Abstract
Introduction
Bisphosphonates and denosumab are the most common antiresorptive drugs (ARs). ARs may cause medication-related osteonecrosis of the jaw (MRONJ) as a side effect, characterized by osteonecrosis and ulceration of the oral mucosa. The pathophysiology of MRONJ remains partly unclear and there is currently no consensus on its multietiological background. Immunomodulatory medications such as corticosteroids and antiestrogens may affect MRONJ onset.
Methods
An in vitro 3D cell culture model of gingival, keratinocytes, and fibroblasts were used to elucidate the pathogenesis of MRONJ. Cell cultures were exposed to ARs, followed by exposures to corticosteroids or antiestrogens. Morphology and proliferation were evaluated.
Results
Among the ARs, alendronate caused the most negative cellular changes, while zoledronate only had a few effects. Denosumab caused more morphological cell atypia and proliferation than other ARs. The combined exposures of individual ARs with corticosteroids had some additional negative effects on gingival cells, whereas ARs with antiestrogen had few effects. The results are partly inconsistent, indicating that 3D cell culture experiments may not be the most suitable method for studying the effects of ARs.
Conclusion
This study suggests that alendronate and denosumab affect gingival cell growth in a 3D cell culture model. These effects are smaller than reported in previous monolayer studies.
Introduction
Medication-related osteonecrosis of the jaw (MRONJ) is a potential complication of antiresorptive therapy. Antiresorptives (ARs) are therapeutically important for patients of several cancers or with high risk of osteoporosis- or osteopenia-related fractures and are therefore important for quality of life. 1
MRONJ is a rare condition with multifactorial etiology that is particularly difficult to treat, especially in cancer patients with a history of high-dose AR treatment. The most used ARs are bisphosphonates (BPs) and denosumab. MRONJ is characterized by osteonecrosis of the jawbone, possible sequestrum formation, and ulceration of the oral mucosa. In addition to ARs, MRONJ patients often receive additional medication for co-existing diseases, such as corticosteroids and antiestrogen. Corticosteroids have immunosuppressive and anti-proliferative properties 2 and antiestrogen can also be immunomodulatory. 3
3D cell culturing is considered a valid alternative to in vivo animal models when studying pathophysiological and pharmacological responses. 4 It has been shown that air-liquid interface 3D systems are more relevant when observing the in vivo situation of mucosal tissue than any other currently available in vitro approach based on submerged cell cultures. 5
For osteonecrosis to be cured, mucosal wound closure must occur. 1 The behavior of mucosal cells concerning MRONJ pathophysiology has been studied in vitro. Previous in vitro studies have concluded that ARs cause cell cytotoxicity, induction of apoptosis, and cell cycle arrest. 6 However, most previous literature has only studied nitrogenous BPs and utilized monolayer cell culturing.7–19 McLeod et al. 20 exposed gingival keratinocytes and fetal foreskin fibroblasts to BPs using a 3D model, concluding that there was no effect of BPs on these cells in the 3D model.
The effects of corticosteroids have been widely studied using monolayer in vitro models, as well as with human and animal in vivo studies. On a cellular level, corticosteroids decrease keratinocyte proliferation, reduce keratinocyte growth factor, and inhibit fibroblast proliferation, migration, and chemotaxis. 21 Estrogen has been shown to directly induce cellular proliferation, differentiation, and growth in gingival keratinocytes and fibroblasts. 22 To our knowledge, the effects of antiestrogen or corticosteroids on human gingival cells have not been studied using a 3D in vitro model.
The aim of this study was to investigate the combined effects of ARs with corticosteroid and antiestrogen on the growth and differentiation of gingival keratinocytes and fibroblasts in a tissue culture model to better understand the mucosal components of MRONJ.
Methods
Pharmaceuticals
Two BPs, zoledronate (Zoledronic acid monohydrate, Sigma-Aldrich Finland Oy, Finland) and alendronate (Alendronate sodium trihydrate, Sigma-Aldrich Finland Oy, Finland), were used at concentrations of 5 μmol/L. These concentrations were selected based on reported tissue drug levels following clinical administration. Pharmacokinetic studies indicate that zoledronate accumulates in bone, binding to bone hydroxyapatite.1,23 Specifically, bioassay data from bone and saliva samples of patients receiving zoledronate revealed BP concentrations ranging from 0.4 to 5 μmol/L, aligning with our used in vitro concentration. 24 Denosumab (XGEVA, Amgen, Thousand Oaks, CA, USA) was used in gradient concentrations of 25 μg/mL, reflecting its estimated concentration in serum following subcutaneous administration.25,26 The co-medications tested included antiestrogen (Faslodex®) and corticosteroid (Di-Adreson-F Aquosum®). The concentrations of antiestrogen (8 ng/mL) and corticosteroid (303 ng/mL) were determined based on reports of drug concentrations in plasma with commonly used dosages.27,28 Additionally, there were negative controls as well as controls only exposed to either corticosteroid or antiestrogen.
Cell cultures
Prior to conducting experiments, a pilot study was performed to minimize errors. A biostatistician (author EL) was also consulted regarding sample size. Cell experiments were conducted on a minimum of three cultures per exposure in three separate experiments. Three different sites on each histological slide of each culture were recorded. The number of cultures per exposure, ‘n (final sample size)’, were as follows: Antiestrogen n = 12 (4), Alendronate n = 9 (3), Alendronate and Antiestrogen n = 12 (4), Alendronate and Corticosteroid n = 12 (4), Control n = 15 (5), Denosumab n = 9 (3), Denosumab and Antiestrogen n = 9 (3), Denosumab and Corticosteroid n = 9 (3), Corticosteroid n = 9 (3), Zoledronate n = 9 (3), Zoledronate and Antiestrogen n = 9 (3), and Zoledronate and Corticosteroid n = 9 (3).
Spontaneously immortalized human gingival keratinocytes (HMK) 29 of passage seventeen and human normal gingival fibroblasts 30 of passage eight were used in the co-cultures. Cell lines were authenticated and tested for mycoplasma contamination (Venor GeM OneStep Mycoplasma Detection Kit, Sigma). Both cell cultures were grown separately in Dulbecco’s Modified Eagle Medium (DMEM) with 1% Penicillin-Streptomycin-Neomycin, 1% L-glutamine, and 10% Fetal Bovine Serum (Thermo Fisher Scientific, Waltham, MA, USA) in standard 250 ml flasks in a humidified incubator with 5% carbon dioxide (CO2) and 95% air at 37°C. No exclusion criteria were set.
Collagen matrix preparation for the air-liquid interface model
Fibroblasts were detached for the experiments with 0.25% trypsin (Gibco BRL) and then suspended in a collagen (Pure Col, Advanced BioMatrix, Inc, CA, USA) 3 mg/ml solution to a concentration of 250,000-300,000 cells/ml. A 500 µl aliquot of the solution was placed in each insert well (Thincert), which were then placed on a six-well plate (Costar, Cambridge, MA). The matrix gel was allowed to solidify for 1-2 h at 37°C in an oven with no CO2 flow. Flavin Adenine Dinucleotide (FAD) medium was placed onto the insert well of the solidified gel along the edges. FAD medium consists of three parts DMEM and one part Ham-F12 (Gibco BRL), supplemented with 10% fetal bovine serum, 4 mM L-glutamine (Gibco BRL), 5 μg/ml insulin (Sigma), 0.18 mM adenine (Sigma), 0.1 nM cholera toxin (Sigma), 5 ng/ml epidermal growth factor (Boehringer Mannheim, Mannheim, Germany), and 0.4 μg/ml hydrocortisone (Sigma). Ascorbic acid, 50 μg/ml, was added to the medium before culture. Plates were transferred to grow in the incubator. Collagen matrices with fibroblasts were cultured in the insert wells for 1 day.
Air-liquid interface Co-culture model of HMKs on matrices with fibroblasts
HMKs were passaged and suspended with DMEM. The cell suspension was aspirated through the needle of a 1 ml syringe. Cells were again suspended in the FAD medium at 400,000 cells/0.5 ml. HMK cell suspension was carefully added (500 µl/well) along the edges of the insert wells. Insert wells were cultured for 2 days until keratinocytes formed a 100% confluent layer on the collagen matrix. The growth medium (FAD) was changed once per day. After the keratinocytes had reached confluence, the co-cultures were lifted, and with the aid of a scalpel blade, the collagen was carefully cut loose from insert well wall edges. The cultures were placed in new six-well plates (Costar) on stainless steel grids and maintained in the incubator. The FAD medium of the co-cultures was replaced once every 2 days. The submerged co-cultures were allowed to grow for 7 days at the air-liquid interface before exposure (tenth day of the experiment).
Each concentration of pharmaceuticals was pipetted (0.5 ml) into the insert well simultaneously with the last medium change. The experiment ended after 24 h of exposure. The exposure time was chosen based on a previous monolayer study 18 with the same pharmaceuticals where both keratinocyte and fibroblast viability and proliferation were already significantly affected after 24 h.
Fixation, preparation, and staining of Co-culture samples
Co-cultures were placed in a transport case, fixed in formalin, and transported in a series of alcohol before embedding into paraffin blocks. The blocks were cut into 20 µm sections. Three sections were taken from each co-culture. Slides were stained with hematoxylin and eosin (HE) and with Ki-67 (Monoclonal Mouse Anti-Human, Dako Omnis, Agilent, Santa Clara, USA). All slides were blinded by one author (HM E-K) prior to analysis. Each slide was assigned a numerical code and the pharmaceutical exposure information was stored in a separate Microsoft Excel file (Microsoft Corp., Washington, USA), which was inaccessible during evaluation. The histopathological evaluations were performed by an oral pathologist (J R) under these circumstances and characteristics recorded. Fibroblast and keratinocyte layers were measured (µm) with ImageJ (Research Services branch, National Institutes of Health, USA) (Figure 1). Pharmaceutical treatment allocations were then unblinded by author HM E-K. The following characteristics were recorded from the epithelial layer/keratinocytes (Figure 2): apoptosis (yes/no), cell atypia (yes/no), keratinization (yes/no), metaplasia (yes/no), keratin pearls (yes/no), possible disorganization of cell layers (0-2), and proliferation (Ki-67) (Figure 3). Additionally, an overall classification of co-culture tissue samples (normal/disorganized/atypia) was recorded (Figure 4). ‘Disorganized’ means morphological, architectural disturbance with cell organization in the structure of the epithelium, ‘atypia’ means pleomorphism at the cellular level resembling dysplastic changes, and ‘metaplasia’ means that the epithelium has adenomatous features. An example of measuring fibroblast and keratinocyte layer depth (μm), measured with ImageJ (National Institutes of Health, USA). Magnification size x40. Ki-67 (Mib-1) proliferation. Positive, brown epithelial cells (blue arrows) were counted (n) and recorded per sample. Statistically significant changes in means are marked, * = p < 0.05. Magnification size x40. Histopathological characteristics from the epithelial layer/keratinocytes: (A): keratinization (red arrow) and keratin pearl (blue arrow), cell atypia (dashed blue arrows) (B): apoptosis (blue arrows) (C): metaplasia where the epithelium has round, adenomatous features (blue arrows). Magnification size x40. Overall classification of co-culture tissue samples as normal/disorganized/atypia. ‘Disorganized’ means morphological, architectural disturbance with cell organization in the structure of the epithelium, ‘atypia’ means pleomorphism at the cellular level resembling dysplastic changes, and ‘metaplasia’ means that the epithelium has adenomatous features. Normal epithelium; disorganized epithelium showing architectural disturbance of the epithelium; dysplastic epithelium shows drop like rete ridges and pleomorphism at the cellular level resembling dysplastic changes. Magnification size x40.



Statistical methods
Continuous variables are summarized with categorical variables and percentages: median, lower and upper quartiles, or range. The association between drug exposure and histopathological characteristics (apoptosis, keratinization, disorganization, cell atypia, metaplasia) was tested with Fisher’s exact test. Differences in fibroblast and keratinocyte cell layer width (in µm) between drugs (control and one drug at a time) and sites were tested with a two-way analysis of variance. A logarithmic or square root transformation was used to fulfill the assumption of normality for studentized residuals.
The threshold for statistical significance was set to 0.05 (two-tailed). The data analysis for this paper was conducted using SAS software Version 9.4 for Windows (SAS Institute Inc., Cary, NC, USA).
Results
3D Co-culture
During the 3D air-liquid interface co-culture conditions, cells formed gingival tissue-like cultures. Human mucosal keratinocytes formed a well-differentiated, multi-layered squamous cell epithelium. Some cultures showed keratinization. Beneath the epithelium, fibroblasts formed regular connective tissue, modeling the lamina propria. The median thickness of keratinocyte layers in the samples with no treatments was 55.43 μm (range 15.30-314.2 μm) and fibroblast layers 50.17 μm (range 0-157.2 μm). These samples are later referred to as control samples.
AR Treatments
Each AR treatment caused at least some cellular changes in gingival tissue-like cultures. Keratinocytes with alendronate treatment showed more keratinization, cell atypia, disorganization, metaplasia, and proliferation than control cultures (each p < 0.012 (Alendronate exposed cultures with keratinisation n = 6 versus control cultures n = 0, atypia n = 4 versus 0, severe disorganization n = 4 versus 0, metaplasia n = 4 versus 0, proliferation (Ki-67) mean 12.89 versus 2.73.)). Zoledronate treatment caused a less disorganized cell pattern of keratinocytes compared to the control culture (all p < 0.003). Denosumab treatment caused atypia (all p < 0.003), increased cell proliferation (all p < 0.025), and had more samples classified as disorganized (p = 0.003) (Denosumab-exposed cultures with atypia n = 5 versus control cultures n = 0, proliferation (Ki-67) mean 10.22 versus 2.73, classified as disorganized n = 7 versus 2.).
Antiestrogen treatment with ARs
Statistically significant differences between AE and ARs compared to controls (p < 0.05), analyzed with two-way analysis of variance (including drug and layer width as factors) and Fisher’s exact test.
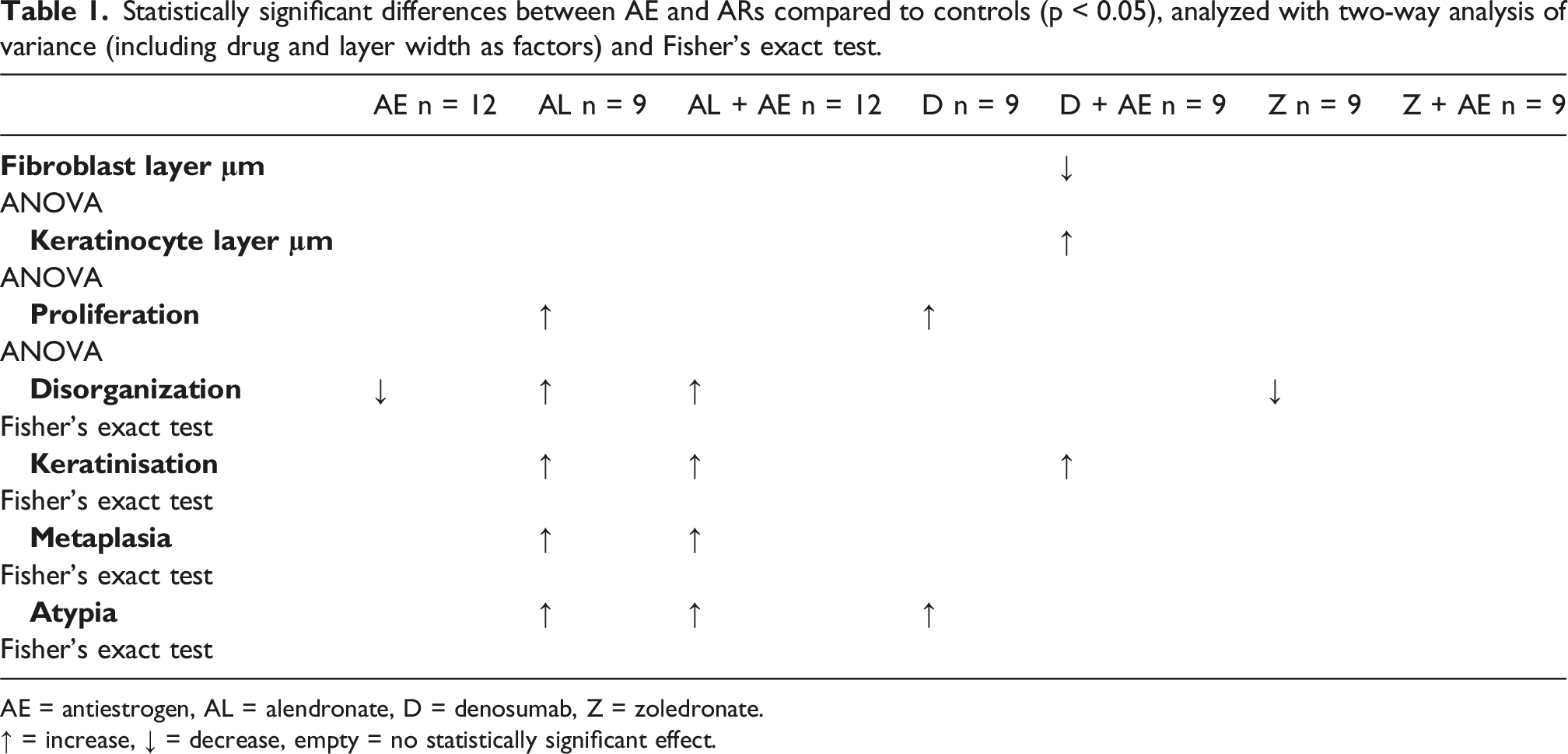
AE = antiestrogen, AL = alendronate, D = denosumab, Z = zoledronate.
↑ = increase, ↓ = decrease, empty = no statistically significant effect.
Corticosteroids with ARs
Statistically significant differences between Corticosteroids and ARs compared to controls (p < 0.05), analyzed with two-way analysis of variance (including drug and layer width as factors) and Fisher’s exact test.

CS = corticosteroid, AL = alendronate, D = denosumab, Z = zoledronate.
↑ = increase, ↓ = decrease, empty = no statistically significant effect.
Zoledronate- and corticosteroid-exposed cells had more apoptosis, keratinization, and disorganization than only zoledronate-exposed cells (all p < 0.049) (Zoledronate- and corticosteroid-exposed cultures’ apoptosis n = 6 versus zolendronate-exposed cultures n = 1, disorganization n = 9. versus 2, keratinization n = 5 versus 0.). Zoledronate with corticosteroids caused more disorganization and keratinization than control cultures (p = 0.003). Samples were also more likely to be classified as ‘disorganized’ in comparison to controls (all p < 0.003). Keratinocyte cell layers were thinner after only zoledronate exposure than zoledronate and corticosteroid exposure (p = 0.019), while there were no differences in fibroblast layer thickness. There was no statistically significant difference between zoledronate- and corticosteroid-exposed keratinocyte cell layer thickness and gingival cell controls.
Corticosteroid with denosumab had a negative effect on proliferation compared to only denosumab exposure (p = 0.002), thus there was no statistically significant difference compared to control cultures. Combined corticosteroid and denosumab exposure lead to a thinner keratinocyte cell layer than only corticosteroid-exposed cultures (p = 0.040) (Corticosteroid- and denosumab-exposed cultures’ keratinocyte layer thickness mean 72.27 μm versus corticosteroid exposed cultures’ mean 121.72 μm.), while there was no statistical difference in cell layer thickness between controls and only denosumab-exposure samples. Samples were also more likely to be classified as ‘atypia’ or ‘disorganized’ compared to controls (all p < 0.0001).
Discussion
AR treatments
All three AR treatments had some effect on 3D gingival cell cultures. Alendronate seemed to have the most effects, causing keratinization, keratinocyte atypia, disorganization, metaplasia, and increased proliferation. These findings are partially supported by Soydan et al., 16 who also reported negative effects of alendronate on fibroblast proliferation and higher apoptosis in keratinocytes. However, the effects of zoledronate treatment in the 3D model were unexpected and are not in line with previous monolayer studies. Zoledronate has been noted to decrease keratinocyte and fibroblast cell viability,7–9,12,18 though two studies concluded that zoledronate did not have negative effects on keratinocytes or fibroblast cells.19,20 Many studies used higher concentrations and longer exposure times and thus observed more pronounced effects, raising questions about the interpretation of results in in vivo conditions. The effects of BPs may be more pronounced in vivo after tooth extraction, as the concentrations can be 100 times higher at extraction sites.23,31 Another explanation may be cell-cell and cell-matrix interactions in 3D cultures, which create signaling pathways that enhance cell survival and resistance, or the presence of more physiological gradients influencing drug metabolism and response. 32 This finding might give some support to the hypothesis that mucosal deterioration might occur secondary to bone necrosis, rather than being an initiating factor. 33
Denosumab treatment alone caused keratinocyte atypia and proliferation at low concentrations. The effects on gingival cells were consistent with previous studies.18,34 In Yuan et al., 34 fibroblasts were tested with almost double the denosumab concentrations as in the present study, resulting in negative cellular effects on wound healing. There are no previous MRONJ studies of human gingival cells in 3D co-culture conditions or on antiestrogen treatment on these cells.
Antiestrogen treatment with ARs
Antiestrogen treatment alone did not affect 3D gingival cell culture. Antiestrogen with alendronate increased keratinocyte metaplasia (p = 0.0046). Antiestrogen with denosumab had some varying, inconclusive effects. Antiestrogen with zoledronate did not cause significant changes. It has been suggested that fibroblast growth factor pathway and estrogen pathway are positively related. 35
Corticosteroids with ARs
Corticosteroids alone caused only a minor decrease in proliferation on 3D cell culture. This is somewhat contradictory to previous literature regarding the negative effect of corticosteroids on fibroblast and keratinocyte layer width. Corticosteroids are known to impair wound healing by inhibiting keratinocyte growth factor and (gingival) fibroblast growth.2,21,36
Alendronate with corticosteroids caused cellular changes compared to controls but not compared to only alendronate exposure. Zoledronate alone showed minor cellular changes, but along with corticosteroid exposure, there were several additional changes, which indicates a negative interaction. Denosumab with corticosteroids also caused changes that were not detectable with only denosumab or corticosteroid exposure, indicating a possible interaction in keratinocytes.
Clinical relevance
The additive negative effects of antiestrogen with alendronate, as well as corticosteroid, seen in our study may explain clinicians’ observations and epidemiological studies’ reports of antiestrogen being a factor in predicting MRONJ risk in patients1,37
Denosumab interaction with corticosteroids caused negative cellular effects, which could be supported by the reported higher risk of skin infections in patients using corticosteroids and denosumab. 25
This study supports the widely discussed notion that MRONJ may not be homogeneous and that drug-specific mechanisms must be considered clinically.
Studies and clinical guideline position papers on AR drug holidays’ effects on MRONJ treatment remain inconclusive. 1 Although this study used a cell culture model, future AR drug holiday studies may wish to consider the possible effects of corticosteroids and/or antiestrogen.
Strengths and limitations
This study was conducted with human gingival keratinocytes and human gingival fibroblasts, making it best suited for modeling the in vivo effects of oral mucosa. The concentrations of ARs represented real-life tissue concentrations.24,38,39 Co-cultures were replicated three times under repeatable conditions with at least three separate co-cultures per exposure to enable reliable statistical analysis. A 24-h exposure was chosen to ensure methodological consistency with our previous monolayer study and to describe early cellular responses. While this duration does not mimic chronic clinical exposure, longer in vitro periods are limited by reduced cell resilience and increased contamination risk.
3D cell culturing also has limitations mimicking in vivo circumstances in the gingiva. However, this research approach is only possible with cell culture methods and is considered viable for pharmacological studies.4,40 Importantly, immune cell functions have been proven to play a role in MRONJ onset 1 and this model does not include blood cells or immune cell functions. Innate or acquired immune dysfunction has been proven to be a part of MRONJ pathogenesis, therefore future studies should integrate immune components to achieve a more physiologically relevant system. Current challenges include microscopy due to large size (compared to monolayer cell cultures) and the questionable ability of different compounds (e.g., antibodies, drugs) to penetrate 3D structures. 3D models are used by various research fields, and methods involving 3D cell cultures still need standardization. 4 In this study, however, an air-liquid interface system was used, which is currently considered the most reliable 3D system. 5 Additionally, this study describes morphological changes but does not aim to explain the mechanisms behind them. There was also some inconsistency in our culture results.
Even though only preplanned statistical analyses were conducted, the number of statistical analyses performed was high, which may increase Type 1 error and the potential for false significant results. Therefore, these results must be interpreted with caution, and further studies are needed to evaluate this 3D method in studying MRONJ.
Conclusion
Among the ARs studied, alendronate exhibited the most pronounced effects on gingival cell morphology and proliferation. Zoledronate, on the other hand, had limited effects. The addition of corticosteroids or antiestrogens to AR treatments resulted in minor additional effects compared to AR treatments alone. Our 3D co-culture model provides valuable insights into the cellular mechanisms underlying MRONJ. Future studies should focus on incorporating immune components to better mimic in vivo conditions and further elucidate the interactions between ARs, corticosteroids, antiestrogens, and bone tissue. The results of this study suggest that ARs impact gingival cell growth and differentiation, contributing to the pathogenesis of MRONJ.
Footnotes
Acknowledgments
The authors thank Katja Sampalahti and Mariia Valkama for their skillful technical assistance.
Ethical approval
This study was conducted using solely established human gingival cell lines. The human gingival keratinocyte and fibroblast cell lines used in this study are well-characterized and previously reported in literature,29,30 along with the ethical aspects of establishing these cell lines and routine authentication and mycoplasma testing conducted. No human participants, identifiable human material, or animals were involved in this study. In accordance with Finnish legislation (Medical Research Act 488/1999) and ethical guidelines, the further use of these established cell lines did not require ethical approval. The authors declare compliance with research integrity guidelines and no use of AI-generated text in the preparation of this manuscript.
Author contributions
Conceptualization: Heidi M Ekholm-Kerppola, Tero Soukka, Jaana Rautava Methodology: Heidi M Ekholm-Kerppola, Jaana Rautava. Formal analysis and investigation: Heidi M Ekholm-Kerppola, Eliisa Löyttyniemi, Jaana Rautava. Writing review and editing: Heidi M Ekholm-Kerppola, Jaana Rautava. Funding acquisition: Heidi M Ekholm-Kerppola. Supervision: Tero Soukka, Jaana Rautava.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Finnish Dental Society Apollonia; 20220072; 22017.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available at a data repository. Ekholm-Kerppola, Heidi, 2024, “Impact of Antiresorptive Therapy and Co-medications in a 3D Gingival Cell Co-Culture Model”, ![]() , Harvard Dataverse.
41
, Harvard Dataverse.
41
