset of lupus (P = 0.145) nor high fat diet feeding (P = 0.905) changed the serum IFN-α level (Figure 6).
Serum interferon alfa (IFN-α) level. No significant serum IFN-α level change was detected.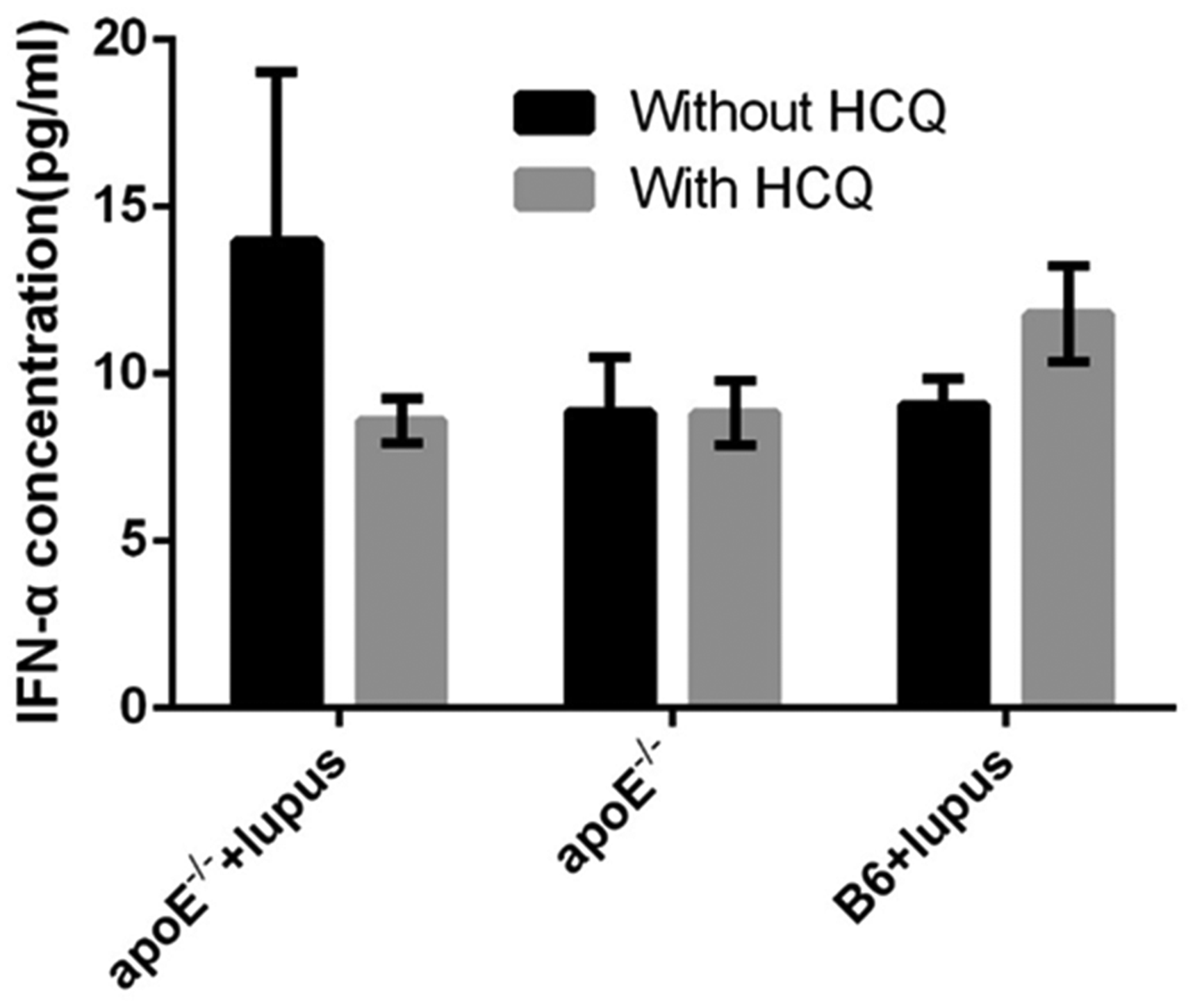
Discussion
SLE is the most common and typical autoimmune disease. Elevated autoantibodies, especially dsDNA antibodies, are vital for the diagnosis of SLE. Previous studies have reported that autoantibodies peaked 6 months after mice were injected with pristine. 14 However, those studies were carried out mainly in BALB/c or C57BL/6 mice. For the first time, we established a lupus atherosclerosis murine model by the injection of pristane in ApoE–/– mice. Six months after pristane injection, serum dsDNA antibodies of both ApoE–/– and C57BL/6 mice were elevated. Meanwhile, pristine-injected mice also had manifestations similar to SLE patients, such as patchy hair loss, skin haemorrhage, urine protein elevation, serum albumin reduction and serum IgG elevation. Atherosclerotic plaques were also observed in the aortas of all ApoE–/– mice, indicating that atherosclerosis models were successfully built in ApoE–/– mice. However, atheroma formation was not observed in C57BL/6 mice, in accord with previous observations. Taken together, lupus combined with the atherosclerosis model were successfully built in ApoE–/– mice, which provided us with an ideal tool to observed the features and mechanisms of lupus atherosclerosis. Our further study was based on this lupus model in ApoE–/– mice.
Atheroma were much more severe in both the aortas and aortic sinus of lupus mice compared to non-lupus mice. This manifestation was in accordance with the observation in humans that CAD patients combined with SLE were younger and had multivessel lesions. 2 Dyslipidemia has long been considered a vital risk factor for atherosclerosis. However, TC and LDL-cholesterol levels decreased in lupus mice. It has also been reported that lipid levels decreased in previously untreated patients with active SLE. 15 We believe that the apparent decreased lipid levels reflect the inflammatory process associated with SLE. The onset of lupus aggravated atherosclerosis while it alleviated hyperlipidemia. Therefore, lupus must have led to more severe atherosclerosis in ways other than traditional risk factors.
We found that inflammatory reactions were activated in lupus mice. Increased leukocyte numbers reflected activated inflammatory reactions in aortas. Macrophages are known for their ability for phagocytosis and are closely connected to the development of atherosclerosis. Furthermore, activated macrophages are also capable of releasing inflammatory factors and attract more inflammatory cells gathered in the atheroma. DCs are thought to be the most powerful antigen-presenting cell. DCs are capable of antigen uptake and presentation. With the help of co-stimulatory molecules, DCs activate downstream immune responses. A previous study found that DC numbers increased inside the atheroma. When stimulated by pathogen antigens, DCs were activated and proliferated, and accelerated atherosclerosis. 16 Besides this, CCL17+ DCs also aggravate atherosclerosis by inhibiting regulatory T cells (Tregs). 17 In this study, we found that both macrophage and DC cell numbers increased in the aortas of lupus mice. Increased DC numbers are partly activated by cell debris caused by pristane. It has been proved that pristane induces cell apoptosis. 18 Increased numbers of apoptotic cells in the plaque have also been observed in other lupus murine models. 19
Immunofluorescence co-localization analysis revealed that more than 97% of macrophages and DCs overlapped. Partial correlation analysis showed that macrophage and DC cell numbers were highly correlated. Again, our observation reinforced the fact that macrophages and DCs overlap to some extent. 16 The same group of cells phagocytose antigens and present antigens to other immune cells consecutively and effectively. Flow cytometry analysis also proved that cells expressing both CD68 and CD11c increased. These dual function cells are likely to play a great role in lupus atherosclerosis.
IgG deposited diffusely inside the atheroma. Additional IgG deposition was observed within the plaques of lupus mice. This observation was in accord with previous reports that IgG and immune complex tend to deposit in connective tissue of SLE patients, especially in the kidneys and skin. In this study, we observed accelerated IgG deposition in atheroma in lupus mice for the first time. It has been reported that IgG may be proatherogenic by the formation of oxLDL-containing complex and subsequent activation of macrophages. 17 We found that macrophage numbers increased simultaneously, so our study confirmed this theory again. Active phagocytosis contributed to excessive IgG within atheroma in lupus mice. However, the composition, source and function of deposited IgG remain to be elucidated in future studies.
We found that lupus mice were characterized by increased IgG and elevated spleen index, which reflects activated immune reactions. Similar results have been reported in other lupus combining atherosclerosis mice models. 20 Splenic lymphocytes and DC numbers change in parallel with aorta cell numbers in lupus mice, indicating that the formation of atheroma is regulated locally inside the atheroma, as well as in immune organs. In lupus mice, lymphocyte numbers were decreased, whereas DC numbers were increased in both the aortas and spleens. It has been reported that pristane effectively induced apoptosis of lymphocytes in vivo and in vitro.21,22 In our study, again we observed reduced splenic lymphocytes after pristane injection. The conventional view is that activated T cells accelerate atherosclerosis. However, a group of T cells called Tregs are vessel protective. Tregs reduced in atherosclerosis-prone LDLr–/– and ApoE–/– mice. 17 We assume that Tregs reduced significantly in lupus mice, and reduced Tregs accelerated atherosclerosis. Cell debris may activate DCs, thus DC numbers increased in the spleen as well as in the aorta.
Hydroxychloroquine treatment alleviated atherosclerosis by reversing abnormal immune status in lupus mice. Hydroxychloroquine has long been used by rheumatologists as an effective anti-SLE medication. In this study, dsDNA antibody reduced significantly in lupus mice administered with hydroxychloroquine. Again, the effectiveness of hydroxychloroquine in the management of SLE has been proved in mice. After taking hydroxychloroquine for 6 months, the atherosclerosis burden in both lupus mice and non-lupus mice reduced significantly. No significant change in blood lipids was detected, contrary to reports that hydroxychloroquine improved lipid metabolism in humans. 23 In lupus mice, hydroxychloroquine administration reversed immune cell number abnormality in aortas and spleen by ramping up lymphocyte numbers and diminishing total leukocytes, macrophages and DC numbers. This can be explained by previous findings that hydroxychloroquine mainly acted through raising the pH of lysosome and interfering with the function of macrophages and DCs. 11 Nevertheless, cell numbers in the aortas of non-lupus mice did not change. We inferred that hydroxychloroquine acts mainly on the abnormal immune system and hydroxychloroquine had little effect on the normal immune system. A previous study observed neither diminished dsDNA antibodies nor lymphocyte numbers change in the spleen after hydroxychloroquine treatment for 5 weeks. 12 We believe this difference was caused by the different duration of hydroxychloroquine administration. In our study, hydroxychloroquine was administered for 6 months. Prolonged use of hydroxychloroquine exerted a more profound impact on the immune system. Although atheroma area and IgG deposition reduced in non-lupus mice taking hydroxychloroquine, blood lipids and immune cell numbers did not change. This suggests that hydroxychloroquine also plays its role in some methods beyond our observations.
Hydroxychloroquine is the most widely used medication for autoimmune diseases. Hydroxychloroquine is effective in delaying the onset of SLE, as well as in reducing blood lipid levels, increasing bone mineral density, anti-cancer and anti-infections. 11 Due to its effectiveness and hypotoxicity, rheumatologists recommend SLE patients to take hydroxychloroquine for a long period of time. It has also been reported that hydroxychloroquine treatment is related to reduced vessel stiffness 24 and less carotid plaque. 25 Our study proved for the first time that hydroxychloroquine alleviates atherosclerosis directly by reversing the immune status abnormality caused by SLE. Hydroxychloroquine even inhibits plaque formation in non-lupus mice, although the mechanisms remain unclear. This result suggests that hydroxychloroquine may be an effective anti-atherosclerosis medication, especially in SLE patients.
Previous studies noted an elevation of IFN-α in the serum of SLE patients, 26 as well as the overexpression of ISGs.27,28 Lymphocytes and DCs activated by IFN-α play important role in the onset of lupus. We detected no significant change of IFN-α in the serum of lupus mice, as has been reported in lupus mice. 29 IFN-α mainly acts within tissues and is not necessarily elevated in the blood. In future studies, we will examine ISG expression in the aorta and observe lupus atherosclerosis severity in IFN-α receptor knock-out mice.
In summary, we have for the first time directly proved that the induction of SLE can aggravate atherogenesis. Although the onset of SLE reduces blood lipids slightly, both innate and adaptive immune deviation contributes to aggravated atherosclerosis in lupus mice. Hydroxychloroquine prevents atherosclerosis progression mainly by reversing the immune status abnormality caused by SLE. Thus, anti-inflammatory, anti-rheumatism therapies and hydroxychloroquine provides a new possible strategy for treating patients with atherosclerosis.
Footnotes
Author contribution
TL and NS performed the experiments, TL analyzed the data and prepared the manuscript. GJS provided suggestions for manuscript revision. SZ assisted with the design of the study. X-WD provided the suggestions for revising the manuscript. SZ helped in the design of the experiments. HN was responsible for the overall design of the study, analysis and interpretation of the data, and manuscript preparation. All authors read and approved the final manuscript.
Data availability
The datasets generated and analyzed for the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key R&D Programs of China, grant/award number: no. 2017YFC1103603 and no. 2017YFA0105204, and CAMS Initiative for Innovative Medicine of China, grant/award number: no. 2016-12M-1-006.
