Abstract
Objective
To conduct a proof-of-concept pilot evaluation of the self-directed format of Walk With Ease (WWE), a 6-week walking program developed for adults with arthritis, in patients with systemic lupus erythematosus (SLE).
Methods
This was a single arm, 6-week pre- and post-evaluation of the self-directed WWE program to assess feasibility, tolerability, safety, acceptability, and effectiveness. Adult patients with physician-diagnosed SLE were recruited to participate during regularly scheduled visits to an academic rheumatology clinic. Self-reported outcomes of pain, stiffness, and fatigue were assessed by visual analog scales (VAS) and the Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-fatigue) scale at baseline and at completion of the 6-week program. Patients also completed a satisfaction survey at the end of the program. Multivariate linear regression models were used to calculate mean changes between baseline and 6-week follow-up scores, adjusting for covariates. Mean change scores were used to estimate effect sizes (ES).
Results
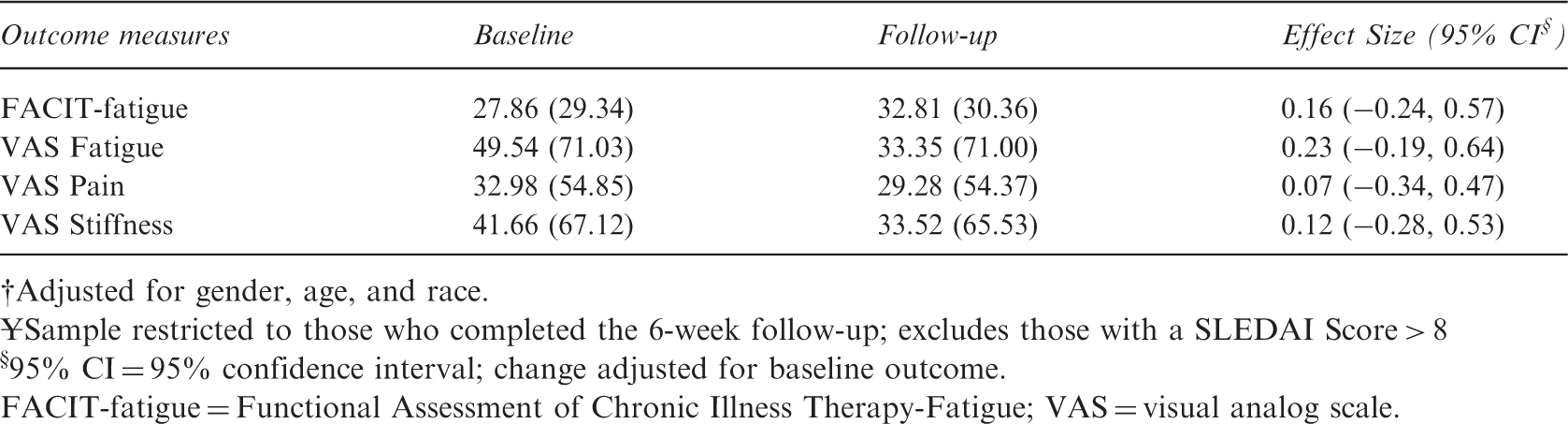
At 6 weeks, 48 of the 75 recruited participants completed the WWE program. Participants experienced modest improvements in stiffness and fatigue (ES = 0.12 and ES = 0.23, respectively, for VAS scores; ES = 0.16 for FACIT-fatigue score) following the intervention. The majority of participants reported satisfaction with the program (98%) and benefitted from the workbook (96%).
Conclusions
The self-directed format of WWE appears to reduce stiffness and fatigue in patients with SLE. It also seems to be a feasible and acceptable exercise program to patients with SLE. Larger studies are needed to confirm these findings.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a systemic autoimmune rheumatic disease that can affect multiple organ systems and is characterized by a fluctuating disease course. A recent systematic review of epidemiologic studies estimates the incidence and prevalence of SLE in North America at 23.1/100,000 person-years and 241/100,000 people, respectively. 1 SLE is more common in women than in men, with a ratio of approximately 6:1. Among ethnic groups in North America, individuals of African descent have the highest incidence (31.2/100,000 person-years), followed closely by Native Americans (30.0/100,000 person-years). 1 The incidences of SLE in North America, per 100,000 person-years are 22.2, 18.0, and 16.7, respectively, among Hispanics, Caucasians, and Asians. 1
SLE disease activity is typically assessed as a composite of clinical features and laboratory data via one of several available validated and reliable instruments, the most commonly used being the systemic lupus erythematosus disease activity index (SLEDAI). 2 However, even among patients with relatively well-controlled disease (e.g., with immunosuppression) and low disease activity based on such assessments, pain, stiffness, and fatigue are common, potentially debilitating symptoms. Musculoskeletal pain affects 50–61% of SLE patients, morning stiffness affects approximately half of patients, and fatigue impacts up to 60–90% of patients with SLE; and the presence of these symptoms is not always correlated with disease activity.3–7 Multiple studies indicate that the presence of fatigue in patients with SLE has instead been correlated with the presence of comorbid conditions such as fibromyalgia and depression, as well as reduced aerobic fitness.6–9 Because these common symptoms do not always correlate with disease activity (active inflammation), non-pharmacologic means to manage them may be a viable option.
A growing body of evidence suggests that exercise as an adjunct therapy in patients with SLE can be effective at reducing pain and fatigue.10–17 For example, Yuen et al. evaluated pain before and after a ten-week Wii Fit exercise program and found that pain severity and total pain rating index were reduced, though the latter finding did not reach statistical significance. 16 In a randomized study of patients with SLE who completed either an isometric exercise or a bicycle exercise program, Bogdanovic et al. found that both groups experienced statistically significant improvements in all areas of quality of life as measured by Short Form 36 (SF36), including body pain, as well as reductions in fatigue based on the Fatigue Severity Scale (FSS). 10
Individuals with SLE tend to be less physically active and more sedentary than individuals without SLE. 18 Factors possibly associated with decreased physical activity among those with SLE include fatigue, pain, stiffness, and more severe depressive and anxiety symptoms.18–20 Patient concern about exercise causing a disease flare is another potential barrier to physical activity. 20 However, multiple studies have shown that disease activity does not worsen with exercise and that fatigue, depression, and physical fitness were, in fact, improved following exercise-based interventions.10,12,14,17 In at least one study, the majority of patients (92%) believed physical activity ultimately would improve SLE symptoms if they could overcome initial hurdles (e.g., concern about exacerbating SLE in the short term). 20 These patients reported that walking was the preferred exercise, as it is “practical,” “sustainable,” and “not strenuous.” 20
Walk With Ease (WWE) is a 6-week evidence-based walking program developed for adults with arthritis that is available in either group (instructor-led) or self-directed formats. 21 This program has been previously validated for use in people with various types of rheumatic and musculoskeletal diseases, such as osteoarthritis and rheumatoid arthritis, and was shown to improve symptoms of pain, stiffness, and fatigue with both formats at 6 weeks.22–24 Further, at one-year follow-up, participants who had completed the self-directed format had sustained significant improvements with regard to pain and stiffness by the visual analog scale (VAS) compared to participants who had completed the group format. 24
Although walking has been studied in SLE patients in a few cases, a dedicated self-directed walking program such as WWE has not been previously evaluated in these patients.11,12,14,15 Our objective was to conduct a proof-of-concept pilot evaluation of the self-directed format of WWE in a new rheumatic disease population, SLE patients. Based on outcomes of WWE in osteoarthritis and rheumatoid arthritis, we hypothesized that participants will experience improvement in pain, stiffness and fatigue symptoms after completion of the 6-week program. We further sought to assess the feasibility, tolerability, safety, acceptability and effectiveness of the WWE program in patients with SLE.
Participants and methods
Participants
Participants were recruited during routine visits to the outpatient rheumatology clinic at the University of North Carolina at Chapel Hill in Chapel Hill, North Carolina. To be eligible for the study, participants had to have a physician diagnosis of SLE, be older than 18 years of age, and engage in only limited walking or other moderate physical activity (i.e., no more than 150 minutes/week). Patients were excluded if they already engaged in moderate physical activity for more than 150 minutes per week, were pregnant, or if their SLEDAI score was >8, indicating high disease activity.
Study design
This was a 6-week pre- and post-evaluation pilot study using the self-directed format of the WWE program, which has its foundation in the social cognitive theory conceptual framework. 25 All participants enrolled received a copy of the WWE workbook and were encouraged to use it as a guide during the program. Participants completed self-report assessments at baseline and at the end of the 6-week program, and a satisfaction survey at the 6-week assessment.
Data collection
After providing written informed consent, the participant completed the baseline self-report questionnaire and received a copy of the WWE workbook. Patient demographic information, including age, sex, and comorbidities was collected. One of the study physicians (SS or KK) calculated the baseline SLEDAI score after the visit based on the clinic note from the study entry visit and the associated laboratory results. SLEDAI scores could not be calculated for some patients because laboratory tests were obtained only if clinically indicated at the time of the visit, not for the sole purpose of calculating a SLEDAI score. The self-report questionnaire used a 100 mm VAS to record the severity of pain, stiffness, and fatigue in the previous seven days, ranging from 0 (not experiencing this symptom) to 100 (“pain/stiffness as bad as it could be” or “fatigue is a major problem”). 26 Fatigue severity in the previous 7 days was also assessed by the FACIT-fatigue scale, a 13-item scale assessing different aspects of fatigue that has been validated in SLE.27,28 FACIT-fatigue is scored on a Likert scale from 4 (not at all fatigued) to 0 (very much fatigued), where lower scores indicate more severe fatigue.
Participants tracked their walking on paper logs provided at study entry. Specifically, participants recorded the duration walked and the distance walked, if known, for each day that they walked. The logs were returned via pre-stamped envelopes at the end of the intervention or were brought back to clinic at the next scheduled follow up visit, except by participants for whom end of study assessments were conducted by phone and who verbally provided the information recorded on their paper logs.
After completing the 6-week walking program, participants completed a self-report follow-up questionnaire along with a satisfaction survey about their experience with the program either mailed or returned during a scheduled follow-up visit. The satisfaction survey addressed whether and how much participants were able to walk during the intervention, confidence that the participant would continue to be physically active following the intervention, how much of the workbook the participant read and whether it was found to be helpful, and whether the participant was satisfied with the program. These end of study assessments were conducted by phone for some participants who were not able to meet in person.
Data analysis
Descriptive statistics were computed for covariates at baseline to characterize the population. Continuous variables were expressed as means and standard deviations and categorical variables as frequency and percentage. All tests were two-sided and considered statistically significant at the p = 0.05 level. All analyses were carried out using SAS software version 9.4 (SAS Institute Inc., Cary, NC).
We used multivariate linear regression models to calculate mean changes between baseline and 6-week follow-up scores, controlling for baseline outcome score and the covariates age, sex, and race. Mean change scores were used to estimate effect sizes (ES), expressed as Cohen's d, which was calculated by comparing the mean change scores from baseline to 6 weeks divided by the pooled standard deviation. 29 The outcomes related to participant satisfaction with the WWE program were reported as percentages.
Ethics
The study was approved by the University of North Carolina at Chapel Hill institutional review board.
Results
Participants
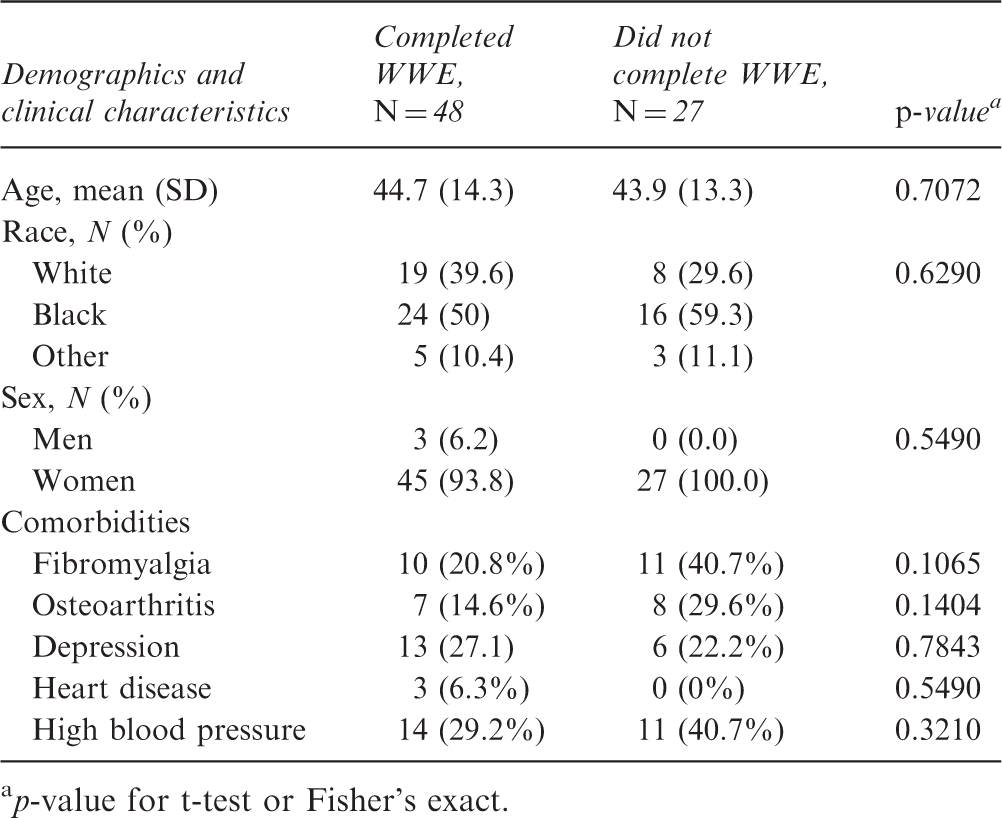
A total of 75 participants were recruited into the study and 48 completed the study (Figure 1). Reasons for study withdrawal among the 27 non-completers included non-compliance with the program, hospitalization, lack of desire to continue the program, intolerance to the exercise, and personal reasons. There were no statistically significant differences in the baseline characteristics or frequency of comorbidities between the participants who completed the study and those who did not (Table 1). We cannot, however, completely rule out the possibility that the differences in frequency of comorbidities between completers and non-completers may have been clinically important, given the numeric disparity.
Study enrollment and retention. Baseline characteristics for SLE WWE completers and non-completers p-value for t-test or Fisher's exact.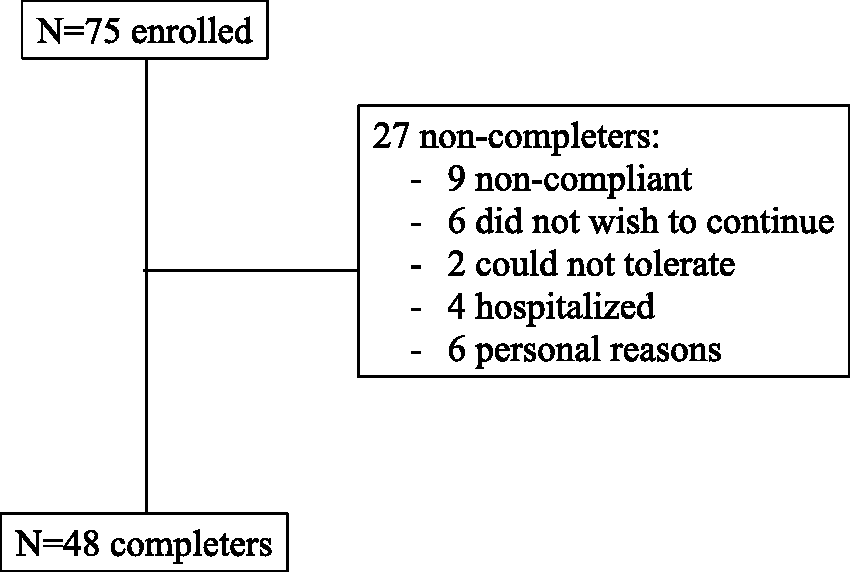
Clinical manifestations of SLE WWE completers

Exercise outcomes
Exercise outcomes
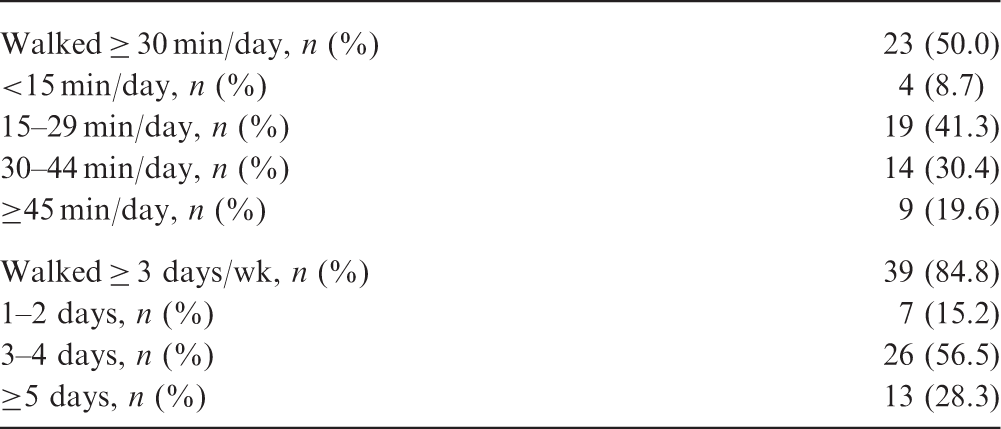
Patient reported outcomes
Adjusted for gender, age, and race.
Sample restricted to those who completed the 6-week follow-up; excludes those with a SLEDAI Score > 8
§95% CI = 95% confidence interval; change adjusted for baseline outcome.
FACIT-fatigue = Functional Assessment of Chronic Illness Therapy-Fatigue; VAS = visual analog scale.
For most outcome variables assessed we did not observe dose-response trends according to time spent walking. Exceptions were observed decreasing SLEDAI score with increasing number of days walked (p = 0.05) and observed decreasing VAS Stiffness with increasing number of minutes/day walked (p = 0.04). There were no significant differences in improvement when compared by gender, race, or comorbidities (data not shown).
Patient satisfaction
Nearly all participants reported that they were satisfied with the program and that they had the confidence in their ability to continue engaging in physical activity (Figure 2). About 96% of participants believed that they benefitted from the workbook, and about two-thirds of participants felt that the workbook had motivated them.
Patient satisfaction with the Walk With Ease program.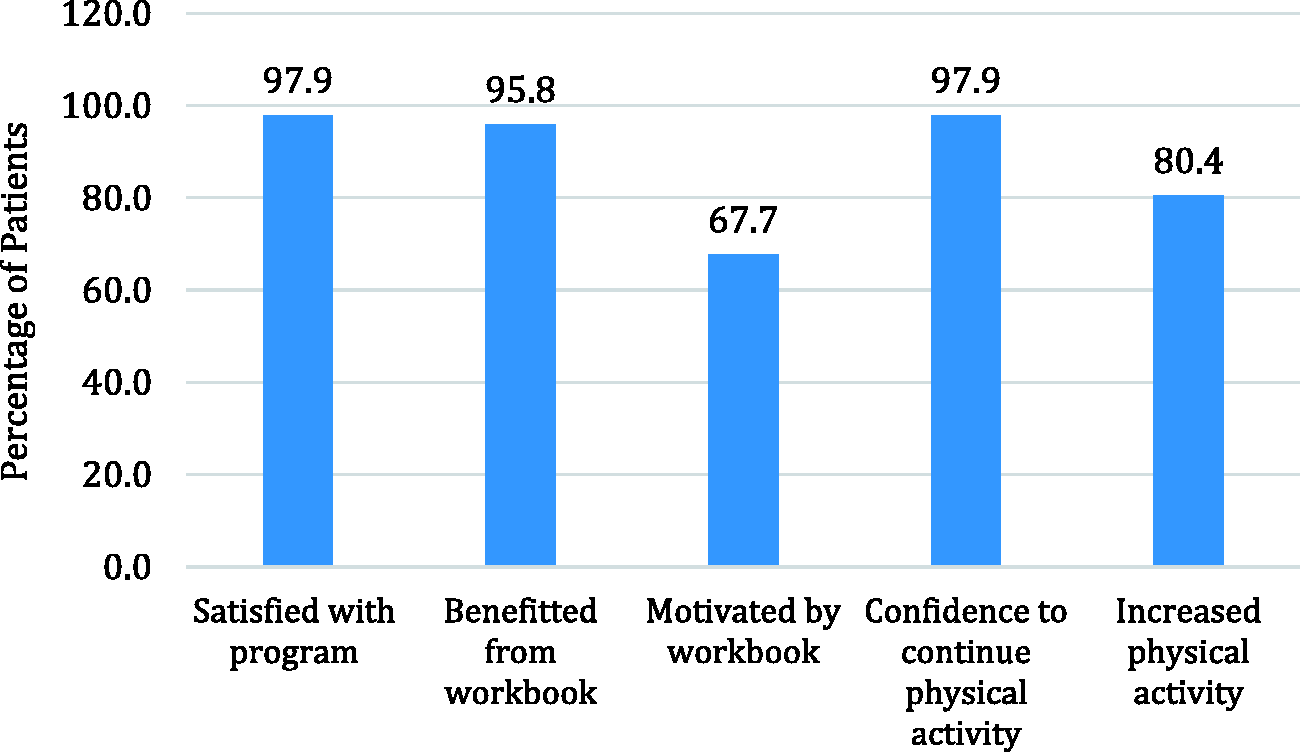
Discussion
Ours is the first study to evaluate the WWE program in SLE patients. The results indicate that the WWE program, previously validated in patients with osteoarthritis and rheumatoid arthritis, can modestly improve symptoms of pain, stiffness, and fatigue in patients with SLE. We also demonstrate that this program is acceptable to patients with SLE. These results are in keeping with the outcomes of other studies that show exercise to be an effective means of reducing pain and fatigue in SLE patients without exacerbating disease activity.10,11,13–16 Our study adds to the literature since we show that a readily available and self-directed walking program (workbook may be purchased online) can successfully reduce symptoms of pain, stiffness, and fatigue in SLE patients and potentially improve quality of life. Importantly, for the potential uptake of this program by patients with SLE, walking has been reported to be a preferred form of exercise by SLE patients. 20
The SLE literature regarding the effect of exercise contains studies of both supervised and predominantly unsupervised exercise programs.11,12,14–16,30 Despite one study concluding that patients derived little benefit from an unsupervised home exercise program, 30 other studies of unsupervised home exercise programs showed statistically significant improvement in fatigue and/or pain by at least one measure, results that are similar to ours.14–16 A small study by Ramsey-Goldman et al. demonstrated good adherence to both supervised and unsupervised exercise regimens among patients with SLE. 13
A Cochrane systematic review from 2005 found that, while patients with cardiovascular disease or chronic obstructive pulmonary disease did better initially with a center-based program, long-term exercise adherence was better with a home-based program. 31 Additionally, Callahan et al. evaluated WWE in group and self-directed formats among patients with arthritis, and reported that patients who completed the self-directed format had gains (in pain and stiffness VAS among other endpoints) sustained over the long term that were not maintained by patients in the group format. 24 These data strengthen the relevance of our study because they indicate that self-directed exercise programs, such as the one piloted in our study, are successful and can have longevity, which is important for maintaining the health benefits of exercise.
Our study has several limitations. The sample size was small; however, this study was designed as a pilot study to assess feasibility and acceptability. We did not plan to assess statistical significance of results, but rather to assess whether symptom improvements were seen. There was also a substantial loss to follow-up, with approximately one-third of participants not completing the study for various reasons. As noted above, there were no statistically significant differences between completers and non-completers. However, the numerical differences in frequency of several comorbidities suggest a potentially clinically important difference which cannot be completely ruled out as a contributor to loss to follow up among some patients. Another limitation was the reliance on patient reporting of walking via an exercise log rather than using a more objective measure of physical activity such as a pedometer. A 2011 study of SLE patients by Yuen et al. found that study participants tended to log more exercise than they had actually performed. 16 Therefore, this method of data collection is likely to have overestimated the amount of exercise the participants performed. We think, however, that this is a reasonable starting point for a pilot study and that it may make our results more widely applicable. Lastly, as we did not plan to have additional study visits, we were not able to calculate a SLEDAI score in most participants following the intervention.
In a future study of WWE in patients with SLE, we would like to confirm our findings in a larger group of participants and to compare an intervention group with a control group. The control group could either be a non-intervention group or a group participating in the instructor-led format of the WWE program. We plan to track exercise/walking more objectively in this future study through the use of pedometers and/or wearable technology/activity trackers (e.g., Fitbit), and to incorporate use of the WWE phone app, which may appeal to technology-savvy SLE patients. It would also be interesting to assess if patients experience improvement in cardiovascular health with the self-directed WWE program, given the markedly increased risk of cardiovascular morbidity and mortality associated with SLE. 32
This pilot study demonstrated that the self-directed format of WWE is acceptable and feasible for patients with SLE. Furthermore, completion of the 6-week program can improve symptoms of pain, stiffness, and fatigue in SLE patients.
Footnotes
Acknowledgments
We would like to thank and acknowledge all the lupus patients who participated in this study. Editorial assistance was provided by Angela Cimmino, funded by the Fund for Excellence in Lupus & Sjogren's at the UNC Thurston Arthritis Research Center.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Saira Z Sheikh has served as a consultant on an Advisory Board for GSK. No other authors have any financial disclosures to report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a pilot study grant from the NC Translational and Clinical Sciences (NC TraCS) Institute supported by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health (grant number UL1TR002489). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.