Abstract
Introduction
Organophosphate pesticides (Ops) like diazinon (DZN) have well-known neurotoxic effects and low-level chronic exposure has been linked to detrimental neurobehavioral impairments and memory deficits. However, it’s not entirely clear how DZN-induced biological changes, particularly in the prefrontal cortex (PFC) contribute to these effects. The purpose of this study is to investigate the impact of DZN exposure on inhibitory avoidance (IA) memory function, amyloid precursor expression (APP), and proinflammatory tumor necrosis factor-α (TNF-α) levels in the rat cortex.
Materials and Methods
Rats were divided into 4 groups and recived 2 mg/kg DZN for 5-days or 12-weeks and two control groups recived the same volume of vehicle. IA memory was assesed using the shuttle box apparatus. Rats were sacrificed and the prefrontal cortex PFC were removed. Real-time PCR and Western blotting were used to messure TNF-α, and amyloid protein precursors gene expression and protein levels.
Results
Our findings indicated that DZN caused body weight loss and a notable decline in performance on the IA memory. Additionally, 5-days exposure increased APP and APLP2 protein levels in the PFC, while 12-weeks exposure decreased these levels. Furthermore, expression of APP and APLP2 gens were decreased in PFC. TNF-α levels increased as a result of 5-days exposure to DZN, but these levels dropped to normal after 12-weeks administration, and this observation was significant.
Conclusion
Taken together, exposure to low doses of DZN leads to disturbances in IA memory performance and also alternations in amyloid beta precursors that can be related to increased risk of Alzheimer’s disease.
Introduction
Diazinon (DZN) is an organophosphate compound that has found extensive use as a pesticide in both agricultural and non-agricultural contexts, primarily for the control of various pests and insects. There has been growing apprehension regarding its environmental impact and potential hazards to human health. The World Health Organization (WHO) classifies DZN as a medium-risk insecticide, and as such, its usage is subject to increasing regulation. 1 DZN achieves its harmful effects by inhibiting acetylcholinesterase, an enzyme vital for the neural functioning of insects. Nonetheless, this mechanism can inadvertently impact non-target species, including humans. 2 Emerging evidence suggests that organophosphate pesticides such as DZN may adversely affect the human brain through additional mechanisms, including inflammation, 3 oxidative stress, 4 disruption of the endocrine system, 5 and alterations in brain lipid metabolism. 6 Notably, long-term exposure to organophosphates, even at levels below those necessary to induce cholinergic toxicity, might trigger these effects. 7
DZN poisoning can manifest in both acute and chronic forms, owing to its nature as an organophosphate pesticide. Acute poisoning generally results from exposure to a substantial quantity of DZN over a brief period and is characterized by the inhibition of acetylcholinesterase, essential for nerve function. 8 Of note, chronic exposure to DZN at lower levels over an extended duration can also have harmful effects. Chronic DZN poisoning is more subtle and can culminate in long-standing health issues such as neurological disorders, which manifest as memory and concentration difficulties, peripheral neuropathy, cognitive impairments, and mood disorders. 9
Furthermore, the Amyloid Precursor Protein (APP) family, consisting of APP and Amyloid Precursor-Like Protein 2 (APLP2), has been implicated in cognitive impairment and Alzheimer’s disease (AD) pathology. APP and APLP2 share structural similarities and are expressed in the brain, with APLP2 demonstrating broader expression. 10 Both proteins are integral to synaptic plasticity and maintenance, especially in relation to neuronal survival and axonal growth. 11
In AD, the dysregulation of APP and APLP2, such as abnormal cleavage, contributes to the formation of beta-amyloid plaques and neurofibrillary tangles. These structures induce neuronal dysfunction, ending up in cognitive impairment. Furthermore, alterations in gene expression and mutations in APP and APLP2 are also contributory factors. 12 On the other hand, the chronic production of tumor necrosis factor-α (TNF-α) by microglia provokes a neuro-inflammatory response associated with AD. TNF-α has been proposed as a main regulator of the immune response in many organs, such as the brain. This inflammation which is mediated through TNF-α may also contribute to amyloid-β (Aβ) plaques and tau protein hyperphosphorylation (tau) identified to accumulate in the brains of AD patients. 13
Another critical aspect is the role of prefrontal cortex (PFC) in AD. 14 This area contributes to a wide variety of brain functions, including high-level cognitive functions, decision-making, problem-solving, planning, and social behavior, and is instrumental in memory processing. As AD progresses, β-amyloid plaques tend to accumulate in various brain regions, including the PFC, leading to a decline in cognitive functions associated with this area.
Despite a great number of investigations done around the neurodegenerative effects of DZN, the fundamental mechanisms of neurodegeneration induced by long-term exposure to this pesticide have not been entirely clarified. In this study, we aim to explore the consequences of both acute and chronic exposure to DZN on inhibitory avoidance memory. Additionally, we investigate the influence of DZN exposure on the levels of TNF-α, APP, and APLP2 within the PFC.
Methods and material
Animals
Adult male Wistar rats in apparently good health, weighing approximately 220 ± 5 (6 weeks of age) was obtained from the animal house at the Institute of Pasteur in Tehran, Iran. Throughout the experiment, rats were kept in cages under standard laboratory conditions (controlled temperature 22 ± 2°C, relative humidity 50 %, and 12/12 h dark-light cycle). Prior to starting the experiments, the rats were allowed to acclimate to these conditions for a period of 1 week. All experimental procedures conducted in this study received ethical approval from the Medical Ethics Committee of Ardabil University of Medical Sciences under the approval code IR. ARUMS.REC.1399.540.
Experimental design
Diazinon, specifically O, O-diethyl O-[4-methyl6-(propane-2-yl) pyrimidin-2-yl] phosphorothioate, was acquired from Alpha Chemistry, Tehran, Iran. A total of 28 animals were included in the study, divided evenly into 4 experimental groups (N = 7). All rat were randomly divided into the four following groups with 8 animal per group and supplemented orally through gavage tube, once a day: Vehicle (olive oil 100 % vehicle treated-group, 0.5 mL, for 5 days), DZN (low dose DZN-treated group, 2 mg/kg per day dissolved in 1 mL olive oil, for 5 days), Vehicle (olive oil 100 % vehicle treated-group, 0.5 mL, 5 days a week for 12 weeks), and DZN (low dose DZN-treated group, 2 mg/kg per day dissolved in 1 mL olive oil, 5 days a week for 12 weeks). DZN dosages were selected according to the previous studies and below the threshold to exerts cholinergic problems.
15
Additionally, daily monitoring was carried out to promptly identify any abnormalities, instances of mortality, and alterations in behavior patterns. For a visual representation of the experimental timeline, refer to Figure 1. Study timeline. PND: Post-natal day, IAM: Inhibitory avoidance memory test.
Inhibitory avoidance memory test
The inhibitory avoidance memory (IAM) test serves as a widely acclaimed technique to analyze fear-driven memory in laboratory rodents (see Figure 2). It employs a shuttle box apparatus, measuring (20 * 80 * 20 cm), with two separate compartments divided by a guillotine door. One compartment is well-illuminated, and the other is kept dark. Both compartments feature stainless steel floors, while the dark compartment additionally includes an external stimulator to deliver electrical shocks. It was ensured that the apparatus was thoroughly cleaned and devoid of any residual odors or debris prior to each session. The experiment consisted of three main phases: habituation, training, and testing. In the habituation phase, the rodent was initially placed in the bright compartment, where it was given 10 s for exploration. The guillotine door was then raised to grant the rodent access to the dark compartment. Once the rodent entered, the door was closed, and the animal was allowed 30 s to explore the dark space. This procedure was repeated after a 30-min interval. Following another 30-min break, the training phase began. The rodent underwent the same steps as in the habituation phase; however, this time, upon entering the dark compartment, it was subjected to an electrical shock, administered at 0.5 mA for 3 s 48 h post-training, the testing phase took place. The rodents were placed in the bright compartment and, after 10 s, the guillotine door was lifted.
16
The shuttle box apparatus. This apparatus is made of two compartments, one dark and the other light, separated by a guillotine door. (Figure drawn in Krita Version 5.1.)
During the subsequent 10 min, the time taken for the rodent to enter the dark compartment, referred to as Step-Through Latency (STL), the cumulative duration spent inside the dark compartment, and the total number of entries into the dark compartment were recorded. A maximum cut-off point of 600 s was evaluated. At all tests, sound was masked with aquatic conditioned.
Molecular assessments
RNA extraction and qRT-PCR
The sense and antisense sequences of the primers used in quantitative reverse transcription-polymerase chain reaction (qRT-PCR) test for evaluating related gene expressions.

Western blotting
The tissue cells were harvested and washed with PBS. The total protein was extracted with RIPA lysis buffer (Santa Cruz Biotechnology) according to the manufacturer’s instructions. Briefly, the tissue was resuspended in a lysis buffer, containing the protease inhibitor, Sodium Deoxycholate, NaCl, SDS, EDTA, Triton NP40 (1%) and Tris-HCL. The suspension was mixed and vortexed for 10 s, then incubated on ice for 10 min. Afterward, the lysates were centrifuged at 12,000 r/min for 10 min at 4°C. Subsequently, 0.12 mL of samples was separated on 5% stacking and 10% running gel on SDS‐PAGE electrophoresis and blotting into the polyvinylidene difluoride (PVDF; Roche Diagnostics) membrane via the semidry immunoblotting. The membranes were blocked with 0.5% Tween 20 in PBS for 2 h on a shaker at room temperature and then incubated with a monoclonal antibody against TNF-α, APP, APLP2, and β‐actin as a reference protein overnight at 4°C. Then, after washing of PVDF, the membranes were incubated with rabbit anti-goat secondary antibody for all proteins. (1:5000; diluted in PBS) for 45 min at RT under the shaking condition. The protein bands were visualized using the electrochemiluminescence kit (ECL advanced reagents) and western blot imaging system.
Statistical analysis
Statistical analysis was performed utilizing GraphPad Prism version 8. The data is presented as the mean ± standard error of the mean (SEM). To assess the significance of variations in data, two-way analysis of variance was employed. In cases where there were significant differences, further post hoc analyses were conducted using the Tukey test to compare between the groups. The threshold for statistical significance was established at p < .05.
Results
Effect of exposure to DZN on body weight gain
Throughout the experiment, there were no occurrences of death for any of the animals being observed. The effect of exposure was analyzed using repeated-measures ANOVA, which revealed a significant impact of exposure to DZN (F
1,12
= 37.334, p = .001) on body weight gain over a period of 12 weeks. Figure 3 depicts the results of the comparison of the two groups - the control group and the group exposed to DZN. The comparison indicates a progressive increase in body weight difference starting from the seventh week (p < .05). The average body weight of the animals decreased drastically as a result of being exposed to DZN (2 mg/kg). (* for p < 0. 05; ** for p < 0. 01 and *** for p < 0. 001). Cont: Control; DZN: Diazinon.
Effects of exposure to DZN on inhibitory avoidance memory test
The effect of day on step through latency (STL) (F
1,28
= 30.023, p = .001), the effect of exposure to DZN (F
1,28
= 240.893, p = .001) and the interaction between the two factors (F
1,28
= 19.029, p = .001) were found to be significant. The effect of group was also found significant (F
3,28
= 96.648, p = .001). A significant decrease in STL was observed in the 5-days and 12-week exposure in comparison to the control groups (p = .001 for both comparisons). This decrease was larger in the 12-weeks DZN group in comparison to the 5-weeks DZN group (p = .001) (see Figure 4(a).). Effect of day on total dark time (F
1,28
= 10.817, p = .003), effect of exposure to DZN (F
1,28
= 242.244, p = .001) and the interaction between the two factors (F
1,28
= 6.180, p = .020) were all significant. The effect of group (F
3,28
= 86.414, p = .001) on total dark time was found significant. Post-hoc analysis resulted in a significant increase in the 5-days and 12-week exposure groups in comparison to controls (p = .001 for both). This increase was larger in the 12-weeks DZN group in comparison to 5-days DZN group (p = .002) (Figure 4(b).). Number of entries into the dark compartment (shown in the graph as # entries into the dark compartment) is shown in Figure 4(c). Analysis failed to show a significant effect of day (F 1,28 = 0.175, p = .679) on number of crossings, but effect of exposure (F
1,28
= 5.630, p = .026) was significant. The interaction between the two factors (F
1,28
= 0.019, p = .890) and the effect of group (F 3,28 = 1.942, p = .150) were insignificant. Post-hoc analysis failed to show a significant observation between the groups. Effect of exposure to diazinon (2 mg/kg) on inhibitory avoidance memory test. Exposure to DZN decreased step through latency and increased total dark time. (*** for p < 0. 001 and ** for p < 0. 01). Cont: Control; DZN: Diazinon.
Effects of exposure to DZN on TNF-α, APP and APLP2 gene expression
Significant changes were observed in mRNA levels between the DZN-treated and control rats as indicated by RT-PCR analysis (n = 5). The results of two-way ANOVA indicated that the effect of exposure to DZN (F
1, 20
= 4.080, p = .060) was insignificant, but the effect of day (F
1, 20
= 64.758, p = .001) was significant on the relative APP expression level. The interaction between these two factors was also significant (F
1, 20
= 64.758, p = .001). Additionally, the group factor had a significant effect (F
3, 20
= 44.532, p = .001). Post-hoc analysis of the experimental groups showed a significant decrease in APP expression levels following 12 weeks of DZN exposure (p = .003). Interestingly, a 5-days exposure period induced an increase in APP expression levels (p = .001) (Figure 5(a).). The APLP2 expression levels were analyzed using two-way ANOVA, which revealed that exposure to DZN had no significant effect (F
1, 20
= 0.831, p = .375). However, day had a significant effect (F
1, 20
= 45.627, p = .001), and there was a significant interaction between the two factors (F
1, 20
= 45.627, p = .001). The effect of group was also found significant (F
3, 20
= 30.695, p = .001). Further analysis showed that APLP2 expression levels decreased after 12-week of exposure to DZN (p = .001), while a 5-days exposure increased APLP2 levels (p = .004), similar to the effect observed with APP (Figure 5(b)). An evaluation of TNF-α expression levels was carried out, and the results showed that DZN exposure (F
1, 20
= 397.684, p = .001), duration of exposure (F
1, 20
= 167.527, p = .001), and their interaction (F
1, 20
= 167.527, p = .001) had a significant effect. The group factor also had a significant effect (F
3, 20
= 244.246, p = .001). Post-hoc analysis indicated that TNF- α levels increased following a 5-days exposure period (p = .001). However, the levels dropped in 12 weeks of exposure (Figure 5(c).). Although the long-term exposure group’s TNF-α levels rebounded, they remained higher (p = .001) (Table 2). Effects of exposure to DZN (2 mg/kg) on (a) APP, (b) APLP2, and (c) TNF-α gene expression in the PFC. A decrease in APP and APLP2 levels was observed in 12-week of exposure to DZN while 5-days exposure increased these levels. On the other hand, 5-days exposure to DZN increased TNF-α levels. (** for p < 0. 01, and *** for p < 0. 001). Cont: Control; DZN: Diazinon. Effects of exposure to DZN (2 mg/kg) on gene expression results. Ns: not significant.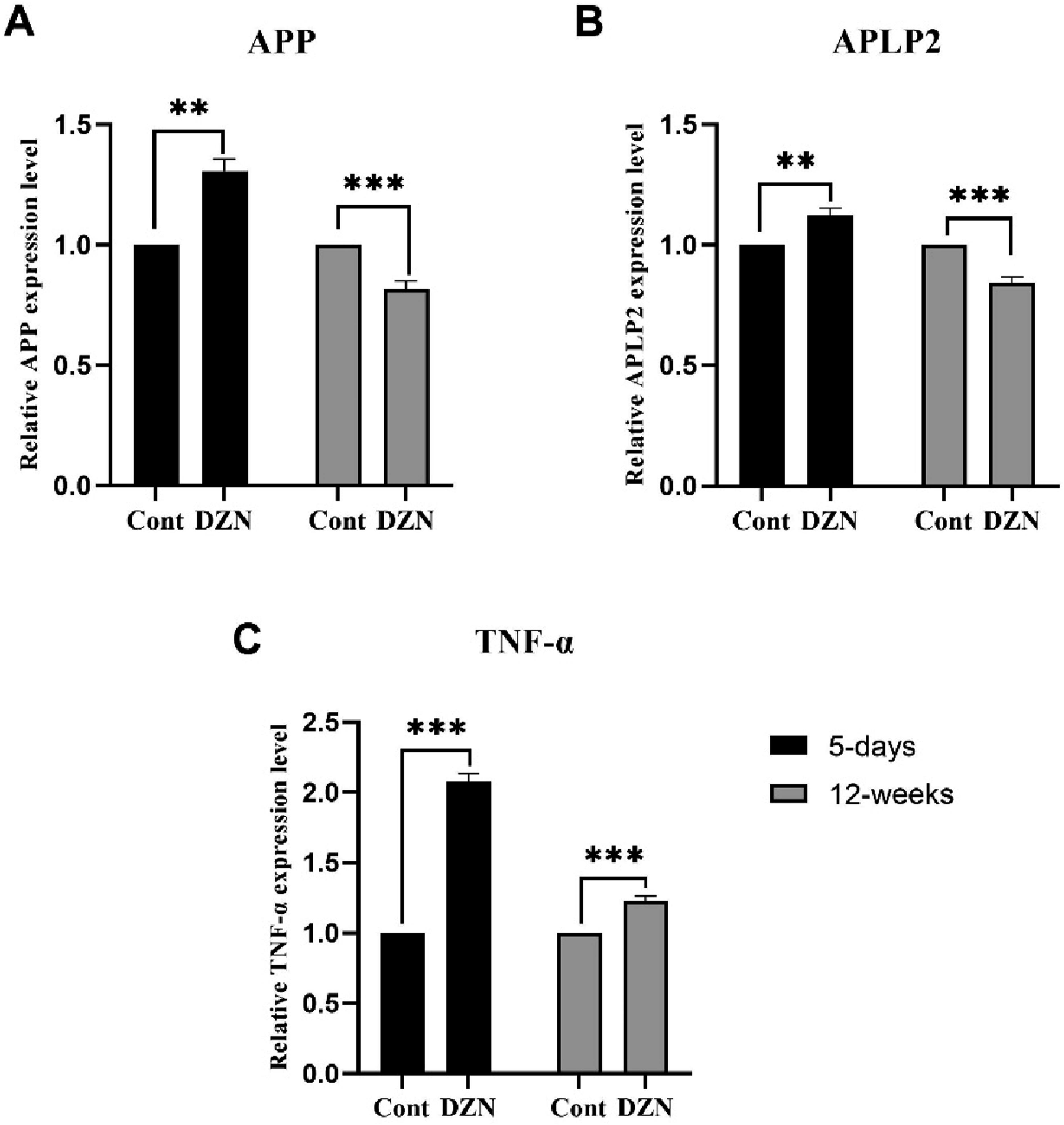

Effects of exposure to DZN on TNF-α, APP and APLP2 protein levels
A series of western blotting experiments (N = 5) were subjected to comprehensive a two-way ANOVA analysis. The findings underscore that duration of exposure significantly influenced APP levels (F 1, 20 = 110.086, p = .001), whereas the impact of exposure to DZN (F1, 20 = 1.116, p = .307) and the interaction between these two factors (F 1, 20 = 8.3419, p = .011) remained statistically insignificant. The influence of group was marked as significant (F3,20 = 39.847, p = .001). Notably, as depicted in Figure 6(a)., the PFC APP level was notably lower in rats exposed to DZN for 12 weeks compared to the control group (p = .05). Conversely, 5-days exposure did not yield any significant impact on APP levels (p = .579). The outcomes of two-way analysis on APLP2 levels indicated a significant effect of duration of exposure (F 1, 20 = 99.357, p = .001), but effect of DZN exposure (F 1, 20 = 0.002, p = .967) was found insignificant, with the interaction between these factors (F 1, 20 = 21.282, p = .001) on APLP2 levels in the PFC being found significant. The impact of the group remained significant (F3,20 = 40.214, p = .001). Exposure to DZN for 12 weeks resulted in a reduction of APLP2 levels (p = .021), while short-term exposure led to an increase in these levels (p = .024), as shown in Figure 6(b). Furthermore, the results of the two-way ANOVA unveiled the significant influence of DZN (F1, 20 = 17.667, p = .001), the effect of exposure duration (F 1, 20 = 4.950, p = .041), and the interaction between exposure duration and DZN exposure (F1, 20 = 10.766, p = .005) on the expression of TNF-α were deemed significant. The impact of group was again highlighted as significant (F3,20 = 11.128, p = .001). The data indicated that a 5-days exposure period led to heightened TNF-α levels in the PFC, in comparison to the control group (p = .001). With prolonged exposure, these levels subsided and became comparable to those observed in the control animals (p > .05), as shown in Figure 6(c). The figure shows the effects of exposure to DZN (2 mg/kg) on protein levels in the PFC. The figure demonstrates a decrease in APP and APLP2 levels after 12 weeks of exposure to DZN (p < .05). 5-days exposure to DZN increased the levels of TNF-α and APLP2. (* for p < 0. 05 and *** for p < 0. 001). Cont: Control; DZN: Diazinon.
Discussion
Prominent neurotoxic effects of DZN through inhibition of acetylcholinesterase activity in nervous system which impair mood, spatial learning, and memory function, are well reported in literature.9,17 Besides the anticholinergic toxicity caused by DZN, there are now a great body of evidence proposing the involvement of non-cholinergic mechanisms in cognitive impairment following long-term exposure with low doses of DZN. 18 There are influential pieces of evidence referring to various cellular and molecular mechanisms which are responsible for the harmful consequences of sub-chronic DZN exposure such as oxidative stress 19 and dysfunctions in neurotransmitter synthesis, degradation, and transport.20,21 Furthermore, accumulating evidence has been obtained from human and experimental studies suggesting that neuroinflammation plays a key role in the pathogenesis of neurodegenerative diseases such as AD.21,22
In this study, we observed that-low doses of chronic oral exposure to the pesticide DZN, below the threshold to significantly affect AchE activity, during adulthood impaired IA memory. Furthermore, the expression of genes associated with AD in brain regions associated with neurobehavioral functions were altered due to chronic exposure to DZN. In addition, we reported an increased expression of TNF-α levels in the PFC following 5 days exposure of DZN.
We determined that at the end of the 12-weeks exposure, DZN-treated rats failed to gain body weight in comparison to control animals. It was assumed that this body mass loss might be associated with a reduction in food and water intake due to a decrease in appetite as a consequence of cholinesterase inhibition in the brain and plasma. Moreover, DZN exposure induced lipids degradation and protein catabolism. In general, this significant weight loss occurs by a lack of nutrients which can cause further metabolic stress, possibly resulting in brain dysfunction. 23 Previous investigations examined levels of OPs that significantly inhibit AChE activity and reported neurobehavioral consequences such as memory impairment in adulthood.24–27 In addition, it is shown that even a brief neonatal exposure to DZN at low levels below the threshold for cholinesterase inhibition can produce long lasting negative effects on glutamatergic function, 28 passive avoidance learning 29 and novel object recognition. 28
Our current findings demonstrate that exposure of rats to the DZN, in a subthreshold dose and in an absence of any signs of overall toxicity, produces alterations in IA memory performance in both 5 days and 12 weeks exposure. Our findings suggest that DZN affect APP systems that support memory function, implying participation of mechanisms other than their common inhibition of cholinesterase.
OPs have also shown to alter the expression of genes associated with synthesis, storage, and degradation of various neurotransmitters systems including cholinergic, GABAergic, serotonergic, glutaminergic, and endocannabinoids. 30 In previous investigations, DZN exposure altered many of the hippocampal genes which are responsible for neurotransmission. For instance, DZN cause a down-regulation in the genes encoding the GABAB1 receptors as a fundamental candidate in the recognition memory formation. 31 In this study, APP and APLP2 mRNA expression were evaluated in the PFC of rats which is believed to be involved in the pathophysiology of AD. 32 Our results indicated that DZN at low doses increases APP and APLP2 mRNA expression in PFC during 5 days of exposure.
It’s important to note that APP-mediated signaling cascade inhibits memory deficit in AD and plays essential role in the pathophysiology of AD, 33 and mediates cognitive functions. 34 According to biochemical and genetic reports, APP processing could be helpful in the progression of therapeutic targets in AD treatment. 35 Our findings support this hypothesis as we observed alternations in APP expression in PFC induced by DZN exposure that could contribute to consequent memory deficit.
Besides downregulation of APP and APLP2 gens, the decrement in related proteins due to DZN exposure was observed confirms that DZN can affect cognitive processing through APP cleavage by activating β- and γ-secretase into APP-like proteins. 36 Interestingly, there has been a growing interest in APLP2 potential role as a biomarker for AD. 37 Although APLP1 seems to be related to synaptic plasticity and activity, the direct contribution of this parameter in AD progression is not stablished. 38
The present study evaluated the level of TNF-α production following short and long-term exposure to DZN. Our results showed that the secretion of TNF-α were increased significantly in PFC after 5-days of exposure with DZN compared to control group, however, in long-term exposure the level of TNF-α was decreased. In agreement with the results of a study by García-Lestón et al. who reported that 148 males who work in jobs that involve lead have revealed significant reductions in the serum of CD4+/CD8+ T cells ratio and in the levels of TNF-α and IFN-γ, while an increase in the proportion of CD8+ T cells and the IL-10 levels were observed. 39 In another study, it was shown that long-term exposure to uranium led to significant changes in the level of cytokines like TNF-α reduction which released by stimulated cells in the mice. 40 Similarly, in mammals, long-term cadmium exposure can result in reduced expression of cytokines, such as IL-1 and TNF-α, and inhibition of macrophage activity. 41
It should be noted that, Th1 cells have a critical role in the formation of cellular immunity and mediate the immune response related to cytotoxicity and local inflammation. 40 Th1 cells also stimulate macrophages to induce their secretion of pro-inflammatory cytokines, so yielding the above-described results, decreased TNF-α secretion-which are adequately explained by the suppression of the production of Th1 cytokines. This study hypothesized that with long time exposure, DZN may cause a macrophage inhibition and systematic shift of Th1 cytokines to Th2 cytokines. This concept should be explored further.
Conclusion
Our current findings demonstrate that exposure of rats to the DZN, in a subthreshold dose and in an absence of any signs of overall toxicity, produces alterations in IA memory performance, proinflammatory cytokines, and APP proteins. One limitation of the present work is the lack of the evaluation of beta-secretase enzyme activity, which could further confirm thar DZN contributes to cognitive impairment through APP cleavage caused by activating β- and γ-secretase into APP-like proteins, APLP1 and APLP2. Furthermore, evaluation of prefrontal cortex dendritic spines neurons could shed more light on the mechanisms by which DZN affects IA memory.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ardabil University of Medical Sciences.
