Abstract
Objective
The study aimed to explore the inhibitory effect of stevioside on colorectal cancer and its molecular mechanism.
Methods
Colorectal cancer cells were selected for functional testing, including the following groups: 0 μM stevioside, 1 μM stevioside, 2.5 μM stevioside and 5 μM stevioside. CCK-8 kit and EdU staining were employed to assess the cell viability. Cell apoptosis was deterred by flow cytometry. Western blot assay was utilized to detect the protein expressions of cleaved-caspase-3, Bax, Bcl-2, E-cadherin and Vimentin. The polarization of macrophage was evaluated through flow cytometry, western blot and immunofluorescence staining. The effect of stevioside on the proliferation of tumor tissue was detected by tumor formation and immunohistochemical staining in nude mice.
Results
Stevioside exhibited a significant concentration-dependent inhibitory effect on the proliferation, migration, and invasion of colorectal cancer cells, while promoting apoptosis
Conclusion
Stevioside possesses the ability to significantly hinder the proliferation of colorectal cancer cells and induce apoptosis, the mechanism of which may be closely related to the regulation of macrophage M1 polarization.
Introduction
Colorectal cancer (CRC) ranks as the third most prevalent malignant cancer and the fourth leading cause of mortality globally. 1 It is reported 2 that approximately 1,600,000 patients worldwide succumbed to colorectal cancer in 2040. Genetic predisposition, environmental pollution, and unbalanced dietary habits are among the key influencing factors of colorectal cancer. 3 Despite significant advancements in targeted therapy and immunotherapy for colorectal cancer, the 5-year survival rate remains unsatisfactory. 4 Therefore, it is imperative to delve deeper into the pathological mechanisms of colorectal cancer and pursue the development of more effective treatment strategies.
Due to the undesirable side effects and limited effectiveness of chemotherapy drugs, traditional Chinese medicine (TCM) has garnered significant attention as a novel anti-tumor approach, renowned for its therapeutic efficacy and unique advantages.5,6 Stevioside, a diterpene glycoside extracted from the stevia species, possesses a wide array of pharmacological benefits, encompassing the regulation of blood glucose levels, 7 blood pressure, 8 anti-inflammatory properties, 9 anti-fibrotic effects, as well as anti-cancer potential. 10 Stevioside significantly alleviates osteoarthritis progression by suppressing key pro-inflammatory cytokines (IL-6 and IL-1β) and attenuating adjuvant arthritis. 11 It has been revealed 12 that stevioside could induce apoptosis in breast cancer MCF-7 cells via ROS-mediated mitochondrial pathways. Chen et al. reported 13 that stevioside exerts inhibitory effects on bladder cancer cell proliferation by activating the GSK-3β and ER stress signaling pathways. Stevioside’s capacity to reprogram the immunosuppressive tumor microenvironment (TME) in cancer underscores its therapeutic potential as a multi-target immunomodulator. The role of stevioside in colorectal cancer and its molecular mechanisms is rarely reported.
Inhibition of proliferation and induction of apoptosis constitute the pivotal molecular mechanisms that drugs employ to eradicate cancer cells. 14 Our study aimed to explore the regulatory impact of stevioside on the proliferation and apoptosis of colorectal cancer. Both in vitro and in vivo experiments have conclusively demonstrated that stevioside exerts a notable inhibitory effect on the proliferation of colorectal cancer cells, while simultaneously promoting their apoptosis. Furthermore, stevioside-induced polarization of macrophage M1 may be one of the molecular mechanisms that play a protective role in repressing cancer. Our results may provide novel insights into the potential therapeutic of stevioside for the treatment of colorectal cancer.
Materials and methods
Cell culture and treatment
HCT116 and HT29 cells were purchased from Wuhan Procell Biotechnology Co., Ltd. The CRC cells were authenticated by STR profiling and confirmed to be free of mycoplasma contamination through PCR-based microbial detection. The cells were cultured in 1640 medium (Sigma, USA) and DMEM (Sigma, USA) containing 10% FBS (Gibco, USA), respectively. The cells were cultured in an incubator maintained at 37°C and 5% CO2 concentration. The cells were divided into four distinct groups: a control group (0 μM), a low-dose group treated with 1 μM stevioside, a medium-dose group treated with 2.5 μM stevioside, and a high-dose group treated with 5 μM stevioside. Following 24 h of culture, CRC cells were collected for subsequent experiments. THP-1 cells (ATCC® TIB-202 TM) were maintained at 37°C in a humidified incubator with 5% CO2. Resting macrophages (M0) were differentiated from THP-1 monocytes with 10 ng/ml PMA (P8139, Sigma-Aldrich) for 24 h.
CCK-8 kit assay
Cell viability was accurately measured using the CCK-8 kit (Beyotime, C0037). The entire procedure was executed precisely in accordance with the kit’s instructions, and the absorbance values were subsequently determined using a microplate reader.
EdU staining
As previously reported, 15 EdU staining was detected using an EdU kit (Thermo Fisher, C10340). In brief, CRC cells were seeded into 24-well plates and treated with different drug concentrations for 48 h. Next, cells were co-cultured with EdU reagent for 3 h, followed by fixation with 4% paraformaldehyde for 15 min and membrane disruption with 0.1% Triton-100 for 15 min. Apollo staining reaction solution was added and incubated for 30 min at room temperature in the dark. Cells were incubated with diluted DAPI for 10 min at room temperature in the dark. After gently washing with PBS, cells were observed under a fluorescence microscope in randomly selected fields and photographed for recording.
Immunohistochemistry staining
Immunohistochemistry staining was performed following a standard protocol. 16 Briefly, after conventional dewaxing and hydration, the antigen is repaired for 3 min and then incubated with the primary antibodies (Ki67, ab16667, 1:200; CD86, ab220188, 1:200; and CD206, CST, 24,595, 1:100) at 4°C overnight. The next day, slides were washed and incubated with HRP-conjugated secondary antibodies for 1 h. Then DAB staining, hematoxylin redyeing, dehydration and sealing. Finally, randomly selected fields were observed and photographed under a microscope.
Flow cytometry
The CRC cells were collected and stained using 5 µL Annexin V and 5 µL Propidium Iodide (Keygen Biotech). Following this, a binding buffer (500 µL) was utilized to resuspend the cells, enabling the detection of apoptotic cells through flow cytometry (Beckman, USA).
Western blot
Proteins were extracted from CRC cells and tissues using RIPA reagent, and protein concentrations were determined with the BCA kit. Subsequently, a western blot analysis was performed according to previously reported methods. 17 The specific primary antibodies included Bax (abcam, ab32503, 1:1000), Bcl-2 (), cleaved-caspase-3 (abcam, ab214430, 1:1000), E-cadherin (CST, 5296,1:1000), Vimentin (CST, 3932, 1:1000), CD86 (ab220188, 1:1000), CD206 (CST, 24,595, 1:1000). GAPDH (abcam, ab8245, 1:1000) was used as an internal control. Finally, Image J software was used to determine the gray value of protein bands.
RT-qPCR analysis
Total RNA was extracted from CRC cells and tissues using TRIzol reagent, and cDNA was synthesized using a reverse transcription kit according to the kit instructions.
RNA purity was assessed by UV spectrophotometry through A260/A280 (1.8–2.1) absorbance ratios. RT-qPCR reactions were performed as follows: 95°C for 30 s, followed by 40 cycles at 95°C for 5 s and at 60°C for 20 s using SYBR Green qPCR assay kit (Takara Bio, Inc.) on an ABI 7500 Real-Time PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc.). GAPDH was used for loading control. The 2-ΔΔCt method was used for quantitative analysis to calculate the relative mRNA expression levels. The following primer sequences were used: TNF-α forward, 5′-GCGACGTGGAACTGGCAGAAG-3′ and reverse, 5′-GCCACAAGCAGGAATGAGAAGAGG-3'; IL-10 forward, 5′-TTCTTTCAAACAAAGGACCAGC-3′ and reverse, 5′-GCAACCCAA GTAACCCTTAAAG-3'; Arg-1 forward, 5′-CAGAAGAATGGAAGAGTCAG-3′ and reverse, 5′-CAGATATGCAGGGAGTCACC-3'; IL-12 forward, 5′-CAG TACACC TGCCACAAAGGA-3'; reverse: 5′-GTGTGACCTTCTCTGCAGACA-3'; GAPDH forward, 5′-GCACCGTCAAGGCTGAGAAC-3′ and reverse, 5′-TGGTGAAGACGCCAGTGGA-3'.
Wound healing assay
CRC cells were seeded at a density of 1.0 × 105/ml overnight. Subsequently, using a fine pipette tip, cells were carefully scratched perpendicularly across the cell culture plate. Following three washes with PBS, a serum-free medium was added for further cultivation. The widths of the scratches were then measured and recorded under an optical microscope at 0 h and 24 h.
Transwell experiment
Transwell assay was performed to detect the cell invasion as previously reported. 18
Tumor transplantation experiment
A total of thirty-two 6-week-old male BALB/c mice were purchased from Beijing Huafukang Bioscience Co., LTD. Mice were housed in specific pathogen-free (SPF) conditions with constant temperature (20 °C–25 °C), humidity (65%) and a 12-h light/dark cycle, where they received standard diet and water. After a week of adaptive feeding, 5 × 106 HCT116 cells in 100 μL PBS were were subcutaneously injected into the right dorsal flanks of Balb/c nude mice (n = 6/group). Subsequently, the mice were randomly allocated into four groups: the control group, low-dose group (2.5 mg/kg stevioside), medium-dose group (5 mg/kg stevioside), and high-dose group (10 mg/kg stevioside). Treatment groups were given by gavage of stevioside for 21 days, the control group received saline. Finally, the blood and tumor tissues were harvested after anesthesia, and the volume was measured using digital calipersand weighed. The animal experiments in the study were approved by the Animal Ethics Committee of The First Affiliated Hospital of Guizhou University of Chinese Medicine (Approval No. GUCHM-20221019). Symptoms such as pain, weight loss, loss of appetite or weakness were set as humane endpoints for the present study. All efforts were made to minimize animal suffering, and euthanasia was performed using an intraperitoneal overdose of pentobarbital sodium to ensure a humane endpoint.
Statistical analysis
Data in this study are presented as mean ± SD. SPSS22.0 software package was used for statistical analysis. The Student’s
Results
The effects of stevioside on the proliferation and apoptosis of colorectal cancer cells
In the present study, to investigate the growth inhibitory effect of stevioside on the colorectal cancer cells, a CCK-8 kit was performed after 24 h, 48 h and 72 h of treatment at various doses (1 μM, 2.5 μM and 5 μM). It (Figure 1(a)) showed the dose-dependent inhibitory effect of stevioside on HCT-116 and HT-29 cells. EdU staining results (Figure 1(b)) showed that compared with the 0 μM group, the EdU positive rate was significantly reduced in the stevioside treatment group ( The effects of stevioside on the proliferation and apoptosis of colorectal cancer cells (a) CCK-8 kit was used to detect the cell viability. (b) EdU staining was performed to detect the proliferation. (c) The apoptosis was determined using flow cytometry (d) Western blot was used to detect the protein expressions of cleaved-caspase-3, Bax and Bcl-2 in colorectal cancer cells. *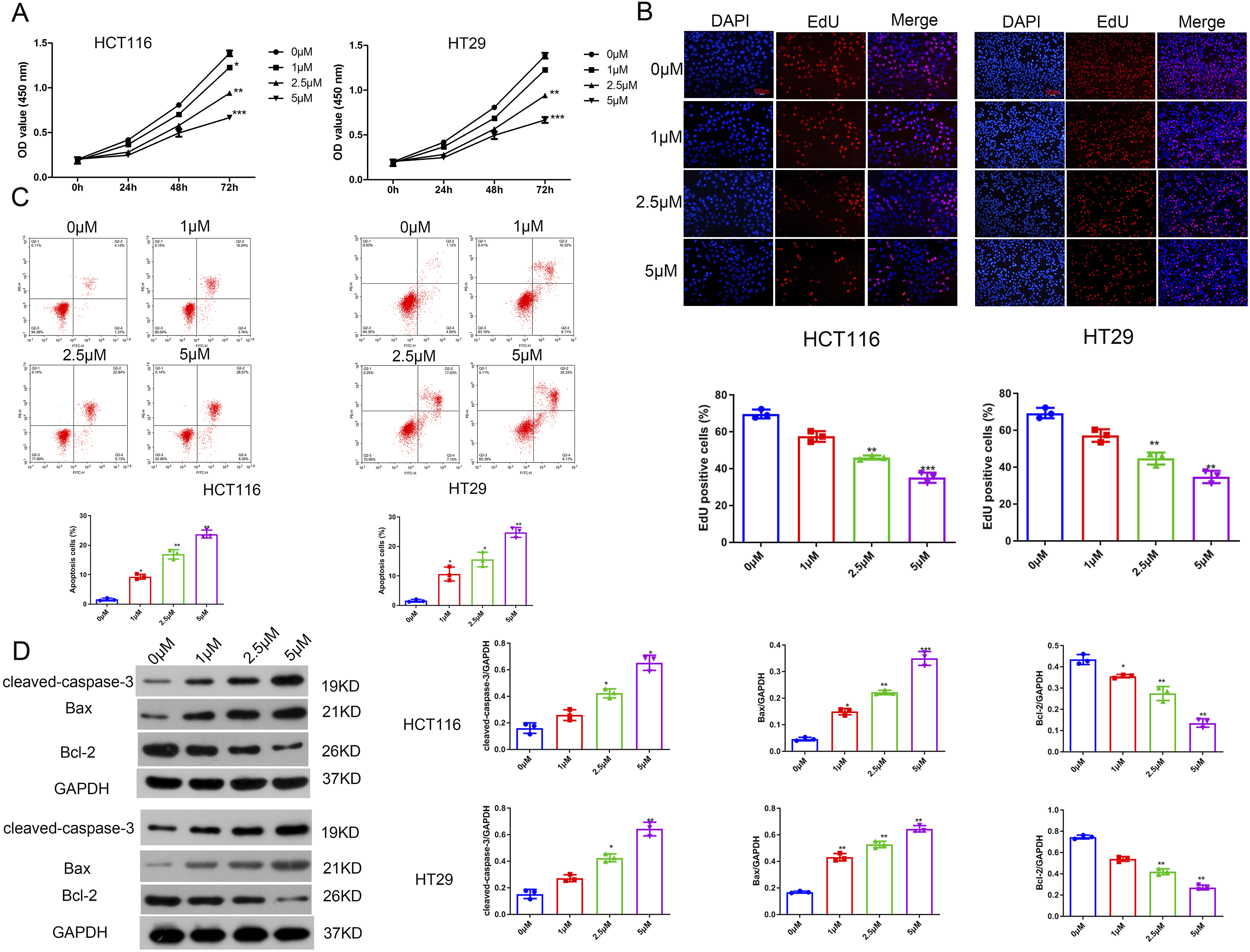
The effects of stevioside on the migration and invasion of colorectal cancer cells
Next, wound healing assay results (Figure 2(a)) showed that compared with 0 μM group, the migration in the stevioside treatment group (2.5 μM and 5 μM) was reduced significantly. And 5 μM stevioside had the most significant effect on the migration of colorectal cancer. Transwell experiment results (Figure 2(b)) showed that the cell invasion ability was significantly reduced in the stevioside treatment group compared with the control group ( The effects of stevioside on the migration and invasion of colorectal cancer cells (a) Wound healing assay was used to detect the migration. (b) Transwell experiment was performed to detect the invasion. (c) The protein expressions of E-cadherin and Vimentin were determined by western blot. *
Stevioside repressed colorectal tumor growth in vivo
To further assess the role of stevioside in colorectal tumor growth Stevioside repressed colorectal tumor growth 
Stevioside induces M1 polarization of macrophages in vivo
Immunohistochemical staining results (Figure 4(a) and (b)) showed that the positive rate of CD86 in the stevioside treatment group was significantly increased compared with the control group. However, the positive rate of CD206 decreased significantly. Interestingly, western blot results (Figure 4(c)) showed the same trend. In addition, RT-qPPCR analysis results (Figure 4(d)) showed that compared with control group, the mRNA expressions of Arg-1 and IL-10 in stevioside treatment group were reduced significantly, while the mRNA expressions of IL-12 and TNF-α were increased significantly. In conclusion, it suggested that stevioside induces M1 polarization of macrophages Stevioside induces M1 polarization of macrophages 
Stevioside induces M1 polarization of macrophages in vitro
THP-1 cells were treated with 10 ng/ml PMA and induced THP-1 cells into M1-TAMs. As shown in Figure 5(a), cells exhibited more pseudopods and round shapes, which is consistent with the features of M2-TAMs after 48-h treatment. PMA-induced M1 polarization was confirmed by flow cytometry results (Figure 5(b)). Flow cytometry results (Figure 5(c)) showed that compared with control group, the positive rate of CD86 in stevioside treatment group was increased significantly. As shown in Figure 5(d), it exhibited that compared with control group, the positive rate of CD206 in stevioside treatment group was reduced significantly. Consistent with the flow cytometry results, immunofluorescence staining results (Figure 5(e)) showed the same trend. RT-qPCR analysis results (Figure 5(f)) showed that compared with control group (0 μM), the mRNA expressions of Arg-1 and IL-10 were reduced significantly in stevioside treatment group, while the mRNA expressions of IL-12 and TNF-α were increased significantly in stevioside treatment group. The above results suggested that stevioside induced M1 polarization of macrophages Stevioside induces M1 polarization of macrophages 
Discussion
In conventional colorectal cancer (CRC) treatment, 5-fluorouracil (5-FU) and oxaliplatin form the cornerstone of systemic chemotherapy.19,20 As a fluorinated pyrimidine antimetabolite, 5-FU mediates its cytotoxic effects through dual mechanisms: irreversible inhibition of thymidylate synthase (impairing DNA synthesis) and erroneous incorporation into RNA molecules. Oxaliplatin, a diaminocyclohexane platinum derivative, generates intra-strand DNA crosslinks while exhibiting superior renal safety profiles compared to conventional platinum agents. Despite chemotherapy has the therapeutic advantages, dose-dependent neurotoxicity and severe gastrointestinal toxicity affect treatment dependence.
Traditional Chinese medicine (TCM) has been widely concerned and recognized in the clinical application of cancer treatment. 21 Considering that traditional Chinese medicine composition is complicated, the exact molecular and role in regulating anti-cancer functions remains ambiguous. However, it has been documented22,23 that stevioside possesses the ability to inhibit the progression of various types of cancer. In the present study, we discovered that stevioside significantly impeded cell proliferation and fostered apoptosis, with the most pronounced effect observed in the 5 μM stevioside (high-dose) group. Furthermore, our findings revealed that stevioside had the capacity to induce macrophage M1 polarization and regulate the tumor microenvironment.
The tumor microenvironment comprised a diverse array of cells, cytokines, and extracellular matrix components, all of which play crucial roles in regulating tumor development.24,25 Increasing reports indicated26,27 that the tumor microenvironment, encompassing immune cells and inflammatory factors, exerts a complex influence on cancer cell invasion, migration, and proliferation. Under varying pathological conditions or due to drug stimulation, macrophages in the tumor microenvironment could be stimulated to undergo polarization towards either the M1 or M2 phenotype.
28
Reports have indicated
29
a positive correlation between lower M1 polarization and the malignancy of tumors. Furthermore, M1 macrophages secrete CD86, CD206, the anti-inflammatory factor (IL-10), and the chemokine (CCL17), which may contribute to their anti-tumor and anti-inflammatory functions. Zhang et al. discovered
30
that miR-203 exhibits anti-tumor effects by enhancing macrophage M1 polarization, thereby establishing its potential as a diagnostic biomarker for prostate cancer. Lactobacillus casei & Lactobacillus reuteri facilitate the M1 polarization of macrophages through the inhibition of TLR4, thus modulating intestinal flora homeostasis and mitigating the progression of pancreatic cancer.
31
Zong et al. found
32
that M1 macrophages promote the expression of PD-L1 in hepatocellular carcinoma cells by regulating IL-1β, further strengthening their tumor-enhancing effects. In this study,
In summary, our findings revealed that stevioside exhibits significant inhibitory effects on the malignant progression of colorectal cancer by inducing polarization of M1 macrophages and augmenting the inflammatory response. This study thus presents a promising novel therapeutic approach for colorectal cancer and may have theoretical guiding significance for the clinical utilization of stevioside.
Nevertheless, our research still has some limitations. Due to the small sample size in this study, the correlation between M1 polarization of macrophages and the tumor microenvironment
Footnotes
Acknowledgments
We are grateful to all participates for their contributions for the present study.
Ethical Approval
The animal experiments in the study were approved by the Animal Ethics Committee of The First Affiliated Hospital of Guizhou University of Chinese Medicine (Approval No. GUCHM-20221019).
Author contributions
Y. B.: Writing, Conceptualization, Methodology, Software Y. W: Data curation, Writing, Original draft preparation. F. Z.: Visualization, Investigation. K. W.: Reviewing and Editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data in the article will be made available by the authors, without undue reservation.
