Abstract
Introduction
This study aimed to assess the acute toxic properties of drilling fluid (DF), a multicomponent substance extensively utilized in oil extraction, through a single intragastric administration in mature male rats, to better understand its potential health risks.
Methods
Acute toxicity was evaluated in mature male rats via a single intragastric administration of DF at doses of 300, 600, 1200, 2400, and 4800 mg/kg. Mortality, body weight changes, and relative organ weights were monitored throughout the study. Biochemical enzyme activities, including ALT, AST, ALP, and LDH, were assessed. Behavioral responses were recorded, and histological examinations of the liver, kidneys, and heart were conducted to evaluate tissue-level effects.
Results
Administration of DF caused significant toxic effects including mortality (2 deaths at 2400 mg/kg; 3 deaths at 4800 mg/kg), changes in body weight, and relative weights of liver (control: 7.12 ± 0.53; 2400 mg/kg: 7.89 ± 0.51; 4800 mg/kg: 9.68 ± 0.67; p < 0.001) and heart (control: 1.24 ± 0.17; 2400 mg/kg: 0.97 ± 0.21; 4800 mg/kg: 0.84 ± 0.08; p < 0.001). Activities of cytolytic enzymes ALT (control: 78.65 ± 4.28; 4800 mg/kg: 89.47 ± 2.62; p < 0.001) and AST (control: 226.18 ± 31.66; 4800 mg/kg: 322.73 ± 8.02; p < 0.001), as well as ALP and LDH, were significantly altered. Behavioral activity was markedly reduced. Histological changes were observed in the liver, kidneys, and heart tissues.
Discussion
These findings demonstrate that DF exhibits marked acute toxicity and causes significant physiological and histopathological damage in mammals, indicating a potential hazard.
Introduction
All stages of oil production technology – including drilling wells, oil extraction, and transportation of raw materials to processing plants – involve the use of numerous chemical reagents.1–3 Overall, the use of chemical reagents increases the efficiency of oil recovery by more than 15%. Drilling fluid (DF) performs several functions simultaneously. They are designed to clean the well, keep the cuttings in suspension, ensure wellbore stability, cool and lubricate the tool, lift the cuttings from the bottom to the surface, and facilitate effective penetration of productive formations.4,5 DF, as a rule, have a complex multicomponent composition which includes a wide variety of inorganic reagents and organic compounds. 6
Hydrocarbon extraction is associated with numerous adverse environmental effects. DF, drilling wastewater, waste drilling fluid (WDF), and drilling cuttings (DC) can pollute the soil, surface, and groundwater. 7 Moreover, populations residing near oil production regions exhibit significantly higher rates of allergic, cardiovascular, and pulmonary diseases, 8 alongside the emergence of new diseases with monofactorial and multifactorial etiologies. Children living near petrochemical facilities also experience significantly impaired health. 9
Occupational chemical exposure led to significant shifts in metabolic processes in workers in the oil production industry. Drillers and operators of underground well repair show lipid metabolism disorders, changes in the activity of cytolytic enzymes, activation of lipid peroxidation processes, and an increase in the level of medium-molecular peptides in the blood serum, confirming the presence of endogenous intoxication syndrome. 10 Workers involved in hydrocarbon extraction have been found to have significant changes in the hepatobiliary system. 11 Professional chemical exposure in the production environment, exacerbated by meteorological, physical, psycho-emotional factors, and insufficient personal and industrial hygiene, contributes to the development of occupational acneiform dermatitis, as well as to relapse and a more severe, progressive course of acne vulgaris. 12
DF employed in hydrocarbon extraction is among the most toxic multicomponent substances in oil production. The toxicity of DF is due to the presence of a wide variety of organic and inorganic components.13,14 As a result of contact with DF, both DC and drilling wastewater become toxic.15,16 Currently, the nature and consequences of pollution of natural environmental objects during well drilling have hardly been studied in practice. The available individual publications on the toxicological assessment of DF and DC are superficial and descriptive, distinguished by the incompleteness of research and the vagueness of developments, and often solve individual private issues. In addition, the depth of theoretical studies lags behind modern requirements dictated not only by the pace and volume of drilling operations in oil production, but also by the methodological requirements of preventive toxicology. Many chemical reagents used to prepare DF do not have complete toxicological characteristics and regulated values of maximum permissible concentrations (MPC) for certain environmental objects, including the air of the workplace.
This study was conducted in compliance with the fundamental principles of Laboratory Animal Science (LAS) and bioethical standards established by Directive 2010/63/EU, as well as the OECD Guidance on Establishing a National Chemicals Register (OECD Publishing, Paris, 2024, https://doi.org/10.1787/bebc508c-en), and in accordance with the principles of the “Three Rs”. The experimental protocol fully adhered to the rules of Good Laboratory Practice (GLP) (GOST 33044-2014, 2021) and followed the ARRIVE 2.0 guidelines to ensure transparency and reproducibility in animal research. Available literature on the toxicity assessment of DF has primarily focused on the test systems involving protozoa, fungi, helminths, and fish.17–20 However, the toxic effects of DF on mammals remain largely unexplored. There is a lack of data regarding the toxic and specific effects of DF, including dose-dependent responses. Conducting studies on adult male rats enables the acquisition of objective data concerning the potential hazards of DF and the underlying mechanisms of its toxic action.
Objective of this study: To investigate the general toxic properties of DF under acute exposure conditions in sexually mature male rats.
Material and methods
Statement on OECD guidelines, 3Rs principles, and animal welfare compliance
This study was conducted in compliance with the ARRIVE 2.0 guidelines, the principles of the “3Rs” (Replacement, Reduction, Refinement), and local ethical regulations (Protocol No. 3 (3/14), 15 March 2023, West Kazakhstan Marat Ospanov Medical University). The acute toxicity assessment followed the OECD Test Guideline No. 420 (Fixed Dose Procedure), which is designed to avoid the classical LD50 approach and does not use mortality as the primary endpoint.
In line with OECD 420, dose levels were selected based on prior LD50 estimates and preliminary exploratory studies to minimise unnecessary testing. Humane endpoint criteria were predefined and applied throughout, including early euthanasia for moribund animals or animals showing severe distress, thereby preventing death from toxicity as an intended endpoint. Observations focused on non-lethal biomarkers (e.g., food and water intake, behaviour, grooming activity, body weight dynamics, biochemical markers, and histology) to derive toxicological information while limiting reliance on lethality data.
The study used six animals per dose group rather than the minimum OECD 420 recommendation to account for increased variability from using outbred rats obtained from multiple sources, and to ensure sufficient tissue samples for detailed histopathological evaluation. This deviation from the minimum was considered necessary to achieve the objectives with scientific validity, but no more animals were used than required. Adaptive monitoring was in place so that no further dosing or additional animals were introduced once the predetermined highest dose level was reached.
Currently, there are no fully validated in vitro or in silico models capable of reproducing the complex systemic and histopathological responses to oil-based drilling fluid mixtures in mammals. An in vivo approach was therefore required. Nevertheless, all possible measures were implemented to reduce animal numbers, refine procedures, and enhance welfare while meeting the scientific goals of the study.
Animals
The general toxic effects of DF were assessed in mature outbred male rats weighing 180–220 g. Animals were randomly assigned to groups, stratified by body weight to minimize variability. The variation in body weight within groups did not exceed 10%. To minimize selection bias, a complete blinding procedure was implemented for researchers and staff. This approach, which included proper randomization and concealment of animal group assignments, allowed differences between groups to be attributed only to random error. Six groups of six animals each were formed. The control group (group 1) consisted of intact animals that were not subjected to any manipulations. Five experimental groups were formed, making a total of 36 male rats. The animals in the experimental groups were given a single intragastric administration of the DF in an acute experiment: group 2 received a single administration of the DF at a dose of 300 mg/kg; group 3 – 600 mg/kg; group 4 – 1200 mg/kg; group 5 – 2400 mg/kg; group 6 – 4800 mg/kg. The dose selection was based on previously conducted studies investigating median lethal doses (LD50), which were found to be relatively high, 21 as confirmed by probit analysis and calculation methods developed by Behrens, Kerber, and Pershin. Based on these data and preliminary exploratory experiments, the specified doses of DF were selected. DF was administered once via intragastric gavage in its original, undiluted form. The administered dose volume was individually calculated based on each animal’s body weight.
Samples of the DF were collected from the Zhanazhol oil field, located in Western Kazakhstan. Under a contractual agreement with the Aktobe Regional Center for Sanitary and Epidemiological Expertise and LLP “Geoplazma”, a chemical analysis of the DF was performed. The analysis confirmed the presence of lead and arsenic, as well as bentonite clay, lime, ferrochrome lignosulfonate, carboxymethyl cellulose, caustic soda, and barium sulfate. Analysis also revealed that the drilling sludge contained phosphorus oxide, silicon dioxide, calcium oxide, sodium oxide, potassium oxide, and bitumoids. The DF used in the experiment was oil-based.
Ethical approval was obtained from the Local Bioethics Committee of the NJSC “West Kazakhstan Marat Ospanov Medical University” on March 15, 2023 (Protocol No. 3 (3/14)).
Animal welfare conditions
Experimental animals were housed under vivarium conditions with free access to food, water, and natural light. The ambient temperature was maintained at 20 to 22°C, in full compliance with sanitary and international standards. Animals were fasted for 12 h prior to DF administration. After the administration of the DF, the general condition of the control and experimental animals was continuously monitored. Signs of toxicity, the number and time of death of animals during the entire observation period, as well as the consumption of food and water were assessed. Observations were performed at multiple time points: for 6 h on the first day, after 24 h and on the following days of the experiment. The duration of observation of the animals was 14 days. During the experiment, not only was the general condition of the animals assessed, but also the condition of the fur and skin, as well as the intensity and nature of spontaneous motor activity. In the experimental animals, motor activity indicators (duration and number of vertical stands) and the emotional component of behavior (number of grooming episodes) were recorded over a 14-day period. Body weight was recorded in all groups before the administration of the substance, weekly during the entire observation period, and expressed in grams. Body weight gain was also determined.
At the end of the experiment, the animals were euthanized in accordance with ethical principles outlined by FELASA, ICLAS, and the AVMA Guidelines for the Euthanasia of Animals (2020, USA). In order to minimize distress, the animals were administered intramuscular xylazine (Xylanit) at a dose of 4 mg per 100 g of body weight, followed by decapitation. After euthanasia, biological samples were collected, body weight and relative organ weights were measured. The following organs were harvested for histomorphological examination: heart, stomach, liver, kidneys, lungs, and spleen.
Biochemical analysis and clinical blood analysis
The activity of the enzymes was determined in the blood, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), C-reactive protein (CRP), gamma-glutamyl transpeptidase (GGTP), using the automatic modular analyzer “Cobas 6000” immunochemical module C 501-1. Biochemical parameters of rat blood serum measured in this study (in particular, the activity of the enzymes ALT, AST, ALP, GGTP, LDH) are presented in the following units of measurement: - U/L (CRP - mg/L).
Reference intervals for these enzymes vary considerably across numerous scientific articles and reference manuals. This trend also applies to other biochemical parameters of peripheral blood. Thus, in the reference manual by B.G. Yushkov et al., the ALT activity of serum in male rats was 102.5 U/L, in female rats – 28.9 U/L; AST activity – in male rats 386.6 U/L, in females – 80.4 U/L; alkaline phosphatase activity in male rats was 290 U/L, in females – 201 U/L (in Wistar rats – 395 U/L). 17 In the scientific article N.G.Voitenko with co-author, the reference values of ALT activity were for male rats – 80–1001 U/L, females – 77–909 U/L; AST – males – 64–940 U/L, females – 45–473 U/L; alkaline phosphatase in male rats – 254–484 U/L, females – 187–334 U/L. 18 Additionally, in the work of V.I.Inchin with co-author, the ALT activity in Wistar rats was 339.8 U/L, AST – 64.8 U/L. 19
Determination of red blood parameters — hemoglobin, erythrocytes, color index, hematocrit, platelets, erythrocyte sedimentation rate (ESR); and white blood parameters — leukocytes, leukocyte profiles, monocytes, lymphocytes, were carried out using the automatic hematology analyzer Sysmex XN550 (II).
Histological examination
Microscopic examination of all representative tissue sections was performed in the morphological laboratory of the histology department of the West Kazakhstan Marat Ospanov Medical University NPJSC. The material was fixed in a 10% solution of buffered formalin. Histological processing of the material was carried out according to the instructions for standard operating procedures of the morphological laboratory. Histological sections 4–5 μm thick were prepared from paraffin blocks using a rotary microtome “Accu-Cut SRV 200” (Sakura Finetek, USA). The sections were stained with hematoxylin and eosin (HE) both manually and automatically using the histological stainer BIO-OPTICA-AUS 240 (Bio-Optica, Milan). Microscopy of histological preparations was performed using a light microscope at magnifications of 50×, 100×, 400×, and 1000×. Histological preparations were examined by using a digital light microscope AxioLab A1 (registration certificate RK-MT-7 No. 009046, country of origin: Germany, state registration date: 17.08.2018). Photography of the preparations was performed with a digital camera, the AxioCam ERc5s. To eliminate subjective bias in the interpretation of results, all histological sections were coded to conceal group identity (investigator blinding).
Statistical analysis and funding information
The results of the studies were analyzed using IBM SPSS Statistics Version 25. For the analysis of qualitative variables, the Pearson Chi-square test (χ2) was used. To compare data measured at different time points, repeated measures analysis of variance (repeated measures ANOVA) was performed. Quantitative data were presented as mean (M) ± standard deviation (SD). A p-value of ≤0.05 was considered statistically significant.
The research materials were obtained within the framework of the scientific project IRN AR19676915, titled “Development of Toxicometric Criteria for the Hazard and Toxicity Assessment of Drilling Fluid and Cuttings.” This scientific grant is funded by the Committee of Science of the Ministry of Science and Higher Education of the Republic of Kazakhstan.
Results
General signs of intoxication
General toxic effects of DF following a single intragastric administration were assessed based on external signs of acute intoxication, eating behavior, body weight dynamics, behavioral reactions, as well as the mortality of experimental animals. Signs of intoxication, such as stiffness, tremor of the front and hind limbs, impaired coordination of movement, and rapid breathing, were recorded in experimental rats receiving DF in doses from 1200 mg/kg to 4800 mg/kg. By the end of the first day of observations, the animals moved little, reacted weakly to external stimuli, and assumed a lateral position. Behavioral parameters, including the duration and number of standing episodes, as well as the number and duration of grooming behaviors, also tended to decrease under conditions of a single intragastric administration of large doses of DF. On day 14, the experimental animals exhibited changes in coat condition, characterized by loss of shine and neatness, resulting in a dull appearance.
In almost all experimental groups, a decrease in feed and water consumption was observed, with the most pronounced changes occurring in groups receiving high doses of DF. On the 1st day of the experiment the amount of feed consumed per day in the experimental groups of animals receiving the DF at a dose of 2400 mg/kg and 4800 mg/kg was 17.7 ± 0.7 g/kg (CI 15.4–20.1) and 15.7 ± 0.5 g/kg (CI 14.2–17.3) per day, respectively, by on the 7th day of the experiment in these experimental groups the feed consumption was 18.2 ± 1.1 g/kg (CI 14.9-21.5) and 16.0 ± 13.1 g/kg (CI 11.7-20.3) per day, respectively, and on the 14th day of the experiment 15.2 ± 0.5 g/kg (CI 13.7–16.8) and 14.7 ± 0.7 g/kg (CI 12.4–17.1), respectively. Changes in feed intake were less pronounced in the experimental groups where the animals were given a single intragastric administration of DF at doses of 300 and 600 mg/kg. Changes in water consumption were also observed: in the experimental groups where the animals received large doses of DF (2400 mg/kg and 4800 mg/kg), daily water intake significantly decreased on the 5th and 7th days of the experiment, followed by a subsequent increase.
Body weight, organ ratios, and mortality outcomes
The pattern of body weight changes in experimental animals following a single intragastric administration indicates the adverse effects of DF. Notably, by day 7 of the experiment, a significant reduction in body weight in the test animals was observed. The most significant changes were identified in the groups where animals received the DF at doses of 1200 mg/kg, 2400 mg/kg, and 4800 mg/kg. By day 14 of the experiment, a trend toward an increase in animal body weight was observed (Figure 1). Changes in body weight dynamics.
Pathophysiological mechanisms regulating eating behavior, metabolic processes, as well as changes in digestive function under conditions of acute intoxication, play an important role in body weight reduction in experimental animals of experimental animals.20,21 Reduced food intake and weight loss under conditions of acute single exposure to DF may be associated with the aforementioned mechanisms and malabsorption syndrome. Chemical effects on the digestive tract may manifest themselves in inflammatory processes in the intestine, a decrease in the synthesis and excretion of bile acids in the liver, which are necessary for the digestion and absorption of lipids, carbohydrates, and proteins.22,23 This may underline reduced feeding and weight loss. The extent, duration, and dynamics of the observed changes in the body weight of experimental animals and the body mass ratio of internal organs in experiments on acute intoxication with certain chemical substances largely depend on the effective dose, the method of its entry into the body, and the duration of the experiment.24,25
In this study, a clear dose-dependent mortality pattern was observed. A single administration of DF at 2400 mg/kg resulted in the death of two rats, occurring on days 3 and 7, respectively. Administration of DF at a dose of 4800 mg/kg resulted in the death of two rats on day 2 and one rat on day 5 of the experiment. No mortality was observed in animals receiving single intragastric doses of 300, 600, or 1200 mg/kg DF.
Changes in organ-to-body weight ratios of internal organs.

Note: *Statistically significant differences between groups were determined using general linear models (GLM); p < 0.05.
Biochemical blood parameters
Changes in enzyme activity in the blood.

Note: ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; CRP: C-reactive protein; GGTP: gamma-glutamyl transpeptidase; LDH: lactate dehydrogenase.
*Statistically significant differences between groups were determined using general linear models (GLM); p < 0.05.
Peripheral blood is known to be a highly functional and morphoplastic system that actively responds to the effects of harmful chemical agents. Analysis of the toxic effect of DF under conditions of single acute intragastric exposure revealed several minor changes in white blood cell counts. The total lymphocyte count increased in experimental groups compared to controls (64.33 ± 1.52, control; experimental groups: 300 mg/kg 66.10 ± 0.95; 600 mg/kg 67.55 ± 1.99 (p = 0.007); 1200 mg/kg 70.37 ± 2.92 (p = 0.023); 2400 mg/kg 69.60 ± 3.89 (p < 0.001); 4800 mg/kg 75.20 ± 3.61 (p < 0.001)). Eosinophil counts were also elevated in experimental groups compared to the control (3.68 ± 0.39, control; experimental groups: 300 mg/kg 4.00 ± 0.54 (p = 0.003); 600 mg/kg 3.85 ± 0.48 (p = 0.002); 1200 mg/kg 3.85 ± 0.48 (p = 0.002); 2400 mg/kg 3.98 ± 0.54 (p < 0.001); 4800 mg/kg 4.47 ± 0.43 (p < 0.001)). In the experimental groups, no statistically significant changes were found in other peripheral blood parameters (hemoglobin, erythrocytes, color index, hematocrit, mean corpuscular volume, platelets, monocytes, ESR) compared to the control.
The analysis of enzyme activity and peripheral blood composition demonstrated that blood, as a highly functional system, responds rapidly to the effects of DF. A highly specific marker of the damaging effects of DF is the determination of the activity of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, lactate dehydrogenase, and gamma-glutamyl transpeptidase. An increase in lymphocyte and eosinophil counts is also a significant indicator in assessing the general toxic effects of DF. Our findings largely correlate with published data, which show that the activity of blood enzymes ALT, AST, LDH, ALP, and GGTP is an important indicator of the toxic effect of chemicals during acute and subchronic exposure.26–28
Histological changes in the organs
When examining histological preparations, the histological appearance of the control group rats was normal. The histological structure of the cardiac tissue of rats that received DF at a dose of 300 mg/kg was similar to that of the control group, except for mild congestion of venules and arterioles. In the experimental groups where the animals received DF at doses of 600 and 1200 mg/kg, moderate interstitial edema and congestion of the myocardial vessels were observed, and at the highest doses (2400 and 4800 mg/kg) of the DF, the most severe circulatory disorders and pathological changes were observed (Figure 2(a)–(f)), (Figure 3). Micrographs of rat heart tissue on day 14 after a single administration of DF. (a) normal histological structure of the heart tissue of control rats; (b) group receiving DF (300 mg/kg), slight vascular engorgement (1); (c) and (d) groups receiving DF (600; 1200 mg/kg), moderate vascular distention of the endomysium (2), moderate interstitial edema (3); (e) and (f) groups receiving DF (2400; 4800 mg/kg), moderate vascular congestion of the endomysium (4), interstitial and perivascular edema (5), severe congestion (6). Hematoxylin and Eosin (H&E) staining. Magnification is 100x and scale bar = 100 µm for a, b, c, d, e, f.
Following a single intragastric administration of the DF (Figure 3), no pronounced microscopic changes were observed in the glandular and non-glandular parts of the stomach wall, except for minor (at doses of 300 mg/kg; 600 mg/kg) and moderate (at doses of 1200 mg/kg; 2400 mg/kg; 4800 mg/kg) vascular congestion, and as the concentration of the DF increased in the compared experimental groups, moderate edema and dystrophic changes in the cells of some gastric glands and moderate desquamation of the simple columnar epithelium were histologically noted in the stomach tissue. Figure 4 presents the pathological changes in the stomach of experimental animals following a single intragastric administration of DF. The most significant histological changes and circulatory disorders were revealed in the groups where the animals received the DF at doses of 1200 mg/kg, 2400 mg/kg, and 4800 mg/kg. Morphological indicators of the heart in animals on day 14 following a single administration of DF. Morphological indicators of the stomach in animals on day 14 following a single administration of DF.

Pathological changes in the liver observed in rats acutely exposed to DF at doses of 2400 and 4800 mg/kg ranged from vacuolar degeneration to necrobiotic changes in isolated hepatocytes. Such liver changes predominated in the central lobular areas (Figure 5(a)–(f)). All identified morphological indicators of liver damage – including circulatory disorders, inflammatory infiltrates, and other pathological changes – were significant and exhibited clear dose dependence (Figure 6). Micrographs of rat liver tissue on day 14 following a single administration of DF. (a) normal histological structure of liver tissue from control rats; (b) group receiving DF (300 mg/kg), slight vascular engorgement (1); (c) and (d) groups receiving DF (600; 1200 mg/kg), distention and moderate congestion of sinusoidal capillaries (2) and central vein (3); (e) and (f) groups receiving DF (2400; 4800 mg/kg), pronounced vascular congestion (4), focal inflammatory infiltrates (5), congestion and dilation of the central vein (6), focal necrosis of hepatocytes (7). Hematoxylin and Eosin (H&E) staining. Magnification is 100x and scale bar = 100 µm for a, b, c, d, e, f. Morphological indicators of the liver in animals on day 14 following a single administration of DF.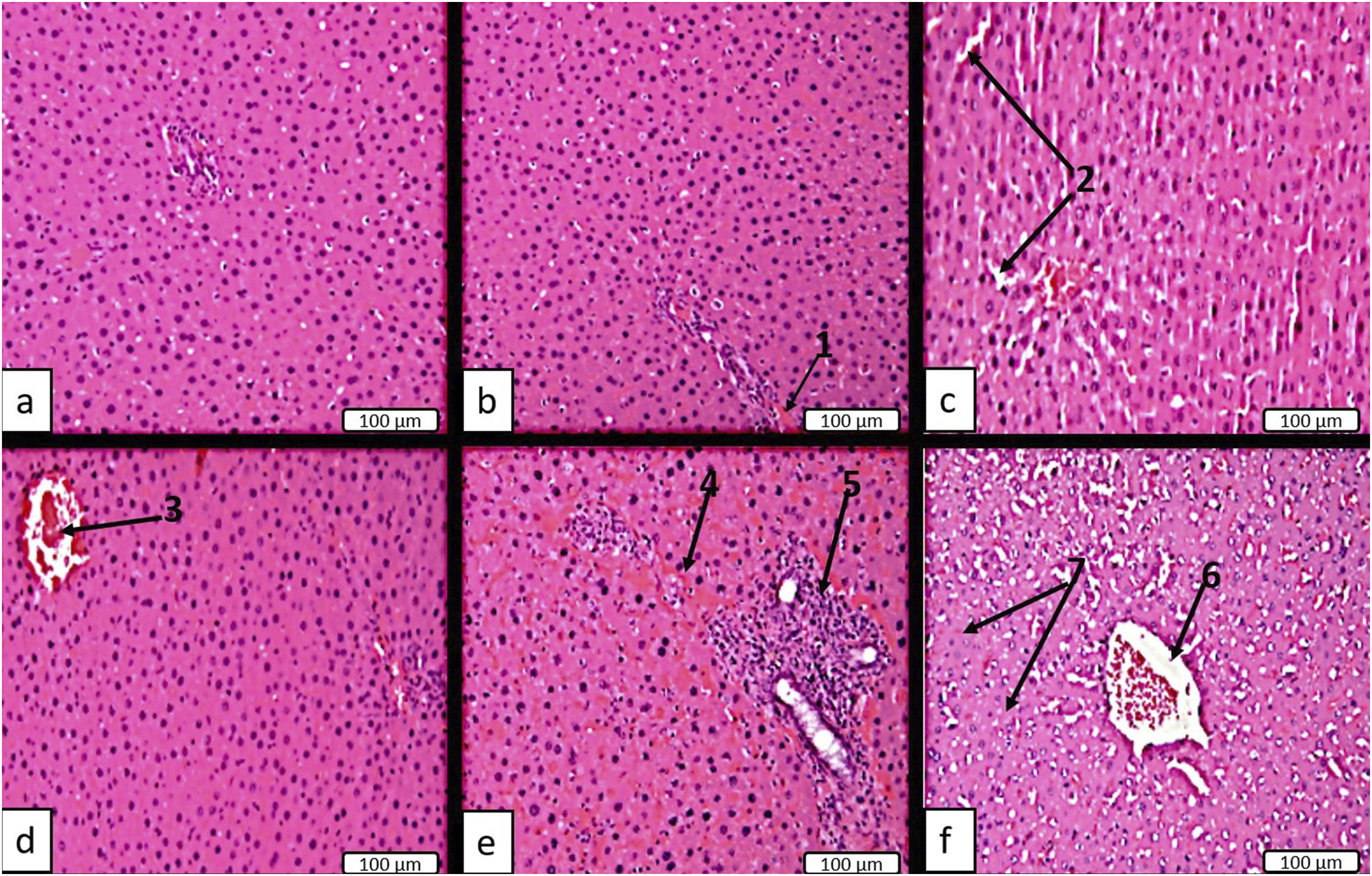

Histological staining of kidney tissues from control rats revealed normal structures, including glomeruli, tubules, interstitium, and blood vessels. Following a single intragastric administration of DF at doses of 300, 600, and 1200 mg/kg, the glomeruli and tubules were unchanged. In contrast, rats in the group receiving DF at doses of 2400 and 4800 mg/kg showed several histopathological changes in the kidneys compared with those in the control group. Тhese findings indicate that the severity of microscopic kidney alterations is primarily dose-dependent (Figure 7(a)–(f)). Micrographs of rat kidney tissue on day 14 following a single administration of DF. (a) normal histological structure of the kidney of control rats; (b) group receiving DF (300 mg/kg); (c) and (d) groups receiving DF (600; 1200 mg/kg), dilation and moderate vascular engorgement (1); (e) and (f) groups receiving DF (2400; 4800 mg/kg), dilation and congestion of vessels (2), expansion of the lumen of the glomerular capsule (3). Hematoxylin and Eosin (H&E) staining. Magnification is 100x and scale bar = 100 µm for a, b, c, d, e, f.
Histological features of the lungs in rats administered DF at doses of 300 and 600 mg/kg were similar to those of controls, except for minor congestion of the vessels of the interalveolar septum. The most significant pathological and inflammatory changes, along with circulatory disorders, were observed at the maximum dose (2400 and 4800 mg/kg) of the DF, in the form of thickening of the interalveolar septa due to pronounced interstitial edema, focal hyperplasia of lymphoid elements, and vascular congestion.
The histological structure of the spleen of rats administered DF at doses of 300 and 600 mg/kg was similar to that of the control group. In several tissue sections, moderate edema along the trabeculae and slight congestion of the trabecular vessels were observed in the sinusoids of the red pulp. The frequency of significant vascular changes was high in rats given DF at doses of 2400 and 4800 mg/kg. In the periarteriolar focal lymphocyte depletion was observed in the periarteriolar lymphoid sheaths (PALS), also known as T-lymphocyte zones, and the germinal centers of the lymph nodes were normal. Moderate dilation of the central arterioles was also observed in several tissue sections.
Histopathological changes were examined in the heart, stomach, liver, kidneys, lungs, and spleen of rats following a single intragastric administration of DF. Pathomorphological reversible changes were manifested in the form of circulatory disorders, from slight to severe vascular congestion, and focal hemorrhage. These were accompanied by moderate edema, focal dystrophy, vacuolar degeneration, and necrobiotic changes in some cells, as well as moderate focal hyperplasia of lymphoid elements in the internal organs. The severity of significant histopathological changes shown in this study increased with increasing concentration of the DF, which may be associated with a direct toxic effect of the DF components on tissues.
Discussion
Given the multicomponent composition of DF, as well as changes in its chemical makeup depending on the technological process and the degree of use, its toxicity and hazard can only be reliably determined through experimental methods. Various aquatic organisms are typically used as test organisms. However, since no single species serves as a universal indicator, bioindicators should be carried out using several test organisms at different levels of biological organization, including hydrobionts, higher plants, microorganisms, and mammals.
In this study, we examined the general toxic effects of DF following a single intragastric administration in sexually mature male white rats. The evaluation of the animals’ general condition, changes in body weight dynamics, internal organ coefficients, and patterns of food and water consumption, as well as the analysis of the severity of toxic manifestations – based on lethal dose data and changes in behavioral responses – indicated a dose-dependent relationship and confirmed that DF exhibits a certain degree of toxicity and hazard.
These findings were supported by observed changes in peripheral blood parameters, serum enzyme activities, and morphological alterations in organs and tissues.
The principles and methodological approaches we used in the experimental design – concerning the standardization of animals by genetic background, health status, environmental consistency and control, as well as body weight, age, and sex – allowed for substantial adherence to the “3R” principles. Additionally, the establishment of inclusion and exclusion criteria, the use of randomization and blinding, and the presentation of data on animal numbers throughout the experiment improved both the internal and external validity of the experimental research.
The present study lays the foundation for further investigations into the toxicity of DF under subchronic and chronic exposure conditions. In such studies, which explore the general toxic and specific effects of oil production waste, the conditions and opportunities arise for applying a broad spectrum of alternative methods, such as in vitro techniques.
In studies of environmental impact during hydrocarbon raw material production, DC are most often analyzed, but there is limited information available regarding the toxicity and hazard of DF. The biological impact of DF depends on its components. At the same time, the reagents used in DF are very diverse, and toxicological information is limited and largely classified by manufacturers. DF are classified by chemical composition into polymeric, inhibitory, salt-saturated water-based, and oil-based types. The higher level of toxic action of hydrocarbon-based DF is due to the toxicity of their dispersion media (oil, diesel fuel) and components of the dispersion phase (surfactants, emulsifiers, flocculants, thickeners, weighting agents, chlorides), as well as the combined action of the reagents included in their composition.13,14,16 The presence of polycyclic and heterocyclic aromatic hydrocarbons in DF may exert a synergistic toxic effect. 29
Available30–34 literature data on the content of heavy metals in DF,35,36 as well as the results of our own research on the chemical composition of DF, demonstrate toxic effects attributable to heavy metals. The effects and consequences of heavy metal exposure have been extensively studied and are associated with the development of cytotoxicity, oxidative stress, immunotoxicity, hepatotoxicity, nephrotoxicity, and neurotoxicity. 37 In the processes of metal absorption and elimination, distribution, metabolism, and excretion play a key role, as they can directly influence the manifestation of their toxicodynamic properties and, consequently, the outcomes of our study.
The observed discrepancy between biochemical and histological findings may reflect differences in the sensitivity of these two approaches. Cytolytic enzymes such as ALT, AST, LDH, GGTP, and ALP are released into the bloodstream during the early stages of cell injury. However, their serum levels do not always correspond to histological alterations, particularly when structural damage remains below the threshold of microscopic detection. In the acute model of DF exposure we developed, their blood exhibited dynamic changes. At early stages, tissue damage may be compensated by physiological mechanisms, leading to reduced enzyme release. In cases of moderate liver or kidney injury, cellular regeneration may also contribute to changes in enzyme activity.38,39
Histological changes such as edema, vascular disturbances, and inflammation are not always associated with cellular necrosis, which causes enzyme release. In the case of interstitial edema or vascular congestion, the damage may be reversible, not leading to a massive enzyme release. Differences in methodological sensitivity, stages of tissue damage, and physiological compensatory mechanisms may explain the discrepancies observed between histological and biochemical findings in our study.40–42
Given the presence of multiple heavy metals in DF, it is essential to consider the combined effects of these chemical elements and the associated biological responses. Such combined effects may play a fundamental role in the toxicokinetics and toxicodynamics of DF in acute chemical exposure experiments. The multicomponent nature of DF can modify its overall toxicity. Studies on combined metal toxicity have shown that mixture effects are difficult to predict. Both synergistic and antagonistic interactions have been observed, 43 leading to contradictory findings across studies, 44 which also depend on metal concentrations. 45 The evaluation of diagnostic markers of early metabolic disorders in oilfield workers10,46 shows, in some cases, there is a mismatch between protein, carbohydrate, and lipid metabolism indicators and cytolytic enzyme activity, as well as parameters of lipid peroxidation activation and the number of medium-molecular-weight peptides. These parameters do not consistently correlate with the severity of endogenous intoxication syndrome. The assessment of the risk of human exposure to radionuclides and heavy metals in samples of oil-based DF shows that the effects may pose not only short-term but also long-term hazards. 47
Conclusion
The observed acute toxicity of DF induces dose-dependent histopathological effects in rats, reflecting its overall toxic impact. The obtained data form a foundation for further investigations into subchronic and chronic toxicity, highlighting potential risks to occupational safety and environmental health. This underscores the urgent need for improved regulation, protective measures, and enhanced safety protocols.
The toxicological findings presented in this article provide valuable insights for researchers and professionals in the oil industry. These findings will aid in developing necessary safety measures to ensure optimal working conditions and protect environmental health. Furthermore, this information will be crucial for predicting medical and ecological outcomes in oil production regions and for implementing comprehensive health protection strategies for the workforce.
Footnotes
ORCID iDs
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Local Commission on Bioethics of the West Kazakhstan Medical University (meeting No. 3 (3/14) dated 15 March 2023).
Author contributions
Conceptualization, A.M., K.B., Zh.K., A.K., S.B., S.S., K.Sh. and G.K.; Methodology, A.M., Zh.K., S.B. and S.S.; Formal Analysis, A.M. and Zh.K.; Investigation, K.B., A.K. and G.K.; Data Curation, K.B. and G.K.; Writing – Original Draft Preparation, A.M., Zh.K. and S.B.; Writing – Review and Editing, A.M., K.B., Zh.K., S.B. and S.S.; Supervision, A.M.; Project Administration, A.M.; Funding Acquisition, A.M. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. AP19676915).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available from the corresponding author upon request.
