Abstract
A pooled post-hoc analysis of the phase 3, randomized, placebo-controlled BLISS trials (1684 patients with active systemic lupus erythematosus (SLE)) was performed to evaluate the effect of belimumab on renal parameters in patients with renal involvement at baseline, and to explore whether belimumab offered additional renal benefit to patients receiving mycophenolate mofetil at baseline. In addition to belimumab or placebo, all patients received standard SLE therapy. Patients with severe active lupus nephritis were excluded from the trials. Over 52 weeks, rates of renal flare, renal remission, renal organ disease improvement (assessed by Safety of Estrogens in Lupus Erythematosus National Assessment–Systemic Lupus Erythematosus Disease Activity Index and British Isles Lupus Assessment Group), proteinuria reduction, grade 3/4 proteinuria, and serologic activity favored belimumab, although the between-group differences in most renal outcomes were not significant. Among the 267 patients with renal involvement at baseline, those receiving mycophenolate mofetil or with serologic activity at baseline had greater renal organ disease improvement with belimumab than with placebo. Limitations of this analysis included the small patient numbers and the post-hoc nature of this pooled analysis. The results suggest that belimumab may offer renal benefit in patients with SLE. Further study is warranted in patients with severe active lupus nephritis.
Introduction
Belimumab is a human immunoglobulin-G1λ monoclonal antibody that inhibits the biologic activity of soluble B-lymphocyte stimulator. Elevated serum levels of soluble B-lymphocyte stimulator protein correlate with disease activity in systemic lupus erythematosus (SLE). 1 Two phase 3 trials – BLISS-52 and BLISS-76 – demonstrated that a significantly greater proportion of patients responded (using the SLE Responder Index (SRI)) to belimumab plus standard SLE therapy than to placebo plus standard therapy. 2,3 The safety profile of belimumab plus standard therapy was similar to that of placebo plus standard therapy in both trials.
Nephritis is a frequent cause of SLE-associated morbidity and mortality. Renal involvement occurs in 40%–70% of all patients with SLE, with end-stage renal disease developing in 10%–20% of all patients. 4–7 Recently, the American College of Rheumatology published guidelines on the screening, treatment, and management of lupus nephritis, summarizing induction and maintenance therapy approaches from severe active to quiescent renal disease. 8
The BLISS trials were not designed to specifically assess the effects of belimumab on renal parameters. 2,3 Patients with severe active lupus nephritis were excluded from the studies, including those patients with proteinuria >6 g/24 h or serum creatinine >2.5 mg/dl, and those who required hemodialysis or high-dose prednisone within 90 days of study initiation. Renal outcomes measured for this post-hoc pooled analysis included renal flare rate (as defined by Alarcón-Segovia et al. 9 ), renal remission rate, Safety of Estrogens in Lupus Erythematosus National Assessment–SLE Erythematosus Disease Activity Index (SELENA/SLEDAI) or British Isles Lupus Assessment Group (BILAG) renal involvement, proteinuria, hematuria, and creatinine. The trials also assessed the rate of normalization of anti-double-stranded DNA (anti-dsDNA) and complement (C3 and C4). These serologic markers are among the indications for a renal biopsy to diagnose lupus nephritis. 8 Levels of anti-dsDNA and C3 and C4 may predict disease activity, disease exacerbations such as flares, and patient response to treatment. 8,10–13 The present post-hoc pooled analysis of the BLISS trials was, therefore, performed to evaluate the effect of belimumab on renal parameters in patients with active SLE and renal involvement at baseline.
Mycophenolate mofetil (MMF) has become a major treatment modality for lupus nephritis. 8,14–16 A proportion of patients in the BLISS trials were receiving MMF at baseline as background SLE therapy. The present analysis, therefore, also examined whether patients with stable renal involvement at baseline who were receiving MMF derived additional renal benefit from belimumab.
Methods
The BLISS studies were multicenter, multinational, placebo-controlled trials in which patients with active SLE were randomized to placebo or to belimumab 1 or 10 mg/kg. 2,3 Corticosteroids, antimalarials, and immunosuppressants, including MMF, were permitted as background standard SLE therapy in addition to the study drug. BLISS-52 (N = 865) was conducted in Latin America, Eastern Europe, and the Asia-Pacific region; BLISS-76 (N = 819) was performed primarily in North America and Europe. The pooled analysis population comprised 1684 patients.
Key inclusion criteria at screening for both studies were SELENA-SLEDAI score ≥6, autoantibody positivity (antinuclear antibody titer ≥1:80 and/or anti-dsDNA ≥30 IU/ml), stable standard SLE therapy for ≥30 days, and without acute renal activity requiring cyclophosphamide or severe active central nervous system lupus. 2,3 Changes to standard immunosuppressive and antimalarial therapy were restricted after 16 weeks of treatment. Prednisone dose was unrestricted for the first 24 weeks, but required a return to within 25% of, or no more than 5 mg above, the baseline dose at week 24, with no further increases for the remainder of the study. Addition of an angiotensin-converting-enzyme inhibitor after 4 months or statins after 6 months was prohibited.
Patients received study treatment for 48 weeks in BLISS-52 and for 72 weeks in BLISS-76. 2,3 The primary efficacy endpoint in both trials was SRI response rate at week 52, defined as a >4-point reduction in SELENA–SLEDAI score, no new BILAG A organ domain score and no more than 1 new B score, and no worsening in Physician’s Global Assessment score compared with baseline. 17
Pooled analysis of renal outcomes data
BLISS-52 and BLISS-76 had nearly identical study designs, patients had similar baseline characteristics, and both studies met the primary endpoint; therefore, data from these studies were pooled to allow a larger sample size for analysis of renal outcomes. 2,3 In this pooled analysis, efficacy endpoints were calculated over 52 weeks and are reported by treatment group for renal outcomes among patients with renal disease at baseline. A renal flare attributable to SLE had to be confirmed by the treating physician and meet at least one of the following three criteria: 1) reproducible increase in 24-hour urine protein to >1000 mg if the baseline value was <200 mg, to >2000 mg if the baseline value was 200–1000 mg, or to more than twice the baseline value if that value was >1000 mg; 2) reproducible increase in serum creatinine of >20% or ≥0.3 mg/dL, whichever was greater, accompanied by proteinuria (>1000 mg/24 h), hematuria (≥4 red blood cells (RBCs) / high-power field (hpf)), or RBC casts; and 3) new reproducible hematuria (≥11–20 RBCs/hpf) or reproducible 2-grade increase in hematuria compared with baseline, associated with >25% dysmorphic RBCs, glomerular in origin, and accompanied by an 800-mg increase in 24-h urinary protein level or new RBC casts.
The definition of patients with renal disease varied with the outcome evaluated, as indicated. The following renal outcomes were evaluated: (1) renal flare rate over 52 weeks in the entire pooled population; (2) renal remission over 52 weeks in patients with baseline proteinuria ≥1 g/24 h, with renal remission defined as erythrocyte count <10 cells/hpf, absence of cellular casts, and proteinuria <1 g/24 h equivalent, without doubling serum creatinine level; (3) normalization at week 52 of anti-dsDNA (from baseline positive (≥30 IU/ml) to negative (<30 IU/ml)) or complement (from low (<90 mg/dl) to normal/high (≥90 mg/dl) C3, or low (<16 mg/dl) to normal/high (≥16 mg/dl) C4); (4) median percent change in proteinuria at week 52 in patients with baseline proteinuria >0.2 g/24 h, percentage of patients with grade 3 or 4 proteinuria at week 52 (>2 g/24 h; estimated from protein/creatinine ratio), and percentage with ≥2-grade worsening in proteinuria to grade 3 or 4; (5) median percent change in creatinine at week 52 from baseline, percentage of patients with grade 3 or 4 (>3 × upper limit of normal (ULN)) elevation, and percentage with a 2-grade elevation at any point during the trial; (6) SELENA-SLEDAI renal organ/item improvement (i.e. negative score shift) in patients with renal organ involvement at baseline (patients who withdrew were considered to have no improvement; the criteria to score four points for proteinuria at screening were proteinuria >1.0 g, or new onset or recurrent proteinuria >0.5 g, with the score retained at baseline unless there was a >0.5-g decrease or decrease to ≤0.5 g from screening to baseline); and (7) BILAG renal organ system improvement in patients who had an A or B score and were scored by the principal investigator as having active renal disease at baseline, with improvement defined as a change from a baseline A or B score to B, C, or D (patients who withdrew or took protocol-prohibited medication were considered to have no improvement).
Proteinuria grades were defined according to the National Institute of Allergy and Infectious Diseases Adult Toxicity Tables used in the BLISS protocols as follows: grade 1, mild, 0.2–1.0 g/24-h equivalent (based on spot urine and protein/creatinine ratios); grade 2, moderate, >1.0–2.0 g/24 h; grade 3, severe, >2.0–3.5 g/24 h; and grade 4, life threatening, >3.5 g/24 h. 18 Creatinine grades were defined according to the same source as follows: grade 1, mild elevation, 1.0–1.5 × ULN; grade 2, moderate, >1.5–3.0 × ULN; grade 3, severe, >3.0–6.0 × ULN; and grade 4, life threatening, >6.0 × ULN.
The following renal variables were also assessed in the subset of patients who were receiving MMF at baseline: renal flare rate throughout the studies, renal SELENA-SLEDAI and BILAG score improvements at 52 weeks in patients with baseline renal involvement, and reductions in proteinuria at 52 weeks.
Statistical analyses
Statistical comparison between treatment groups was performed only for selected variables. The likelihood ratio test (or Fisher’s exact test when ≥25% of expected cell counts in the contingency table were <5) was used to compare each belimumab group with the placebo group for the following values: normalization of serology (anti-dsDNA, and C3 and C4) that was abnormal at baseline, differences in BILAG renal improvement rates in patients with BILAG renal involvement and MMF use at baseline, and rate of 2-grade shift in proteinuria. The Wilcoxon rank-sum test was used to compare each belimumab group with the placebo group for median percent change in proteinuria and mean change in creatinine from baseline to week 52. The log-rank test was used to compare the time to first renal flare and the time to first renal remission.
Results
Patients
Baseline SLE characteristics and renal involvement
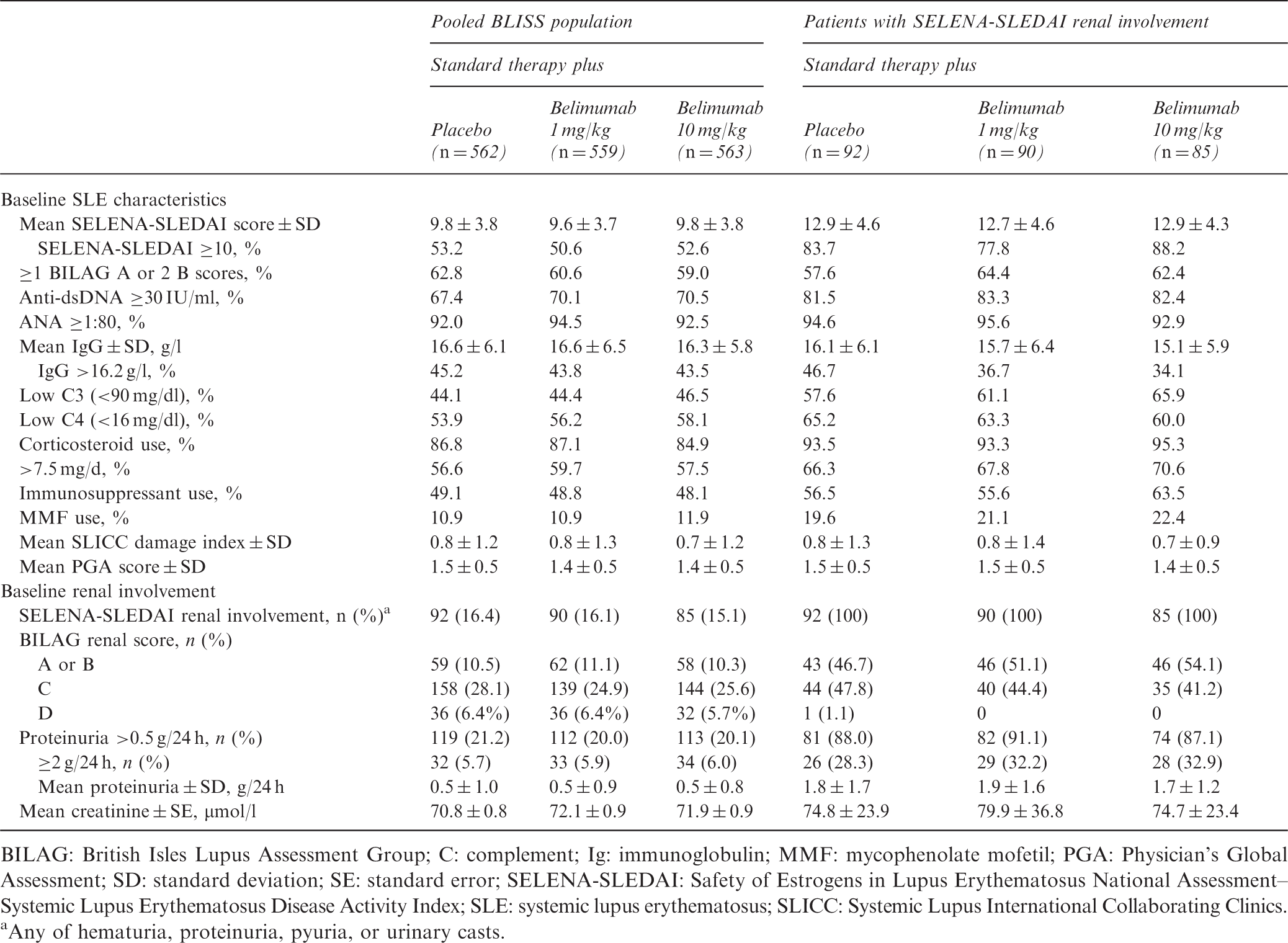
BILAG: British Isles Lupus Assessment Group; C: complement; Ig: immunoglobulin; MMF: mycophenolate mofetil; PGA: Physician’s Global Assessment; SD: standard deviation; SE: standard error; SELENA-SLEDAI: Safety of Estrogens in Lupus Erythematosus National Assessment–Systemic Lupus Erythematosus Disease Activity Index; SLE: systemic lupus erythematosus; SLICC: Systemic Lupus International Collaborating Clinics.
Any of hematuria, proteinuria, pyuria, or urinary casts.
SELENA-SLEDAI and BILAG renal organ scores
Of patients with SELENA-SLEDAI renal involvement at baseline, numerically higher proportions treated with belimumab had SELENA-SLEDAI renal improvement, including hematuria and proteinuria, at week 52 (Figure 1(A)). Data are not shown for urinary casts (≤3/group) due to the small numbers of patients. Within the subgroup of 267 patients with SELENA-SLEDAI renal involvement, a greater percentage of the subset of patients with low C3/C4 levels and anti-dsDNA positivity at baseline (n = 182) had renal organ system and item improvements with belimumab 10 mg/kg vs. standard therapy alone (Figure 1(B)). This result with belimumab 10 mg/kg was more pronounced (12.8%) than in the overall population with SELENA-SLEDAI renal involvement at baseline (7.0%).
Renal improvement at week 52 in patients with renal involvement at baseline. Safety of Estrogens in Lupus Erythematosus National Assessment–Systemic Lupus Erythematosus Disease Activity Index renal organ system and item improvement in (A) pooled population and (B) subgroup with low complement levels and anti-double-stranded DNA positivity at baseline. (C) British Isles Lupus Assessment Group renal organ system improvement in patients with renal A or B score and active renal disease at baseline.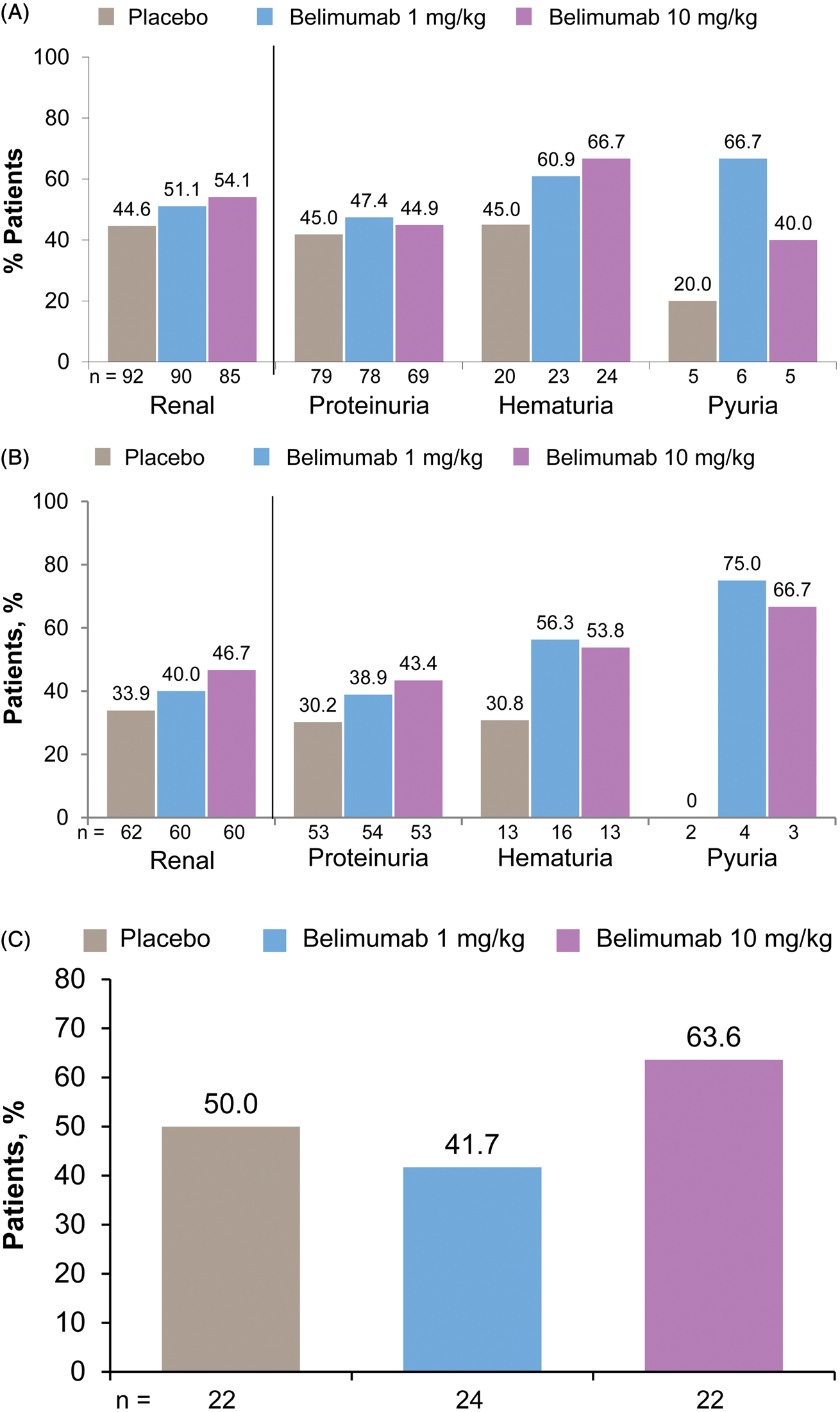
Of patients with a BILAG renal A or B score at baseline, there was no significant difference between groups in the proportions of patients with BILAG renal organ improvement at week 52 (Figure 1(C)).
Renal remission and flares
In patients with baseline proteinuria ≥1 g/24-h equivalent (n = 220), renal remission rates were numerically higher and time to first renal remission was numerically shorter in the belimumab-treated groups (Figure 2(A)). In the overall pooled population, patients receiving belimumab vs. placebo had numerically lower renal flare rates (Figure 2(B)). Belimumab treatment resulted in fewer renal flares defined by increases in proteinuria: 2.1% and 1.1% of patients in the belimumab 1-mg/kg and 10-mg/kg treatment groups vs. 3.0% in the placebo group. Over 52 weeks, no renal flares defined by increases in hematuria were reported and similar numbers of flares defined by increases in serum creatinine were noted across treatment groups (0.4%–0.5%).
Renal remission and flare, and changes in proteinuria over 52 weeks in the pooled population. (A) Renal remission rates and median times to first renal remission in patients with baseline proteinuria ≥1 g/24 h. (B) Renal flare rates in the full pooled population. (C) Median percent reductions in proteinuria in patients with baseline proteinuria >0.2 g/24 h (n = 643). *p < 0.05; +
p < 0.01; #
p < 0.001.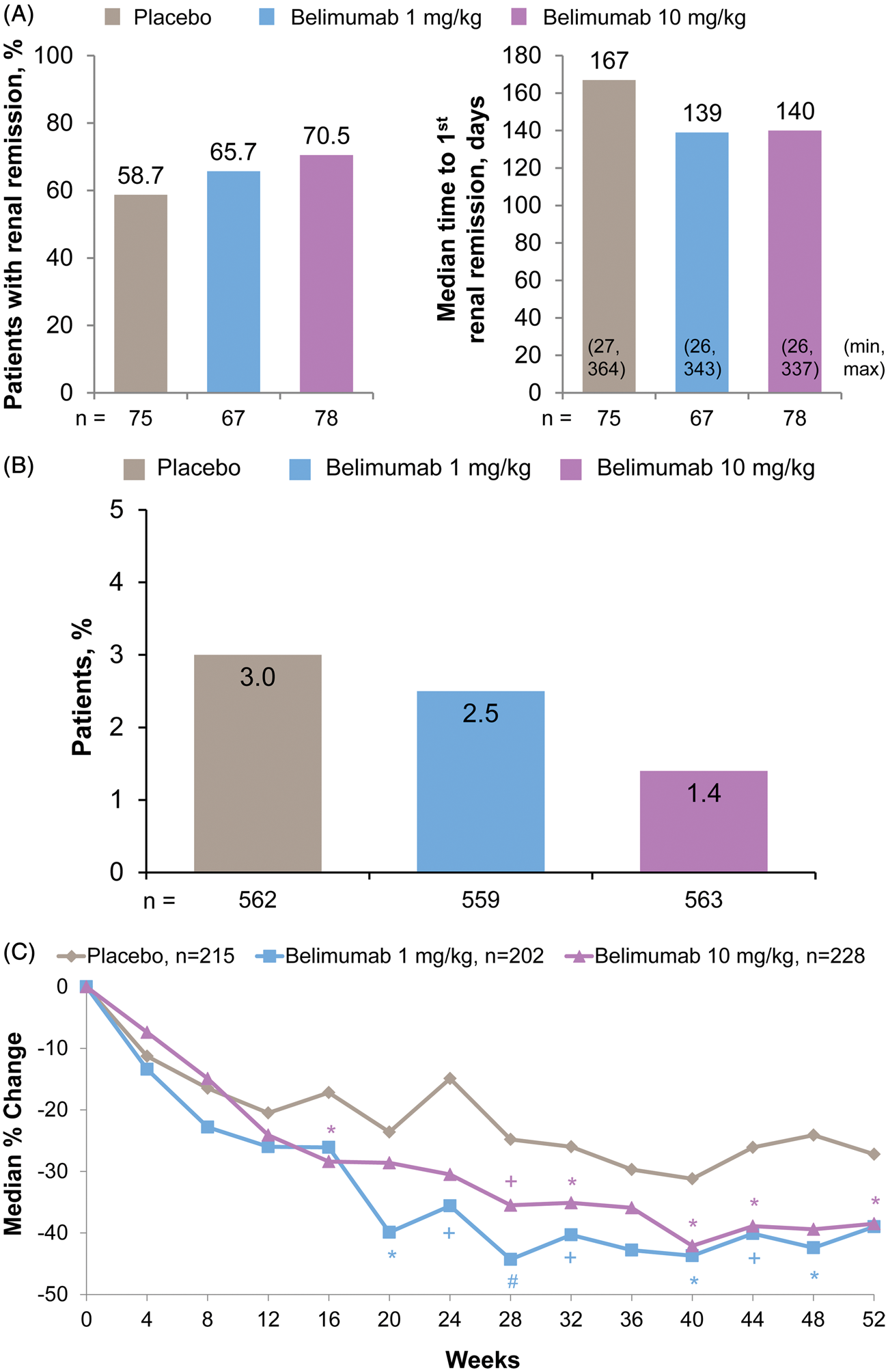
Normalization of serology
Normalization of serology, and proteinuria and creatinine changes, in pooled population over 52 weeks
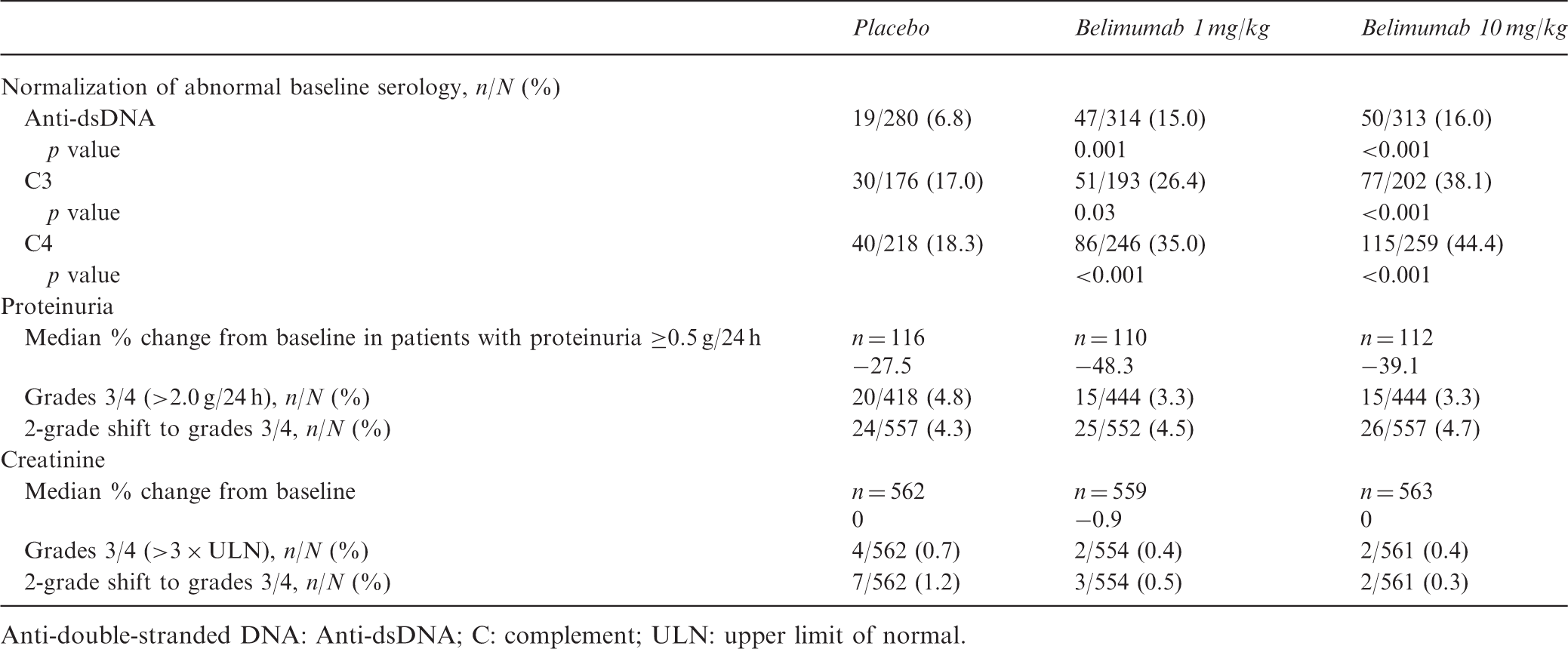
Anti-double-stranded DNA: Anti-dsDNA; C: complement; ULN: upper limit of normal.
Proteinuria
In patients with baseline proteinuria >0.2 g/24 h (n = 645), those receiving belimumab had numerically or significantly greater median percent reductions in proteinuria during weeks 12–52 than those treated with placebo (Figure 2(C)); this was also the case in patients with baseline proteinuria >1.0 g/24 h (n = 218). Grade 3/4 proteinuria developed at some point over 52 weeks in a numerically smaller proportion of patients receiving belimumab vs. placebo (Table 2). The rates of ≥2-grade worsening in proteinuria to grade 3 or 4 were similar between treatment groups.
Creatinine
In the overall pooled population, median reductions in creatinine at week 52 compared with baseline were minimal and similar between treatment groups (Table 2). The proportions of patients with grade 3/4 creatinine elevations at some point during the studies and the rates of ≥2-grade worsening were numerically lower with both belimumab doses plus standard therapy than with placebo plus standard therapy.
Patients receiving MMF at baseline
The proportions of patients in the total population with renal involvement at baseline who were also receiving MMF were as follows: 21% (56/267) with SELENA-SLEDAI renal involvement, 16% (29/179) with BILAG renal A or B scores, 16% (89/545) with BILAG renal C or D scores, and 22% (76/344) with proteinuria >0.5 g/24 h.
Of patients receiving MMF at baseline, 29.6% had ≥1 SELENA-SLEDAI renal item, 15.3% had a BILAG renal A or B score, 40.2% had a BILAG renal C score, and 40.2% had proteinuria >0.5 g/24 h. Patients receiving MMF were numerically more likely to have the following SELENA-SLEDAI renal items than those not receiving MMF: proteinuria (26.5% vs. 11.6%), hematuria (7.4% vs. 3.5%), vasculitis (9.5% vs. 6.2%), low complement (68.3% vs. 60.6%), and increased anti-dsDNA (75.7% vs. 68.2%). Patients receiving MMF were more likely to have SELENA-SLEDAI renal involvement at baseline than those not taking MMF as background SLE therapy; renal domain involvement regardless of treatment group in patients with vs. without MMF was 29.6% vs. 14.1% (Figure 3). Involvement of the other SELENA-SLEDAI organ domains was otherwise generally balanced between patients treated and not treated with MMF at baseline.
Safety of Estrogens in Lupus Erythematosus National Assessment–Systemic Lupus Erythematosus Disease Activity Index organ system involvement at baseline in pooled population with and without mycophenolate mofetil (MMF).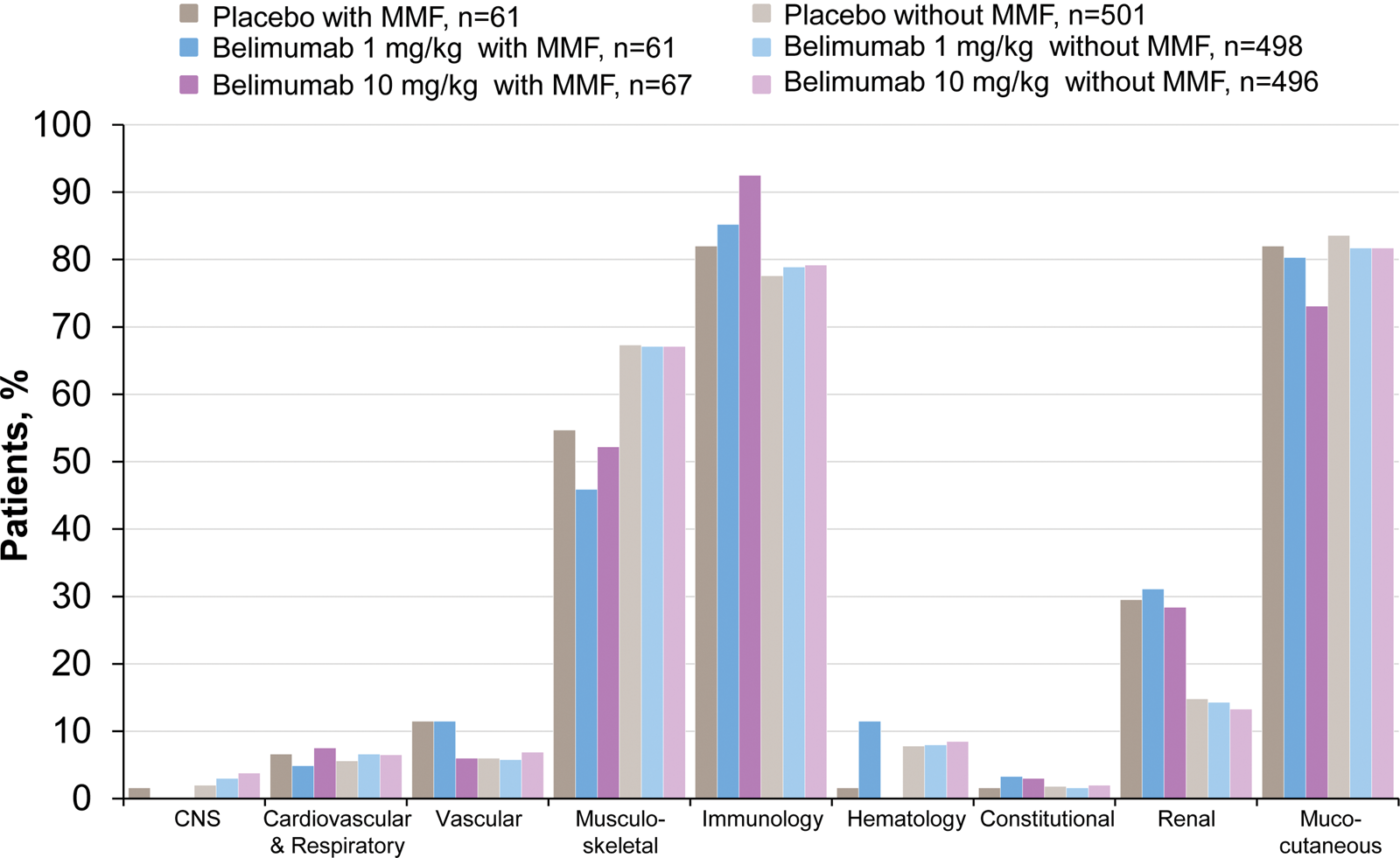
Renal improvement with belimumab in patients receiving MMF at baseline
Renal improvement in patients receiving mycophenolate mofetil at baseline

BILAG: British Isles Lupus Assessment Group; SELENA-SLEDAI: Safety of Estrogens in Lupus Erythematosus National Assessment–Systemic Lupus Erythematosus Disease Activity Index.
By week 52, BILAG renal improvement was seen in 20.0% (7/35), 32.4% (11/34), and 30.6% (11/36) of patients treated with placebo, and belimumab 1 and 10-mg/kg, respectively, who had BILAG renal A–C scores and were receiving MMF at baseline. In the small numbers of patients with BILAG renal A or B scores at baseline, a similar effect was seen. The proportions of patients receiving MMF at baseline who experienced renal flares during the 52-week study were 4.9% (3/61), 4.9% (3/61), and 1.5% (1/67) with placebo, and belimumab 1 and 10-mg/kg, respectively. With the exception of one renal flare defined by an increase in serum creatinine in the placebo group, all other renal flares in patients receiving MMF at baseline were defined by increased proteinuria.
Discussion
Renal involvement is a common cause of morbidity in SLE. 4–7 The BLISS trials excluded patients with acute lupus nephritis, and the effect of belimumab therapy on renal abnormalities was not a prespecified endpoint of these studies. 2,3 This post-hoc pooled analysis was, however, conducted to derive information about the potential effect of belimumab on renal parameters and in patients who were receiving MMF therapy at baseline.
Numerically more patients receiving belimumab with SELENA-SLEDAI renal organ/item (i.e. hematuria or proteinuria) involvement at baseline showed renal improvement at week 52. Among the subgroup with a BILAG renal A or B score and active renal disease at baseline, proportionally more patients treated with belimumab 10-mg/kg had BILAG renal improvement, although the differences were not statistically significant. As noted in a previous pooled analysis of the BLISS trials by Manzi et al., 19 smaller proportions of patients with BILAG renal worsening were noted with belimumab 1-mg/kg (4.9%) and 10-mg/kg (6.1%) treatment vs. placebo (7.5%).
Analysis of a subgroup of the pooled BLISS population with generally stable active renal disease indicated that patients receiving standard therapy plus belimumab 10-mg/kg had a higher rate of renal remissions compared with standard therapy alone, and the time to first renal remission was numerically shorter with belimumab. In addition, there was a numerically lower renal flare rate with belimumab 10-mg/kg compared with belimumab 1-mg/kg or placebo. Median percent reduction in proteinuria at week 52 was numerically greater with either belimumab treatment compared with placebo. The numerically greater decrease in proteinuria with belimumab was observed after week 12 through week 52 in patients with proteinuria >0.2 g/24 h.
A higher proportion of patients with hypocomplementemia and anti-dsDNA antibody positivity at baseline in the renal subgroup of the pooled population showed improvement in SELENA-SLEDAI renal organ domain/renal items. In this subset, the proportions of patients achieving normalization of serologies at week 52 were significantly higher in each belimumab group than with placebo (p < 0.05). Monitoring lupus nephritis disease activity includes following anti-dsDNA titers, complement levels, and proteinuria. 8 Increases in serologic activity and proteinuria have been associated with lupus nephritis, general SLE flares, and worsening disease activity. 10–13 Normalization of C3 or anti-dsDNA, irrespective of SLE therapy, was a predictor of reduced risk of severe SLE flare, and normalization of C4 was a predictor of an SRI response in the BLISS trials. 20
Mycophenolate mofetil is commonly used in the treatment of lupus nephritis. 8 Data from this pooled analysis suggest that belimumab may provide additional benefit in patients receiving MMF therapy with baseline SELENA-SLEDAI renal involvement, including improvement in renal biomarkers, and may be useful in patients who have shown improvement with standard therapy, including MMF, but continue to have some degree of renal abnormality. The small patient numbers and post-hoc nature of this analysis, however, limit definitive conclusions.
The limitations of this analysis include the small numbers of patients with generally chronic stable baseline renal involvement and the smaller numbers with baseline renal involvement plus MMF use. Further, patients with severe active lupus nephritis were excluded from the BLISS trials. The post-hoc nature of these analyses also limits extrapolation of the findings. The difference in renal outcomes between the placebo group and either belimumab treatment group was significant only for normalization of baseline serology, and for renal SELENA-SLEDAI improvement in patients with baseline renal involvement receiving MMF. The BLISS trials were not designed and not powered to detect a difference in renal outcomes between treatment groups for the base populations reported in this pooled analysis. The findings are, therefore, inconclusive, but suggest that belimumab plus standard therapy may have a beneficial impact on renal outcomes in patients with SLE.
In summary, further studies in patients with lupus nephritis and severe active renal disease are required to document a benefit of belimumab in patients with renal involvement receiving standard lupus nephritis induction and maintenance therapy.
Footnotes
Acknowledgment
Editorial support was provided by Eileen McCaffrey and Eleanore Gross of BioScience Communications, New York, USA, and was funded by GlaxoSmithKline, Uxbridge, Middlesex, UK, and Human Genome Sciences, Rockville, Maryland, USA.
Funding
This work was supported by GlaxoSmithKline and Human Genome Sciences.
Conflict of interest
Drs Dooley, Aranow, and Ginzler have received consulting fees and research support from GlaxoSmithKline and Human Genome Sciences. Drs Houssiau and D’Cruz have received consulting fees from GlaxoSmithKline and Human Genome Sciences. Dr Roth is employed by and owns stock in GlaxoSmithKline. Drs Cooper, Zhong, and Freimuth are employed by and own stock in Human Genome Sciences.
