Abstract
Peptide therapeutics hold attractive potential. However, the proper stabilization of such therapeutics remains a major challenge. Some peptides are marginally stable and are prone to degradation. Therefore, in addition to chemical modifications that can be introduced in their sequence, a wide variety of excipients are added in the formulation to stabilize them, as is also done routinely for protein therapeutics. These substances are supposed to suppress peptide/protein aggregation and surface adsorption, facilitate their dispersion and additionally to provide physiological osmolality. Particular attention has to be paid to the choice of such excipients. Here we highlight the observation that in certain clinical situations, an excipient that is not totally inert can play a highly damaging role and mask (or even reverse) the beneficial effect of a molecule in clinical evaluation. This is the case, for instance, of trehalose, a normally safe excipient, which notably has proven to act as an activator of autophagy. This excipient, although used efficiently in several therapeutics, adversely impacted a phase IIb clinical trial for human and murine lupus, a systemic autoimmune disease in which it has been recently discovered that at the base line, autophagy is already abnormally enhanced in lymphocytes. Thus, in this particular pathology, while the peptide that was tested was active in lupus patients when formulated in mannitol, it was not efficient when formulated in trehalose. This observation is important, since autophagy is enhanced in a variety of pathological situations, such as obesity, diabetes, certain neurological diseases, and cancer.
Introduction
According to the Merriam-Webster dictionary, an excipient is “a usually inert substance (as gum arabic, syrup, lanolin, or starch) that forms a vehicle (as for a drug or antigen); especially: one that in the presence of sufficient liquid gives a medicated mixture the adhesive quality needed for the preparation of pills or tablets”. In reality, a number of classical excipients show notable side effects, and these are clearly mentioned on the accompanying package leaflet of drugs. For example, in individuals suffering from phenylketonuria, aspartame or polyethylene glycol can provoke digestive disturbances. This is a serious but long-lasting and rather well-controlled aspect of drug usage that is carefully taken into consideration by pharmaceutical companies and supervised by competent international authorities. What is much less appreciated is the possible influence of such ‘safe’ excipients when associated with newly developed compounds that enter into clinical trials. This is the case, for example, of trehalose, a non-hygroscopic alpha-linked disaccharide formed by two molecules of glucose, used as an excipient in formulated drugs.
Trehalose is approved by the US Food and Drug Administration and received the GRAS (Generally Recognized as Safe) rating in 2000. It is also approved in Europe, Japan, Taiwan and Korea, and is Kosher certified. This disaccharide is present as protector of protein integrity in several commercially available therapeutic products, notably monoclonal antibodies, including Herceptin®, Avastin®, Lucentis®, and recombinant proteins such as Advate® in hemophilia. 1 It is also used in food and cosmetic products. This non-reducing sugar exists under free form in plants, algae, insects, fungi and micro-organisms, sometimes in abundant amounts, but not in mammals. It is ingested daily in the normal human diet and is metabolized by the trehalase enzyme residing in the intestinal villi. The ability of trehalose to stabilize aggregation-prone proteins is currently exploited to design novel strategies in the treatment of polyglutamine diseases, such as Huntington’s chorea and Alzheimer’s disease.2,3 Trehalose may also improve bone metabolism and prevent osteoporosis.4,5 There is, therefore, no apparent intrinsic risk in the use of trehalose, and potential applications are widely investigated.
Trehalose, however, has been reported to be an autophagy enhancer.3,6 Autophagy is an essential mechanism by which a cell degrades its own cytoplasmic components through the lysosomal machinery. This tightly regulated homeostatic process is central in the cell response to stress, and also in controlling diverse aspects of immunity and inflammation.7–9 Enhancing autophagy can be beneficial (in cancer or certain neuronal diseases, for example) but also detrimental; in any case, it is not neutral. It is worth mentioning that in this regard trehalose has been used experimentally as autophagy enhancer, as 3-methyladenine is classically used to inhibit autophagic activity in in vitro experiments.10,11 It has also been applied as facilitator for intramuscular administration of naked plasmid to improve transgene expression. 12
When the excipient plays the role of an active substance and can interfere with a potential drug
Drop-out rates and responder rates in phase IIb clinical trials of Lupuzor and Forigerimod
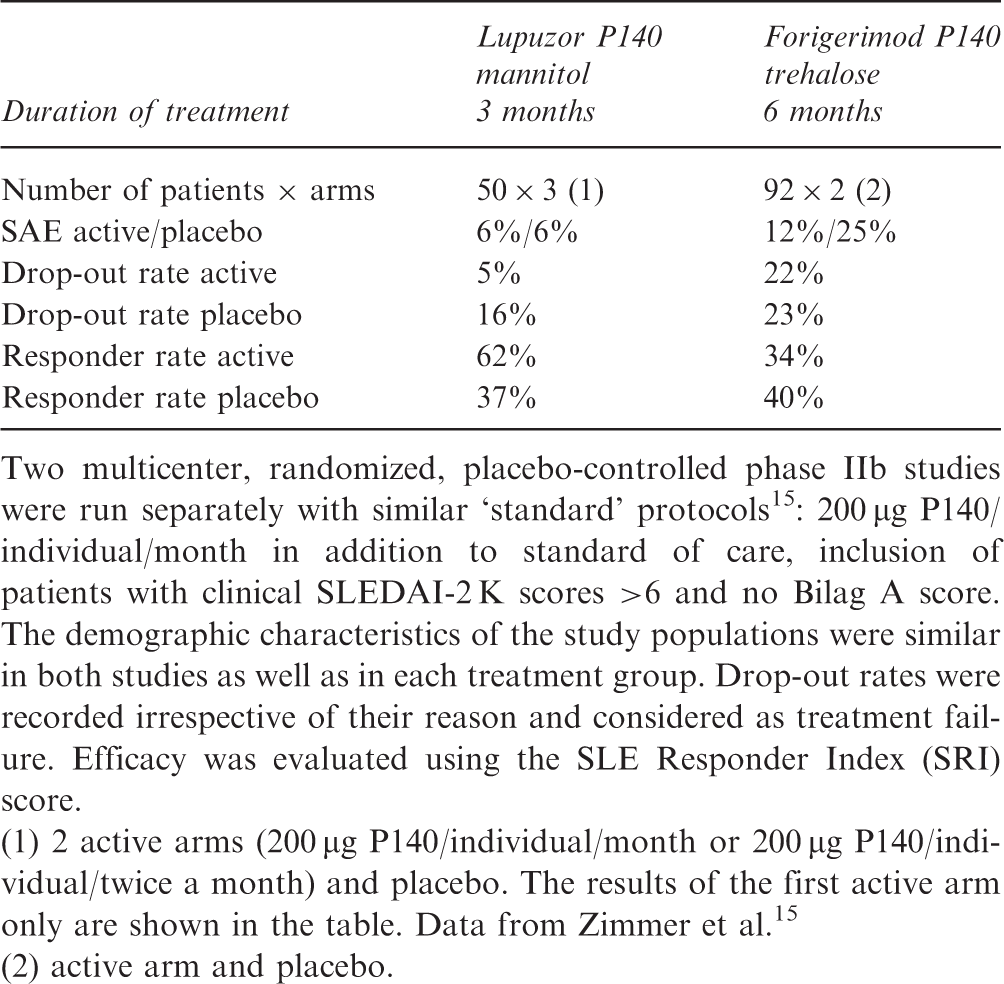
Two multicenter, randomized, placebo-controlled phase IIb studies were run separately with similar ‘standard’ protocols 15 : 200 µg P140/individual/month in addition to standard of care, inclusion of patients with clinical SLEDAI-2 K scores >6 and no Bilag A score. The demographic characteristics of the study populations were similar in both studies as well as in each treatment group. Drop-out rates were recorded irrespective of their reason and considered as treatment failure. Efficacy was evaluated using the SLE Responder Index (SRI) score.
(1) 2 active arms (200 µg P140/individual/month or 200 µg P140/individual/twice a month) and placebo. The results of the first active arm only are shown in the table. Data from Zimmer et al. 15
(2) active arm and placebo.
Our increasing knowledge of the mechanism of action of P140 peptide in mice and human cells allows us to propose a reasoned explanation for the grounds on which trehalose, and not mannitol, counteracts the P140 effect in a lupus setting. Our recent data raised in MRL/lpr-prone mice showed that P140 peptide targets autophagy,
17
and more particularly chaperone-mediated autophagy (CMA) (unpublished observation). Notably, we have demonstrated that the P140 inhibitory effect on CMA results from its ability to alter the integrity of the HSC70/HSPA8-HSP90 heterocomplex of lysosomal chaperones (unpublished observation). Expression of HSC70 and LAMP-2 A, the two main CMA components, which is increased in MRL/lpr B cells, is down-regulated after P140 treatment. Taking all these findings together, we proposed that the effect of P140 on the HSPA8-HSP90 heterocomplex leads to endogenous (auto)antigen processing interference followed by a much weaker loading to MHCII molecules, leading in turn to a lower activation of autoreactive T cells and B-cell help (Figure 1). P140 peptide behaves as a modulator (not as an immunosuppressor)
18
of the autoimmune response, whose main effect is thus to weaken MHCII autoantigen presentation through a cellular mechanism depending on the autophagy process. This set of findings is central, since we showed recently that macroautophagy (the best characterized type of autophagy) is abnormally enhanced in T lymphocytes from lupus mice and patients,
19
and that cross-talk exists between macroautophay and CMA.
20
Therapeutic targets in systemic lupus erythematosus. A range of target molecules and corresponding therapeutic agents currently under investigation are illustrated. Belimumab (Benlysta) is the first FDA-approved treatment for lupus in over 50 years. In contrast to the other agents, including Belimumab, P140/Lupuzor targets the up-stream autoreactive response by acting as a modulator of antigen presentation by major histocompatibility complex (MHC) class II molecules.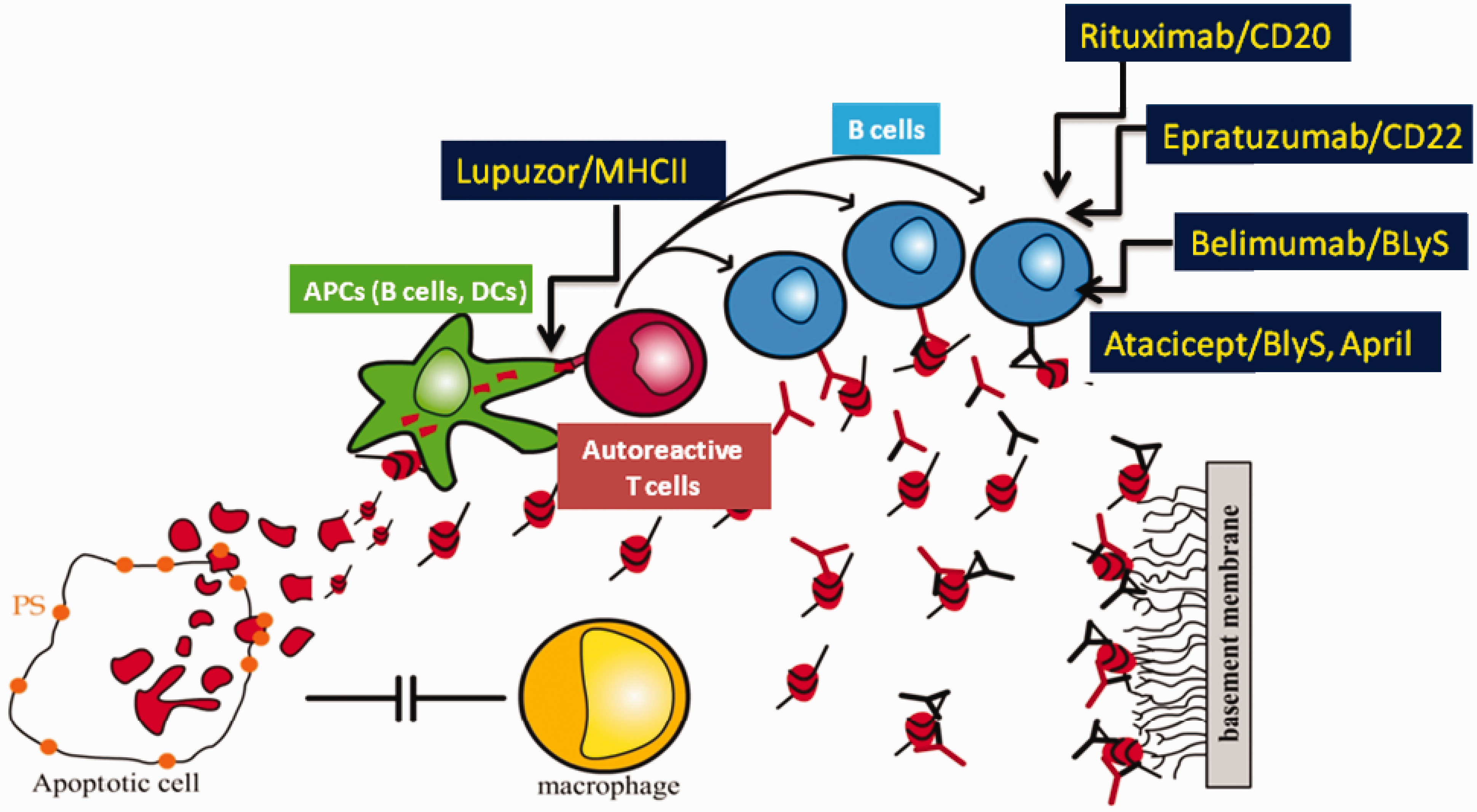
In this context of pathologically elevated macroautophagy activity, any component that reduces this enhanced activity might be potentially favorable, while any activator would be rather harmful. It is the case that trehalose, which is a known mTOR-independent activator of the macroautophagy pathway,3,21 further activates a mechanism that is already deregulated towards an excess of activity. 19
Trehalose virtually shuts down the immunomodulatory properties of the P140 peptide in MRL/lpr mice
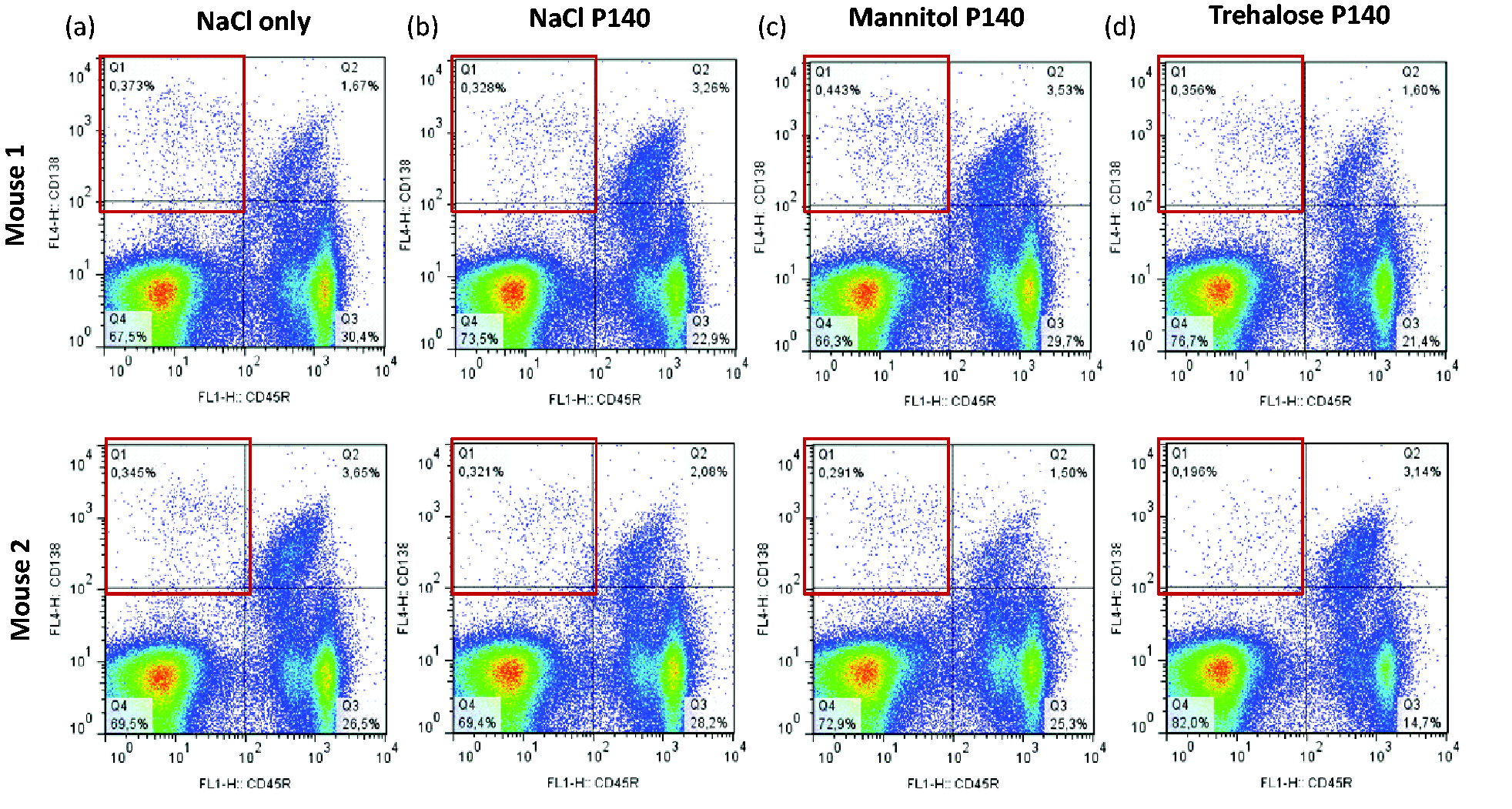
The results with Forigerimod were disappointing because both the brake (P140 peptide) and the accelerator (trehalose) pedals were pressed down. If the levels of circulating cytokines, as measured using non-quantitative kit RayBio® cytokine antibody arrays, were not significantly different in the peripheral blood of MRL/lpr mice that received the peptide in mannitol and trehalose (four consecutive administrations by the intraperitoneal route; two mice per group; measurements at day 5), the adverse impact of trehalose was clearly notable when other parameters were followed. Thus, the capacity of P140 peptide to reduce the greatly increased peripheral cellularity in lpr-bearing MRL mice was significantly reduced when the peptide was injected intravenously in the trehalose formulation compared with a mannitol formulation (Figure 2(a)). The level of circulating CD45R/B220-CD138+-expressing plasmocytes as measured by flow cytometry analysis remained low in all groups of mice with, however, unequal results in mice that received P140 in trehalose (Figure 3). This high variability of responses was also observed when the level of peripheral white cells was measured after administration of P140 peptide in trehalose as compared with mannitol (Figure 2(a)).
22
In vivo effect of trehalose administrated alone or associated to P140 peptide, on MRL/lpr lupus mice. Effect on the viability of CD45R/B220-CD138+ plasmocytes of P140 peptide administrated in different media to MRL/lpr lupus mice.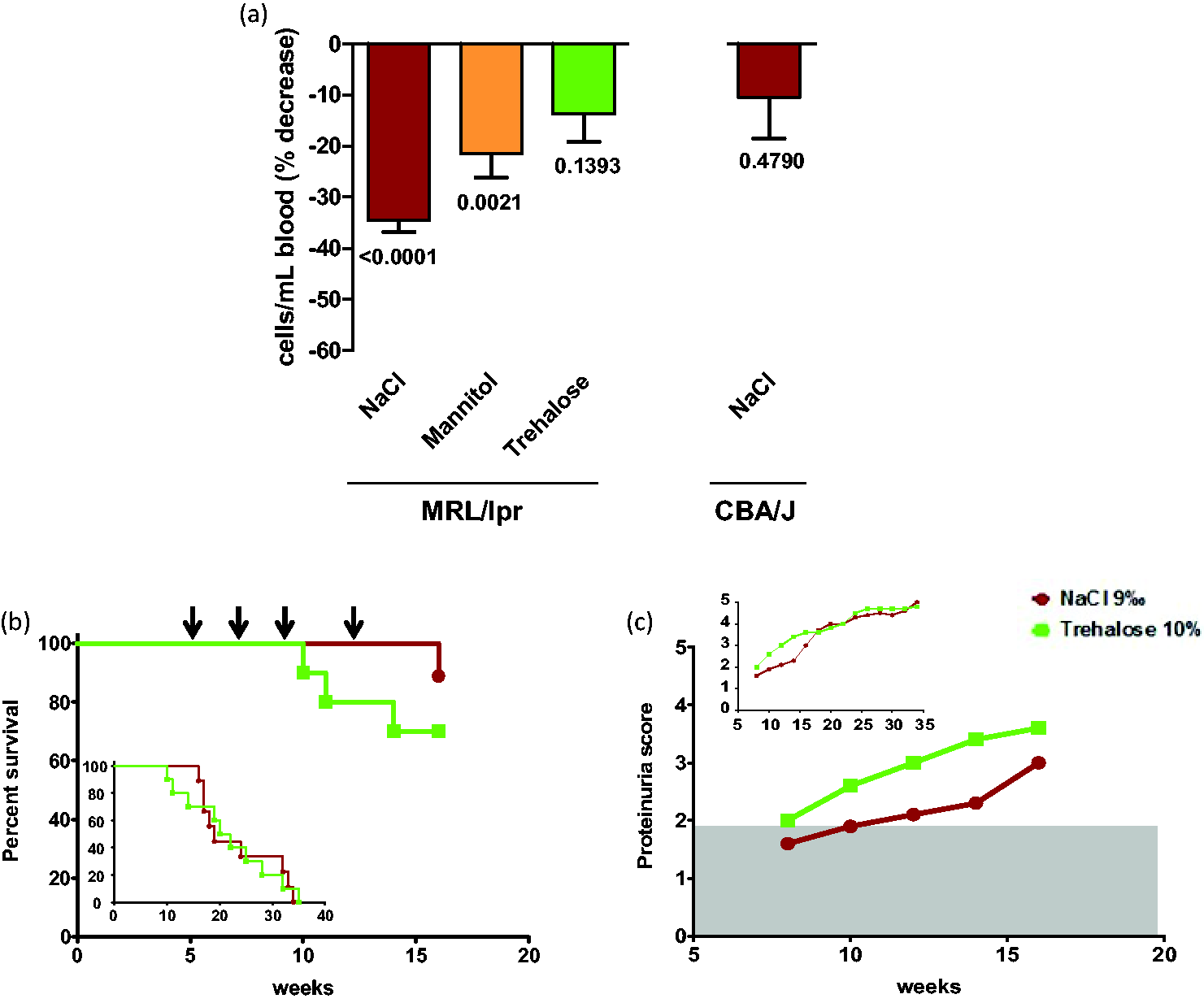
We also discovered that trehalose administrated alone tends to accelerate lupus disease in lupus-prone mice (proteinuria and survival) when compared with saline only (Figure 2(b) and (c)). This harmful effect disappeared after the cessation of trehalose administration.
A similar effect of trehalose on P140 peptide in a human setting?
In the open-label follow-up of 11 patients given Forigerimod (CEP-3075) following the CEP-33457 phase IIb clinical trial, our group (DJW) observed that, unlike Lupuzor, there was no evident response for several months. Approximately 6 months later clear-cut improvements in response measures were observed, suggesting that at this stage the negative influence of trehalose was overcome. These observations, as well as the clinical data of the CEP-33457 phase IIb clinical trial indicating that the peptide in trehalose tended even to be less effective than placebo (Table 1), were closely corroborated by data generated ex vivo using human peripheral cells (buffy coats). While, in a dose-dependent manner, P140 peptide in saline provoked a down-expression of HLA molecules at the surface of B cells (as also found in MRL/lpr B cells),
17
P140 in trehalose, on the contrary, had no effect on HLA cell expression even when the P140:trehalose ratio was increased (Figure 4). Taken together, these converging findings are indicative of a negative effect of trehalose when associated with P140 peptide (Forigerimod), a result that was predicted when considering the metabolic pathway that is modulated by the bioactive peptide P140. Furthermore, beside this effect as an autophagy activator
3
that is undesirable in lupus conditions, trehalose might also exert its adverse effect through another of its physicochemical properties; that is, its capability to substitute water molecules around proteins/membranes. This property, which is put forward to explain how trehalose displays its bioprotective action,1,2 might transiently alter the folding and mobility of the P140 peptide and also of partner macromolecules, the interaction of which is critical in its mode of action.
Effect of trehalose on the capacity of P140 peptide to decrease HLA expression at the surface of human B cells.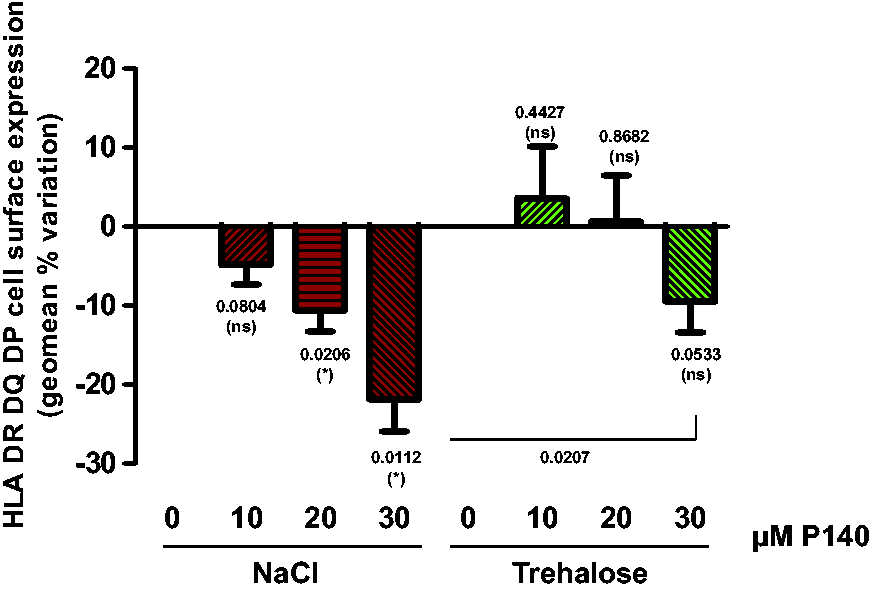
Concluding remarks
Trehalose is not an innocent compound. It displays some active properties which can directly or indirectly alter the behavior of potential therapeutics evaluated in clinical trials. Although it is exploited with success in a number of pharmaceuticals, 1 special attention should be paid to the risk of trehalose impacting negatively on the success rate of a drug candidate evaluated in trehalose. It may well be a matter of an inappropriate excipient.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
DJW was a consultant for Cephalon and SM a consultant for ImmuPharma.
Acknowledgements
We thank Nicolas Schall for help with experimental analyses. Research in SM’s laboratory is financially supported by the French Centre National de la Recherche Scientifique, Région Alsace, the Laboratory of Excellence Medalis (ANR-10-LABX-0034), Initiative of Excellence (IdEx), Strasbourg University, and ImmuPharma France.
