Abstract
Food safety may be one of the major concerns of the global society in the forthcoming decades. Analytical vibrational spectroscopy is expected to become a major tool used for controlling the food quality at every stage of its production, storage and delivery. Near-infrared and infrared spectroscopy have rapidly been evolving in analytical applications over the last decades with strong hyphenation to numerical and statistical methods of analysis of complex data, which are known as chemometrics. Analytical spectroscopy has reached a remarkable value for both industrial and institutional laboratories nowadays. However, the routinely used methods of analysis do not attempt to interpret the analysed spectral information in physicochemical sense. Therefore, analytical routines seldom take advantage of the molecular background underlying the properties of analysed sample. In the present article, we review the most recent accomplishments that evidence the progress which may be achieved when that background becomes actually available. We focus on the example of infrared and near-infrared spectra simulation applied to melamine, one of the most infamous food adulterant. This sheds light on the correspondences between infrared and near-infrared region observed earlier in the analytical papers dealing with detection and quantification of melamine in food products.
Keywords
Introduction
Melamine (IUPAC: 1,3,5-Triazine-2,4,6-triamine) has attracted particular attention after the outbreak of the milk adulteration scandal in 2008.
1
This disastrous event had huge impact, with 290,000 people affected with 51,900 hospitalized in China only.
2
This had global implications on the food industry, food production, supply chains and legal regulations.
3
Afterwards, the amount of scientific publications concerned with melamine has constantly been growing on year-to-year basis (Figure 1), reaching almost 1000 articles in 2018, according to the Web of Science database.
4
The stimulus for analytical chemistry in responding to the scandal was particularly strong. The focus on non-invasive, easily applicable, fast and inexpensive techniques has promoted techniques based on vibrational spectroscopy as an adequate tool in such application.
Number of scientific publications in the years 1995–2018 (and 2019 until April) concerning melamine; source: Web of Science (topic including ‘melamine’).
4
Approximately 88% of these records are scientific articles and their annual distribution looks exactly similar.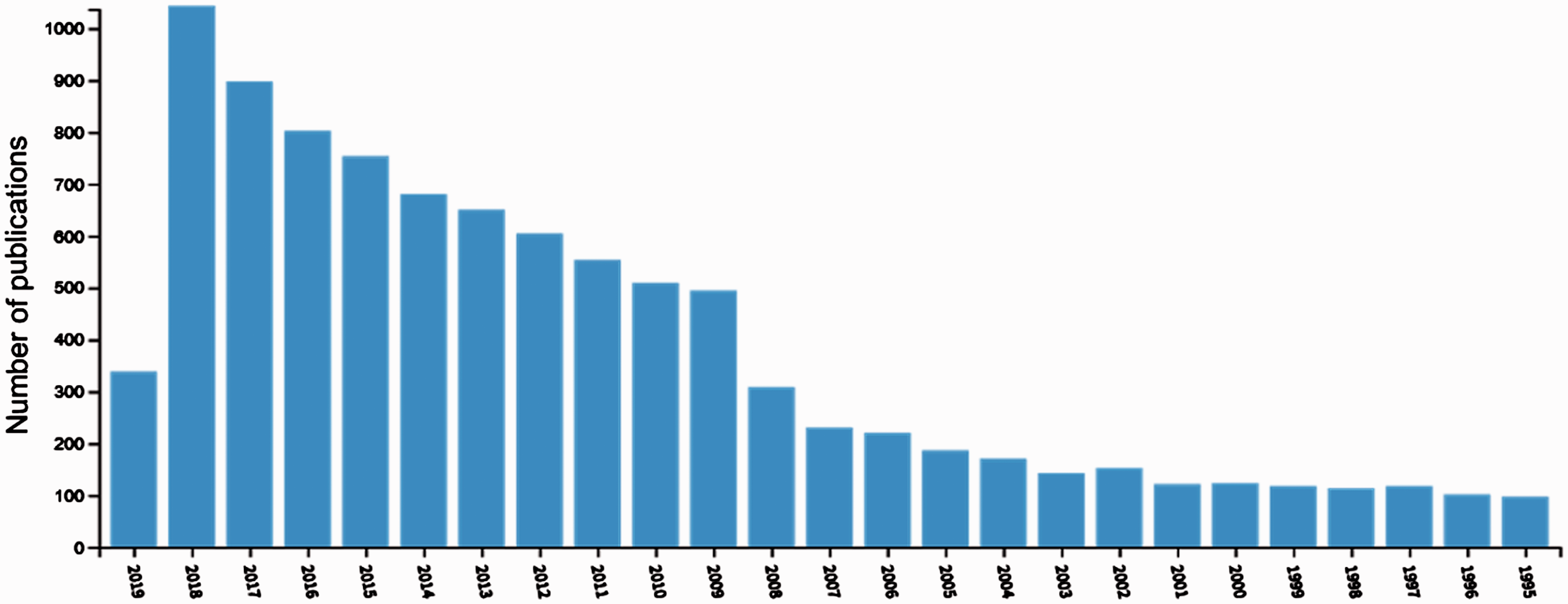
Vibrational spectroscopy (infrared, IR; near-infrared, NIR; and Raman) is gaining particular significance as a food quality control method; a number of factors contribute to this fact. 5 Among those techniques, in selected applications either of them may be equally useful. However, in many cases, NIR spectroscopy is being favoured. Detection and quantification of melamine content in milk powder and similar samples are one of such fields, where IR and NIR techniques are being very commonly used concurrently. Interestingly, the literature tends to give superiority to NIR spectroscopy, in particular for the sample with high content of melamine. 6
The nature of IR and NIR spectra differs in a very specific way.7,8 NIR bands originate from overtone and combination transitions. What matters the most from the point of view of practical interpretation of the spectra, is that the number of individual vibrational contributions in NIR region for any sample significantly surpasses the number of those in IR region. Hence, band overlapping is much more significant in the former. As a result, the complexity of NIR spectra extends far beyond the IR and Raman spectra. On the other hand, intermolecular interactions occurring in some solid state samples may often impact the vibrational spectra heavily, and their bands may differ substantially from those of the same molecules but isolated in an arbitrarily arranged chemical environment (e.g. KBr pellet for IR measurements, or solution in CCl4 for NIR ones). The interaction of the analysed molecules with the matrix molecules in complex samples may lead to a very similar spectral feature as those observed in solid state molecules.
Previously we have discussed the molecular and vibrational mechanisms leading to the particular difficulties in the interpretation of the observed NIR bands. 9 IR spectra generally feature much lesser overlapping, and the major bands frequently remain non-convoluted individual contributions. However, molecular vibrations are highly sensitive to a number of properties among which the interaction with the chemical surroundings should be accounted as being one of the most meaningful. This impacts IR spectra as well. However, comparative studies that attempt to obtain more general overview of these phenomena in IR and NIR region are immensely scarce. Substantial difficulties in theoretical modelling of IR spectra of solids, as well as NIR spectra in general, have been a hindrance which prevented a proper exploration of this area.
However, recent investigation of melamine in powder as reported by Grabska et al. 10 has evidenced the potential which stems from the application of advanced methods of computational chemistry in order to obtain far-reaching answers on the scientific questions outlined above.
The fundamental difference of how IR and NIR bands of melamine are affected by the molecule’s chemical environment
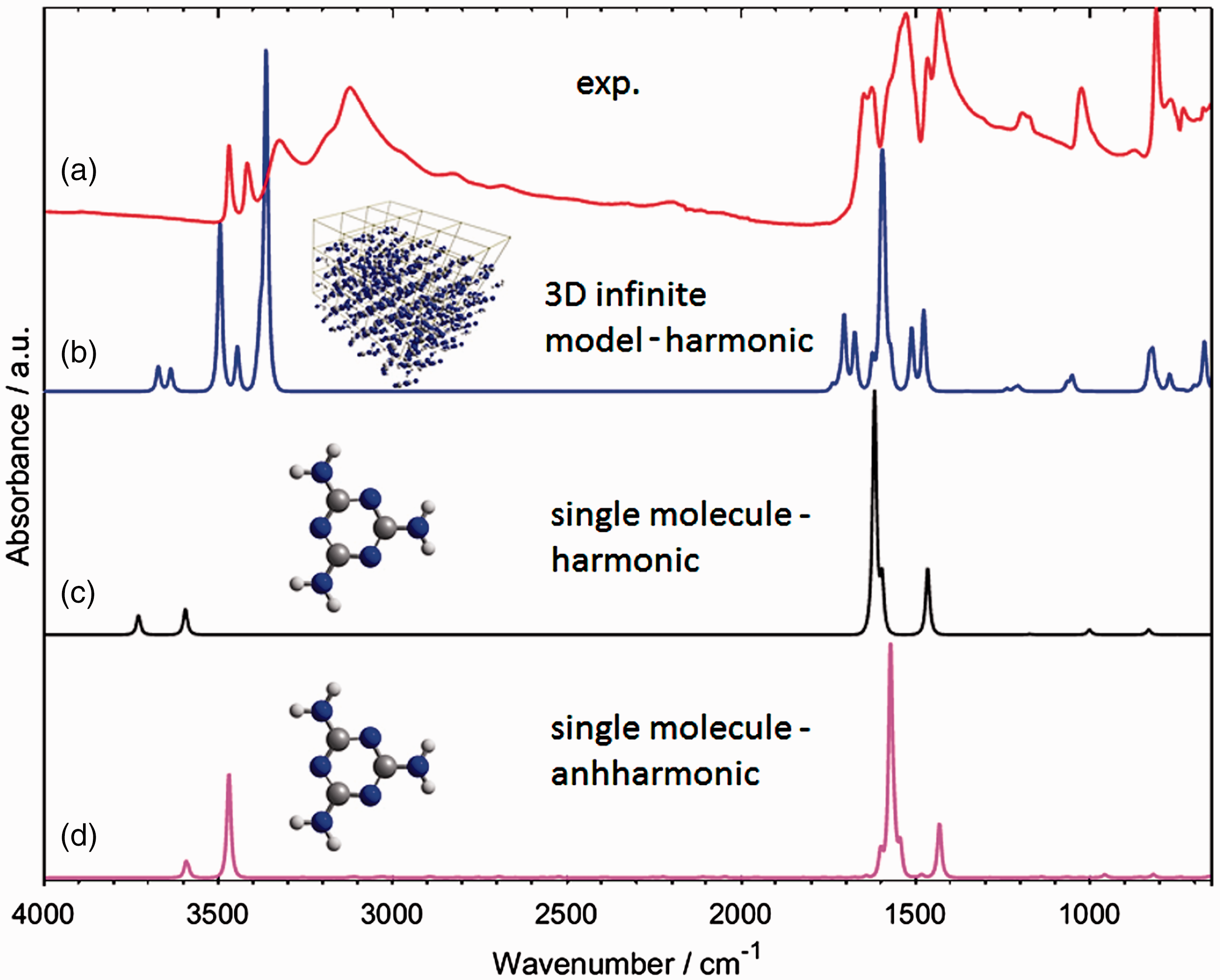
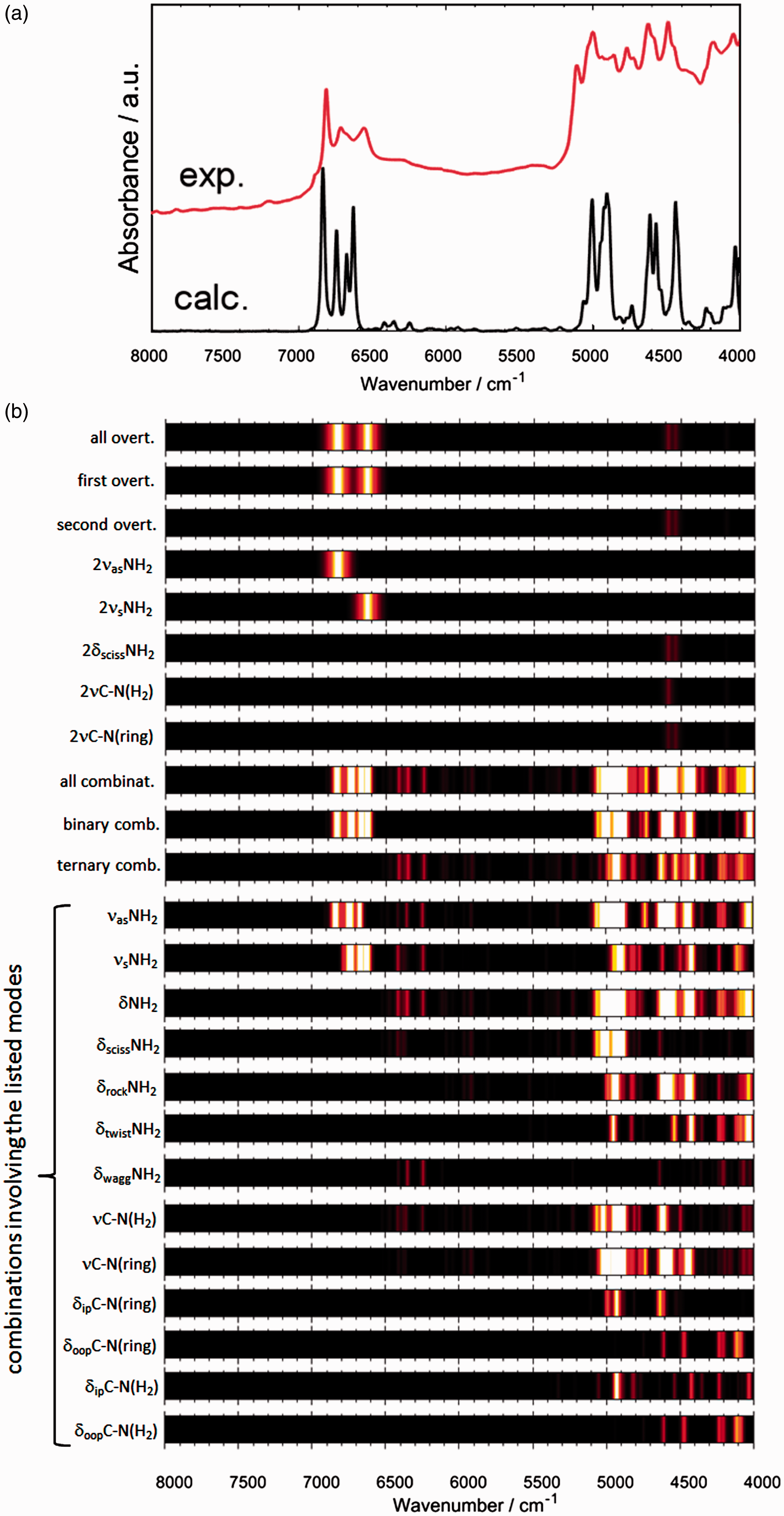
Melamine is a highly interesting molecule from multiple scientific point of view. In the context of vibrational spectroscopy, one may notice its heterocyclic ring and the existence of three NH2 groups (Figure 2). These polar, hydrogen-bonding capable groups allow to expect that molecules of melamine tend to interact with the chemical neighbourhood. Indeed, this can easily be seen in the IR spectrum of melamine (Figure 3(a)), which cannot be reproduced computationally by using the model of a single molecule (Figure 3(c) and (d)).
10
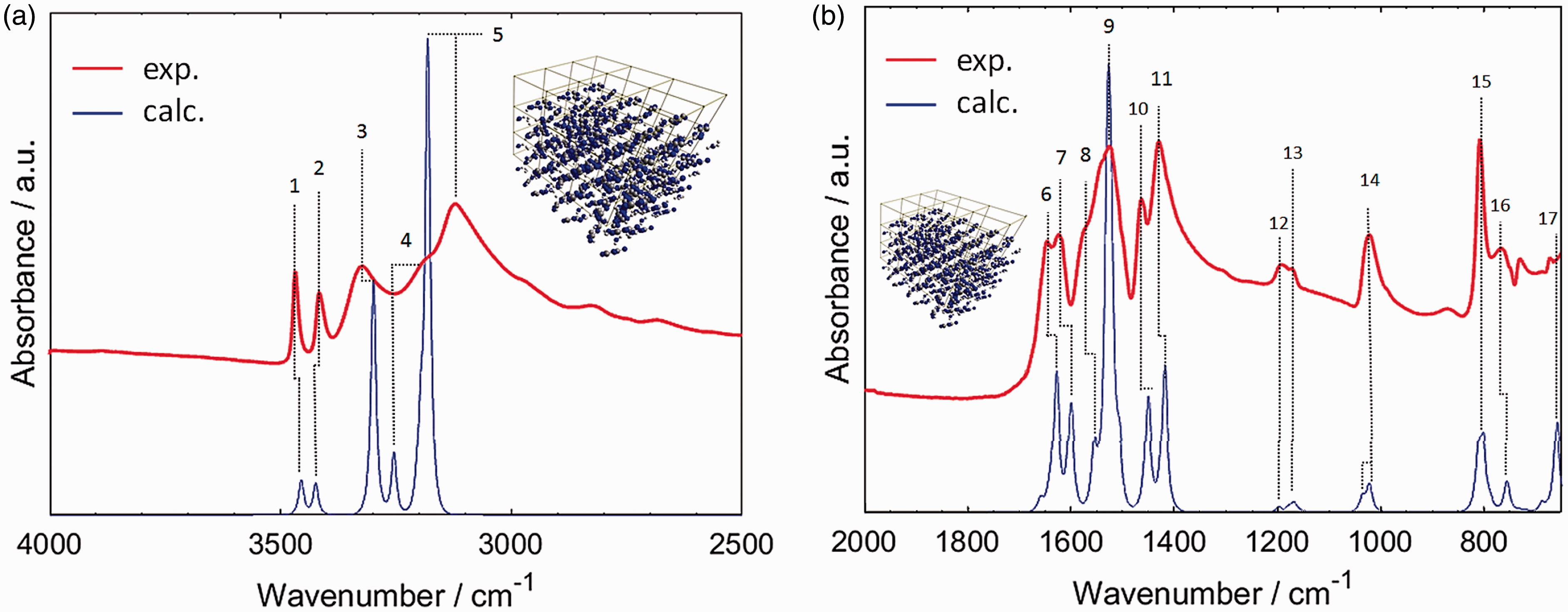
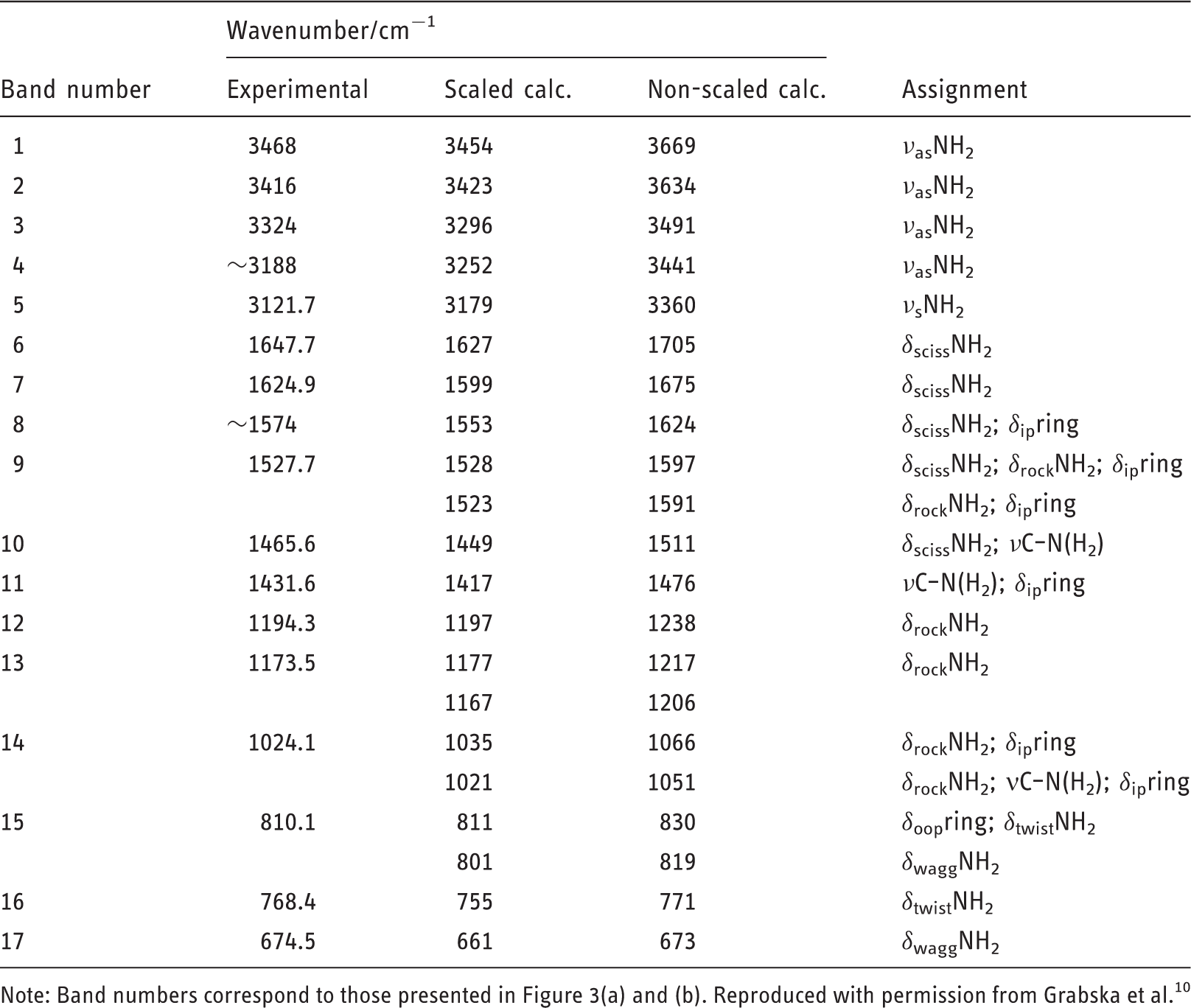
Regardless of whether one would use harmonic (Figure 3(c)) or advanced anharmonic approximation (Figure 3(d)), the resulting simulation lacks key features observed in the experimental spectrum (Figure 3(a)). Clearly, neglecting of the chemical neighbourhood of the molecule of melamine leads to an entirely improper description of its vibrations as they appear in solid state sample. In contrast, by reproducing the effectively infinite periodic structure of crystalline melamine, one can achieve almost perfect reproduction of IR bands (Figure 3(b)) observed in the experiment. This may be achieved even in harmonic approximation, which after applying empirical scaling to account for anharmonic effects allows for detailed and unambiguous assignment of almost all IR bands observed for melamine in powder (Figure 4 and Table 1).
The molecular structure of melamine. (a) Single molecule; (b) single unit cell; (c) 3 × 3 × 3 supercell. Reproduced with permission from Grabska et al.10 Experimental (a) and simulated (b–d) IR spectra of melamine. Simulated spectra are based on molecular models of largely different complexity. One may notice that a single molecule model, regardless whether harmonic or anharmonic approximation is being applied, does not allow to correctly reproduce the observed features. The experimental spectrum was measured in ATR; melamine sample was polycrystalline (powder). Reproduced with permission from Grabska et al.10 Band assignments in the ATR-IR spectrum of polycrystalline melamine based on spectra simulation. Band numbers correspond to those presented in Table 1. (a) 4000–2500 cm−1 region; (b) 2000–650 cm−1 region. Reproduced with permission from Grabska et al.10 Contributions to NIR spectrum of polycrystalline melamine as unveiled in spectra simulation. Reproduced with permission from Grabska et al.10 Band assignments in the experimental ATR-IR spectrum of crystalline melamine. Note: Band numbers correspond to those presented in Figure 3(a) and (b). Reproduced with permission from Grabska et al.10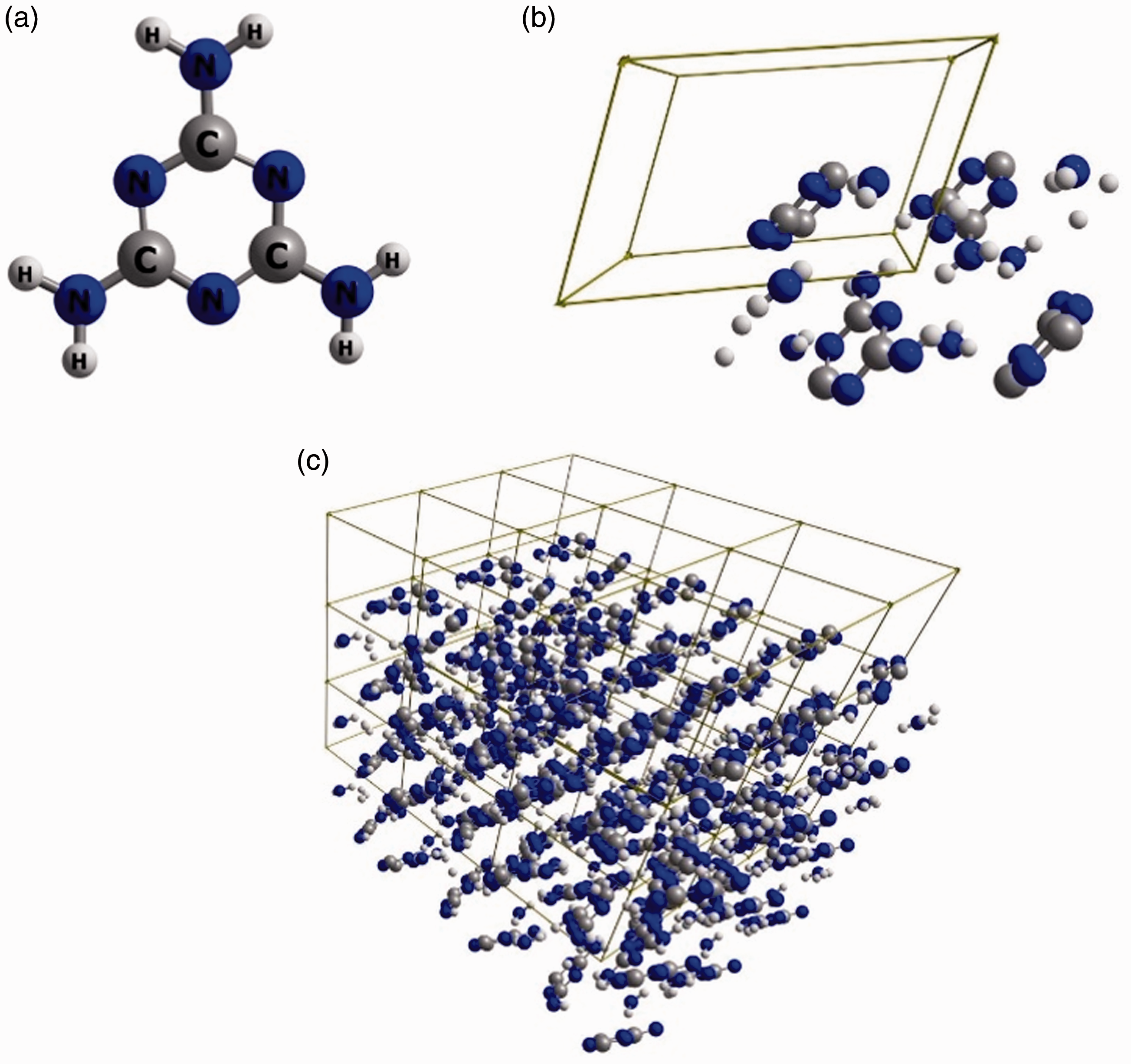
Interestingly, the observations reported by Grabska et al.
10
for IR region of polycrystalline melamine stood in contrast with what has been noticed in NIR region. Surprisingly, a single molecule model has allowed to reproduce NIR spectrum of powdered melamine very well (Figure 5). All major bands observed in the experimental spectrum have been reflected in the simulated one. Some differences could be seen in the lower NIR region, between ca. 5200 and 4000 cm−1, which is populated almost entirely by combination bands. The simulation included the following types of NIR transitions:
first overtones, 2ν second overtones, 3ν binary combinations, νx + νy ternary combinations of νx + νy + νz type ternary combinations of 2νx + νy type
This covers the vast majority of the vibrational transitions, which may be meaningful in 8000–4000 cm−1 region of NIR spectra.
To improve the analysis of the convoluted patterns common for NIR spectra, Grabska et al.10 have presented the relative contributions into the NIR spectrum of the vibrational modes of melamine in the form of color-coded intensities (Figure 5). This effectively deconvolutes the NIR spectrum of melamine, yielding unparalleled level of insight into the spectral origins.
The concluded reasons for the observed disparity between how simulated IR and NIR spectra depend on the molecular model have been as follows. The IR bands of melamine are much more sensitive to the molecule’s chemical neighbourhood. Neglecting the neighbourhood by the simple models (e.g. single molecule) reduces largely the accuracy of description of those vibrations. In contrast, NIR bands seem to be much less depending on the chemical environment, in particular to the adjacent crystalline planes in the structure of melamine. Further details may be found well described in the original report by Grabska et al. 10 What seems the most essential here, is what this and presently continuing studies may bring to analytical spectroscopy. It has been known previously that in the quantitative analysis of melamine, NIR spectroscopy starts to gain superiority over IR spectroscopy for higher content melamine in the sample. It seems reasonable, in light of the finding reported by Grabska et al. 10
The mechanism standing behind this observation may be similar to the one which we have reported earlier for thymol. 11 The vibrations sensitive to intermolecular interactions may be less useful for quantitative studies. However, this is an early assumption and further studies are needed to explore this interesting field.
Prospects for future: Finding synergy between analytical and theoretical spectroscopy
In Huck Lab (lead by Professor Christian W. Huck), we are now conducting an Austrian Science Fund (FWF)-funded research project aiming at a further exploration of the intriguing area connecting basic spectroscopy, computational chemistry and analytical science. One of our aims is to unveil the relationship between the influential factors as recognized in predictive chemometrics and the molecular background. Our recent results gathered for thymol and melamine, both of which have frequently been focused on in analytical spectroscopy, suggest that a significant improvement in our understanding of what exactly ‘happens underneath’ may be obtained. This still remains an unexplored field, on which we now focus our attention. The continuation of this study requires involving a wider spectrum of samples in order to yield more generalized conclusions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this
