Abstract
Objective
Bronchopulmonary dysplasia (BPD), a common chronic lung disease in premature infants, is frequently complicated by pulmonary hypertension (PH), a dangerous condition characterized by elevated pulmonary vascular pressure. At present, the most classic and commonly used animal model for BPD involves exposure to high oxygen concentrations, but existing studies based on this model have seldom addressed whether it recapitulates the pathological features of PH.
Methods
In the present study, we established a rat model of BPD complicated with PH (BPD-PH). Histological hematoxylin and eosin (H&E) staining, enzyme-linked immunosorbent assay (ELISA), flowcytometry, reverse transcription–quantitative PCR (RT-qPCR), wound healing assay, transwell migration and invasion assays, transcriptome RNA-sequencing and GO analysis were performed to evaluate this BPD-PH model.
Results
Our study demonstrated that rats with BPD-PH exhibited characteristic alveolar simplification and features of pulmonary hypertension, including increased mRVP, RV/LV + S and RV/WT. BPD-PH rats had enhanced inflammatory response and increased accumulation of immune cells in lung. Moreover, differentially expressed genes in the BPD-PH rat model were depicted and we found Leucine-rich alpha-2-glycoprotein 1 (Lrg1) was significantly upregulated by 19.32 fold (p < 0.001). LRG1 affected migration, invasion and transforming growth factor-β (TGF-β) signaling activation in primary alveolar epithelial cells and pulmonary endothelial cells. Finally, our BPD-PH model was used for the evaluation of two commonly used drugs for BPD and PH, Dexamethasone and Sildenafil. These two drugs were effective in alleviating lung injury, inflammatory response and hypoxia-induced PH in the rat model of BPD-PH.
Conclusion
Our data indicate that we established a novel BPD-PH model in rats, and it provided a new choice for the future research for BPD complicated with PH.
Introduction
Bronchopulmonary dysplasia (BPD) is a chronic lung disease that predominantly affects preterm infants, characterized by inflammatory injury to the developing lungs and impairment of alveolar and vascular growth. These injuries ultimately cause an aberrant reparative response leading to persistent airway and pulmonary vascular diseases which may affect adult lung function. As a multifactorial condition, BPD arises from a combination of risk factors, most notably extreme prematurity (gestational age <28 weeks) and low birth weight.1,2 Nearly 80% of infants with gestational age at 22 to 24 weeks will develop BPD, while only 20% of infants born at 28 weeks are diagnosed with BPD. 3 In addition, almost 95% of infants with BPD have a very low birth weight (less than 1500g). 4 Additional contributing factors include mechanical ventilation and associated injuries, oxygen toxicity, inflammation, maternal smoking, chorioamnionitis, and intrauterine growth restriction. 5 Current management strategies emphasize minimizing ventilator-induced lung damage by avoiding intubation where possible, and employing pharmacologic agents such as caffeine and corticosteroids, and inhaled of nitric oxide. 6 At present, the pathogenesis of BPD is still not fully elucidated. A better understanding of BPD pathogenesis may help to develop novel therapeutic strategies.
Pulmonary hypertension (PH), characterized by elevated pulmonary vascular pressure, represents a particularly severe complication of BPD. It is estimated that nearly 25% of premature infants with moderate or severe BPD will develop PH (BPD-PH). 7 Among them, 47% of these infants die in 2 years’ time after first diagnosis with BPD-PH. 8 The pathophysiological hallmarks of BPD-PH include impaired alveolar gas exchange, abnormal vascular remodeling, and a reduction in pulmonary vessel density. The risk factors for developing BPD-PH overlap considerably with those for BPD itself, encompassing extreme prematurity, mechanical ventilation, hyperoxia, and inflammation/sepsis. 9 Although targeted PH medications are used, their application in infants remains off-label, and current management strategies are largely guided by expert consensus rather than robust clinical trial evidence. Sildenafil, a drug inhibits the breakdown of cyclic GMP, becomes the mainstay of contemporary BPD-PH therapy. 10 Other therapeutic strategies include inhaling of NO and supplemental of oxygen. Nonetheless, a significant gap exists, as prospective or randomized controlled trials for BPD-PH are still scarce. Besides, animal models are needed to obtain pharmacokinetic, pharmacodynamic and safety data for any pharmacotherapy used in BPD-PH treatment preclinically.
Accumulating evidence indicates that intermittent and prolonged hypoxia represents a key contributor to the development of PH in chronic lung diseases, including BPD. 11 The underlying mechanism of why hypoxia causes PH and the time course of hypoxia-induced PH have been intensively studied in a variety of animal models, including newborn animals.12,13 In the present study, we established a novel rat model of BPD‐PH, which exhibited enhanced pulmonary inflammation accompanied by increased accumulation of immune cells in the lung. Transcriptomic profiling further identified differentially expressed genes in BPD-PH rats, most notably a significant upregulation of LRG1, which functionally influenced migration, invasion, and TGF-β signaling activation in both primary alveolar epithelial cells and pulmonary endothelial cells isolated from these animals. Moreover, this novel BPD-PH model was employed to evaluate the efficacy of two commonly used pharmacological agents for BPD and PH, demonstrating its utility as a valuable preclinical tool for investigating disease mechanisms and therapeutic interventions.
Materials and methods
Establishment of BPD-PH rat model
The Sprague-Dawley rats (n = 10) were purchased from Weitong Lihua Lab animal Tec. Co. Ltd (China) and maintained at an average temperature of 22°C in a typical 12-12 h cycle of light/dark at 55 ± 10% humidity. Animal procedures were reviewed and approved by the Animal Management Committee of Shenzhen Children’s Hospital (Approval NO.: 202205232). The BPD-PH rat model was built according to previous reports.
14
Briefly, newborn rats (postnatal day 1, P1, n = 20) from several litters were mixed and randomly divided into four groups, the air group (A), low oxygen group (AL), BPD group and BPD-PH group using a random number generator. An atmospheric oxygen concentration control chamber (Shanghai Yuyan Instruments #S1007, China) was used to conduct hyperoxia and hypoxia experiments. Rats were kept in the chamber and exposed to different concentrations of oxygen as depicted in Figure 1(a). The pups in A group were exposed to indoor air (21%O2). The pups in AL group were exposed to 1 h hypoxia (13%O2) and 23 h normoxia (21%O2) daily. The pups in BPD group were exposed to hyperoxia (60%O2) daily. The pups in BPD-PH group were exposed to 1 h hypoxia (13%O2) and 23h hyperoxia (60%O2) daily. These treatments were started immediately after birth and lasted for 14 days. To avoid oxygen toxicity in the dams and to eliminate maternal effects between groups, dams were rotated every 24 h between normoxia and hyperoxia chambers to prevent maternal effects. All morphometric analyses and quantifications were performed by investigators blinded to experimental group allocation. Dexamethasone (Selleck# S1322) and Sildenafil (Selleck #S4684) were obtained from Selleck (USA) and dissolved in 1% DMSO + 5% PEG300 + 5% Tween 80 + 89% deionized water. The 1% DMSO + 5% PEG300 + 5% Tween 80 + 89% deionized water was used as vehicle control in animal experiments. Dexamethasone and Sildenafil were administered by intragastric gavage at 100 μL per animal at the same time each day. Drug solutions were prepared fresh daily. BPD-PH rats were treated with Sildenafil (Silde), or the combination of Sildenafil and Dexamethasone (Dexam) at day P7 to P14 daily. Rats were euthanized by injection of pentobarbital sodium (50 mg/kg i.p.) at the end of drug treatment. Predetermined humane endpoints included weight loss >20% from baseline, severe respiratory distress [respiratory rate >200 breaths/min with labored breathing], inability to ambulate, or lethargy unresponsive to stimulation. A total of 50 Sprague-Dawley rats (10 adult and 40 newborn) were used in our study. No animals met early euthanasia criteria during the study. Animals were excluded if dams cannibalized pups, pups exhibited congenital abnormalities, or technical issues occurred during catheterization. No animal was excluded. Establishment and characterization of a rat model of BPD-PH. (a) the schematic diagram of BPD-PH rat model. B-C, H&E staining of lung parenchyma (×200 magnification; scale bar = 100 μm) from rats in A, AL, BPD and BPD-PH group at day 14 after hyperoxia/hypoxia treatment (b) Images were acquired using an Olympus BX51 microscope equipped with a Olympus DP70 camera at ×200 objective magnification. Representative images from n = 5 animals per group. Average chord length was shown (c) Arrows indicated typical structure of pulmonary alveoli. D-F, mean right ventricular pressure (mRVP) (d) the ratio of right ventricle weight to left ventricle plus interventricular septum weight (RV/LV + S) (e) and the ratio of right ventricle weight to wall thickness (RV/WT) (f) were evaluated at day 14 after hyperoxia treatment. n = 5 animals per group. G-I, H&E staining of small pulmonary arteries (×400 magnification; scale bar = 50 μm) from rats in A, AL, BPD and BPD-PH group at day 14 after hyperoxia/hypoxia treatment were shown (g). Images were acquired using an Olympus BX51 microscope equipped with a Olympus DP70 camera at ×400 objective magnification. Representative images from n = 5 animals per group. Ratios of vascular thickness (h) and lumen area (i) were shown. J-K, KL-6 (j) and VEGF (k) in serum samples of rats in each group at day 14 after hyperoxia treatment were measured by ELISA assay. n = 5 animals per group. Student's t-test was used for statistical analysis between two groups. n = 5 for each group. n.s., not significant.
Immunohistochemistry (IHC) staining
Following overnight fixation in 4% formalin, lung tissue samples were processed through paraffin embedding, sectioned at a thickness of 5 μm, and stained with hematoxylin and eosin (H&E) to assess histomorphological changes. Images were obtained using an Olympus BX51 microscope (Tokyo, Japan) equipped with Olympus DP70 camera using OLYMPUS Stream software 2.4 and analyzed by Image J software.
Enzyme-linked immunosorbent assay (ELISA)
The levels of KL-6 (Biorbyt #orb782217, USA), VEGF (Abcam #ab100786, USA), TNF-α (Abcam #ab236712, USA), IL-17A (Abcam #ab214028, USA), IL-6 (Abcam #ab234570, USA) and IL-1β (Abcam #ab255730, USA) were determined by ELISA kits as protocol described. In brief, samples were sequentially incubated in antibody-precoated plates, followed by successive additions of biotinylated detection antibodies, streptavidin-horseradish peroxidase (HRP) conjugate, substrate solution, and finally a stop solution. The optical density of each well was then measured at 450 nm using a microplate reader. All samples were performed in triplicate (technical replicates).
Right ventricular hypertrophy measurement
The rats were anesthetized with sodium pentobarbital, then a polyethylene catheter was inserted into the right ventricle through the right jugular vein, then mean right ventricular pressure (mRVP) was measured using a PowerLab 8/35 multichannel biological signal recording system (AD Instruments, AUS). Following hemodynamic assessment, the animals were euthanized with pentobarbital sodium (50 mg/kg, i.p.). Cardiac tissues were then harvested, and the right ventricle (RV), left ventricle (LV), and interventricular septum (S) were carefully dissected and weighed. The mean right ventricular pressure (mRVP), the ratio of right ventricle weight to left ventricle plus interventricular septum weight (RV/LV + S), and the ratio of right ventricle weight to wall thickness (RV/WT) were calculated.
Transcriptome RNA-sequencing and GO analysis
Total RNA was isolated using TRIzol reagent (Takara, Japan) and subsequently purified with the Ribo-off rRNA Depletion Kit (Vazyme #N406, USA). cDNA libraries were constructed with the VAHTS Universal V8 RNA-seq Library Prep Kit for Illumina (Vazyme #NR605, USA) and sequenced on an Illumina HiSeq 2500 platform (Illumina, USA). Gene expression levels were quantified in reads per kilobase per million mapped reads (RPKM). Differentially expressed genes were identified using a threshold of |log2 fold change| ≥ 2.0 and an adjusted p-value < 0.05. Significantly enriched signaling pathways were assessed through Gene Ontology (GO) analysis using the DAVID 6.8 tool. All experiments included three biological replicates per group. The raw sequence data reported in this paper have been deposited in the Genome Sequence Archive in National Genomics Data Center, China National Center for Bioinformation/Beijing Institute of Genomics, Chinese Academy of Sciences (GSA: CRA022737) that are publicly accessible at https://ngdc.cncb.ac.cn/gsa.
Reverse transcription–quantitative PCR (RT-qPCR)
Total RNA was isolated using TRIzol reagent (Takara, Japan) according to the manufacturer’s instructions. Subsequently, 1 μg of the extracted RNA was reverse-transcribed into complementary DNA using a High-Capacity RNA-to-cDNA kit (ThermoFisher Scientific, #4387406, USA). Quantitative PCR was carried out with PowerUp SYBR Green Master Mix (ThermoFisher Scientific, #A25742, USA) on an ABI 7900HT quantitative PCR system (Applied Biosystems, USA). Gene expression calculated by 2-ΔΔCt method. GAPDH was used as internal control. The primers used in this study were listed in Supplemental Table 1. All samples were performed in triplicate (technical replicates).
Cell culture
Primary alveolar epithelial cells of newborn rats from 3 independent litters (n = 3 biological replicates) were isolated as previously described.15,16 Briefly, lung tissues were cut into pieces, digested with collagenase (1 mg/mL) and DNase (10 mg/mL), and passed through 70 μm strainer. Primary pulmonary endothelial cells from newborn rats were isolated as previously described. 17 Endothelial cells were separated by incubating with CD31 microbeads (Miltenyi Biotec #130-109-680) and selecting via magnetic separation according to the manufacturer’s instructions. Cells were cultured in DMEM (Hyclone, USA) supplemented with 10% fetal bovine serum (FBS, Hyclone, USA) and 1% penicillin/streptomycin (Hyclone, USA) at 37°C in a humidified atmosphere containing 5% CO2.
Lentivirus production and transduction
Lentiviral particles were produced via co-transfection of HEK293 T cells using packaging plasmids pMD2G and psPAX2 at a 5:3:2 mass ratio as previous described, 18 with Lipofectamine 2000 (Invitrogen) as the transfection reagent. The virus-laden supernatant was harvested at 24, 48 and 72 h post-transfection. For virus transduction, cells were exposed to lentiviral particles overnight in the presence of 10 μg/mL polybrene (Solarbio, #H8761), followed by selection with 2 μg/mL puromycin for 4 days.
Cell viability assay
Cell viability was assessed using the CCK-8 Cell Counting Kit (Beyotime, China), following the manufacturer’s protocol. Briefly, cells were plated at a density of 2500 cells per well in 96-well plates and subjected to designated treatments. Post-treatment, 10 µL of the CCK-8 reagent was added to each well, followed by a 2-h incubation period at 37°C. Absorbance at 450 nm was then determined using a Wellscan MK3 microplate reader (Labsystems, Finland). All samples were performed in triplicate (technical replicates).
Wound healing assay
Primary alveolar epithelial cells were seeded in 6-well plates and cultured to a confluence of 90%. A sterile plastic tip was used to scratch the cell monolayer. Cell debris was washed away by PBS. Images were obtained at 0 and 24 h by Olympus BX51 microscope (Tokyo, Japan). The width of the scratched gaps was measured by Image J software. Wound closure was calculated by the equation: Wound closure (%) = (1- width at 24 h/width at 0 h) × 100 %. All samples were performed in triplicate (technical replicates).
Transwell migration and invasion assays
Transwell migration and invasion assays were performed as previous described using transwell chamber (8 μm pore size, Millipore, USA). 19 In transwell migration assay, 5.0 × 104 cells were seeded into the upper chamber in serum-free condition. The low chamber was filled with 600 μL DMEM medium supplemented with 10% FBS. Cells were allowed to migrate or invade for 24 h, then cells at the bottom of the transwell chamber were fixed by 4% paraformaldehyde for 15 min at room temperature, and stained with crystal violet for 15 min at room temperature. To evaluate cell invasion, the transwell chamber was pre-coated with Matrigel (BD, USA). Images of migration or invasion cells were taken by Olympus BX51 microscope (Tokyo, Japan) equipped with Olympus DP70 camera using OLYMPUS Stream software 2.4. All samples were performed in triplicate (technical replicates).
Tube formation assay
To prepare the assay surface, each well of a 96-well plate was covered with 50 µl of Matrigel. Subsequently, primary pulmonary endothelial cells were plated onto this Matrigel substrate at a density of 1.5 × 104 cells per well. Following a 6-h incubation period, Olympus BX51 microscope (Tokyo, Japan) equipped with Olympus DP70 camera using OLYMPUS Stream software 2.4 was employed to capture images. The total length was quantified using the Angiogenesis Analyser plugin within the ImageJ software. All samples were performed in triplicate (technical replicates).
Flow cytometry
Lung tissue samples were minced and digested with dissociation buffer (2 mg/ml collagenase IV and 0.02 mg/ml DNase, Sigma, USA) in DMEM medium containing 10% FBS and 1% penicillin/streptomycin (Hyclone, USA) at 37°C for 1 h with agitation. Single cell suspensions were passed through 70 μm strainer. A total of 1.0 × 106 cells were stained with flowcytometry antibodies at 4°C for 30 min. Flow cytometry was performed using a B.D. Influx cell sorter (BD Bioscience, USA) and analyzed by Flowjo v10 software (Treestar, USA). The antibodies used in flow cytometry were: CD45 (#17-0461-82, 1: 200), CD3 (#CD3-2-FITC, 1: 200), CD8a (#25-0084-82, 1: 200), CD4 (#12-0040-82, 1: 200), CD25 (#46-0390-82, 1: 200), FoxP3 (#12-5773-82, 1: 200), Ly6C/6G (#25-5931-82, 1: 200) and F4/80 (#MA1-91124, 1: 200) were purchased from ThermoFisher Scientific (USA). All samples were performed in triplicate (technical replicates).
Western blot
Cultured cells and tissue samples were lysed using RIPA lysis buffer (Beyotime, China) containing a protease inhibitor cocktail (Sigma, USA). The protein concentration of each lysate was determined with a bicinchoninic acid (BCA) assay kit (Beyotime, China). Equal amounts of protein (20 μg) were separated by 8%-12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and subsequently transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, USA). The membranes were blocked by 5% non-fat milk, incubated with first antibodies at 4°C overnight and corresponding second antibodies at room temperature for 1 h. Western blot bands were revealed by ECL kit (ThermoFisher Scientific, USA). The antibodies used in this study were list below: LRP1 (Cell signaling technology #26387, 1: 1000) and GAPDH (Cell signaling technology #2118, 1: 1000). Western blots were performed on samples from n = 3 independent experiments. Band intensities were quantified using ImageJ software (version 1.52a, NIH) and normalized to GAPDH loading control. Relative protein expression was shown as fold-change compared to control.
Statistical analysis
Data normality was assessed using the Shapiro-Wilk test. For normally distributed data, parametric tests (t-test, ANOVA) were used. For non-normally distributed data, non-parametric alternatives (Mann-Whitney U test, Kruskal-Wallis test) were employed. All datasets met normality assumptions. For experiments with multiple comparisons, p values were adjusted using Bonferroni correction. Sample size (n = 5 per group) was based on previous hyperoxia-induced pulmonary hypertension studies demonstrating adequate statistical power with comparable group sizes.20,21 Exact p-values are reported when p > 0.001; p-values < 0.001 are reported as p < 0.001. Data were shown as mean ± SD and p < 0.05 was regarded as statistically significant.
Results
Establishment and characterization of a rat model of BPD-PH
Previously studies indicate that intermittent periods of hypoxia may contribute to the development of PH in BPD, 11 thus we tried to establish a rat model of BPD-PH by exposing newborn rats to a long period of hyperoxia (60% O2, 23h) and a short period of hypoxia (13%O2, 1h) every day for 2 weeks. Hyperoxia is a well-established method to induce BPD in newborn rats. 16 In our study, newborn rats were randomly divided into four groups, then exposed to hypoxia (13%O2), normoxia (21%O2) or hyperoxia (60%O2) as indicated for 14 days (Figure 1(a)). Histological assessment revealed that lungs from both the BPD and BPD-PH groups exhibited hallmark features of alveolar simplification, including enlarged airspaces, increased mean chord length, and a reduction in terminal airspace count and secondary septa, compared with the air control (A) and air plus low oxygen (AL) groups, confirming successful establishment of hyperoxia-induced BPD (Figures 1(b) and 1(c)). Compared with air group (A), the lung tissue in air + low oxygen group (AL) showed no significant morphological changes in pulmonary alveoli, indicating that exposing to a short period of hypoxia had no apparent influence on alveolar simplification (Figures 1(b) and 1(c)). To confirm the successfully induce of PH in BPD, mRVP, RV/LV + S, and RV/WT were evaluated. Compared with rats in BPD group, rats in BPD-PH group showed increased mRVP, RV/LV + S and RV/WT, indicating that hypoxia-induced PH model was successfully built in rats of BPD-PH group (Figures 1(d)–1(f)). The pathophysiological changes in the pulmonary arteries during PH development were evaluated. As shown in Figure 1(g), we observed significant increase in the ratio of vascular wall thickness (Figure 1(h)) and the ratio of vascular lumen area (Figure 1(i)) in small pulmonary arteries of BPD-PH group compared to BPD group, indicating pulmonary venous remodeling in BPD-PH rats. KL-6 is a marker for pulmonary injury. 22 In our study, KL-6 was evidently upregulated in rats of BPD group compared with those in air group (A) (Figure 1(g)). Notably, a further increase in KL-6 was observed in the BPD-PH group compared to the BPD group (Figure 1(j)), suggesting aggravated lung damage. VEGF is another biomarker of BPD and plays a central role in both lung development and function. 23 We found that VEGF was upregulated in rats of both BPD and BPD-PH groups compared with those in air group (A) (Figure 1(k)). Taken together, these results indicated the successful built of a rat model of BPD-PH.
BPD-PH rats have enhanced inflammatory response and increases accumulation of immune cells in lung
Mounting evidence underscores the critical role of inflammation in the pathogenesis of BPD-PH.24,25 To evaluate the inflammatory profile in our experimental model, we measured the mRNA expression of key pro-inflammatory cytokines in lung tissues. Firstly, the mRNA levels of pro-inflammatory cytokines TNF-α, IL-17A, IL-6 and IL-1β were dramatically increased in lung tissues of BPD and BPD-PH group compared with that in air group (A), suggesting enhanced inflammatory response in lung tissues of BPD and BPD-PH group (Figure 2(a)). Furthermore, the expression of these cytokines was markedly higher in the BPD-PH group than in the BPD group, suggesting a more severe inflammatory state in rats with superimposed pulmonary hypertension (Figures 2(a)–2(d)). Similarly, The levels of key pro-inflammatory cytokines, including TNF-α, IL-17A, IL-6, and IL-1β, were measured in lung tissues using ELISA. Compared to the air control group (A), rats in the BPD group exhibited significantly elevated cytokine levels, and these concentrations were further increased in the BPD-PH group, indicating an intensified inflammatory state in the latter (Figures 2(e)–2(h)). The above results indicated that BPD-PH rats had enhanced inflammatory cytokine production. Next, the accumulation of immune cells in lung was evaluated by flow cytometry. The gating strategy of flow cytometry was depicted (Figure 3(a)). We found that the accumulation of CD45+ Leukocytes, CD3+CD8+ Cytotoxic T cells (Cyto T), CD25+FoxP3+ regulatory T cells (Tregs), Ly6C+Ly6G+ Neutrophils (Neu) and Ly6G-F4/80+ Macrophages (Mф) was enhanced in BPD group compared with that in air group (A) (Figures 3(b)–3(g)). In addition, the accumulation of CD45+ Leukocytes, CD3+CD8+ Cyto T, Ly6C+Ly6G+ Neutrophils and Ly6G-F4/80+ Mф was increased, while the accumulation of CD25+FoxP3+ Tregs was decreased in BPD-PH group compared with that in BPD group (Figures 3(b)–3(g)). Collectively, our data indicated that BPD-PH rats had enhanced inflammatory response and increased accumulation of immune cells in lung. PH-BPD rats show enhanced inflammatory cytokine production. A-D, relative mRNA levels of Tnfa (a), Il17a (b), Il6 (c) and Il1b (d) in lung tissues of each group at day 14 after hyperoxia treatment were evaluated by RT-qPCR. n = 5 for each group. E-H, the levels of pro-inflammatory cytokines TNF-α (e), IL-17A (f), IL-6 (g) and IL-1β (h) in lung tissues of each group at day 14 after hyperoxia treatment were evaluated by ELISA assay. n = 5 for each group. Student’s t-test was used for statistical analysis between two groups. n = 5 for each group. *p<0.05. PH-BPD rats exhibit increasing accumulation of immune cells in lung. (a) The gating strategy in flow cytometry analysis was shown. B-G, the accumulation of CD45+ Leukocytes, CD3+CD4+ T helper cells (CD3+CD4+ Th), CD3+CD8+ Cytotoxic T cells (Cyto T), CD25+FoxP3+ regulatory T cells (Tregs), Ly6C+Ly6G+ Neutrophils (Neu) and Ly6G-F4/80+ Macrophages (Mф) in lung tissues of each group at day 14 after hyperoxia treatment was evaluated by flow cytometry. Student's t-test was used for statistical analysis between two groups. n = 5 for each group. *p<0.05.

Identification of differentiated expressed gene in BPD-PH rat model
The pathogenesis of BPD-PH is complex and involves diverse genetic factors. To uncover genes potentially implicated in this pathophysiology, we performed transcriptomic RNA sequencing on lung tissues from a rat model of BPD-PH. Differentiated expressed gene was defined as |log2 Fold Change| ≥ 2 and p < 0.05. Compared with lung tissues from rats of air group (A), a total of 636 upregulated genes and 565 downregulated genes were identified in lung tissues of BPD-PH rats (Figure 4(a), supplemental table 2). Kyoto Encyclopedia of Genes and Genomes (KEGG) and gene ontology analysis were conducted to evaluate significant influenced signaling pathways or gene ontology (GO) categories. NF-κB signaling, mitotic spindle, β-Catenin signaling, TGF-β signaling, Jak-Stat3 signaling, G2/M checkpoint, Apoptosis, Notch signaling, Myogenesis and Angiogenesis were the top ten most affected signaling pathways or GO categories (Figure 4(b)). The top 20 upregulated and top 20 downregulated genes were depicted in the heatmap (Figure 4(c)). Among them, Leucine-rich alpha-2-glycoprotein 1 (Lrg1), which is involved in angiogenesis and inflammation, ranked the sixth most upregulated genes and is reported to associate with BPD in previous study.
26
The elevated expression of LRG1 was further confirmed at both the mRNA and protein levels via RT-qPCR and western blot analyses, demonstrating a marked increase in lung tissues from BPD rats compared to the air control group (A), with a further upregulation observed in the BPD-PH group (Figures 4(d) and 4(e)). Collectively, these findings identify key differentially expressed genes in the novel BPD-PH rat model. Identification of differentiated expressed gene in PH-BPD rat model. (a) volcano map showed differentially expressed genes between lung tissues in rats of BPD-PH group and Air group. (b) enrichment analysis of significant affected signaling pathways or GO categories based on the KEGG and gene ontology. (c) the heatmap depicted the top 20 upregulated and downregulated genes between lung tissues in rats of BPD-PH group and Air group by transcriptome RNA sequencing. D-E, LRG1 expression in lung tissues of rats in each group was evaluated by RT-qPCR (d) and western blot (e). Student's t-test was used for statistical analysis between two groups. Statistical comparisons among multiple groups were performed using One-way ANOVA. *p<0.05.
LRG1 affects migration and TGF-β signaling activation in PH-BPD rats
To evaluate the potential influence of LRG1 in BPD-PH model, we ectopically expressed LRG1 in primary alveolar epithelial cells and pulmonary endothelial cells isolated from newborn rats. Accumulating evidence indicates that infants with BPD exhibit impaired alveolarization, reduced cellular migration, and compromised injury repair.
16
LRG1 has been previously reported to participate in the regulation of cell migration and angiogenesis in various cell types.27,28 Based on these findings, we hypothesized that LRG1 might influence the migratory and invasive capabilities of alveolar epithelial cells. Consistent with this hypothesis, forced expression of LRG1 significantly enhanced the wound-healing capacity of primary alveolar epithelial cells compared to vector-transfected controls in a wound healing assay (Figures 5(a) and 5(b)). In both transwell migration and invasion assays, ectopic expression of LRG1 significantly enhanced the migratory and invasive capacities of primary alveolar epithelial cells relative to vector-transfected controls (Figures 5(c) and 5(d)). TGF-β signaling plays a central role in lung morphogenesis and development of BPD.
29
In our study, LRG1 overexpression increased the phosphorylation of SMAD5 and SMAD2/3 in primary alveolar epithelial cells, suggesting the activation of TGF-β signaling (Figures 5(e) and 5(f)). Similarly, LRG1 exerted pronounced pro-migratory and pro-invasive effects in pulmonary endothelial cells, where forced expression promoted cell growth, wound closure, migration, and invasion (Figures 6(a)–6(e)). Moreover, LRG1 overexpression markedly enhanced the angiogenic capacity of pulmonary endothelial cells in an in vitro tube formation assay, as reflected by increased total tube length compared to the control group (Figures 6(f)–6(g)). In addition, LRG1 overexpression also increased the phosphorylation of SMAD5 and SMAD2/3 in pulmonary endothelial cells. Taken together, we found that LRG1 promoted migration, invasion and TGF-β signaling activation in primary alveolar epithelial cells and pulmonary endothelial cells from PH-BPD rats. LRG1 facilitates migration and TGF-β signaling activation in primary alveolar epithelial cells from PH-BPD rats. A-B, primary alveolar epithelial cells were transduced with LRG1 expression lentivirus or empty vector (Vector) control, then seeded in 6-well plates for wound healing assay (a). Wound closure was shown (b). C-D, primary alveolar epithelial cells were transduced with LRG1 expression lentivirus or Vector control, then used for transwell migration and invasion assay. Representative images (c) and relative migration and invasion cell numbers (d) were shown. E-F, primary alveolar epithelial cells were transduced with LRG1 expression lentivirus or Vector control, then collected lysates for western blot (e). Relative protein expression normalized to GAPDH (f) was shown. Student's t-test was used for statistical analysis between two groups. *p<0.05. LRG1 promotes growth, migration, invasion, tube formation and TGF-β signaling activation in pulmonary endothelial cells from PH-BPD rats. A, primary pulmonary endothelial cells were introduced with LRG1 expression lentivirus or empty Vector control, then seeded in 96-well plates for cell viability assay (a). B-E, primary pulmonary endothelial cells introducing with LRG1 expression lentivirus or empty Vector control were used for wound healing assay (b-c) or transwell assays (d-e). Wound closure (c) and relative migration and invasion cell numbers (e) were shown. F-G, primary pulmonary endothelial cells introducing with LRG1 expression lentivirus or empty Vector control were used for in vitro tube formation assay. Representative images (f) and relative tube length (g) were shown. H-I, primary pulmonary endothelial cells were transduced with LRG1 expression lentivirus or Vector control, then collected lysates for western blot (h). Relative protein expression normalized to GAPDH (i) was shown. *p<0.05.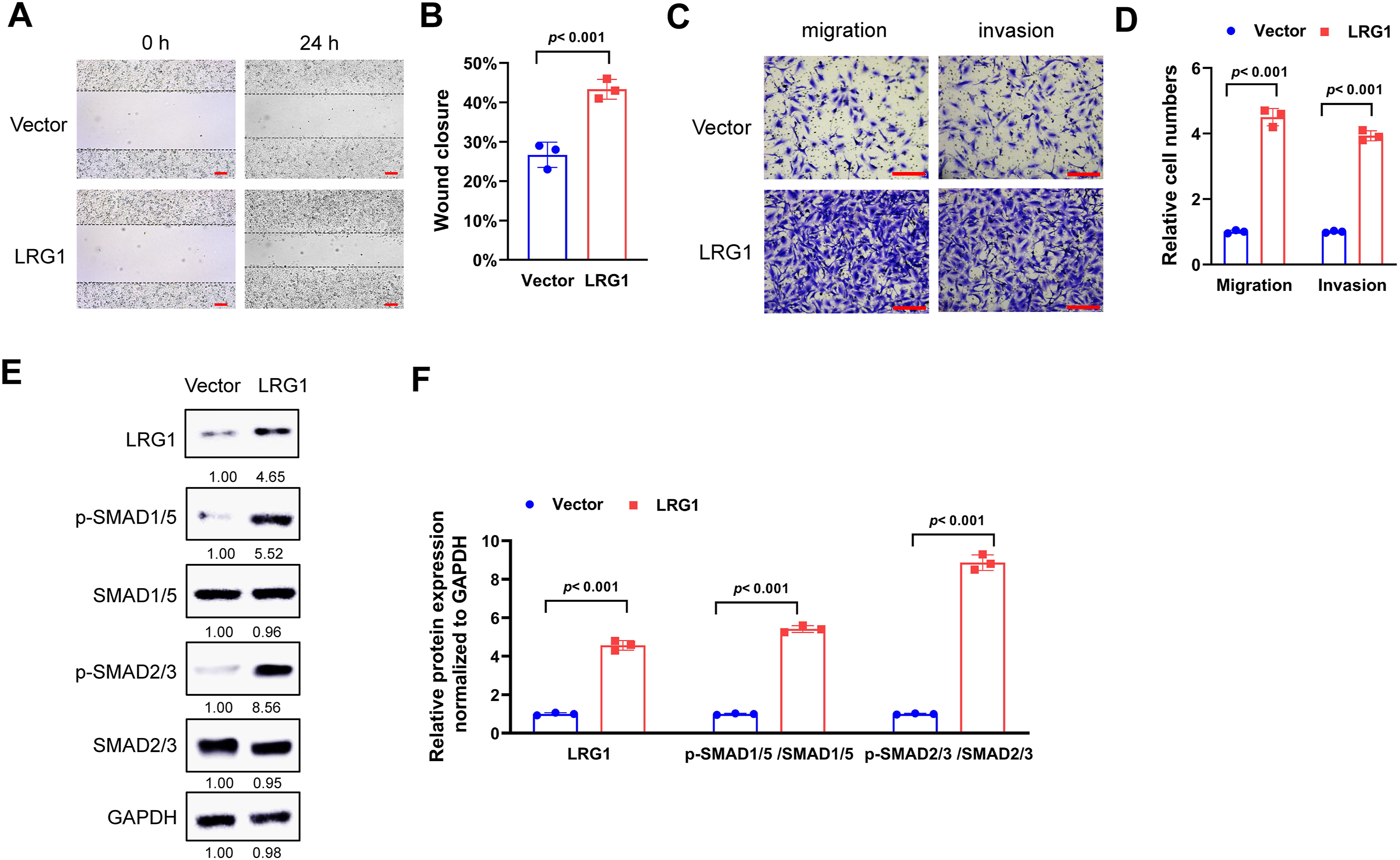

Evaluation of PH and BPD drugs in the rat model of BPD-PH
To assess the pharmacological relevance of the novel BPD-PH rat model established in this study, two commonly used drugs for BPD and PH were evaluated. Sildenafil is an FDA approved drug for the treatment of PH, while Dexamethasone is an anti-inflammatory drug for preventing BPD. In our study, BPD-PH rats were treated with Sildenafil (Silde), or the combination of Sildenafil and Dexamethasone (Dexam) as indicated. In H&E staining, BPD-PH rats showed characteristics of alveolar simplification, such as enlarged alveoli, increased chord length and reduced number of terminal airspace and secondary septa, but these situations were alleviated by the treatment of Sildenafil or the combination of Sildenafil and Dexamethasone, indicating that Sildenafil and Dexamethasone were effective in alleviating lung injury in the rat model of BPD-PH (Figures 7(a) and 7(b)). Administration of Sildenafil, either alone or in combination with Dexamethasone, significantly attenuated key indicators of pulmonary hypertension in BPD-PH rats, as demonstrated by reduced mRVP, RV/LV + S, and RV/WT compared to vehicle-treated BPD controls, indicating successful amelioration of hypoxia-induced PH (Figures 7(c)–7(e)). Furthermore, ELISA revealed that the elevated levels of pro-inflammatory cytokines (TNF-α, IL-17A, IL-6, and IL-1β) observed in lung tissues of the BPD group relative to air controls were markedly suppressed by treatment with Sildenafil alone or in combination with Dexamethasone (Figures 7(f)–7(i)). Importantly, these therapeutic regimens showed no significant effect on body weight compared to the vehicle control, suggesting a favorable safety profile with no overt cytotoxicity (Figure 7(j)). Taken together, these findings support the utility of the present BPD-PH rat model as a robust preclinical platform for evaluating potential pharmacotherapies. Evaluation of PH and BPD drugs in the rat model of PH-BPD. A-E, BPD-PH rats were treated with 5 mg/kg Sildenafil (Silde), or the combination of 5 mg/kg Sildenafil (Silde) and 0.2 mg/kg Dexamethasone (Dexam) at day P7 to P14 daily, then lung parenchyma (×200 magnification; scale bar = 100 μm) were stained with H&E staining (a). Images were acquired using an Olympus BX51 microscope equipped with a Olympus DP70 camera at ×200 objective magnification. Representative images from n = 5 animals per group. Arrows indicated typical structure of pulmonary alveoli. Average chord length was shown (b). Mean right ventricular pressure (mRVP) (c), the ratio of right ventricle weight to left ventricle plus interventricular septum weight (RV/LV + S) (d), and the ratio of right ventricle weight to wall thickness (RV/WT) (e) were evaluated. F-I, the levels of pro-inflammatory cytokines TNF-α (f), IL-17A (g), IL-6 (h) and IL-1β (i) in lung tissues of each group were evaluated by ELISA assay. n = 5 animals per group. (j) the body weight of BPD-PH rats was monitored every 3 days to assess drug toxicity. Data are presented as mean ± SD (n = 5 per group). Student's t-test was used for statistical analysis between two groups. *p<0.05.
Discussion
A variety of animal models have been developed to recapitulate the key pathological features observed in the lungs of premature infants with BPD. 30 These models mostly depend on injurious stimuli such as mechanical ventilation, oxygen toxicity or infection and sterile inflammation in mice, rats, rabbits, pigs and nonhuman primates. 31 For example, a hyperoxia-induced BPD model in neonatal mice is constructed to evaluate the mechanic roles of GSDMD in the pathogenesis of BPD and ROP. 32 In large animals such as preterm lamb, prolonged ventilation supporting with oxygen-rich gas is used to recapitulate the injury of immature postnatal lung of BPD. 33 In our study, we employed a hyperoxia-induced BPD model in rats to simulate impaired lung development. Despite the small size compared with preterm human babies, rat models of BPD are predominant models used in research for their relatively short gestation times and readily available and easily to maintain. Our BPD-PH rats had higher level of mRVP, RV/LV + S and RV/WT compared with BPD rats, suggested that we successfully built the BPD-PH model. Actually, hypoxia is commonly used to induce PH in rodent models.20,21 Thus, it was reasonable to induce PH in BPD rats by a short period of hypoxia. Our model combines intermittent hypoxia with hyperoxia to mirror clinical BPD-PH, provides an early-phase preclinical model to study BPD-PH as a synergistic disease entity rather than isolated conditions. A key limitation of this model is the simultaneous induction of BPD and PH via a combined hyperoxia-hypoxia insult from birth, which contrasts with the clinical progression in preterm infants where PH typically manifests after the initial BPD diagnosis. This discrepancy in the disease timeline represents a significant translational constraint. Moreover, this study used both male and female pups, and sex-specific effects were not evaluated. Future research should explore potential sex differences in BPD-PH development.
A growing body of evidence indicates that hyperoxia can trigger the accumulation of inflammatory cells within the developing lungs of preterm infants, a process implicated in the pathophysiology of bronchopulmonary dysplasia. In the present study, Ly6C+Ly6G+ Neutrophils and Ly6G-F4/80+ Mф were most abundant and enriched in BPD-PH rats. Indeed, neutrophils and macrophages are recognized as key drivers of inflammation in BPD, supported by clinical observations such as a significantly elevated neutrophil-to-lymphocyte ratio in infants who develop BPD compared to those who do not. 34 Neutrophil extracellular traps (NETs) have been found to be elevated in BPD infants and exacerbated BPD-like injury in neonatal mouse models by suppressing the WNT/β-catenin signaling pathway. 35 In a hyperoxia-induced mice model of BPD, hyperoxia promotes M1 macrophage polarization, activates macrophage-derived IL-6/STAT3 signaling in vivo, then finally leads to loss of epithelial cells in BPD lung. 36
In the present study, transcriptomic RNA sequencing was employed to identify genes potentially involved in the pathogenesis of BPD-PH, and found LRG1 was prominently upregulated and affected migration, invasion and TGF-β signaling activation in primary alveolar epithelial cells and pulmonary endothelial cells from PH-BPD rats. These findings not only support the utility of the BPD-PH model for uncovering novel genes implicated in disease development but also highlight LRG1 as a key vasculopathic mediator known to regulate angiogenesis through modulation of canonical and non-canonical TGF-β pathways in a cell- and context-dependent manner. 37 LRG1 has been implicated in mediating vascular damage across a range of pathological conditions, including pulmonary diseases.38,39 As LRG1 was upregulated in our BPD-PH rat model, we hypothesized that LRG1 likely contributes to the pathogenesis of BPD-PH. This model was further employed to assess the therapeutic efficacy of two commonly used drugs, Sildenafil and Dexamethasone, both of which demonstrated effectiveness in mitigating lung injury in BPD-PH rats. These results indicated that the rat model of BPD-PH was feasible for drug evaluation for BPD-PH treatment.
Conclusion
In summary, we established a rat model of BPD-PH, which showed characteristics of alveolar simplification and symptoms of PH. Our data indicated that we established a novel BPD-PH model in rats, and it provided a new choice for the future research for BPD complicated with PH.
Supplemental material
Supplemental Material - Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications
Supplemental Material for Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications by Suixin Liang, Xin Wang, Yemei Wu in Human & Experimental Toxicology
Supplemental material
Supplemental Material - Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications
Supplemental Material for Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications by Suixin Liang, Xin Wang, Yemei Wu in Human & Experimental Toxicology
Supplemental material
Supplemental Material - Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications
Supplemental Material for Establishment of a rat model of bronchopulmonary dysplasia complicated with pulmonary hypertension and its potential applications by Suixin Liang, Xin Wang, Yemei Wu in Human & Experimental Toxicology
Footnotes
Authors’ contributions
All authors guaranteed the integrity of the entire study. Suixin Liang and Xin Wang conducted the experiments. Data were analyzed by Yemei Wu. Manuscript was prepared by Suixin Liang. All authors had read and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Sanming Project of Medicine in Shenzhen (Grant no. SZSM202311027), Guangdong High-level Hospital Construction Fund, and Clinical key specialty construction project of Guangdong Province. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files. Numerical data for all figures were added in supplemental table 3. The raw sequence data reported in this paper have been deposited in the Genome Sequence Archive in National Genomics Data Center, China National Center for Bioinformation/Beijing Institute of Genomics, Chinese Academy of Sciences (GSA: CRA022737) that are publicly accessible at ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
