Abstract
Introduction
Testicular toxicity commonly manifests as impaired spermatogenesis and testicular atrophy. Bisphenol A (BPA), a commonly used organic plasticizer, negatively affects sperm parameters, hormonal levels, and fertility. Octreotide (OCT), a somatostatin analog, was originally used to treat acromegaly, carcinoid tumors, vasoactive intestinal peptide-secreting, and growth hormone-secreting tumors. OCT has demonstrated potential therapeutic properties beyond its traditional use in endocrine disorders. We hypothesized that OCT would attenuate BPA-induced testicular damage through its anti-inflammatory, anti-oxidant, and anti-autophagic properties. This study aimed to assess the mechanisms of BPA-induced testicular toxicity and evaluate the ameliorative effects of OCT.
Methods
Forty adult male Sprague Dawley (SD) rats were randomly assigned to four equal groups: group1: saline control, group2: dimethyl sulfoxide (DMSO) vehicle, group3: BPA-treated, and group4: BPA + OCT-treated. Treatments were administered for 4 weeks. Sperm count, testicular weight, serum testosterone, lactate dehydrogenase (LDH), alkaline phosphatase (ALP), total antioxidant capacity (TAC), testicular levels of malondialdehyde (MDA), tumor necrosis factor-alpha (TNFα), Beclin-1 (BECN-1), Microtubule-associated proteins 1A/1B light chain 3A (MAP1LC3A/LC3), mammalian target of rapamycin (M-TOR), histopathological examination, apoptotic index, and Johnson’s score were assessed.
Results
BPA administration significantly impaired spermatogenesis, reduced serum testosterone and TAC, and increased MDA, TNFα, and autophagy-related markers, along with histopathological damage of testis. Co-treatment with OCT mitigated these effects by improving sperm parameters, hormone levels, oxidative stress markers, inflammatory cytokines, and testicular histology.
Discussion
The findings suggest that OCT exerts a protective effect against BPA-induced testicular toxicity, through its anti-inflammatory, antioxidant, and autophagy-modulating properties. OCT may offer therapeutic potential in mitigating BPA-induced testicular toxicity.
Introduction
Bisphenol A (BPA) is an environmental pollutant widely used in industry. It can be found in epoxy resins (such as food packaging and metal can-lining) and polycarbonate plastic products used to manufacture refillable drinking bottles, plastic utensils, and safety equipment. BPA can be found in carbonless thermal receipt paper, which can be frequently touched in our daily lives. 1
After liberation from various commonly used products, 2 BPA is absorbed and spread by the circulating blood. 3 BPA is claimed to induce multiple pathological conditions such as reproductive organ toxicities, developmental anomalies, neurological disorders, behavioral changes, metabolic abnormalities, and cancers. Its toxic effect on the reproductive system occurs via different mechanisms. 4 BPA is a xenoestrogen that stimulates estrogenic activity, leading to a decrease in testosterone levels due to decreased free androgenic hormonal activity that inhibits testicular steroidogenesis. Recent studies reported that exposure to BPA is always associated with low semen quality through cross-reacting with estrogen receptors. 5
Exposure to BPA liberates reactive oxygen species (ROS), including hydroxyl radical, hydrogen peroxide, and superoxide anion. 6 Over production of ROS leads to oxidative damage to membrane lipids, proteins, and nucleic acids. 7 The levels of ROS in semen show an inverse correlation between the percentage of motile sperm and the outcome of pregnancy. 8 This oxidative stress can stimulate apoptosis and autophagy pathways, leading to more impairment of spermatogenesis.9,10 Beclin1 (BECN-1) and light chain3 (LC3) have been widely used to monitor the number of autophagosomes and autophagic activity. Hence, BECN-1 and LC3 are commonly used as markers of ongoing autophagy. 11
Dimethyl sulfoxide (DMSO)-only vehicle control is essential to distinguish any potential effects of the solvent itself from those of the treatment compounds. DMSO is widely used as a solvent due to its ability to dissolve a broad range of substances and its generally low toxicity at small concentrations. 12
Octreotide (OCT) is a somatostatin analog initially used to treat acromegaly, carcinoid tumors, vasoactive intestinal peptide-secreting tumors, and growth hormone-secreting tumors. 13 Recently, mounting evidence demonstrated the remarkable ameliorative effect of OCT, through autophagy modulation along with abrogation of underlying cellular apoptosis. 14 For instance, reports have demonstrated that Octreotide could suppress apoptosis of liver and kidney in the hepatic ischemia-reperfusion injury animal model via autophagy enhancement. Moreover, octreotide preserved the myocardial tissue structure, restrained neutrophil infiltration and apoptosis in doxorubicin-induced cardiomyocyte toxicity as a consequence of improving the anti-oxidation capacity of cardiomyocytes via activating the nuclear factor erythroid 2 (Nrf2) pathway. 15
The hypothesis guiding this study is that Octreotide, a somatostatin analog known for its anti-inflammatory and cytoprotective properties. Recent research has demonstrated that octreotide has an organ-protective effect through various mechanisms, as anti-inflammatory effect by downregulation of inflammatory mediators and cytokines including Tumor necrosis factor alpha (TNFα) and Interleukin 1β (IL-1β), lowering of endotoxin levels, and direct cyto-protective properties. 16 Based on the above-mentioned findings, OCT may exert an ameliorative effect against testicular toxicity induced by BPA, a widely used industrial chemical with known endocrine-disrupting and reproductive toxic effects. The selection of BECN-1, Microtubule-associated proteins 1 A/1B light chain 3A (MAP1LC3A/LC3), and mammalian target of rapamycin (M-TOR) as molecular targets reflects an assumption that BPA-induced testicular damage may involve dysregulation of autophagy and cell survival pathways.
This is the first study to investigate the ameliorative effects of OCT on BPA-induced testicular toxicity by examining its impact on oxidative stress, inflammation, apoptosis, autophagy, and histopathological changes.
Aim of the work
The aim of this study was to evaluate the possible ameliorative effect of OCT on testicular toxicity induced by BPA in male SD rats by examination of sperm parameters, hormonal profile, oxidative state, autophagy and histopathological changes.
Materials and methods
Drugs and chemicals
Bisphenol A (BPA, CAS Number: 80-05-7) (Catalog Number: 02,183) was obtained from Loba Chemie Pvt. Ltd (Mumbai, India). It was dissolved in dimethyl sulfoxide (DMSO, CAS Number: 67-68-5) (Catalog Number: D2650) was obtained from Sigma Aldrich Company (St Louis, Missouri, USA).
Octreotide (Octreotide acetate, CAS Number: 83,150-76-9) (Catalog Number: O1014) was obtained from Sigma-Aldrich (St Louis, Missouri, USA). It was dissolved in sterile water for injection before administration. Unless otherwise described, other used chemicals were obtained from El-Gomhouria Company for Drugs, Egypt.
Experimental animals
The experiment was conducted in the medical pharmacology department of the faculty of medicine at Tanta University. 40 adult male Sprague Dawley (SD) rats (weighing 150-200 g) were housed in clean, well-ventilated cages with controlled conditions (constant temperature of 25 ± 3°C and 12 h light/dark cycles). They had a standard diet and water ad libitum. All animal experiments were approved by the Research Ethics Committee, Faculty of Medicine, Tanta University, Egypt (Approval no. 36239/12/22) and were conducted in accordance with ARRIVE 2.0 guidelines. All procedures were carried out in accordance with the principles of humane animal care. The number of animals used was the minimum required to achieve statistical significance, and every effort was made to minimize animal suffering.
Randomization and blindness
Animals were randomly allocated into four equal groups (10 rats for each) as follows. • Group 1 (saline control group) was injected intraperitoneally (IP) with saline 1 ml/kg/day 4 times per week (Sunday, Monday, Wednesday, and Thursday). • Group 2 (DMSO vehicle group) was injected IP with a vehicle of DMSO in a dose of 1 ml/kg/day, once daily for 4 times per week for 4 weeks.
17
• Group 3 (BPA-treated group) was injected IP with BPA, which was prepared by dissolving BPA in DMSO to form stock solutions to final concentration (45 mg BPA/1 ml DMSO), was given in a dose of 50 mg/kg/day, 4 times per week (Sunday, Monday, Wednesday, and Thursday) for 4 weeks.
18
The dose was chosen according to a pilot study performed before the main experiment. • Group 4 (BPA + OCT-treated group) was injected IP with BPA and OCT. OCT was given in a dose of 20 µg/kg/day 4 times per week (Sunday, Monday, Wednesday, and Thursday) for 4 weeks.
15
All drugs were administered according to the dosing schedule described in experimental design in Figure 1. Summary of the experimental design.
Histopathological assessments were performed by an investigator blinded to the treatment groups.
Assessment of body weight (gram)
The body weight of rats was recorded at the beginning of the experiment (Initial body weight) and recorded at the end of the experiment (Final body weight) after 4 weeks, and then the percent change (%) of body weight was calculated as follows:
Percent change in body weight gain = [(Final body weight − Initial body weight)/Initial body weight] × 100.
Blood and tissue sampling
At the end of the experiment, all the rats were anesthetized by diethyl ether, 19 blood was collected by intra-cardiac puncture then allowed to clot for 10 min at room temperature and then centrifuged for 20 min at a speed of 3000 rpm to obtain serum for measuring of (serum levels of testosterone hormone, Testicular enzymes; lactate dehydrogenase (LDH), Alkaline phosphatase (ALP) and Total antioxidant capacity (TAC). Both testes were isolated and washed with cold phosphate buffer saline. The right testis was weighed and then fixed in formalin 10% to be processed for examination of histopathological changes by light microscope. The left testis was divided into two parts; one part of the left testis was homogenized in 5 ml Phosphate-buffered saline (PBS). The homogenates were centrifuged for 20 min at 3000 rpm at 4°C. 18 The supernatant was collected and frozen at −80 C till used for measurement of the following tissue parameters (TNF-α, BECN1, LC3 and mTOR). The other part of the left testis was homogenized in 5 ml potassium phosphate buffer. The homogenate was centrifuged for 20 min at 3000 rpm 4°C. 20 The supernatant was collected and frozen at −80 C until used to measure MDA.
Assessment of the weight of the right testis (gram)
After dissection, the right testis was washed in cold PBS, then dried on filter paper, and the weight of the testis was measured in grams.
Assessment of the Sperm count (million/ml)
Epididymis was isolated and washed with cold phosphate buffer saline after dissection. Epididymis was minced and immersed immediately with 5 ml of 0.9% saline in a Petri dish for 10-20 min to allow sperms to swim out into the saline. 21 A sample (20 microns) was taken from the seminal fluid by micropipette and placed under the coverslip of the Neubauer hemocytometer chamber, which allows sperms to settle down for 5 min and then examined under the light microscope for assessment of sperm count. Sperm count was performed in the five small squares of the red blood cell count. The equation used was:
Sperm count = (number of sperm counted x Dilution)/(number of squares x 4) in million/ml
Assessment of biochemical parameters
Spectrophotometric assay of serum testicular enzymes (LDH and ALP), TAC in serum and MDA level in tissue by kits obtained from Spectrum Diagnostic, Egyptian company for biotechnology (SAE) (Catalogue No. 283 002, AP 10 20, TA 25 13, MD 25 29) respectively.
Serum testosterone hormone level, tissue TNFα, BECN1, LC3, and M-TOR were determined by ELISA kits from Sun Red Biotechnology Company, Shanghai, China. (Catalogue No. 201-11-5126, 201-11-0765, 201-11-1689, 201-11-1701, 201-11-4241) respectively. All the procedures were conducted according to the manufacturer’s instructions.
Histopathological examination
After dissection, the right testis was immediately fixed in 10% formaldehyde. Paraffin sections were done (5 µm) from paraffin tissue blocks and stained with hematoxylin and eosin (H and E) and then slides were examined under the light microscope for histopathological changes. (i) Testicular biopsy score according to Johnsen’s score: Ten different preparations for each group were prepared with 5 μm thickness. One hundred tubules were evaluated for each group under the 20x objective lens according to the criteria specified below.
22
Table 1 (ii) The percent of apoptosis within the spermatogenic cells per 1000 cells (Apoptotic index %) was inspected to identify the apoptotic cells, and then the apoptotic index (AI) was then calculated as follows:
23
Histological scoring of testicular biopsies of studied groups at the end of the experiment (after 4 weeks).

Apoptotic Index (AI) = (Number of apoptotic cells per section/Total number of cells per section) × 100.
Statistical analysis
Statistical analysis was performed using (Graph Pad Prism version 5 for Windows 10, Graph Pad Software, Inc.). Shapiro-Wilk test for normality was performed. Quantitative parametric data were presented as mean ± SEM (standard error of the mean) and were analyzed by one-way ANOVA (F) test followed by the Tukey-Kramer multiple comparisons test. Quantitative non-parametric data were presented as median (maximum-minimum) and interquartile range (IQR). They were analyzed using the Kruskal-Wallis test with the Mann Test to compare each group. The significance was considered at values of p < 0.05.
Results
Effect of octreotide on body weight, testicular weight, and sperm count
Comparison of biochemical, histological, and apoptotic parameters across the studied groups at the end of the experiment (after 4 weeks).
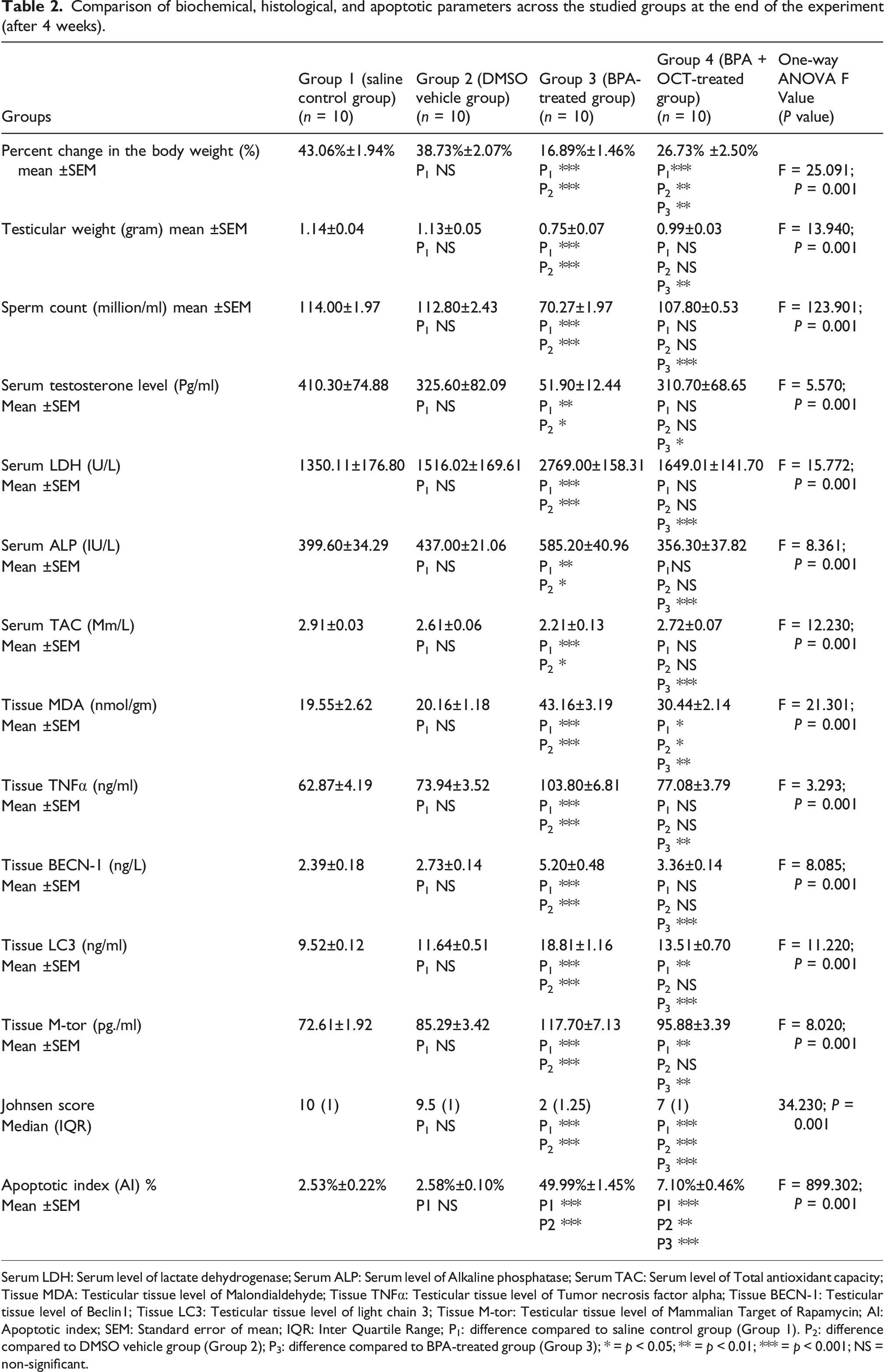
Serum LDH: Serum level of lactate dehydrogenase; Serum ALP: Serum level of Alkaline phosphatase; Serum TAC: Serum level of Total antioxidant capacity; Tissue MDA: Testicular tissue level of Malondialdehyde; Tissue TNFα: Testicular tissue level of Tumor necrosis factor alpha; Tissue BECN-1: Testicular tissue level of Beclin1; Tissue LC3: Testicular tissue level of light chain 3; Tissue M-tor: Testicular tissue level of Mammalian Target of Rapamycin; AI: Apoptotic index; SEM: Standard error of mean; IQR: Inter Quartile Range; P1: difference compared to saline control group (Group 1). P2: difference compared to DMSO vehicle group (Group 2); P3: difference compared to BPA-treated group (Group 3); * = p < 0.05; ** = p < 0.01; *** = p < 0.001; NS = non-significant.

Effect of Octreotide on: (a) Body weight, (b) testicular weight, (c) sperm count, (d) serum levels of testosterone, (e) serum levels of lactate dehydrogenase, (f) serum levels of alkaline phosphatase, (g) serum levels of total antioxidant capacity and (h) tissue levels of malondialdehyde in different studied groups. Serum levels of testosterone were measured by ELISA. Serum levels of lactate dehydrogenase, alkaline phosphatase, total antioxidant capacity and tissue levels of malondialdehyde were measured spectrophotometrically. Data were presented as mean ± SEM (n=10). Data were analyzed by one-way ANOVA followed by post-hoc Tukey’s multiple comparison test. Serum LDH: Serum level of lactate dehydrogenase; Serum ALP: Serum level of Alkaline phosphatase; Serum TAC: Serum level of Total antioxidant capacity; Tissue MDA: Testicular tissue level of Malondialdehyde; SEM: Standard error of mean; IQR: Inter Quartile Range; P1: difference compared to saline control group (Group 1); P2: difference compared to DMSO vehicle group (Group 2); P3: difference compared to BPA-treated group (Group 3); * = p < 0.05; ** = p < 0.01; *** = p < 0.001; NS = non-significant.
Effect of octreotide on serum testosterone levels
Administration of OCT showed a significant increase in serum testosterone as compared to group 3 and a non-significant difference as compared to group 1 and group 2. Figure 2(d).
Effect of octreotide on oxidative stress parameters (LDH, ALP, TAC and MDA)
Compared to the BPA-treated group (group 3), OCT administration resulted in a significant decrease in LDH, ALP, and MDA, along with a significant increase in TAC. There was no significant difference in LDH, ALP, and TAC when compared to Groups 1 and 2, while OCT administration led to a significant increase in MDA as compared to these groups. Figure 2(e)–(h).
Effect of octreotide on inflammatory biomarker (TNFα) and autophagic biomarkers (BECN-1, LC3 and M-TOR)
OCT administration resulted in a significant decrease in the levels of TNFα, BECN-1, LC3, and M-TOR as compared to Group 3, with no significant difference observed when compared to Groups 1 and 2. Figure 3(a)–(d). Effect of Octreotide on Tissue levels of Tissue levels of (a) tumor necrotic factor α, (b) BECN-1, (c) LC3, (d) M-tor, (e) histological score of testicular lesions according to Johnsen’s score and (f) percent of apoptosis within the spermatogenic cells per 1000 cells (Apoptotic index %) in different studied groups. TNFα, BECN-1, LC3, and M-tor were measured by ELISA. Data were presented as mean ± SEM (n=10). They were analyzed by one-way ANOVA followed by post-hoc Tukey’s multiple comparison test. Histological score of testicular lesions according to Johnsen's score was presented as median (maximum-minimum) and interquartile range (IQR). They were analyzed by Kruskal-Wallis test with the Mann Test to compare each group. Tissue TNFα: Testicular tissue level of Tumor necrosis factor alpha; Tissue BECN-1: Testicular tissue level of Beclin1; Tissue LC3: Testicular tissue level of light chain 3; Tissue M-tor: Testicular tissue level of Mammalian Target of Rapamycin; AI: Apoptotic index; SEM: Standard error of mean; IQR: Inter Quartile Range; P1: difference compared to saline control group (Group 1); P2: difference compared to DMSO vehicle group (Group 2); P3: difference compared to BPA-treated group (Group 3); * = p < 0.05; ** = p < 0.01; *** = p < 0.001; NS = non-significant.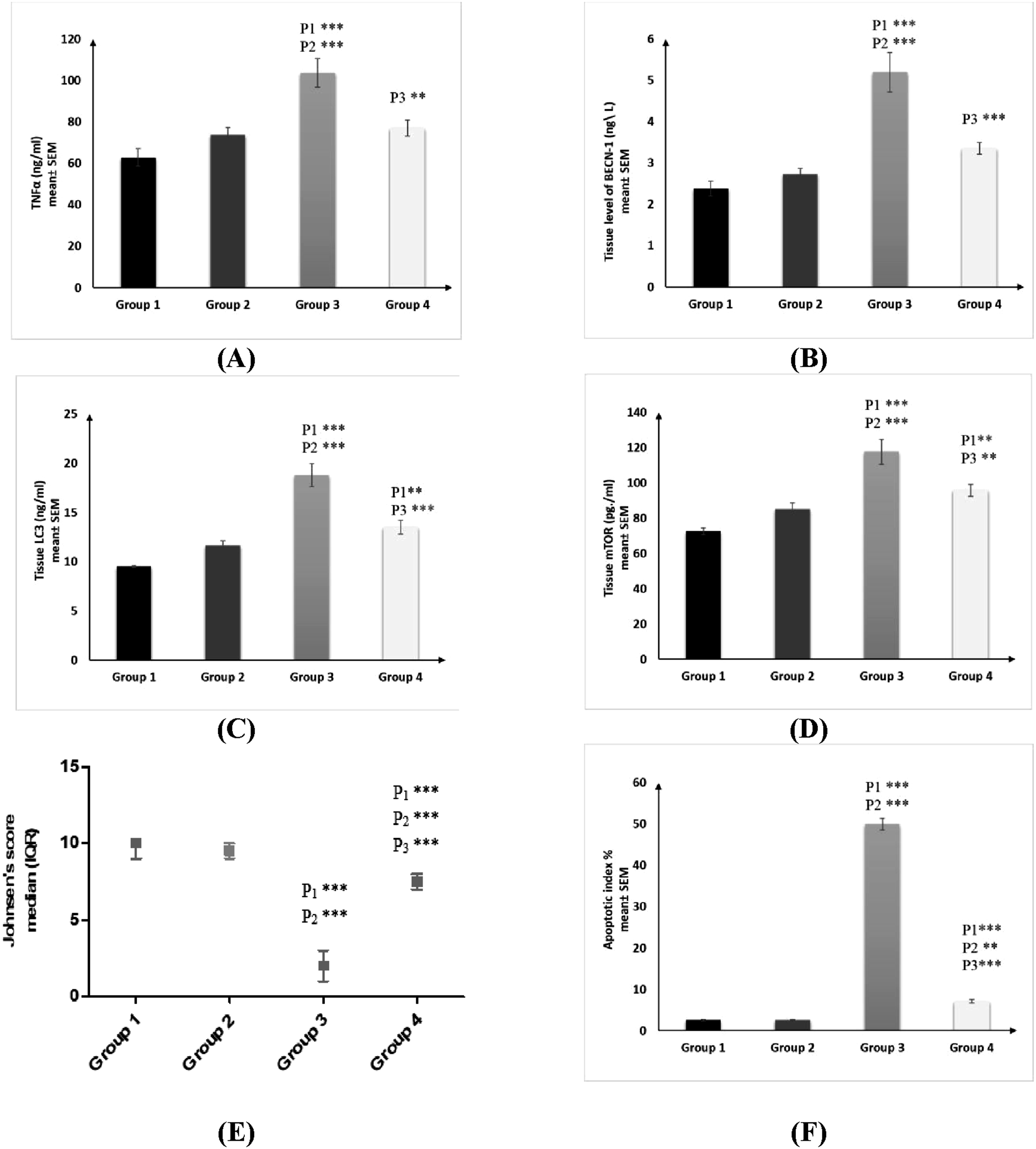
Effect of octreotide on testicular histopathological score and apoptotic index
There was a significant increase in the histopathological score of testicular lesions according to Johnsen’s score following OCT administration, as compared to Group 3, and a significant decrease as compared to Groups 1 and 2. In contrast, there was a reversal in the trend for the apoptotic index. Figure 3(e) and (f).
Effect of octreotide on histological findings
There was marked improvement in the degenerative changes that occurred in group 3 and increase in the sperm count within the lumen of seminiferous tubules. Figure 4. Histopathological examination of the testicular tissues stained with H&E (X 200), bar = 50 µm. (a) Group 1: showing normal seminiferous tubules lined with spermatogenic cell layers (white arrowheads) with the presence of numerous free sperms within their lumen (SP) and with normal Leydig’s cells (LC); (b) Group 2: showing normal seminiferous tubules lined with spermatogenic cell layers (white arrowheads) with numerous sperms within their lumen (SP) and with normal Leydig’s cells (LC); (c) and (d) Group 3: showing marked degenerative changes within the testicular tissues associated with severe atrophy of the seminiferous tubules with complete necrosis of the spermatogenic cells (white arrowheads) and metaplastic stratification of the lining epithelium (black arrowhead); (e) and (f) Group 4: showing a remarkable decrease in the degenerative changes within the spermatogenic cells (white arrowheads), with a significant increase in the sperm count within the lumen (SP) and mild interstitial edema (black arrowhead) and normal Leydig’s cells (LC).
Histopathological findings
Histopathological findings of group 1, group 2, group 3, and group 4 were enumerated in this figure (Figure 4).
Discussion
Testicular toxicity in rats commonly manifests as an impairment of spermatogenesis and is often accompanied by some atrophic change in the testis, Sertoli, and Leydig cells24.
Bisphenol A (BPA) and its substitutes are becoming the global emerging environmental pollutants 25 that have numerous adverse effects on fertility. 26 Also, in rodents, it can decrease testis weight, sperm count, and motility even with low doses. 27
OCT is a synthetic somatostatin octapeptide that primarily inhibits growth hormone secretion. 28 Recent studies have demonstrated that OCT exerts a cytoprotective effect through several mechanisms, including down-regulation of inflammatory mediators, suppression of endotoxin levels, and antioxidant effect. 29
In the present study, there was a significant decrease in body weight, testicular weight, and sperm count in group 3 as compared to group 1 and group 2, and this was in agreement with Sharma et al., 30 who reported that the decrease in body weight was due to the toxic effect of BPA. Also, Ehigiator et al. 31 reported that the reduction in body weight may be attributed to appetite suppression induced by BPA, inhibition of the hypothalamic-pituitary-gonadal (HPG) axis by BPA and they also reported the decrease in the sperm count was due to inhibition of androgen production, Sertoli cells function and impairment of steroidogenesis. This was in contrast with Tekin et al., 32 who reported that administration of BPA showed no significant difference in body and testicular weight between the experimental groups. This discrepancy may be due to differences in treatment duration—14 days versus 4 weeks in our study. The dose of BPA used in this study was 50 mg/kg/day, representing the lowest observed adverse effect level (LOAEL), the lowest tested dose of a substance reported to cause harmful health effects in humans or animals. 33 It was associated with testicular toxicity and impaired fertility, which was confirmed by a pilot study performed before our main experiment.
In the current study, BPA administration showed a significant decrease in serum testosterone levels as compared to group 1 and group 2. This result was in agreement with Hatipoglu et al., 34 Mendiola et al. 35 as well as Zirkin et al., 36 who reported that this decrease is due to BPA-estrogenic activity that inhibits testicular steroidogenesis, also interference with cholesterol metabolism which is the initial raw material for Leydig cells to synthesize testosterone.
OCT administration showed a significant increase in testosterone levels as compared to group 3. This effect exerted by OCT closely associated with the preservation of Leydig cell structure and function. Leydig cells are highly susceptible to oxidative stress and inflammatory damage, which impair steroidogenesis. 37 OCT exerts cyto-protective effects through its anti-oxidant and anti-inflammatory properties, primarily mediated via activation of somatostatin receptors (SSTRs).38,39 This preservation of cellular integrity maintains the expression and activity of key steroidogenic enzymes, thereby supporting testosterone biosynthesis.
This was in agreement with Hussain ALDulaimi. 40 who explained that this increase in testosterone level was due to the ability of OCT to preserve Leydig cell function and reduce oxidative stress.
In the present study, BPA administration showed a significant increase in serum levels of ALP and LDH as compared to group 1 and group 2, and this result was in agreement with Mahdavinia et al., 41 who reported that this decrease was due to inflammation and necrosis caused by BPA. Also, its ability to induce oxidative stress by generating superoxide anions that affect lipid peroxidation, damage cellular membranes, and increase the level of toxic metabolites. This result was contrary to Gao et al., 42 who reported that LDH levels decreased in all BPA-treated groups, indicating spermatogenesis dysfunction. LDH is a commonly used biomarker for spermatogenesis disorder. It has been found to exist in spermatogenesis and plays an active role in the energy supply through glucose metabolism. 43
OCT administration showed a significant decrease in serum ALP and LDH levels as compared to group 3, and this was in agreement with Aziz et al. 44 and Zigam et al., 38 who reported that this decrease was due to restoration of testicular function, anti-inflammatory and antioxidant effects of OCT.
Oxidative stress has been involved in the pathogenesis of testicular toxicity through overproduction of pro-inflammatory cytokines as TNF α, overproduction of free oxygen radicals and reduction of the activity of antioxidant enzymes (catalase, superoxide dismutase, glutathione peroxidase, glutathione reductase, and glutathione-S-transferase) also, increased ROS production interferes with the mitochondrial energy metabolism causing degeneration and necrosis.36,42,45
In the present study, oxidative stress was assessed by measuring the level of TAC and MDA. BPA administration showed a significant decrease in TAC as compared to group 1 and group 2 and a significant increase in MDA level as compared to both groups, and this was in agreement with Hatipoglu et al. 34
OCT administration showed a significant increase in TAC and a significant decrease in MDA levels as compared to group 3, and this was in agreement with Aziz et al. 44 and Unsal et al., 46 who reported that this effect was due to OCT anti-oxidant effect and its ability to protect testicular tissue from oxidative damage.
In the current study, BPA administration showed a significant increase in the TNFα level compared to group 1 and group 2, which was in agreement with the results of Khalifa et al., 47 Tekin et al., 32 and Hatipoglu et al. 34 who reported that BPA could cause immune system malfunction and increased pro-inflammatory cytokine levels (such as TNF-α), leading to mononuclear cell infiltration, edema, and hyperemia of testicular tissue. Also, the estrogenic activity can alter the expression of Nuclear Factor-КB (NF-КB), increasing cytokine secretion.
OCT administration showed a significant decrease in TNFα level as compared to group 3, and this was in agreement with the results of Tian et al. 48
LC3 is a key protein marker for autophagy, which is involved in the formation of autophagosomes, which are double-membrane vesicles that engulf cellular components destined for degradation. 49 Also, M-TOR and BECN1 are responsible for autophagosome biogenesis, maturation, and regulation of autophagy.50,51
In the present study, BPA administration showed a significant increase in LC3, M-TOR, and BECN1 compared to group 1 and group 2. BPA has been shown to induce oxidative stress, which can trigger autophagy as a protective mechanism by increasing LC3 levels. 52
Also, BPA triggers the recruitment and activation of Phosphoinositide 3-kinase (PI3K). This lipid kinase phosphorylates phosphoinositides to generate phosphatidylinositol-3,4,5-trisphosphate (PIP3). It inhibits the tuberous sclerosis complex (TSC1/TSC2), 53 which is a negative regulator of the mTOR pathway leading to the activation of the small GTPase, which in turn activates the mTOR complex 1 (mTORC1). 54
BECN1 forms a multiprotein complex with various interactors that collectively activate autophagosome formation, underscoring its vital role in autophagy induction. Furthermore, BECN1 forms a multiprotein complex with various interactors that activate the class III phosphatidylinositol 3-kinase (PI3KCIII). This complex interplay highlights the central role of Beclin-1 as a master regulator of autophagy. 55 These results were in agreement with the results of Khan et al., 56 Zhang et al. 57 and Park et al. 58
OCT administration showed a significant decrease in LC3, BECN1, and M-TOR levels compared to group 3. The observed reduction in LC3 and BECN1 suggests suppression of excessive autophagy. 59 In the context of BPA-induced testicular injury, heightened autophagy can become maladaptive, leading to degradation of essential cellular components. 51 Thus, OCT’s inhibitory effect may reflect a protective mechanism against autophagy-mediated cell damage.
OCT has an inhibitory effect on autophagy 60 as it binds to specific SSTRs, 61 particularly SSTR2 and SSTR5, which are expressed in testicular tissue. 62 This interaction activates phosphotyrosine phosphatases, which suppress receptor tyrosine kinase signaling, leading to reduced PI3K activity. 63 Consequently, the production of PIP3 is decreased, limiting AKT phosphorylation and downstream activation of mTOR.64,65 As mTOR is a key regulator of cell growth and autophagy, its inhibition disrupts autophagosome formation. 66 This cascade also lead to inhibition of the growth factor-mediated signaling pathway 67 resulting in suppression of Phosphoinositide 3-kinase/Activated protein kinase/Mammalian target of rapamycin (PI3K/AKT/M-TOR) pathway resulting in a reduction of phosphorylation of M-TOR and Beclin-1.68,69 This inhibition results in a decrease in LC3 level. 60
In the present study, BPA administration showed a significant increase in the apoptotic index compared to group 1 and group 2. The apoptotic index is quantitatively determined by counting apoptotic cells and apoptotic bodies, with the resultant percentage of apoptotic cells within a given cell population serving as a surrogate marker for the incidence of apoptosis.70,71 BPA exposure disrupts mitochondrial function, releasing pro-apoptotic factors and activating the intrinsic pathways and apoptosis regulators as B-cell leukemia/lymphoma 2 protein (BCL-2) family proteins. 72 It also upregulates p35 mitogen-activated protein kinase (P35-MAPK) and Tumor suppressor gene (P53) levels, which promote apoptosis. 32 Also, Gao et al. 42 reported that BPA can induce apoptosis due to its oxidative stress and inflammatory effects.
OCT administration showed a significant decrease in the apoptotic index compared to group 3. This is due to the activation of SSTRs that upregulate the expression of anti-apoptotic proteins 73 and the inhibition of the mitochondrial-mediated apoptosis pathway. 74
In the current study, BPA administration showed a significant decrease in the histological Johnsen’s score as compared to group 1 and group 2, which was in agreement with Hatipoglu et al. 34 This result was ameliorated by the administration of OCT, which supported its satisfactory results.
Conclusions
The current study supported the hypothesis that oxidative stress, inflammation, apoptosis, and autophagy are mechanisms believed to play a critical role in the pathogenesis of BPA-induced testicular toxicity. Our findings support the therapeutic potential of Octreotide in mitigating BPA-induced testicular damage, though further studies are needed to confirm these effects and elucidate clinical relevance.
This study has several limitations. First, the use of single-dose OCT administration may not accurately reflect the effects of repeated or long-term clinical use, potentially limiting the applicability of the results to real-world scenarios. Future studies should explore a range of doses to better understand therapeutic windows. Second, the sample size was limited, which may affect the statistical power and generalizability of the findings. Additionally, while rodent models provide important preclinical insights, they do not fully replicate the complexity of human physiology, particularly in the context of disease mechanisms and pharmacodynamics. Lastly, the study focused on short-term outcomes; long-term effects of OCT exposure were not assessed, and future research should investigate potential delayed or cumulative impacts.
Footnotes
Acknowledgment
Thanks to my great supervisors, Prof. Dr Fleur Fathi Abd El-Monem, Dr Rasha Osama El Esawy, and Dr Ola Mousa Salem, for their valuable guidance and kind supervision. Special thanks to Prof. Dr Wailed Abdo, Professor of Veterinary Pathology, Faculty of Veterinary Medicine, Kafr Elsheikh University, for helping me carry out the histopathological examination.
Ethical considerations
All animal experiments were approved by the Research Ethics Committee, Faculty of Medicine, Tanta University, Egypt (Approval no. 36239/12/22) and were conducted in accordance with ARRIVE 2.0 guidelines.
Author contributions
Funding
The authors received no financial support for this article’s research, authorship, and/or publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
