Abstract
Introduction
Pancreatic cancer and cervical cancer are among the most common cancers. Brown algae have anti-inflammatory, anti-cancer, anti-fungal, antioxidant, and immune-boosting properties. This study investigated the antioxidant properties and the effect of brown algae extract on pancreatic and uterine cancer cells.
Materials and methods
In this study, Cervical (Hela) and pancreas (Paca-2) cancer cell lines were examined. The algae materials were extracted by sequential maceration method and amount of fucoxanthin content in the sample was determined by using High Performance Liquid Chromatography (HPLC) system. The cytotoxic effect of different concentrations of brown algae was measured by the MTT assay. All statistical calculations for comparing IC50 were analyzed using Graph Pad Prism software.
Results
the algal sample contained an average of 102.52 ± 0.12 μg of fucoxanthin per 100 g. IC50 for 2, 2-diphenyl-1-picrylhydrazyl (DPPH) and hydrogen peroxide free radical scavenging activity for methanolic extract was 2.02 and 11.98 ± 0.13 respectively. Brown algae in all fractions inhibited cell growth and survival. In Hela cell lines, the methanolic extract was the most effective inhibitor, while in Paca cell lines, hexane and methanolic extracts were particularly potent. The methanolic extract was more toxic than other fractions on Hela and Paca cell lines.
Conclusion
This study highlights brown algae extracts strong anticancer effects on uterine and pancreatic cancer cells, suggesting its potential as a natural anticancer drug. Different fractions of the extract showed superior apoptotic and cytotoxic effects, with higher concentrations leading to increased apoptotic effects and reduced survival rates of cancer cells.
Introduction
Despite preventive and therapeutic measures, cancer remains the second most frequent cause of death worldwide, after cardiovascular diseases.1–3 Tumors are masses of cells that undergo uncontrollable proliferation and can invade other parts of the body, leading to various complications.4,5 The origin of cancer is undetermined; however, genetic and environmental components might be involved.6,7 The most common types of cancer are lung, gastric, bowel, liver, breast, and prostate cancers, with blood, brain, and lymph node cancers being the most common types in children. 8
Pancreatic cancer is relatively rare, with risk factors including smoking, obesity, diabetes, and rare genetic conditions. 9 Unfortunately, pancreatic adenocarcinoma, the most common type of pancreatic cancer, typically has a poor prognosis, with only 25% of people surviving for a year after diagnosis and 5% surviving for 5 years. Early detection can improve the 5-year survival rate to about 20%. 10
Cervical cancer originates in the cervix and is primarily of the squamous cell carcinoma subtype. This cancer is usually asymptomatic in its early stages, and symptoms may include vaginal bleeding and pelvic pain or pain during sexual intercourse. 11 HeLa cancer cells are suitable cell lines for proliferation characterized by unlimited reproduction and the ability to withstand long passages. In Iran, cervical cancer is the second most common cancer among women after breast cancer and is the leading cause of death after ovarian cancer. 12
Using natural substances and extracts taken from nature to induce cell death (apoptosis) in cancer cells is one of the most common treatments to prevent or stop cancer. It has gained recognition due to the increasing incidence of cancer in developing and developed countries.13–15 Research has developed a targeted approach to the biochemical prevention of cancer that aims to stop or reverse the progression of cancer cells without the toxic doses of nutrients and drugs. 16 Medicines of natural origin, including vinca alkaloids, taxanes, podophyllotoxin, camptothecin, and anthracyclines, are used against cancer, and half of the internationally approved antineoplastic drugs are natural drugs or their derivatives. Many of these drugs emerge based on the knowledge gained from studies on naturally available small molecules and macromolecules. 17
Algae are rich in biologically active compounds that interest the pharmaceutical industry. 18 Brown algae is a major group of seaweed that has shown potential anti-cancer properties through research into crude extracts. 19 Fucoxanthin, a carotenoid structure found in brown algae, has various biological activities, including anti-cancer potentials related to its free radical scavenging activity.20,21 Brown algae have been the subject of phytochemical research, leading to the discovery of over 500 new metabolites. Nizamuddinia zanardinii (Schiffner) P.C.Silva (Family: Alariaceae) is a brown alga in the Oman Sea (Chabahar-Tang in Iran), with no reports on its chemical compounds and their effects. 22
Anti-cancer effects of different algae on human breast and colorectal cancer cells found that the compounds present in seaweed can combat these cancers, suggesting that seaweed can be a potential anti-cancer agent. 23 In a separate study, the anticancer properties and ability to induce cell apoptosis were investigated using Gracilaria algae extract. The results indicated that the algal extract increased the lifespan of mice with Ehrlich ascites tumor (EAT) cells and significantly inhibited tumor growth. 24 The marine resources, particularly algae in the south of Iran, notably Chabahar, are valuable but need to be explored with proper planning. 25 Given the global prevalence of uterine and pancreatic cancers, as well as the proven potential effects of brown algae on various cancers, this study aimed to investigate the cytotoxicity of the hydroalcoholic extract of the brown alga N. zanardinii on the uterus (Hela) and pancreas (Paca-2) cancer cell lines.
Materials and methods
Chemicals and materials
Hela and Paca-2 cancer cell lines were prepared by the Pasteur Institute of Iran (Iran). Methanol HPLC grade, quercetin, Gallic acid, 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2, 4, 6-tripyridyl-s-triazine (TPTZ), Folin–Ciocâlteu, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) and fucoxanthin were purchased from Sigma Aldrich (USA). FITC‐Annexin V/propidium iodide (PI) apoptosis detection kit was purchased from Thermo Fisher Scientific (USA). Vitamin C, aluminum chloride, and Dimethylsulfoxide (DMSO) were purchased from Merck (Germany). Dulbecco’s Modified Eagle’s medium (DMEM) and fetal bovine serum (FBS) were purchased from GIBCO (USA). n-Hexane, methanol, and all other chemicals and solvents were of the highest grade commercially available.
Algae materials and extraction
Brown algae N. zanardinii was collected from Tang-Chabahar village (geographical location: 21–25 latitude and 54–59 longitude). A voucher specimen was deposited at the Herbarium of the Faculty of Pharmacy, Mazandaran University of Medical Science, Iran (voucher No: P17-51). Algal samples were cleaned of foreign matter and then, rinsed with sterile water to remove any associated debris. After that algae sample was dried under shade for 6 days and ground into a coarse powder. The algae materials were extracted by sequential maceration method with n-Hexane and then methanol at room temperature, followed by hot water extraction at 90°C for 6 h. The extracts were concentrated under reduced pressure using a rotatory evaporator after filtration. To obtain the dry powder of the extracts, a freeze dryer was employed at a temperature of −45 Celsius for 3 days, after which the extract was pulverized. Finally, the dry extract was sealed in a glass container and stored in the refrigerator, away from heat and light. 26
Preliminary phytochemical screening
The hydro-alcoholic extract of N. zanardinii was screened for the presence of various phytochemicals including carotenoids, alkaloids, sterols and triterpenoids, saponins, anthraquinone glycosides, tannins, and flavonoids. 27
Measurement of total flavonoid content
The total flavonoid content was assessed using the aluminum chloride colorimetric method with slight modifications. In this method, a methanolic sample extract solution (0.5 mL) was combined with 1.5 mL of methanol, 0.1 mL of 10% anhydrous aluminum chloride in methanol, 0.1 mL of 1 M potassium acetate, and 2.8 mL of distilled water. After a 30-min incubation at room temperature, the absorbance of the samples was measured at 415 nm. A calibration curve was prepared using standard concentrations of a methanolic solution of quercetin. The total flavonoid content was then expressed as equivalents of quercetin per gram of dried algae extract. 28
Measurement of total phenolic content
Total phenol content was determined by the Folin- Ciocalteu reagent using standard spectrophotometry and standard curve drawing with some modifications. A sample solution of 0.5 mL was mixed with 2.5 mL of 0.2 N Folin–ciocâlteu reagent. After 5 min, 2 mL of 20% sodium carbonate solution was added, and the mixture was vigorously shaken. The absorbance of the samples was measured at 760 nm after incubating for 2 h at room temperature using a double beam Perkin Elmer UV/Visible spectrophotometer. A calibration curve was created using standard concentrations of gallic acid. The total phenolic content was expressed as equivalents of gallic acid per gram of dried algae extract. 29
Determination of antioxidant capacity
Diphenyl-1-picrylhydrazyl assay
DPPH assay is a common method to evaluate the radical-scavenging activity of chemicals. The dark purple color of DPPH (in the stable form) will be lost when the antioxidants reduce it.
The hydrogen donating or radical scavenging ability of the n-hexane, methanol, and water extracts of mentioned algae was measured as follows:
The amount of 1.5 mL of 0.15 mM DPPH in methanol was added to 1.5 mL of different concentrations of the extract and ascorbic acid as the standard compound. Then the mixtures were incubated at room temperature in the dark for 30 min. Finally, the absorbance was measured at 517 nm. The percentage of inhibition of DPPH radical was calculated according to the following formula:
Scavenging of hydrogen peroxide
A solution of hydrogen peroxide with a concentration of 40 mM was prepared in phosphate buffer at pH 7.4. The concentration of hydrogen peroxide was determined by measuring its absorption at 230 nm using a spectrophotometer. Extracts were added to a solution of hydrogen peroxide (0.6 mL, 40 mM). After 10 min, the absorbance of hydrogen peroxide at 230 nm was measured against a blank solution that contained phosphate buffer without hydrogen peroxide.
31
The absorbance values of the control (
FRAP assay
The determination of total antioxidant activity was performed using the Ferric Reducing Antioxidant Power (FRAP) assay, which stands for Ferric Reducing Antioxidant Power assay. This method, with a modification of the Benzie and Strain method, is based on the reduction of Fe (III) to Fe (II). The FRAP colorimetric method involves the reduction of colorless ferric-tripyridyltriazine (Fe (III)-TPTZ) to a blue-colored complex called ferrous-tripyridyltriazine (Fe (II)-TPTZ). To prepare the working FRAP reagent, 25 mL of 300 mM acetate buffer (pH 3.6), 2.5 mL of 10 mM TPTZ solution in 40 mM HCl, and 2.5 mL of 20 mM Fe
Analysis of fucoxanthin content by high performance liquid chromatography
The amount of fucoxanthin content in the sample was determined by using a KNAUR LPG system HPLC (Germany) equipped with a KNAUR UV spectrophotometric detector, so reversed-phase high-performance liquid chromatography (RP-HPLC) with methanol–acetonitrile (7:3 v/v) as the mobile phase at a flow rate of 1.0 mL/min. The RP-HPLC analyses were performed at 28°C using a C18 column (250 × 4.6 Nucl Eo DUR 100-5 C18). Initially, an aliquot of sample was dissolved in the mobile phase and filtered using a 0.22-μm membrane filter. Then, a portion of the filtered sample was subjected to HPLC analysis. The detection wavelength for fucoxanthin was set at 450 nm. To quantify the fucoxanthin content, a calibration curve prepared using an authentic standard was employed. 32
Cell culture
Experiments were carried out with Hela and Paca-2 cancer cell lines. Cells were cultured in DMEM with 10% fetal bovine serum, 100 μg/mL streptomycin, and 100 IU/mL penicillin. Cell lines were grown in an incubator at 37°C and 5.0%
Cell viability test
The cell viability assessment was conducted using the colorimetric MTT assay, which is a widely used method to measure cell viability and proliferation. In this assay, the reduction of a yellow dye called MTT to purple formazan by the mitochondrial enzyme succinate dehydrogenase reflects the metabolic activity and viability of cells. After exposing the cells to various concentrations of test compounds for 72 h, the culture medium was removed. Then, 50 μL of MTT solution was added to each well and incubated for 72 h at 37°C. The MTT solution was then discarded, and the intracellular purple formazan crystals were solubilized by adding a lysis buffer. Following solubilization, the optical density (OD) was measured at wavelengths of 490 nm and 630 nm using a microplate reader (Stat Fax 2100, USA). 33
Apoptosis assay
To assess the apoptosis pattern, the researchers isolated peripheral blood mononuclear cells (PBMCs) from all subjects using density gradient centrifugation on Ficoll-Histopaque. After isolation, the PBMCs were washed twice with phosphate-buffered saline (PBS). The viability of the isolated cells was determined to be greater than 95% using trypan blue staining. To examine cell apoptosis, a double-staining method was employed using a FITC-Annexin V/propidium iodide (PI) apoptosis detection kit from Thermo Fisher Scientific. The protocol recommended by the manufacturer was followed. Briefly, 1 ×
Statistical analysis
Results are expressed as mean ± Standard deviation (SD). The data was analyzed by one-way ANOVA followed by a Bonferroni post hoc test for multiple comparisons using Graph Pad Prism software (version 2.01. Graph Pad Software Inc. San Diego, California). A p-value less than 0.05 was considered significant.
Results
Phytochemical test results

Total phenol and flavonoid content
Total phenol compounds are reported as Gallic acid equivalents by reference to a standard curve (y = 0.0027x + 0.081,
Antioxidant assays
DPPH and hydrogen peroxide scavenging activity and reducing power in methanolic, hexane, and aqueous extract.

Determination of fucoxanthin by high-performance liquid chromatography method
After injecting various extract concentrations, the maximum absorption peak was observed at 445 nm between 8.5 and 8.7 min, indicating fucoxanthin with maximum intensity. The HPLC chromatogram for standard fucoxanthin resulting from a concentration of 125 μg/ml of the standard is shown in Figure 1. Analysis of fucoxanthin by HPLC resulting from a concentration of 125 μg/ml of standard.
Figure 2 illustrates that adding diluted concentration from the standard to the hexane sample increased the peak height corresponding to the absorption region of fucoxanthin. This suggests that the absorption peak of the sample matched fucoxanthin, and its intensity increased with the standard. As shown in Figure 3, after adding a diluted concentration of the standard to the hexane sample, an increase in the peak height present in the absorption region of fucoxanthin is observed. By calculating the area under the standard curve (y = 0.20115x - 0.04479, Analysis of fucoxanthin by HPLC obtained from the concentration of 1 mg/mL hexane extract. Analysis of fucoxanthin by HPLC obtained from 10 μL of the sample with 1 M concentration and low concentration of Fucoxanthin standard.

The cytotoxicity of different algae extracts on paca and hela cancer cell lines
The results of the exposure to aqueous extracts of algae in Paca and Hela cell lines showed that cisplatin and the extract in all doses except 10 μg/ml in the HeLa cell line significantly reduced cancer cell viability that was significantly different from the control group (p < .0001) The results of the exposure to methanolic extracts of algae in cancer cell lines decreased more in all doses of the extract, which showed a significant difference between the doses of algae extract and the control group (p < .0001). Also, cisplatin (positive control group) had a significant difference compared to the control group, which was also significantly different from each other (p < .0001). Paca and Hela cancer cell lines with different concentrations of hexane extract were evaluated for survival. The cytotoxic effect of algae hexane extract on the HeLa cell line was stronger than Paca, and in the HeLa cell line, almost all doses of algae hexane extract reduced cell viability, which compared to the control group was significantly different. (Figure 4) For the Hela cancer cell line, the survival percentage at a concentration of 1000 μg/mL was 14.19% ± 3.89% for the methanolic extract, 32.52% ± 2.87% for the aqueous extract, and 21.85% ± 3.35% for the hexane extract. Similarly, the survival percentages for the Paca cell line were 39.15% for the methanolic extract, 50.55% for the aqueous extract, and 36.18% ± 7.25% for the hexane extract. Cell survival rate (percentage) of paca and Hela cell lines in the exposure of different doses of algae aqueous extract (A.E), methanolic extract (M.E), and hexane extract (H.E). ****p < .0001, significant difference compared to the control group ***p < .001, significant difference compared to the control group *p < .05, significant difference compared to the control group. Note: the experiment was repeated three times independently with similar results.
Apoptosis level
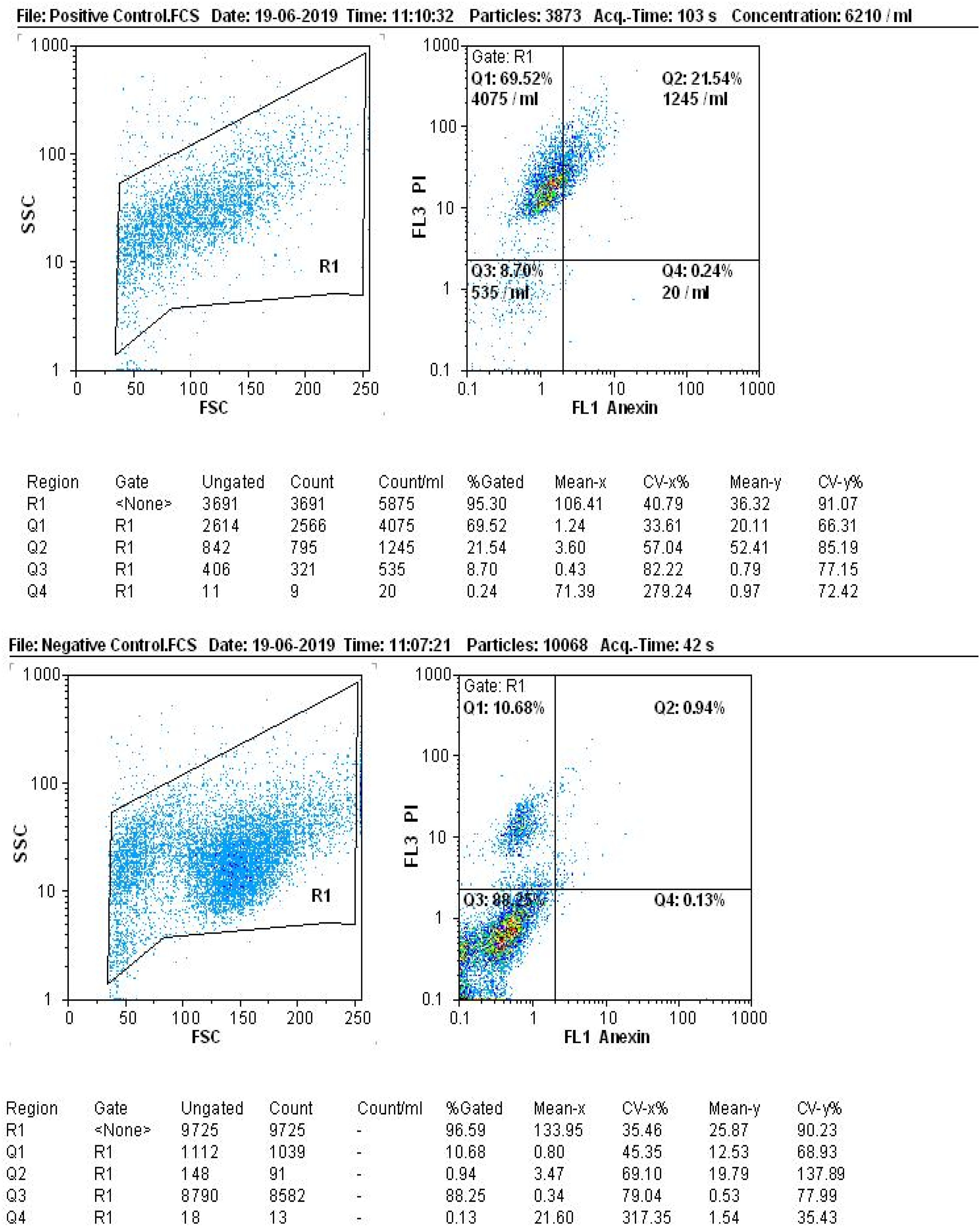
After preparing cell suspensions from the treated cell lines, the assessment of apoptosis incidence in cancer cells revealed that the high dose (1000 mg/mL) of hexane extract resulted in 0.07% primary apoptosis and 10.65% late-stage apoptosis or necrosis in the HeLa cell line. The primary apoptosis rate in the Paca cell line was 0.13%, and the late-stage apoptosis was 7.53% (Figure 5). Similarly, the methanolic extract at a dose of 1000 mg/mL induced 0.01% primary apoptosis and 26% late-stage apoptosis or necrosis in the HeLa cell line, whereas in the Paca cell line, the primary apoptosis rate was 0.02% and the late-stage apoptosis was 34.55% (Figure 6). Notably, the methanolic algae extract exhibited a stronger effect than the hexane extract at a concentration of 1000 mg/mL, virtually matching the efficacy of cisplatin (Figure 7). These results indicate that high-dose apoptosis induction can effectively control cancer cells. The apoptosis rate of hexane extract on HeLa and Paca cell lines using flow cytometry. The apoptosis rate of the methanolic extract on HeLa and Paca cell lines using flow cytometry. The apoptosis rate of aqueous extract and cisplatin on the HeLa and Paca cell lines using flow cytometry.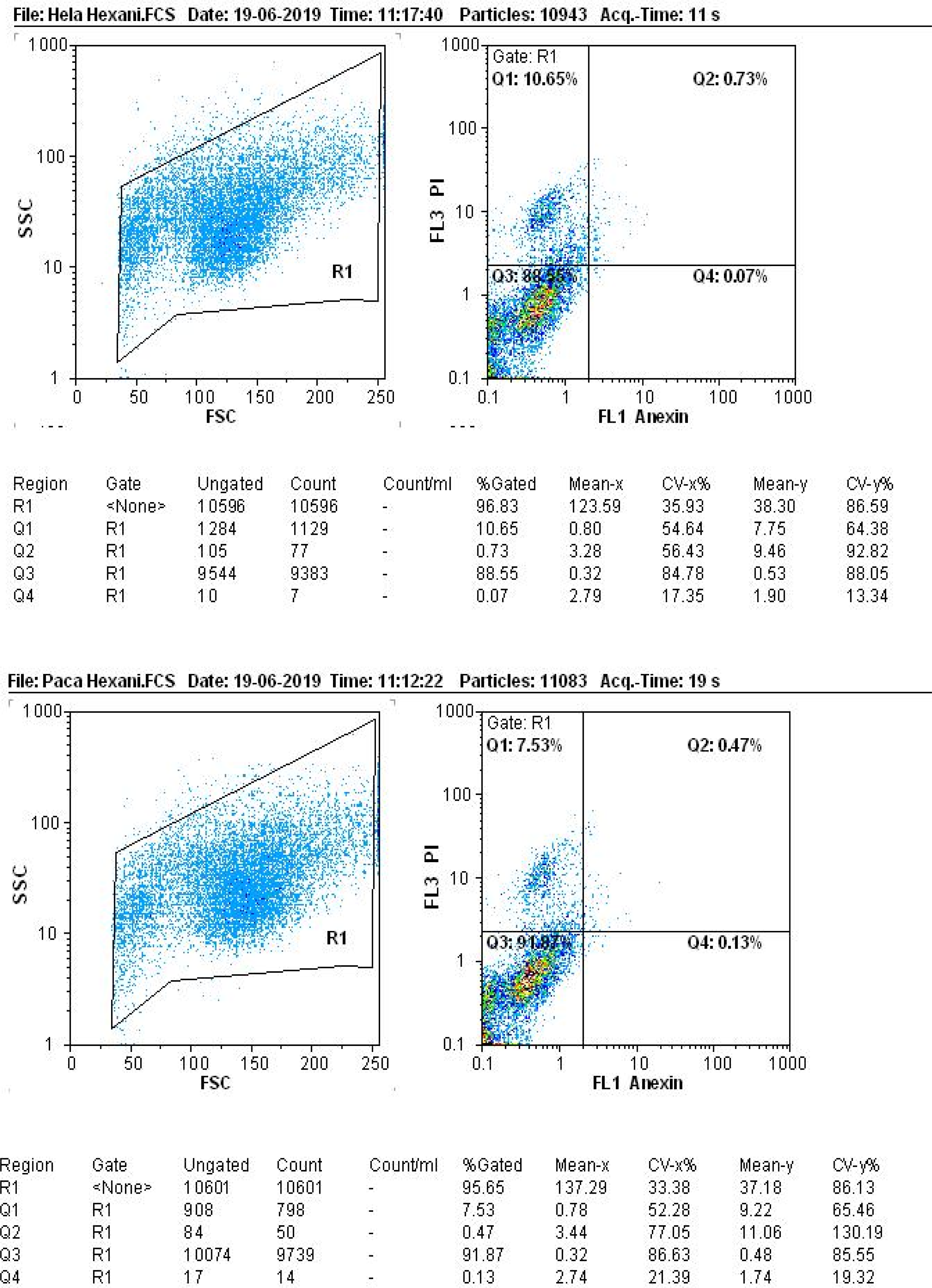

Discussion
The discovery of potent natural products extracted from microorganisms, animals, and plants that effectively target tumor cells while sparing healthy cells is a significant breakthrough in science. 35 Various methods have been developed to assess the effectiveness of natural anti-cancer drugs, with colorimetric assays being widely used due to their ease of use and reliability. 36 Algae, particularly seaweed, are abundant sources of bioactive compounds such as phycocyanin, terpenes, fucosterol, and phenols, demonstrating anticancer properties. 37 These compounds exhibit diverse mechanisms of action, including inhibiting cancer cell growth and metastasis and inducing cancer cell death. 38 This study aimed to investigate the viability and apoptosis rates of HeLa and Paca cancer cells treated with different concentrations of algae extract compared to the untreated cells.
The results of the present study demonstrated that the survival rate of the control samples ranged from 98% to 100%. These findings indicate an inverse relationship between the survival rate of cancer cells and the concentration of extracts. As the concentration of extracts increased, the survival of cancer cells decreased significantly in all extracts at concentrations of 1000, 500, and 100 μg/mL. The extracts had the greatest effect on Hela and Paca cancer cells, resulting in lower survival rates. All fractions of the brown algae exhibited growth and survival inhibition in the cell lines. Specifically, the methanolic extract showed stronger inhibitory effects in the HeLa cell line, while the hexane and methanolic extracts were more significantly effective in the Paca cell line. The methanolic extract demonstrated higher toxicity compared to other fractions in both HeLa and Paca cell lines, which was corroborated by the flow cytometry results. In a study, Methanolic extracts from Pulsatilla patens roots disrupt cancer-related signaling pathways in HeLa cells, inducing apoptosis and inhibiting mitosis. This marks the first identification of active compounds impacting 13 signaling pathways, including transcription factors in cancer processes. The extracts modulate key signaling molecules, preventing excessive proliferation, and inhibiting HeLa cells, hinting at a promising therapeutic mechanism against cancer. 39
The inhibitory activity of the N. zanardinii extract was assessed in a study by measuring its effect on DPPH and superoxide free radicals and results showed a high scavenging ability for superoxide radicals (21.05%), while the DPPH inhibitory activity was 2.3%. 40 In the current study, a higher level of superoxide radical scavenging ability was observed, and the DPPH inhibitory activity was comparable with a slight difference.
Several studies have documented the inhibitory effect of macroalgae on alpha-amylase.41–43 In this study, all samples exhibited alpha-amylase inhibiting activity, and the IC50 values ranged from 1.43 mg/mL to 0.31 mg/mL. These results are consistent with previous findings by Senthil et al., who reported IC50 values ranging from 60 to 83 mg/mL for the inhibition of alpha-amylase in aqueous extracts of Gracilaria cortical, Gracilaria edulis, Ulva lactuca, and Sargassum polycystin. 44 Similarly, Kumar and Soda observed enzymatic inhibition with IC50 values of 81, 73, and 94 mg/mL for ethyl acetate extracts of Gracilaria gracilis, Preciosa Chondrococcus, and Hornemanni Chondrococcus, respectively. 45 It is worth noting that variations in extraction methods, solvents, and algal species across different studies may have contributed to the differences in results. 46 For instance, Lee et al. reported that the IC50 value for alpha-amylase inhibition in the aqueous extract of Ascophyllum nodosum was 10 times higher than that of the alcoholic extract of the same alga. 47 Studies focusing on specific compounds or employing purer extraction techniques have reported more distinct results. For example, Lee et al. reported an IC50 value of approximately 90 mg/mL for alpha-amylase inhibition by phlorotannin extracts from the brown alga Ecklonia cava. 48
In a published study no fucoxanthin content was reported for the brown macroalgae Hicica phosphiformis using a similar HPLC technique to the one employed in the present study, although the specific absorption amount was not provided. 49 Also in a separate study, several Japanese brown macroalgae for fucoxanthin identification and quantification were investigated. They found that 18.7% of every 100 g of xanthophyll in macroalgae contained fucoxanthin, while zeaxanthin accounted for 0.60% of the xanthophyll content. The amount of fucoxanthin per 100 g of xanthophyll in the alga Undaria pinnatifida Suringar was 11.1%. For the macroalgae Eisenia bicyclis, the fucoxanthin content was 7.7% based on 100 g of xanthophyll, whereas Sargassum fulvellum and Hizikia fusiformis Okamura exhibited fucoxanthin contents of 6.5%, 2.2% respectively.
Interestingly, the mentioned study’s maximum absorption peak at 450 nm/10 min closely resembled fucoxanthins absorption time in the Nizamuddinia algae of the present study. 50 In the brown macroalgae Laminaria japonica, where the mobile phase consisted of methanol and water, maximum absorption of fucoxanthin was observed at 10 min using HPLC at 450 nm. The fucoxanthin content on the dry weight of the pigment for U. pinnatifida and S. fusiform was measured at 5.3 mg/100 mg and 10.99 mg/100 mg respectively, indicating a high percentage of fucoxanthin in S. fusiforme, which was lower than the N. zanardinii macroalgae examined in the present study. 51 Another study also reported a different absorption time of 48 min at a wavelength of 445 nm, which significantly differed from the current study findings. 52 Hydroperoxy sterol derived from the methanolic extract of the brown alga N. zanardinii has cytotoxic effects on MCF7, A549, HepG2, and MDBK cancer cell lines, and the compounds exhibited cytotoxicity and apoptotic activity. 53 Additionally, cytotoxicity of the Sargassum glaucescent’s methanolic, chloroform, ethyl acetate, and hexane extracts on the breast cancer cell line (MCF-7) and colorectal cancer cell line (HT-29) at concentrations of 250, 125, 500, and 1000 μg/mL using MTT and Trypan Blue methods were assessed. The results demonstrated a decrease in cell viability with increasing extract concentrations. The methanolic extract at a concentration of 1000 μg/mL exhibited the most significant effect compared to the control and lower extract concentrations. 54 Cytotoxic activity of several brown algae species (Sargassum swartzii, Cystoseira myrica, Colpomenia sinuosa) on cancer cell lines HT-29, Caco-2, T47D, MDA-MB468, and NIH 3T3 revealed that the extracts of these brown algae species collected from the Persian Gulf exhibited apoptotic and cytotoxic properties. 55 Antitumor properties of a brown algae called Padina pavonia were examined on uterine and breast cancer cell lines and methanolic extract of P.pavonia displayed cytotoxic activity with an IC50 of 86.45 μg/mL for uterine cancer cells and 74.59 μg/mL for breast cancer cells. This extract also exhibited apoptotic properties, supporting the cytotoxicity of the algae extract. 36
Various brown algae collected from the British coast exhibited strong antioxidant and antitumor activity on three tumoural cell lines including Daudi, Jurkat, and K562. 56 In a previous study, researchers investigated the apoptotic effects of a brown algae compound, Fucoxanthin, on the HL-60 cell line. The study revealed that this compound induced cell cycle disruption and apoptosis in the HL-60 cell line. 57
Several other studies have also investigated the impact of brown algae on different cancers and consistently highlighted its effectiveness in inhibiting cancer cell growth. These studies include the research conducted by Kim et al. (2010), 58 Vishchuk et al. (2013), 59 Ahn et al. (2014), 60 Kalimuthu et al. (2014), 61 and Aravindan et al. (2015). 62
These findings are consistent with the cytotoxicity and apoptotic activity obtained in this study and further support the results.
Recent studies have indicated that chemical drugs have significant side effects and high costs. Common cancer treatments such as chemotherapy and radiation therapy often lead to adverse effects, such as nausea, vomiting, hair loss, decreased white blood cells, and weakened immune response. Considering the various beneficial properties of seaweed, including its antitumor, anticoagulant, antioxidant, and antiviral activities, it is plausible to consider brown algae as a potentially valuable resource for the future treatment of uterine and pancreatic cancer. 63
Conclusion
In conclusion, the results of this study demonstrate the significant cytotoxic effect and anti-tumor properties of brown algae extract against uterine and pancreatic cancer cells. The findings suggest the potential for further exploration and utilization of the extract in the development of natural anti-cancer drugs. Different fractions of the extract showed superior apoptotic and cytotoxic effects, with higher concentrations leading to increased apoptotic effects and reduced survival rates of cancer cells. These results highlight the importance of considering brown algae extracts for pre-clinical and clinical studies in drug development. Future research should focus on isolating and purifying the active component, evaluating its structure and anti-cancer activity, conducting animal studies, exploring gene expression related to apoptosis (Bcl2 and Bax genes), and investigating the specific pigments present in the algae extract.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

