Abstract
Introduction
Tamoxifen (TMX) shows promise in treating breast cancer, but it faces challenges such as poor solubility, instability, and incomplete release when targeting tumors. Additionally, TMX therapy’s toxicity is a critical issue in breast cancer treatment. This study aimed to assess the impact of hyaluronic acid (HA)-coated TMX-loaded solid lipid nanoparticles (HA-TMX-SLNs) on MCF7 breast cancer cells.
Methods
Solid lipid nanoparticles (SLNs) were prepared using hot homogenization. The HA-TMX-SLNs and TMX-SLNs were characterized and evaluated through transmission electron microscopy (TEM). Cytotoxicity was assessed using the MTT assay, and Western blot analysis was utilized to identify key factors in the cell cycle and apoptosis.
Results
The nanoparticles (HA-TMX-SLNs) demonstrated approximately 55% loading efficiency after 100 h. HA-TMX-SLNs exhibited lower cytotoxicity in MCF7 cells compared to other treatments. Significant decreases in expression levels of cyclin-dependent kinase (CDK) 4, Cyclin D1, CDK2, and Bcl2 were observed after treatment with HA-TMX-SLNs, along with an increase in cleaved/procaspase-7.
Discussion
The in vitro release study showed that HA-coated SLNs consistently released the drug into the media under controlled conditions. Furthermore, HA-TMX-SLNs exhibited cytotoxic effects, increasing apoptosis and inhibiting cancer cell proliferation. These findings suggest that HA-TMX-SLNs effectively deliver TMX to breast cancer cells.

Introduction
The most common cancer in women is breast carcinoma.1,2 In recent decades, the incidence of breast cancer has increased alarmingly. Despite medical advances, breast cancer remains the second most common and deadliest cancer for women. 3 It is predicted that by 2040, there will be more than three million breast cancer cases annually, with over one million deaths.4,5 Steroid hormone receptors (HRs) play a significant role in breast cancer prognosis, and endocrine therapy is vital for breast cancer treatment. ER-positive, PR-positive, and ER/PR-positive breast cancer cells are various combinations of estrogen receptor-positive (ER-positive), progesterone receptor-positive (PR-positive), and both.6,7 Almost all breast cancers are estrogen receptor-positive, and more than half are both ER-positive and PR-positive. 8 Several treatments for breast cancer include radiological interventions and surgery. Postsurgical prevention of breast cancer involves the use of TMX as a medical treatment. 9 TMX, a widely used cancer drug, is highly effective at inducing apoptosis in cancer cells. 10 It triggers apoptosis through a mechanism that is not dependent on the ER, such as inhibiting protein kinase C-mediated pathways. 11 TMX notably affects the expression of Cyclin D and cMyc and promotes the expression of caspases-6, 7, and 9. Additionally, TMX triggers the expression of p53 and p21, which activate G0-G1-phase cell cycle arrest. 12 Recent findings have shown that TMX can activate caspase-3. 13 Furthermore, TMX can regulate the intracellular cascade and inhibit mitochondrial respiration, eventually leading to apoptosis. TMX is a highly effective drug for inducing cancer cell apoptosis through various mechanisms. 14 Its ability to modulate mutations in tumor cells makes it a promising candidate for cancer therapy. 15 TMX therapy displays potential for treating breast cancer however it encounters obstacles like low solubility, instability, and incomplete release when aimed at tumors. Additionally, the toxicity of TMX therapy poses a significant concern in breast cancer treatment. 16 Prolonged use of TMX in therapy has been reported to lead to resistance in breast cancer cells. 17 Anticancer drugs administered through systemic chemotherapy affect both cancer cells and healthy cells in the body, leading to various side effects.18,19 Moreover, these drugs have low biostability and are easily eliminated by the immune system or other physiological reactions in the bloodstream. 20
Drug delivery systems can help address this issue. To address these challenges, nanocarriers have been engineered to create targeted drug delivery systems. 21 Targeted delivery to specific tissues can maximize drug benefits and minimize toxicity through this method.22,23 The main objective of drug delivery is the possibility of targeted delivery to tumors, minimizing potential problems such as side effects, incomplete release, and toxicity, and increasing the ability of drugs to overcome issues such as insolubility, instability, and resistance.24,25 Drug delivery systems consist of formulations, approaches, and technologies designed for specific purposes. The delivery of anticancer agents to combat drug resistance by SLNs has been the focal point of various research groups since the early 1990s. 26 SLNs are commonly employed in breast cancer treatment as nanocarriers to increase drug therapeutic efficacy.27,28 SLNs have been utilized to enhance the effectiveness of cancer therapy by improving drug delivery through the bloodstream. 20 The advantages of SLNs include small particle size, good stability, controlled drug release, biocompatibility, and drug targeting. Additionally, it has been demonstrated that SLNs can overcome multidrug resistance (MDR) by bypassing drug efflux transporters, ultimately enhancing drug therapeutic efficacy and safeguarding against the exposure of combined drugs to the external biological environment.29,30 The high surface energy of SLNs allows them to clump together and interact with plasma proteins indiscriminately, leading to immune opsonization and swift removal of these particles from the system. This interaction can potentially limit the circulation time of SLNs in the body, affecting their efficacy as drug-delivery vehicles. 22
SLNs can readily accept various materials due to their surface properties, enabling targeted therapeutics. 31 The surface modification of SLNs is a simple process for transferring different target molecules. The surface of the SLNs can be coated with HA to overcome this limitation. 32 By attaching carbohydrates to nanoparticles, chemotherapy drugs can be specifically delivered to cancer cells. HA is biodegradable, biocompatible, nontoxic, nonimmunogenic, and noninflammatory. 33 As a natural linear polysaccharide, HA has gained the approval of researchers. HA serves as a drug delivery option due to its ability to bind to the cell receptor cluster of differentiation 44 (CD44), which is often overexpressed on the surface of tumor cells. 34 HA, a natural component found in all species, possesses unique characteristics that make it a promising drug carrier. 35 The development of nanocarriers based on HA has been demonstrated to be advantageous for treating tumors bearing CD44 receptors in terms of improved pharmaceutical delivery, enhanced therapeutic effectiveness, and a significant inhibitory effect on tumor growth. 34 Recent studies have highlighted the significant impact of HA and CD44 on drug resistance in cancer cells. 32 HA enhances nanoparticle uptake by interacting with the CD44 receptor. 36 Extensive research on targeted release, dermal absorption, and drug delivery has shown that HA facilitates the uptake of drug carriers by cancer cells through receptor-mediated endocytosis.23,37,38 Men et al. expressed that HA is a simple way to increase drug absorption in CD44-overexpressing cells by loading a biofunctionalized cationic polymer with doxorubicin. Also, in vivo tests revealed that the inhibition of the growth of tumors more efficiently was confirmed and reduced side effects compared with free drugs. 39 Li et al. demonstrated that HA can boost the absorption of doxorubicin-loaded carbon dots in breast cancer cells. Their comparative analysis with free HA indicated that the HA-enhanced nanoparticles were taken up through binding to the CD44 receptor, leading to increased accumulation of the nanoparticles in tumor tissue. 40 The literature indicates that 1%–5% of nanoformulation drugs can accumulate in target tissues, whereas only 0.01% of free drugs can reach the target tissue. 41 A novel chemotherapeutic nanocomposite, HPCID, incorporates HA and doxorubicin. Treatment with HPCID in combination with sonodynamic therapy in 4T1 xenograft-bearing mice restrained tumor growth and pulmonary metastasis without causing systemic toxicity. 42 Another study by Zhong et al. showed that doxorubicin-loaded cross-linked HA-Lys-LA10 nanoparticles exhibited significant accumulation in tumors and effectively suppressed tumor growth with minimal side effects in vivo 43
The challenges in current breast cancer treatments include low solubility, instability, and incomplete release of drugs like TMX when targeted at tumors. Additionally, the toxicity and development of resistance to TMX therapy pose significant concerns. Anticancer drugs administered through systemic chemotherapy also affect healthy cells, leading to various side effects and low biostability. HA-TMX-SLNs address these gaps by utilizing nanocarriers to create targeted drug delivery systems, maximizing drug benefits and minimizing toxicity. The novelty of this study lies in the use of HA-coated TMX-SLNs, which enhance the impact of TMX on breast cancer cells, demonstrating significant accumulation in tumors and effectively suppressing tumor growth with minimal side effects. Hence, the effects of TMX-loaded SLNs and HA-coated TMX-SLNs were assessed on breast cancer cells to evaluate the efficacy of HA-coated TMX-SLNs in enhancing the impact of TMX on breast cancer cells.
Materials and methods
Materials
TMX (BioSera) was freshly dissolved in methanol and stored in the dark at 4°C. Dulbecco’s Modified Eagle’s Medium (DMEM), penicillin, and streptomycin were purchased from Gibco (Thermo Fisher, USA), as was fetal bovine serum (FBS). The 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl tetrazolium bromide (MTT) reagent was also obtained from Gibco.
Caspase-7 (#9492) from Cell Signaling Technology, cyclin-dependent kinase (CDK) 2 (E-AB-40095) from Elabscience Biotechnology Inc., CDK4 (DCS-35) from sc-23896, cyclin D1 (A-12) from sc-8396, Bcl-2 (N-19) from sc-492, GAPDH (6C5) from sc-32233, mouse anti-rabbit IgG-HRP from sc-2357, m-IgGκBP-HRP from sc-516102 (all from Santa Cruz Biotechnology), and the SuperSignal® Molecular Weight Protein Ladder (84785) from Thermo Scientific were obtained. Octadecylamine, Tween 80, stearic acid, and HA were purchased from Merck. The essential kits were obtained from their respective suppliers’ websites.
Preparation of TMX-loaded SLNs
Using the hot homogenization technique, SLNs containing octadecylamine, Tween 80, stearic acid, and TMX were prepared. Initially, TMX-loaded SLNs were produced with stearic acid and octadecylamine, following Grillone et al., with slight modifications. 44 Stearic acid and octadecylamine were mixed in a 1:1 ratio and then dissolved in chloroform. TMX (10 mg/mL) was added at 80°C as the mixture melted. Tween 80 was gradually added dropwise to the melted lipid-drug mixture. A homogenizer (Mtops SR30, Korea) was used to homogenize the mixture at 25,000 r/min for 15 minutes. To enhance the physical properties of the SLNs, 6% stearic acid was employed in this study. Subsequently, the SLNs were lyophilized and stored in a container at 4°C.
Preparation of HA-coated TMX-SLNs
To make HA-SLN-TMX, the SLN-TMX suspension was mixed with HA solution (at 15 mg/mL) and stirred. The suspensions were then centrifuged at 10000 r/min for 1 h. 45
SLN and HA-SLN characterization
The HA-coated SLNs and SLNs were characterized by studying their particle size, zeta potential, and morphology. The particle size and zeta potential of the SLN and SLN-HA dispersions were estimated using a DLS spectrophotometer (Microtrac NANO-flex® II, Germany) and Zeta-check (Microtrac Zeta-check, Germany) at room temperature. To enhance signal clarity, the samples were mixed with deionized water. TEM (Philips EM 208S, Germany) was utilized to conduct morphological analyses of the SLNs and HA-SLNs. A carbon grid was used to dry the diluted sample with the help of air. Following staining with uranyl acetate, the fixed sample was evaluated by a TEM apparatus (1%) by weight.
FT-IR analysis using Fourier transform infrared spectroscopy
The functional groups involved in the biosynthesis of SLN nanoparticles were determined by preparing and analyzing KBr pellets using Fourier transform infrared spectroscopy (FT-IR; JASCO Ltd, Tokyo, Japan) in the wavenumber range of 500–4000 cm−1. 46
In vitro TMX release study
In vitro evaluation was performed to assess the release of the drug (TMX) from HA-SLNs using the dialysis method. Methanol was used to enhance TMX solubility. Subsequently, 400 µL of the HA-coated TMX-SLN solution (0.02 g/mL, diluted in dH2O) was introduced into the dialysis bag. The dialysis bags were then placed in 120 mL of release medium (composed of PBS and 1% ethanol, with a pH of 7.4) at 37°C and stirred at 100 r/min. At specified time points (0.5, 1, 2, 3, 6, 12, 24, 48, 72, and 100 h), 500 µL of the dialysate was removed and replaced with an equal volume of release medium. The concentration of TMX in the dialysate samples was determined using spectrophotometry.
Cell culture
The human breast cancer cell line MCF7 (NCBI code: C135) was acquired from the Pastor Institute in Iran and seeded in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin at 37°C with 5% CO2. In vitro conditions, MCF7 cells were divided into four groups according to the following treatments: (1) control group (no drug); (2) free TMX group; (3) TMX-SLN group; and (4) HA-TMX-SLN group.
MTT assay
To evaluate the viability of MCF7 cells treated with HA-TMX-SLNs, an MTT assay was performed following the method described by Zarei et al..47,48 MCF7 cells were seeded in 96-well plates at a density of 2 × 104 cells per well and incubated overnight. The following day, the cells were exposed to various concentrations (10-500 µg/mL) of free TMX, TMX-SLNs, and HA-TMX-SLN formulations for 48 h after refreshing the medium. After the incubation period, MTT reagent was added, and the plates were incubated at 37°C for an additional 30 minutes. The formazan concentration was assessed by measuring the absorbance at 570 nm using an ELISA reader (MPR+ Hiperion, Germany). The percentage of cell viability was calculated by comparing the average absorbance of the treatments to the absorbance of the control, as shown in Equation:
Note that the specific concentrations (10–500 μg/mL) and duration (48 h) used in the cytotoxicity study were selected based on a combination of literature review and preliminary experiments. The concentration range was chosen to encompass the reported IC50 values for TMX in MCF7 cells, which typically fall between 10 and 100 μg/mL.49,50 The 48-h duration was chosen because it is a common time point for assessing cytotoxicity and provides sufficient time for cells to respond to treatments.
In the drug release study, a concentration was carefully selected at different times to ensure that the formulations were not saturated with TMX while allowing for measurable release over the 48-h period. This duration was chosen because it is a reasonable time frame for assessing the initial burst release and subsequent sustained release of TMX from SLNs and HA-TMX-SLNs.
In the cytotoxicity study, untreated MCF7 cells served as the control condition to assess the effects of treatment. Under the same conditions and medium as the treated cells, untreated cells were incubated without test substances. This allowed us to determine the natural variability in cell viability and identify any potential artifacts or biases in the assay.
Western blotting analysis
The Cells were plated in a 6-well plate and cultured with HA-TMX-SLNs (50 µg/mL), TMX-SLNs (100 µg/mL), and free TMX (1 µg/mL) for 48 h. The cells were then washed once with PBS. Whole-cell extracts were prepared by lysing the cells with lysis buffer [20 mM Tris (pH 7.5), 150 mM NaCl, 1 mM EDTA, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris, and protease and phosphatase inhibitor cocktails] for 30 minutes on ice. Protein concentration was determined by the Bradford assay. 51 Equal amounts of total protein (40 µg) were resolved by 10% SDS-PAGE and transferred to PVDF membranes by electroblotting. The membranes were probed with specific antibodies against CDK2 (1:1000), CDK4 (1:1000), Cyclin D1 (1:1000), Bcl2 (1:1000), Caspase-7 (1:1000), and GAPDH (1:1000) at 4°C overnight. Detection was accomplished using corresponding horseradish peroxidase (HRP)-conjugated secondary antibodies, with GAPDH serving as the internal control. Signal densities were quantified using a densitometric program (ImageJ).52,53
Statistical analysis
In the present study, we utilized GraphPad Prism (version 8.0, La Jolla, CA, USA) to analyze the numerical data. Three replicates were performed for each experiment, and all data are reported as means ± standard error of the mean (SEM). One-way ANOVA, along with Tukey’s multiple comparison test, were used to identify significant differences between groups. Statistical significance was defined as p < .05.
Results
Characterization and surface properties of the SLN formulation
The mean diameter, zeta potential, and morphology of the TEM-loaded SLN were evaluated. Subsequent tests included in vitro drug release and cytotoxicity assessments. The dynamic light scattering (DLS) and zeta potential analyses of the SLNs and HA-SLNs are depicted in Figure 1. The data indicated that the SLNs had an average size of 181.1 ± 57.794 nm with a polydispersity index (PDI) of 0.499 ± 0.044 and a surface charge of −43.667 ± 0.208 mV. In contrast, HA-SLN exhibited an average size of 516.533 ± 104.33 nm, a PDI of 3.860 ± 0.121, and a zeta potential of −7.767 ± 0.306 mV. These findings confirmed that coating HA onto the SLN surface resulted in an increase in nanoparticle diameter and a decrease in zeta potential. TEM analysis revealed the morphology of both SLNs and HA-SLNs, as presented in Figure 1(a). The results illustrated that the TEM-loaded SLNs and HA-TEM-loaded SLNs displayed acceptable dispersity and spherical morphology. TEM micrographs of SLNs and HA-TMX-SLNs. TEM micrographs (scale bar: 300 nm) of the TMX-SLNs (a) and HA-coated SLN (b) show that the particles have a regular spherical shape.
FT-IR results
FT-IR spectra of HA, SLN, TMX, and HA-TMX-SLN were recorded within the wavelength range of 500-4000 cm−1, as shown in Figure 2. The FT-IR spectra of HA, SLN, TMX, and HA-TMX-SLN were compared and analyzed to identify any differences or similarities in their spectral patterns. These samples showed nearly identical spectral patterns. FT-IR spectra of HA, TMX, SLN, and HA-TMX-SLN. The FT-IR spectra of HA, TMX, SLN, and HA-TMX-SLN are shown in the figure, with the overlapping spectra presented. Two characteristic peaks are observed in the spectra of TMX, SLN, and HA-TMX-SLN: O-H stretching vibrations and aromatic C-C stretching. The prominent peaks of HA-TMX-SLN are observed at 3305, 2119, and 1636 cm−1, which match the peaks found in the spectra of HA (3434, 1642 cm−1), SLN (3282, 2053, 1641 cm−1), and TMX (3321, 2043, 1661 cm−1). The nearly identical spectral patterns of these samples confirm their uniform composition.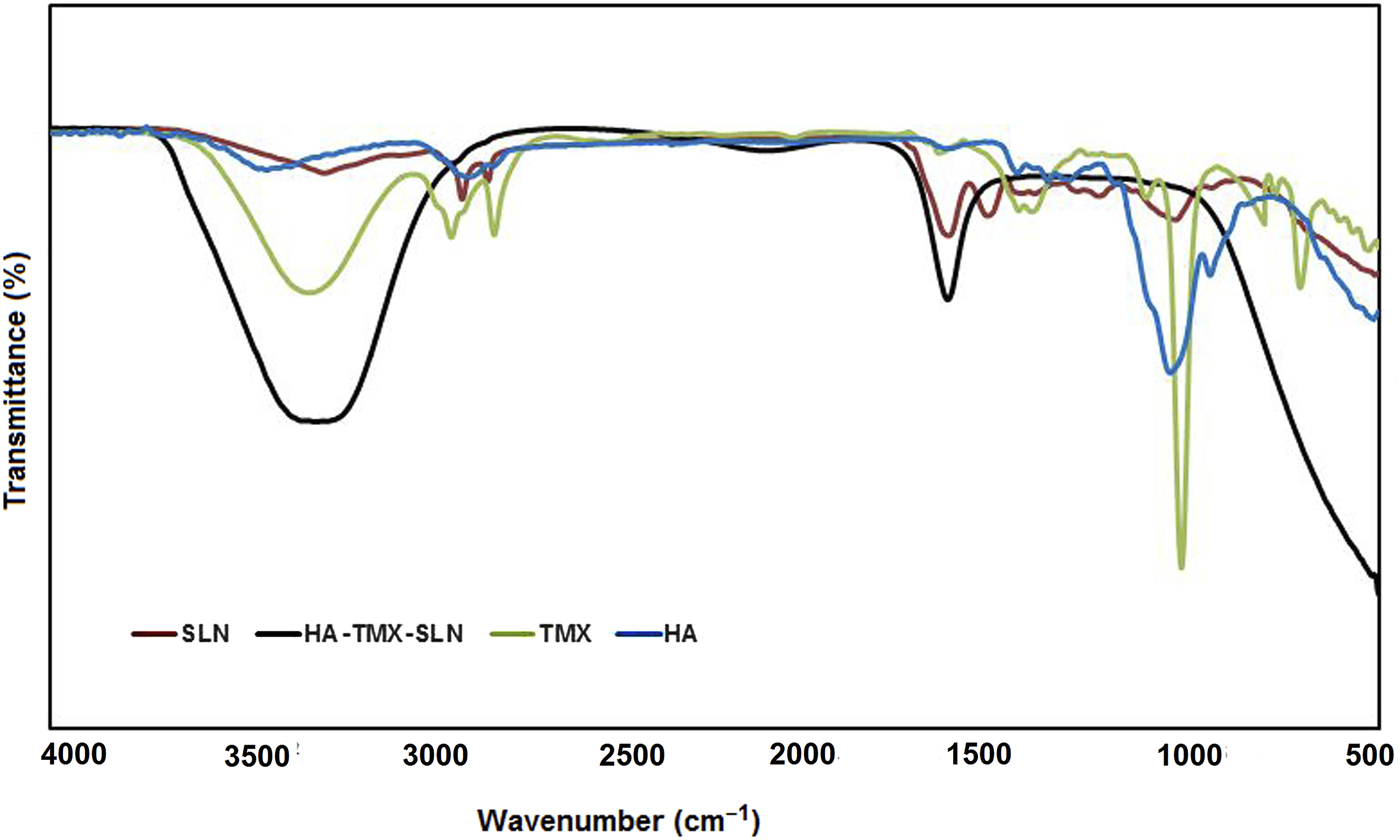
The absorption band of TMX in the FT-IR spectrum at 3321 cm−1 is attributed to broad O-H stretching vibrations. The bands at 2945, 2831, and 2522 cm−1 are associated with aromatic C-H stretching. Additionally, the bands at 1775, 1661, and 1449 cm−1 correspond to carboxylic C = O, aromatic C-C, and C = C ring stretching, respectively. The absorptions at 1242 and 1113 cm−1 are attributed to aromatic C-O stretching. 54 Peaks at 2324, 2225, and 2043 cm−1 were also observed.
The HA absorption band at 3434 cm−1 is attributed to the –OH stretch, while the band detected at 2904 cm−1 is caused by the –CH stretch. Another characteristic band at 1642 cm−1 is attributed to the C = O-NH stretch. The bands at 1452 cm−1 are associated with C-O and C = O stretching. Furthermore, the peak at 1050 cm−1 is attributed to the C-O-C stretch. 55
For SLN, the peaks appeared at 3282 cm−1 (O-H stretching), 2917 and 2855 cm−1 (C-H stretching), 1636 cm−1 (C = O stretching), 1530 cm−1 (C-H stretching), 1242 cm−1 (C-O stretching), and 1053 and 1036 cm−1 (C-O-C stretching). 56
In the spectrum of HA-TMX-SLN, an OH-associated peak at 3305 cm−1 was noted. Notably, a sharp peak for C-C aromatics was observed at 1636 cm−1 in these spectra. Peaks at 2119, and 492 cm−1 were also observed.
The specific peaks of HA-TMX-SLN were observed at 3305, 2119, and 1636 cm−1, matching those found in the HA (3434, 1642 cm−1), SLN (3282, 2053, 1641 cm−1), and TMX (3321, 2043, 1661 cm−1) spectra. The FT-IR spectrum of HA displayed strong peaks and patterns that were predominant in the HA-TMX-SLN spectrum, indicating the presence of HA on the SLN surface.45,54 This finding suggested that TMX was correctly loaded onto the SLN and that HA was coated on the SLN surface. The spectra of these three samples showed nearly identical patterns, confirming the successful synthesis of HA-coated SLNs.
In vitro release of drugs
The release of TMX (drug) from the HA-TMX-SLN formulation was examined at pH 7.4 (as shown in Figure 3). The release was biphasic, including a slow initial phase that released 39.71% of the drug in 12 h, followed by a second stage in which changes in domain percentage were recorded. Two phases can be distinguished in the release profiles: the initial burst-release phase and the sustained-release phase. According to the data, the drugs released in the second phase reached saturation after 48 h (∼50%) and 100 h (∼55%). Drugs cannot spread into the media through the HA layer, which acts as a barrier. Cumulative drug release from HA-coated SLNs. The figure shows the time-associated cumulative release profiles of TMX from HA-TMX-SLNs at pH 7.4 and 37°C. The data are presented as the means ± SEM (n = 3, p values <0.05). The results indicate that, in the second phase, the drug release reached saturation levels of approximately 50% at 48 h and 55% at 100 h. The presence of the HA layer acts as a barrier, effectively limiting the spread of drugs into the media. This highlights the importance of considering the HA layer in drug delivery strategies, as it plays a crucial role in controlling the release of active pharmaceutical ingredients.
The effect of HA-TMX-SLNs on cell viability
The viability assay demonstrated the effectiveness of the HA-coated TMX-SLN formulation against MCF7 cells (p values <0.05, n = 3). As shown in Figure 4, we observed the viability of cancer cells exposed to various formulations, including HA-TMX-SLNs and TMX-SLNs. We evaluated TMX concentrations of 10, 25, 50, 75, 100, 250, and 500 µg/mL. At a 50 µg/mL TMX dose, HA-TMX-SLNs exhibited a 52.91% viability rate after 48 h of treatment. In contrast, at the same dose, TMX-SLNs showed 86.32% less cytotoxicity than HA-TMX-SLNs. These results can be attributed to the HA coating on the SLNs; hence, cells treated with HA-TMX-SLNs at 50 µg/mL died more frequently than those treated with TMX-SLNs at the same concentration. However, free TMX at 10 µg/mL had a similar cytotoxic effect on MCF7 cell viability (∼5%) at doses of 50 or 500 µg/mL after 48 h of treatment, as depicted in Figure 4. Thus, these results further support the efficacy of HA-coated SLNs. Additionally, MTT results indicated that free SLNs had no significant effect on cell viability. In vitro cytotoxicity of free TMX, TMX-loaded SLNs, and HA-TMX-SLNs to MCF7 cells. The figure presents the cytotoxicity of free TMX, TMX-loaded SLNs, and HA-TMX-SLNs to MCF7 cells through both XY (a) and bar graphs (b). MCF7 cells were treated with different concentrations of TMX, TMX-SLNs, and HA-TMX-SLNs (10–500 µg/mL) for 48 h. The data are presented as the mean ± SEM. p values less than 0.05 were considered significant (*p < .05, **p < .01, ***p < .001, ****p < .0001 vs. control). Our results show that at a concentration of 50 µg/mL, HA-TMX-SLNs exhibited a viability rate of 52.91% after 48 hours, which is significantly lower than the viability rate of 86.32% observed for TMX-SLNs. This suggests that the HA coating on the SLNs leads to a substantial increase in cell death in HA-TMX-SLNs compared to TMX-SLNs at the same concentration.
Effect of HA-TMX-SLNs on cyclin D1, CDK4, CDK2, Bcl2, and caspase-7 protein expression
Most human cancers show alterations or mutations in cell cycle checkpoints. Cyclin D1 and CDK4 are central regulators of the G1-S phase transition in breast epithelial cells. The cell cycle transition from the S phase to the G2 phase is driven by CDK2, which ends in the S phase.. 57
In the HA-TMX-SLNs group, there was a significant decrease in the relative expression of Cyclin D1 (0.16-fold) compared to the TMX-SLN (0.67-fold) and TMX (0.92-fold) groups (Figure 5(a), p < .05). Additionally, CDK4 levels were reduced in the HA-TMX-SLN group (0.27-fold) compared to the TMX-SLN and TMX groups (0.49- and 0.81-fold, respectively) (Figure 5(b), p < .05). Moreover, CDK2 protein expression (0.13-fold) in the HA-TMX-SLN treatment group was significantly lower than in the TMX-SLN (0.45-fold) and TMX (0.66-fold) groups (Figure 5(c), p < .05). Furthermore, Bcl2 protein expression decreased by 0.51-fold in the HA-TMX-SLN treatment group compared to the TMX-SLN (0.66-fold) and TMX (0.75-fold) groups (Figure 5(d), p < .05). Shows the results of western blot analysis, which examines the expression of specific proteins in MCF7 cells treated with different forms of tamoxifen (TMX). The proteins examined include Cyclin D1, CDK4, CDK2, Bcl2, and caspase-7 (both pro and cleaved forms). The proteins examined include Cyclin D1 (A), CDK4 (B), CDK2 (C), Bcl2 (D), and caspase-7 (E) (both pro and cleaved forms). The expression levels of these proteins were normalized to the level of GAPDH (F), band was used to adjust the intensity of the other protein bands, ensuring that the results are accurate and comparable. The figure presents images of the western blot gels, with each panel showing the expression of a specific protein. The data are presented as the mean ± standard error of the mean (SEM), which provides a measure of the variability in the results. To determine the statistical significance of the results, we used p values to compare the expression levels of each protein in the different treatment groups. We considered p values less than 0.0001 to be statistically significant, denoted by *p < .05, **p < .01, ***p < .001, and ****p < .0001 compared to the control group. The original images of the western blot gels can be found in the supporting information, Figures S1-S6.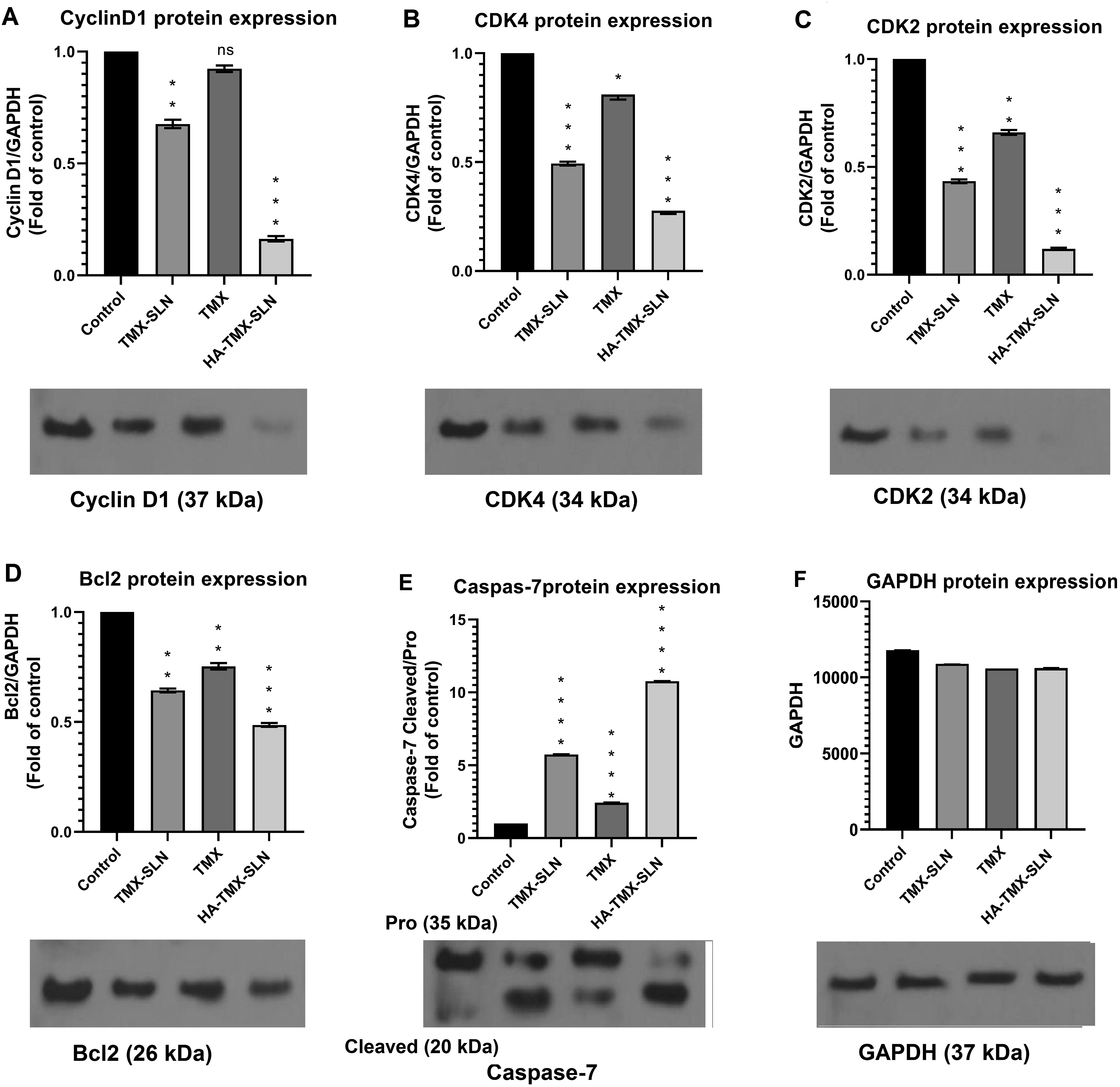
In the HA-TMX-SLN treatment group, there was a significant increase in the Cleaved/Pro caspase-7 protein ratio (10.77-fold) compared to the TMX-SLN and TMX treatment groups, which showed increases of 5.74-fold and 2.43-fold, respectively (Figure 5(e), p < .05). The original images of the western blot gel can be found in Supporting Information (Figurs. S1-S6).
The results indicated that the expression levels of CDK4, Cyclin D1, CDK2, and Bcl2 were significantly lower in MCF7 cells treated with HA-TMX-SLNs compared to those treated with free TMX or TMX-SLNs. Additionally, the cleaved/procaspase-7 ratio was notably higher in the HA-TMX-SLN treatment group than in the other groups. Protein expression levels were normalized to GAPDH (p < .05), and the fold change was calculated relative to control (Figure 5).
Discussion
The development of HA-coated TMX-loaded SLNs (HA-TMX-SLNs) presents a promising approach to improving tamoxifen delivery and efficacy in breast cancer therapy. The observed lower cytotoxicity of HA-TMX-SLNs in MCF7 cells compared to traditional treatments suggests that HA-TMX-SLNs may offer a safer alternative with reduced side effects, which is crucial for enhancing patient compliance and quality of life during treatment.58,59 Furthermore, the significant decrease in expression levels of key cell cycle regulators and anti-apoptotic proteins indicates that HA-TMX-SLNs not only facilitate drug delivery but also modulate critical pathways involved in cancer cell proliferation and survival.60–62 This dual mechanism of action could be leveraged to design combination therapies that enhance tamoxifen therapeutic efficacy while minimizing adverse effects.
The present study proposes HA-modified HA-TMX-SLNs for breast cancer therapy, marking the first investigation into the delivery of TMX-SLNs to HA-coated breast cancer cells in vitro. The SLN formulation, consisting of octadecylamine and stearic acid, demonstrates favorable physicochemical properties for drug delivery. TMX-loaded SLNs show desirable characteristics including zeta potential, mean particle size, and PDI. FT-IR data confirmed the successful synthesis of HA-coated SLNs, while TEM images of TMX-SLNs and HA-TMX-SLNs revealed spherical, uniform nanoparticles. The findings of our study highlight the effective incorporation of TMX into HA-coated SLNs for sustained drug release in breast cancer chemotherapy. Our results demonstrate that the HA-TMX-SLN formulation exhibits a slow initial drug release phase, with 39.71% of TMX released within the first 12 h, followed by a saturation phase where approximately 50% of the drug is released by 48 h. This sustained release profile is crucial for enhancing therapeutic efficacy while minimizing side effects.
Comparative analyses reveal that TMX release kinetics from HA-TMX-SLNs are significantly slower than that observed with conventional TMX-SLNs. This matches the findings of How et al. (2013), 63 where a consistent release rate of TMX was observed from a nanostructured lipid carrier over 24 h, culminating in a total release of 96.5%. Notably, this sustained release mechanism suggests that HA-coated SLNs not only prolong the drug’s half-life but also potentially improve its bioavailability and targeting capabilities due to the affinity of HA for CD44 receptors overexpressed in cancer cells. Additionally, when comparing our results to those of Shen et al. (2015), 64 it is evident that HA-TMX-SLNs outperform various formulations, including free paclitaxel solutions and conventional SLNs. Shen et al. reported a higher release of free paclitaxel (62.14%) during the initial 12 h, indicating a rapid release profile that might lead to periodic high drug concentrations and subsequent clearance from the system. In contrast, the HA-TMX-SLN formulation’s slower release minimizes potential toxicity and supports a more consistent therapeutic effect over time. Furthermore, the significant improvement in drug loading capacity and delivery efficiency of the HA-coated SLNs positions them as a promising platform for drug delivery systems. 65 Other studies focusing on SLNs and nanocarriers have also reported similar benefits, emphasizing the importance of biocompatibility and targeted delivery systems in enhancing treatment outcomes in breast cancer therapies.18,66
The results of our study indicate that HA-TMX-SLN treatment significantly enhances apoptosis in MCF7 breast cancer cells, showcasing a marked cytotoxic effect compared to TMX-SLNs, free TMX, and SLNs. The notable decreases in cell viability (79.79%, 52.91%, and 24.12% at 25, 50, and 75 µg/mL, respectively) underscore the efficacy of HA-coating in improving the therapeutic potential of TMX. In comparison to similar studies on nanoparticle-based drug delivery systems, our findings highlight the importance of CD44-targeting mechanisms in enhancing drug delivery and cytotoxic efficacy. For instance, studies by Tran et al. (2017) 45 and Mero & Campisi (2014) 67 have demonstrated the role of HA in targeting CD44 receptors, which are often overexpressed in various cancer cell types. Our results corroborate these findings, suggesting that the enhanced interaction with CD44 not only facilitates increased drug accumulation but also enhances subsequent apoptotic pathways.
These findings align with previous studies that have highlighted the role of HA in targeting CD44 receptors, which are often overexpressed in various cancer cell types. For example, Tran et al. (2017) 45 reported that HA-SLN formulations exhibited increased cytotoxicity in MDA-MB-231 and BT-47 cells compared to unbound drug formulations. Our results corroborate their findings, suggesting that the enhanced interaction with CD44 not only facilitates increased drug accumulation but also enhances subsequent apoptotic pathways. Moreover, our data reflect a differential response between HA-TMX-SLNs and TMX-SLNs, where the latter displayed negligible changes in apoptosis across the tested doses (92.04%, 86.32%, and 74.09%). This discrepancy signifies the importance of HA in promoting drug delivery and cytotoxic efficacy, likely due to its ability to mediate endocytosis via CD44 interactions. Further supporting our conclusions, Mero & Campisi (2014) 67 demonstrated that HA-modified nanoparticles significantly improved drug uptake in CD44-expressing cells, leading to enhanced therapeutic outcomes. The observed reduction in MCF7 cell viability after treatment with HA-TMX-SLNs versus free TMX also aligns with research indicating that HA-coated carriers can overcome biological barriers, exhibiting superior bioavailability and tumor targeting capabilities. Furthermore, the exploration of SLNs in cancer therapy has been extensively documented, with several studies indicating the potential of lipid-based nanoparticles for improving drug solubility and stability. Our findings contribute to this growing body of literature by emphasizing the additional advantage of using HA-functionalized systems, which not only enhance therapeutic loading but also facilitate cellular uptake in cancerous tissues.
In our study, HA-TMX-SLNs significantly reduced cell viability and promoted apoptosis by downregulating key apoptotic proteins. Similar studies have established the implications of TMX in apoptosis and cell cycle regulation. Chambard et al (2007) 12 demonstrated that TMX induces cell cycle arrest as well as apoptosis in cancer cells, correlating with our findings. However, our use of HA-coated SLNs could enhance the bioavailability and selective targeting of TMX, leading to more pronounced effects on apoptosis than seen with free TMX alone. Moreover, impaired apoptosis noted in many cancer types, including breast cancer, has been linked to chemotherapy resistance. 68 This underscores the urgent need for more effective delivery systems that facilitate apoptosis, such as the HA-TMX-SLNs we investigated. The observed decrease in cell viability in response to HA-TMX-SLNs parallels findings by Zhong et al, 43 who reported similar outcomes with HA-functionalized nanoparticles delivering chemotherapeutic agents. Their research suggested that HA enhances uptake by receptor-positive cancer cells, further enhancing the drug’s efficacy. Furthermore, studies like those by Liu et al. (2021) 69 have highlighted that HA-modified drug delivery systems not only improve cancer cell targeting but also modulate the tumor microenvironment, thereby facilitating a more substantial apoptotic response. Our study aligns with these findings, indicating that the modified delivery system could lead to a more significant reduction in cell cycle progression and increased apoptosis.
Our investigation into the roles of Bcl2 and cleaved caspase-7 in breast cancer treatment revealed significant insights into apoptotic processes influenced by different treatment regimens. The negative correlation between Bcl2 expression and cleaved caspase-7 levels aligns with previous studies that highlight Bcl2 as a prominent anti-apoptotic factor, where lower levels of Bcl2 and heightened presence of cleaved caspase-7 serve as indicators of effective apoptosis induction. This is consistent with findings by Namjoo et al. (2023), 57 suggesting that these protein expression patterns can serve as favorable prognostic factors in breast cancer therapies. The observed increase in cleaved/procaspase-7 in the HA-TMX-SLN treatment group reinforces the notion that encapsulation in SLNs may enhance therapeutic efficacy. Such findings echo previous studies, such as the work by Shen et al. (2015), 64 which demonstrated that nanoparticle-based delivery systems improve drug uptake and thereby augment apoptosis in cancer cells. Furthermore, the involvement of CDKs and their regulatory cyclins in cell cycle progression is critical in understanding the therapeutic landscape in breast cancer. Our measurements indicate reduced levels of CDK4, Cyclin D1, and CDK2 in MCF7 cells, which correlate with reports from Chen et al. (2003) 70 and Susanti & Tjahjono (2021), 71 which illustrate that inhibition of these proteins can cause cell cycle arrest at various checkpoints, thereby enhancing sensitivity to apoptotic stimuli. Notably, the reduced activity of CDK2 observed in our study further corroborates the impacts of DNA damage checkpoints and highlights its role in promoting apoptosis. The findings regarding cell cycle analysis, specifically the decreased proportion of MCF7 cells in the G0/G1 phase upon treatment with HA-PTX-SLNs, suggest a potential mechanism by which these treatments mediate their effects. This is similar to observations by Güney Eskiler (2018), 72 where alterations in cell cycle distribution were evident following therapeutic interventions, emphasizing the necessity of understanding cell cycle dynamics in optimizing treatment strategies.
Regarding potential limitations of the study, we acknowledge that the lack of in vivo validation is a significant limitation. Future studies should aim to investigate the efficacy of HA-TMX-SLNs in animal models and ultimately in clinical trials. Additionally, the study’s focus on a single breast cancer cell line (MCF7) may not be representative of all breast cancer subtypes. Future studies should aim to investigate the efficacy of HA-TMX-SLNs in multiple breast cancer cell lines and patient-derived xenograft models.
In terms of future directions for research, we suggest exploring the potential of HA-TMX-SLNs in combination with other therapeutic agents, such as chemotherapy or immunotherapy, to enhance their therapeutic efficacy. Additionally, the development of HA-TMX-SLNs with targeted delivery to specific breast cancer subtypes or patient populations could further improve their clinical relevance.
Conclusions
This study reveals that HA-coated TMX-SLNs may offer significant advantages in cancer treatment. Specifically, the HA-TMX-SLN formulation enhances the release of TMX and increases its apoptotic activity against MCF7 breast cancer cells by targeting the CD44 receptor. This targeted drug delivery system has the potential to improve treatment efficacy and cytotoxicity. The incorporation of HA into SLNs demonstrates high biocompatibility and represents a promising approach to cancer therapy.
While our findings are promising, they are not without limitations. A major limitation is the use of a single breast cancer cell line (MCF7) and the lack of evaluation of other breast cancer subtypes. Additionally, in vitro studies may not fully translate to in vivo settings, and further investigation is needed to confirm the efficacy and safety of HA-TMX-SLNs in animal models and clinical trials.
To address these limitations, future research should aim to expand the study to include multiple breast cancer cell lines and subtypes, as well as conduct in vivo studies to evaluate the therapeutic potential of HA-TMX-SLNs. Furthermore, exploring the optimal HA-TMX-SLN formulation and dosing regimen will be crucial to maximizing its therapeutic benefits. By addressing these limitations, future research can build upon our findings and provide a more comprehensive understanding of HA-TMX-SLNs in breast cancer therapy.
Supplemental Material
Supplemental Material - Enhancing breast cancer treatment: Evaluating the efficacy of hyaluronic acid-coated tamoxifen-loaded solid lipid nanoparticles on MCF7 cells
Supplemental Material for Enhancing breast cancer treatment: Evaluating the efficacy of hyaluronic acid-coated tamoxifen-loaded solid lipid nanoparticles on MCF7 cells by Niloufar Ghayoumipour and Hossein Ghafouri in Human & Experimental Toxicology.
Footnotes
Acknowledgments
The authors thank the University of Guilan for their kind financial support.
Author contributions
Niloufar Ghayoumipour: Investigation, Formal analysis, Data curation, Writing –original draft, Methodology. Hossein Ghafouri: Conceptualization, Supervision, Project administration, Writing – review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the author(s) did not use any tools.
Supporting information
The original western blot images are provided in the Supporting Information File.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

