Abstract
Ozone, an allotrope of oxygen, is enjoying an increasing interest in the setting and management of the medical adjunct treatment, which is called, maybe too simplistically, “ozone therapy”. Ozone is not a medicine, so the word therapy does not properly fit this gaseous molecule. Like many natural compounds, for example plant flavonoids, even ozone interacts with aryl hydrocarbon receptors (AhRs) and, at low doses, it works according to the paradoxical mechanism of hormesis, involving mitochondria (mitohormesis). Ozone, in the hormetic range, exerts cell protective functions via the Nrf2-mediated activation of the anti-oxidant system, then leading to anti-inflammatory effects, also via the triggering of low doses of 4-HNE. Moreover, its interaction with plasma and lipids forms reactive oxygen species (ROS) and lipoperoxides (LPOs), generally called ozonides, which are enabled to rule the major molecular actions of ozone in the cell. Ozone behaves as a bioregulator, by activating a wide population of reactive intermediates, which usually target mitochondria and their turnover/biogenesis, often leading to a pleiotropic spectrum of actions and behaving as a tuner of the fundamental mechanisms of survival in the cell. In this sense, ozone can be considered a novelty in the medical sciences and in the clinical approach to pharmacology and medical therapy, due to its ability to target complex regulatory systems and not simple receptors.
Introduction
On September 22nd 1896, Nikolas Tesla 1 patented the first electro-mechanical device to produce large quantities of pure ozone at one’s own disposal, warranting the possibility to use this triatomic gas in many research and technological fields, including medicine. The story of ozone, a gaseous compound which will be widely used in medical treatment, boasts, therefore, a “beautiful mind” as its father and forerunner, which would represent a possible warranty for an enlightened, promising and challenging future.
To date, the huge expectation on the “magnificent ozone” as a promising compound able to treat any ailment and illness, as a panacea against challenging diseases, deflated slightly, although ozone retrieved many encouraging results, particularly in recent years, due to some prejudice on its controversial effectiveness. The main reason of this prejudicial view comes from the consideration that the majority of physicians are not fully persuaded that a toxic compound, a pollutant and a xenobiotic, may have curative properties. 2 Ozone is not a pharmaceutical drug, it is a triatomic gas, yet, as many other gaseous compounds in the organism, such as CO or NO, even ozone might have a cellular target to exert its function, at least in theory. At the best of our knowledge, no specific ozone receptor, so to enable ozone in exerting its biochemical and signaling action on cells, has been reported so far. Notwithstanding, the literature is particularly clear in reporting that some approaches with which ozone is used as a medical adjunct treatment 3 should be completely forbidden, due to the damaging effect of this gas in certain circumstances. 4 Although anti-oxidant enzymes and oxidative-stress buffering systems are somewhat present also in the lungs,5,6 in the respiratory parenchyma, ozone rapidly disrupts the surfactant, by its strong oxidative reaction with lipids and proteins, so preventing the correct alveolar function of gas exchange and therefore leading lungs to the functional collapse.7,8 Ozone is therefore toxic via inhalation, whereas it is less noxious in those biological liquids that are fully equipped with a powerful anti-oxidant system. Yet, any bioactive compound coming from the outside, called xenobiotic, is a potentially toxic compound, and often its ability to work as a medical effective substance depends on both doses and administration routes. Hormesis, as explained further, is the way by which many xenobiotics behave as positive modulators of the cell survival.
However, though the absence of a receptor-mediated targeted activity may appear as an easy to address pitfall, ozone pleiotropism, which sets ozone close to other xenobiotics such as bioflavonoids, inorganic xenobiotics and other hormesis-causing substances, has long time represented a weakness in the valuable consideration of the many biochemical potentials of ozone in medicine, just due to its absence of a “true” target specificity. The major hallmark of ozone is that a toxic compound, in certain conditions, exerts a beneficial action, a landmark which is the core meaning of hormesis.
On the other hand, ozone is widely used for disinfection, just because of its toxicant nature, a hallmark therefore aside from hormesis.
For example, in periodontitis and gingival inflammatory disorders, ozone is widely used to reduce microbial biofilms. 9 In this circumstance, ozone exerts its pro-oxidant and anti-bacterial activity. 10 Actually, ozone has primarily a pro-oxidant nature, which only at low doses triggers the production of reactive oxygen species (ROS) at a level that makes these reactive compounds signaling molecules, more than damaging agents.
Which is, therefore, the “real” nature of ozone? Is it a Janus-like compound? A change in paradigm in biomedical sciences may be urgently needed to shed light on this molecule in medicine.2,11,12 How toxicants, including physical energies, and able to trigger an oxidative stress response, can be a benefit for the cellular economy, promoting cell survival and leading to health recovery?
This question is of paramount importance, as medical sciences should overcome the orthodox pharmacology represented by the “ligand-receptor” model and try to provide insightful evidence on how potentially toxic xenobiotics are able to elicit a complex, pro-survival response in the cell, even attempting to shed a light on which kind of intriguing message it brings up to our knowledge on evolutionary pathways.
A possible explanation can be obtained by investigating the crucial role of mitochondria in the biological activity of a defined range of ozone doses.
When medical ozone is in contact with biological fluids, such as blood and plasma, it generates ozonides, namely oxidized substances coming from the oxidative reactions of free oxygen radicals, such as •OH- (hydroxyl radical) or •OOR- (peroxyl radical), this latter coming from the interaction of ozone with lipids, on different macromolecules, such as lipoptoteins.13–15 Final oxidized products are malonyl-dialdehyde (MDA) and electrophilic alkenals from poly-unsaturated fatty acids (PUFAs), such as 4-hydroxynonenal (4-HNE) and 4-hydroxyhexenal (4-HHE), from ω6-PUFA and ω3-PUFA, respectively.16,17
Mitochondria, with their ability in building up a new associated structure with intracellular membranes called Mitochondria-Associated Membrane (MAM) system, particularly crucial for autophagy and mitophagy, 18 is the actual “target” of ozone. Therefore, the new medicine, if any, should consider a new class of heterogeneous compounds able to act on the “mitochondria complex” as a target, rather than a receptor.
A first “change of paradigm” in introducing ozone in medicine, at least as an adjunct treatment in conventional therapy and conventional pharmacology, regards the functional consideration of toxic by-products, such as reactive oxygen species (ROS) or 4-HNE, to show some examples. These molecules, directly coming from ozone treatment, work as “signaling” factors, able to finely regulate fundamental steps in cell survival, replication, differentiation and growth, usually via the mitochondria involvement.19,20 In this perspective, they behave as physiologically tuned modulators of the major cellular functions, thereby affecting numerous systemic processes and symptoms such as immunity, infections, cardiovascular function, pain, fatigue, wound repair, neurological activity, degenerative diseases, aging and inflammation.21,22 This is why ozone is prejudicially considered “too” generic to have some distinct medical property, as a pharmaceutical drug can yet do, and therefore often neglected as a treatment proposal.
In aging, ozone exerts its ability in eliciting pro-survival mechanisms, which usually are induced by low doses of this compound, 23 and which may activate complex regulatory systems involving mitochondria turnover and other regulatory molecules usually associated with stress such as H2O2 and lactate. 24
Most of the finesse in using ozone in the so-called “ozone therapy”,25,26 which is not a therapy but a medical adjunct treatment, lies on the physician’s ability in tailoring sound and effective protocols with proper ozone dosages and sound, standardized administration routes. The purpose is to elicit the production of ROS and other mediators, such as 4-HNE, in a way able to induce their “signaling” property, rather than intrinsic toxic activity.
The mechanism by which a xenobiotic, a plant polyphenol or a chemical pollutant, such as ozone, act beneficially in the cell is known as “hormesis”.16,27 Likewise polyphenols and other phenol-derivatives, even ozone (O3), though being a simple gaseous molecule, participates in the hormetic mechanism by interacting with the aryl-hydrocarbon receptors (AhRs),28,29 which translate the xenobiotic-stimulus into a hormetic response in mitochondria, where AhRs are present. 30 In this sense, ozone may work as a natural compound.
Ozone in immunity, pain and inflammation. Role of mitophagy
How ozone can work as a beneficial molecule?
A first leading concept regards the doses of ozone used in the adjunct treatment known as ozone therapy. Ozone is usually administered in patients, via auto-haemotherapy (infusion of ozonated autologous blood known as oxygen-ozone major autohaemotherapy, O2-O3-MAHT), from a balanced mixture with oxygen, as this approach has a practical reason. Following Henry’s law, it is better to calculate the concentration of ozone in a gas, such as oxygen, from which ozone is generated, as O3 concentration in a liquid is particularly sensitive to temperature variations and % weight of ozone (calculated on oxygen). At 298 K (25°C), 1 atm, 1% O3 on O2 (in weight) is about 7.824 μg/mL O3. Therefore, when a protocol of ozone therapy claims to use 45 μg/mL O3, it refers to a gaseous mixture O3/O2, corresponding to 5.75% O3 (in weight on O2).
The different, despite few, Scientific Societies investigating the biomedical and clinical action of medical ozone, established protocols and methodologies on how much ozone must be used in the several treatment options, usually on the basis of sound meta-analyses or Consensus Conferences. The Italian Scientific Society of Oxygen-Ozone Therapy (SIOOT), in Bergamo (Italy), uses a range of ozone from 10 to 50 μg/mL (very rarely 60-80 μg/mL), 31 in the different approaches (or administration routes), with which ozone is introduced in the patient, minor autohemotherapy, infiltration of ozonated blood, major autohemotherapy, rectal ozone, topical ozone in lipophilic media, ozonated water or oil. 26 In the oxygen-ozone major autohemotherapy (O2-O3-MAHT), 32 where usually 45 μg/mL O3 (in the O2-O3 gaseous mixture) are introduced in the autologous blood (200 mL), previously withdrawn in a sterile bag and then reintroduced, the “actual” concentration of ozone entering the blood, depends on the medium (plasma water) and on blood temperature (about 37°C). So, following Henry’s law, if in air 1% O3 = 7.824 μg/mL O3 at 25°C, then 45 μg/mL O3 in the preparation mixture are 5.75% O3 (weight), yet in water and at 37°C the ozone concentration should drop to less than 8 μg/mL, actually 7.165 μg/mL, following the many online available conversion tables.
At this dose, we calculated that, as only 2% of ozone is converted into 4-HNE, a plasma concentration very close to 3.0 μM 4-HNE can be produced (3.67 μM 4-HNE). This occurs because, in a single session of O2-O3-MAHT, four vol of 45 μg/mL O3 are used (7.165 μg/mL x 4 = 28.66 μg/mL). if considering that, for each O3 molecule, one molecule of 4-HNE is formed (at least theoretically), 2% corresponds to 0.573 μg/mL 4-HNE, finally corresponding to 3.67 μM 4-HNE. A dose close to 3.0 μM 4-HNE has been reported to prevent the activation of NLRP3 and the maturation of IL-1ß and IL-18. 33
Medical ozone is widely recognized as an anti-oxidant molecule, pertinently, if ozone shares the same receptors of natural flavonoids, which are considered anti-inflammatory molecules.34,35
The activation of the Nrf2/Keap1/ARE pathway, held by the activation of ROS as signaling molecules, 36 controls the regulation of haeme oxigenase-1 (HO-1), one of the major anti-inflammatory targets, and, by regulating also the expression of NF-κB, exerts an anti-inflammatory action. 37 Interestingly, 4-HNE plays an inhibitory effect on pyroptosis and on the inflammasome (NLRP3) activation, by directly binding to NLRP3 and preventing its interaction with NEK7. 33 Therefore, anti-oxidant ability and anti-inflammatory potential of ozone, are closely intertwined.
The role of mitochondria in this scenario is fundamental.
Any mitochondrial dysfunction, including impairments in mitochondria autophagy (mitophagy), leads to the activation of NLRP3, which, once activated, promotes the functional maturation of pro-inflammatory cytokines, such as IL-1ß and IL-18. 38 The role of mitophagy is crucial, to prevent an inflammatory response and induce a correct immune mechanism.39–42 The electrophilic molecule 4-HNE, produced by ozonides from ozone, has a panoply of functions, as it is used to regulate mitochondria functionality at physiological levels.43,44
The endogenous 4-HNE, which is used as a signaling molecule to regulate mitochondria stability, undergoes a clearance by scavenging enzymes such as aldehyde dehydrogenase, aldose reductase and the many glutathione-S-transferases triggered by the anti-oxidant response. 44 As 4-HNE acts as a signaling molecule, by regulating also the rate of -SH groups in proteins and the dynamic of redoxins, 44 at low doses it activates the pathway Nrf2/Keap1/ARE, so contributing, again, to an anti-oxidant and anti-inflammatory function.44,45
Dysfunction in mitochondria is a leading hallmark of many chronic immune diseases, and in many of these ones, ozone therapy reported promising and encouraging outcomes. This may occur in autoimmune diseases, such as Sjögren syndrome46,47 and multiple sclerosis,48–51 but also in chronic fatigue syndrome,52,53 fibromyalgia54–56 and others.57,58
If low doses (hormetic) ozone, via its metabolites, such as 4-HNE, has an anti-inflammatory potential, it exerts also a reduction in painful symptomatology, including other discomforts such as fatigue, as occurring in some debilitating chronic diseases, including post-COVID.59,60 Furthermore, medical ozone at low doses, by activating the Nrf2-mediated antioxidant system, may exert a fundamental role on the reduction of neuropathic pain, probably by increasing the levels of glutathione, which in turn inhibits NF-κB activation.61,62
Just in COVID-19, the adjunct medical treatment with ozone reported several encouraging results, particularly from SIOOT,59,63–66 inasmuch ozone, in the dose range 30-45 μg/mL, showed the ability to recover the O2 saturation % indexes and inhibit the increase of inflammatory biomarkers. 63
The anti-oxidant potential of low doses of ozone, which activate ROS-mediated signaling,
36
is preludial to the anti-inflammatory and immune-modulatory role of ozone treatment. Figure 1 shows the main anti-inflammatory role of 4-HNE in an oxygen-ozone major autohemotherapy (O2-O3-MAHT). Cartoon showing the major anti-inflammatory action held by the ozone-derived 4-HNE. (a) The oxygen-ozone mixture is prepared into a 50 mL polypropylene syringe, leading to a final concentration of ozone into oxygen as defined in the text and introduced in an autologous blood collection sterile bag (SANO3); (b) the ozonated blood is slightly mixed with a proper device for few minutes, according to defined protocols, from SIOOT and Multiossigen (BG), then (c) introduced into the same patient. Ozone released into the blood produces ozonides, (d) the main of which (4-HNE) induces the formation of ROS as signaling molecules, modulating the activity of Nrf2 (see text) but also inhibiting the formation of the NLRP3 (inflammasome) complex from the adaptor molecule apoptosis-associated speck-like protein containing a CARD (ASC), the sensor molecule NLRP3 and pro-caspase 1 (e), inhibiting also pyroptosis (f). PYD = Pyrin domain; CARD = caspase recruitment domain; NACHT = NACHT domain; LRR = LRR domain; Cap-1 = Cap-1 domain. The alkenal 4-HNE, by inhibiting the activation of NLRP3, inhibits inflammation.
Ozone, oxygen and the microcirculation
The effect of ozone on the anti-oxidant responsiveness of the cell, induces a series of mechanisms leading to the inhibition of the pro-inflammatory response, which could be seen as a prompt and smooth reaction to molecular alarmins, generated either from xenobiotics, damage associated molecular patterns (DAMPs) or pathogen-associated molecular patterns (PAMPs). ROS regulate the activation of the Nrf2/Keap1/ARE pathway 36 and once Nrf2 is activated, the NF-κB-mediated pro-inflammatory pathway is attenuated, whereas damaged cells undergo apoptosis. 67 Therefore, slight or weak stimuli by toxicants, trigger a protective, pro-survival response, which might be caused by the energetic need of cells to prevent any burdensome response, if the alert is weakly impacting on the cell economy as a whole. The activation of the Nrf2/Keap1/ARE induces mitophagy,68,69 therefore promoting health mitochondria function and biogenesis. Upon an anti-oxidant response, cells activate the anti-inflammatory “program” and the removal of damaged cells, even red blood cells using eryptosis via the action of 4-HNE. 70
On the other hand, the excessive stimulation of ROS and Nrf2, may cause a transient state of “hyperoxia”, which ultimately paradoxically induces the expression of HIF-1α, the hypoxia inducible factor. 71 The same indirect increase in 2,3-diphosphoglycerate (2,3-DPG) reported as an effect of ozone-mediated hyperoxia,2,16 may be caused by ozone-induced MDA, as this ozonide by-product is able to glycate haemoglobin and glycation increases 2,3-DPG in erythrocytes.72–74
The activation of the Nrf2/Keap1/ARE pathway leads to the involvement of HIF-1α, as a cross-talk between Nrf2 and HIF-1α has been reported.75–77
Actually, a mechanism of O2 sensing, is regulated by ROS, which are in turn elicited by O3 and stabilize the activity of HIF-1α in the induced hypoxia. 78 This evidence suggests a close relationship between ROS signaling, Nrf2-mediated response and microcirculation (i.e., the biology of endothelia and their response to ischemia/reperfusion injury).
For example, HIF-1α induces the upregulation of fundamental angiogenetic genes, such as VEGF,79,80 therefore impacting on the cardiovascular function, tissue oxygenation and endothelial rejuvenation, a systemic and relatively late action of medical ozone, which may account for its beneficial evidence in clinical investigation.
The relationship between Nrf2 and HIF-1α are interestingly complex and plays a paramount role in the management of oxygen in cells and tissues. HIF-1α and Nrf2 (nuclear factor erythroid 2-related factor 2) are both transcription factors involved in cellular responses to stress, including oxidative stress and hypoxia. While HIF-1α primarily responds to low oxygen levels (hypoxia), Nrf2 is primarily activated by oxidative stress and electrophilic compounds. Yet, both factors co-participate in the regulation of oxygen availability to cells.
Both HIF-1α and Nrf2 play critical roles in cellular adaptation and protection against stress, but they have distinct mechanisms of action and target different sets of genes. HIF-1α regulates the expression of genes involved in oxygen homeostasis, angiogenesis, glucose metabolism, and cell survival, 81 and promotes the adaptation of cells to low oxygen levels by enhancing oxygen supply, glycolysis, and angiogenesis. HIF-1α is also involved in the immune response and inflammation. It acts by binding to hypoxia response elements (HREs) on target gene promoters. 82
On the other hand, Nrf2 is a key regulator of the cellular antioxidant response. It controls the expression of a wide range of genes involved in cellular defense against oxidative stress, detoxification, and the removal of harmful reactive oxygen species (ROS). Nrf2 activation leads to the transcriptional upregulation of genes encoding antioxidant enzymes, phase II detoxification enzymes, and other cytoprotective proteins. 83 Nrf2 functions by binding to antioxidant response elements (AREs) in the promoters of target genes. Interestingly, there is some crosstalk between HIF-1α and Nrf2 signaling pathways. 81
Studies have suggested that under certain conditions, HIF-1α and Nrf2 can influence each other's activity and target gene expression. For example, both factors can interact with the Kelch-like ECH-associated protein 1 (Keap1), a negative regulator of Nrf2, leading to the activation of Nrf2-dependent genes. Additionally, in some cellular contexts, HIF-1α can enhance Nrf2 activity, resulting in increased antioxidant defense. 84 The precise interplay and regulatory mechanisms between HIF-1α and Nrf2 are complex and context-dependent, and more research is needed to fully understand their relationship. 85 Nevertheless, their cooperative or competitive interactions contribute to the overall cellular response to stress, promoting cell survival and maintaining redox homeostasis in various physiological and pathological conditions.
The interplay HIF-1α/Nrf2 causes the release of nitric oxide (NO) from endothelia, one of the numerous effects of ozone pre-conditioning, acting on experimental ischemia/reperfusion (I/R) injury. 86 Actually, overexpression of Nrf2, which could occur during the ozone therapy, generates a “pseudo-hypoxia”, leading to a delay in the HIF-1α proteolysis, as Nrf2 tends to stabilize the existence of HIF-1α. 85 Ozone induces NO from endothelia and a close cross talk between HIF-1α and NO was reported elsewhere.87,88 NO should inhibit HIF-1α but the ongoing existence of ROS lowers this ability, leaving the pseudo-hypoxic stimulus to work as a promotion of the microvasculature function.
The role of ozone treatment on microcirculation and cardiovascular function should account on the Nrf2-HIF-1α-NO axis, therefore.89–91 This is particularly evident in the role exerted by the major ozone-derived ozonide, i.e., 4-HNE, which exerts a fundamental positive and hormetic action in microcirculation.92,93 Once nitric oxide release is activated, by eNOS, NO should act as a sort of feedback regulator of HIF-1α. 94
Ozone in tissue remodelling and rejuvenation
Ozone is able to reduce inflammation but also to act as a biostimulator. 95 As an anti-inflammatory agent, ozone is enabled to reduce neuropathic pain and any other form of painful event due to inflammation, such as low back pain, often due to disc herniation96–98 and in musculo-skeletal disorders.99–101 Disc herniation is a serious painful concern, particularly in the elderly. The nucleus pulposus is a gel-like substance located in the centre of an intervertebral disc, which is the cushioning structure between the vertebrae in the spine. Disc herniation, also known as a slipped or herniated disc, occurs when the nucleus pulposus protrudes or bulges out through a tear or weakened area in the outer layer of the disc called the annulus fibrosus. When a disc herniation occurs, the nucleus pulposus can put pressure on the nearby spinal nerves, leading to various symptoms. The extent of symptoms depends on the size and location of the herniation, as well as the degree of nerve compression.
Some common symptoms of a herniated disc include either a) pain, i.e., the herniated disc can cause localized pain in the area of the spine where the disc is affected, e.g., if the herniation is in the lower back (lumbar spine), it can cause lower back pain; or b) radiculopathy, i.e., when the herniated disc compresses a spinal nerve, for which it can result in radiculopathy, which is pain, numbness, tingling, or weakness that radiates along the path of the affected nerve. 102 For instance, a herniation in the lumbar spine may cause pain and numbness in the buttocks, legs, and feet (sciatica).
During inflammation in disc herniation, several changes occur within the intervertebral disc. The inflammatory response is triggered by the leakage of the gel-like nucleus pulposus material from the herniated disc, which acts as an irritant to surrounding tissues.103–105 As a consequence, an infiltration of an immune population of cells rapidly occurs. 104 In response to the leaked nucleus pulposus material, immune cells, such as macrophages, neutrophils, and lymphocytes, migrate to the site of inflammation. These immune cells release inflammatory mediators, such as cytokines and chemokines, which further contribute to the inflammatory response.
The regulation of this inflammatory milieu is of paramount importance. 105
Degeneration may follow an increased vascularity. 106 The herniated disc becomes more vascularized during inflammation. Blood vessels in the surrounding tissues proliferate and extend into the disc, bringing in more immune cells and nutrients required for tissue repair. This leads to the immediate release of inflammatory mediators by immune cells, such as tumour necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and prostaglandins, contributing to the inflammatory process. These mediators can increase pain sensitivity, cause tissue damage, and promote further inflammation.
Therefore, degenerative changes can be reported, as chronic inflammation in the intervertebral disc can lead to degenerative changes. The inflammatory mediators can induce the production of enzymes, such as matrix metalloproteinases (MMPs), which degrade the extracellular matrix components of the disc, including collagen and proteoglycans. This degradation weakens the disc structure and can contribute to further disc degeneration104,105 and nerve sensitization.
The anti-inflammatory role of ozone is particularly insightful in this context.
Previous reports would suggest that ozone might rebuild the annulus fibrosus due to its contributing in the synthesis and arrangement of type I collagen.107–109 The ability of ozone to reduce inflammation in the nucleus pulposus, of the intervertebral disc, contributes to the regeneration of this district, usually composed by water, proteoglycans (with aggrecan) and type II collagen, probably switching off apoptosis and promoting to wound healing mechanisms.110–112
The degeneration of the intervertebral disc is fundamentally an inflammatory process, usually mediated by pro-inflammatory cytokines and by degradation products of intervertebral disc cells, including innate cells recruiting cytokines (IL-4, IL-12, IFN-γ, IL-6) and metalloproteinases. 112 Moreover, inflammation leads to the production of TNF-α and IL-1ß, which activates nitric oxide (NO), nerve growth factors and cyclooxygenase 2, then generating painful symptoms. 113
Ozone, as an anti-inflammatory agent, and enabled to act onto type I collagen synthesis and organization, may be at the foundation of the successful treatment of intervertebral disc-related pain, even promoting the macrophage skewing from M1 (inflammatory) to M2 (reparative). 114
The possibility that ozone promotes the intervertebral disc rebuilding, lies on the ability of medical ozone to act as a rejuvenating and remodelling agent, besides to its well-known anti-oxidant and anti-inflammatory potential.
In cosmetic applications, ozone has been suggested to have rejuvenating effects on the skin. Ozone generators are sometimes used in facial treatments, where a controlled amount of ozone is applied to the skin. It is believed that the oxidative properties of ozone can promote blood circulation, increase oxygen supply to the skin, and stimulate collagen production. These effects may contribute to a more youthful appearance and improved skin texture. However, it's important to exercise caution when considering ozone therapy or cosmetic treatments involving ozone. Ozone is a highly reactive gas and should be used under controlled conditions by qualified professionals. The concentration and duration of ozone exposure must be carefully regulated to avoid potential adverse effects. 115
While there is promising research suggesting the rejuvenating and reparative properties of ozone, further studies are needed to establish its efficacy, safety, and optimal applications in different settings. Consulting with medical or skincare professionals who have expertise in ozone treatment can provide more specific information and guidance based on the most current research and best practices.
Effect of ozone therapy on coagulation and fibrinolysis
To date, even in Italy, the use of medical ozone to treat inflammatory and chronic pain is progressively expanding. A simple search on Pubmed allows to retrieve more than 4,000 issues regarding ozone therapy, therefore interest in this medical approach raised fundamental questions about its impact in thrombosis research. Ozone is widely used both in private and public medical clinics, despite there is yet a concerning lack of standardization in methods and therapy protocols.116,117 Scientific Societies, such as the Italian Scientific Society of Oxygen Ozone Therapy (SIOOT), are challenging the issue since long time in order to expand the debate and lead to a consensus guideline, joining professionals and expertise worldwide. 12
Clinicians adopting the major oxygen-ozone autohemotherapy (O2-O3-MAHT) as a therapy for inflammatory pain or fatigue, 116 are currently wondering if ozone used in this practice may have side effects on the patient’s blood coagulation/fibrinolytic system, particularly because of the ongoing increase in immune thromboembolic events started with COVID-19 pandemic, even in an asymptomatic fashion. Despite they did never report any side effect regarding the use of ozone on the plasma thrombotic system, the increase in the rate of thromboembolic events, aside from ozone, in the widest population worldwide following COVID-19 pandemic, raised some concern. Ozone exposure alters the physiological pattern of blood coagulation and the endothelial-platelet cross talk, 118 but this evidence seems to refers to ambient ozone, not to the medical ozone used in the ozone therapy. 119 The substantial difference between the environmental ozone (which is a pollutant) and the medical ozone (which is used as an adjunct therapy), lies fundamentally on ozone dosages and administration routes.117,118
The purported interaction of ozone with the patient’s peripheral blood, fades off within few minutes at the normal body temperature and during an O2-O3-MAHT, therefore other mediators are to be addressed, such as free radicals and lipid peroxides, which have yet very short lifetimes.
Anyway, at the dose range usually employed in an O2-O3-MAHT, i.e., 30-50 μg/mL, recent data suggest that ozone might generate microparticles from erythrocytes, leukocytes, endothelial cells and platelets and moreover increases the prothrombin time (PT), the activated partial thromboplastin time (APTT) and the D-dimer, whereas decreased fibrinogen. 120 Notwithstanding, a study carried out a few years ago reported that microparticles are physiologically present in the circulating blood to prevent pro-thrombotic events in normal and physiological conditions of health. 121 While elsewhere published data reported that ozone via O2-O3-MAHT triggered the release of microparticles from erythrocytes (102.0 ± 90.0 SD MPs/μl), from platelets (73.4 ± 52.5 SD MPs/μl), from leukocytes (34.3 ± 29.9 SD MPs/μl) and from endothelial cells (42.6 ± 31.1 SD MPs/μl), microparticles, isolated from fresh blood samples and identified by flow cytometry in healthy subjects, were almost higher in number.120,121 Actually, microparticles originating from platelets [(237 x 106/L, i.e., 237/μL (median; range 116-565)], erythrocytes (28 x 106/L; 13-46), granulocytes (46 x 106/L; 16-94) and endothelial cells (64 x 106)/L; 16-136), were similar or even higher than microparticles elicited by ozone, so ascertaining that ozone and its oxidized metabolites do not exert any adverse effect on these systems.120,121 Briefly speaking, previous reports have highlighted that, despite ozone may generate microparticles during its introduction into the circulating blood, the concentration of these particles is within the physiological level of particles usually produced during routine life.120,121 In this sense, despite the existence of some report challenging the issue of microparticles, 122 the ozone used within the hormetic range can be considered safe. The slight excess in damaged erythrocytes during a O2-O3-MAHT undergoes an immediate clearance by eryptosis induced by the ozone-triggered 4-HNE. 70
Boczkowska-Radziwon and colleagues, reported that O2-O3-MAHT at 30 μg/mL ozone increased D-dimer to 211.6 ± 107.8 µg/mL, respect to controls (183.6 ± 70.2 μg/mL), yet the physiological range of D-dimer includes this dose (145-348 µg/mL). The same can be said about PT, APTT and fibrinogen, which do not change significantly upon ozone medical treatment. 120
It should be noted, at the best of our knowledge, that there are no case reports or case studies published so far about the existence of adverse effects from ozone therapy on the endothelium-platelet-coagulation or cardiovascular system. On contrast, some Polish nephrologists demonstrated that in dialysis patients with peripheral arterial occlusion disease, O2-O3-MAHT at 50 μg/mL ozone, did not influence blood coagulation parameters, namely PT, APTT and fibrinogen. 119 Furthermore, other authors demonstrated that, again at 50 μg/mL O3, ozone did not cause any inflammatory reaction per se, and did not alter, compared to untreated controls, inflammation parameters such as C-reactive protein (CRP) or IL-6. 121
The use of ozone therapy does not seem to interfere with the blood coagulation/fibrinolytic system in humans, therefore. Furthermore, thrombin regulates the growth of vascular smooth muscle cells and the production of heat shock protein through the JAK/STAT system. 123 Ozone uses the same mechanism of the JAK/STAT3 pathway to prevent ischemia/reperfusion injury, by regulating the production of the heat shock protein hsp70, thus assuming even a cardioprotective role. 124
Finally, some evidence reported that ozone can modify, by oxidation, the biochemistry of fibrinogen, so impairing its function. 125 According to the existing literature and our calculations, this occurs with ozone doses sensitively higher than the ones used in the ozone therapy protocols. This evidence must be declined according to the actual quantity of ozone used in O2-O3-MAHT compared to the one used elsewhere. 125
In this circumstance, we have calculated that as fibrinogen has a median of 3,000 mg/L (200-400 mg/dL), an ozone concentration of at least 375 μg/mL is requested to induce a fibrinogen oxidation able to significantly disturb the process of blood coagulation, at least following the dosages used by authors reporting an inhibitory effect of ozone on fibrinogen.125,126 The dose of ozone in ozone therapy is within the protection range of the total antioxidant capacity (TAC) which is at least 160 μg/mL, according to Bocci et al. 127 Therefore, any oxidative activity of ozone is immediately buffered, within 5 min at 37°C, by the patient’s anti-oxidant response, with which ozone and its oxidized metabolites undergo a rapid clearance from the whole blood. This would mean that the concern regarding the damaging ability of ozone is negligible, in the therapy ranges used for ozone therapy.
Yurina et al., reported oxidative modification in 300 μg/mL fibrinogen using a calculated dose of 1,200-4,800 μg ozone within 1 mL used in the experimental setting (25-100 μM O3 for 1 μM fibrinogen), 125 yet the dose of ozone used in the O2-O3-MAHT from SIOOT/Multiossigen protocols is 45 μg/mL, and the range is 10-80 μg/mL, it is very difficult a damaging action of ozone on fibrinogen in the ozone therapy condition.16,118,125
In contrast with previously published data, 120 ozone has been reported to reduce plasma D-dimer in immuno-thromboembolic pathologies such as COVID-19,128,129 therefore, the ability of the ozone therapy to modulate the complex interplay endothelia/platelets/coagulation, should rely on other indirect mechanisms aside from the direct effect of ozone. 118
We attempted to assess this discussion in our experience. A logistic regression made on data collected in our clinical labs using a median dose of ozone in O2-O3-MAHT of 45 μg/mL compared to already considered O3 doses, 125 i.e., 2,500 μg/mL O3, on D-dimer, fibrinogen and PT (INR), gave a Chi square = 55.3517 (p < .0001) (Hosmer-Lemeshow test chi square = 1.3258), with differences between O3 in O2-O3-MAHT and higher O3 dose (multiple regression analysis). Actually, we confirmed that using a very high dose of O3, i.e., 2.500 μg/mL O3, which is comprised in the range indicated above 1.200-4.800 μg/mL O3, differences resulted in D-dimer (fourfold increase, p = .0004372), fibrinogen (54%, decrease, p = .001127) and PT (INR) (1.5-fold increase, p = .0003899) (data with SPSS v 24.0 statistic software), assessing that only high doses in the logistic regression did not fail in altering the coagulation parameters investigated.
Further research is needed, anyway
The scant number of studies and clinical trials about the in vivo side effect of ozone therapy on the coagulation system, should deserve caution to finalize this commentary as a plain confirmation of ozone safety. Notwithstanding, the negligible dose range used in ozone therapy and its rapid clearance, makes very difficult to argue ozone therapy as a safety concern.
Future remarks in medicine. Which novelty ozone can represent for medical adjunct treatment?
Two major conditions can be defined in the biological activity of ozone as a chemical molecule, i.e., the eustress condition (beneficial) and the distress condition (noxious). Figures 2 and 3 summarize these two circumstances. Cartoon showing the beneficial activity of oxygen-ozone treatment and the pathways involved. Low, regulated doses of ozone, in the range of hormesis, cause the 4-HNE mediated switching off of pro-inflammatory signals in innate immune cells and, at the same time, the induction of the anti-oxidant signaling system Nrf2. The activation of Nrf2 inhibits the activation of NF-κB. A low and moderate production of ROS activates the haeme oxygenase 1 (HO-1), which is associated with the expression of NO and the development of microcirculation. Other factors, such as the hypoxia factor HIF-1α, induced by ROS, contributed to the microvasculature enlargement, via the release of angiogenetic factors, a mechanism then switched off by NO. Red cross = inhibition. Top arrows = negative (red) and positive (green) promotion (upregulation). Bottom arrows: downregulation. HIF-1α: hypoxia inducible factor 1 alpha; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf2: nuclear factor erythroid 2–related factor 2; ROS: reactive oxygen species; VEGF: vascular endothelial growth factor. Cartoon showing the deleterious activity (distress) of high doses of ozone, in the mixture oxygen-ozone out from hormesis range, even via the 4-HNE mediator. Ozone causes inflammation by activating the NLRP3 complex formation, downregulating Nrf2 due to NF-κB upregulation and inhibiting mitophagy in mitochondria, which will release mitochondria DNA, induce unfolded protein response (UPR) and disrupt mitochondria network in innate immune cells. This will lead to a marked inflammatory response ruled by the NF-κB pathway and cytokines. Red cross = inhibition. Top arrows = negative (red) and positive (green) promotion (upregulation). Bottom arrows: downregulation. Red arrows: negative effect for the cell. Green arrow: positive effect for the cell. MTNs: metallothioneins; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf2: nuclear factor erythroid 2–related factor 2; ROS: reactive oxygen species; UPR: unfolded protein response.
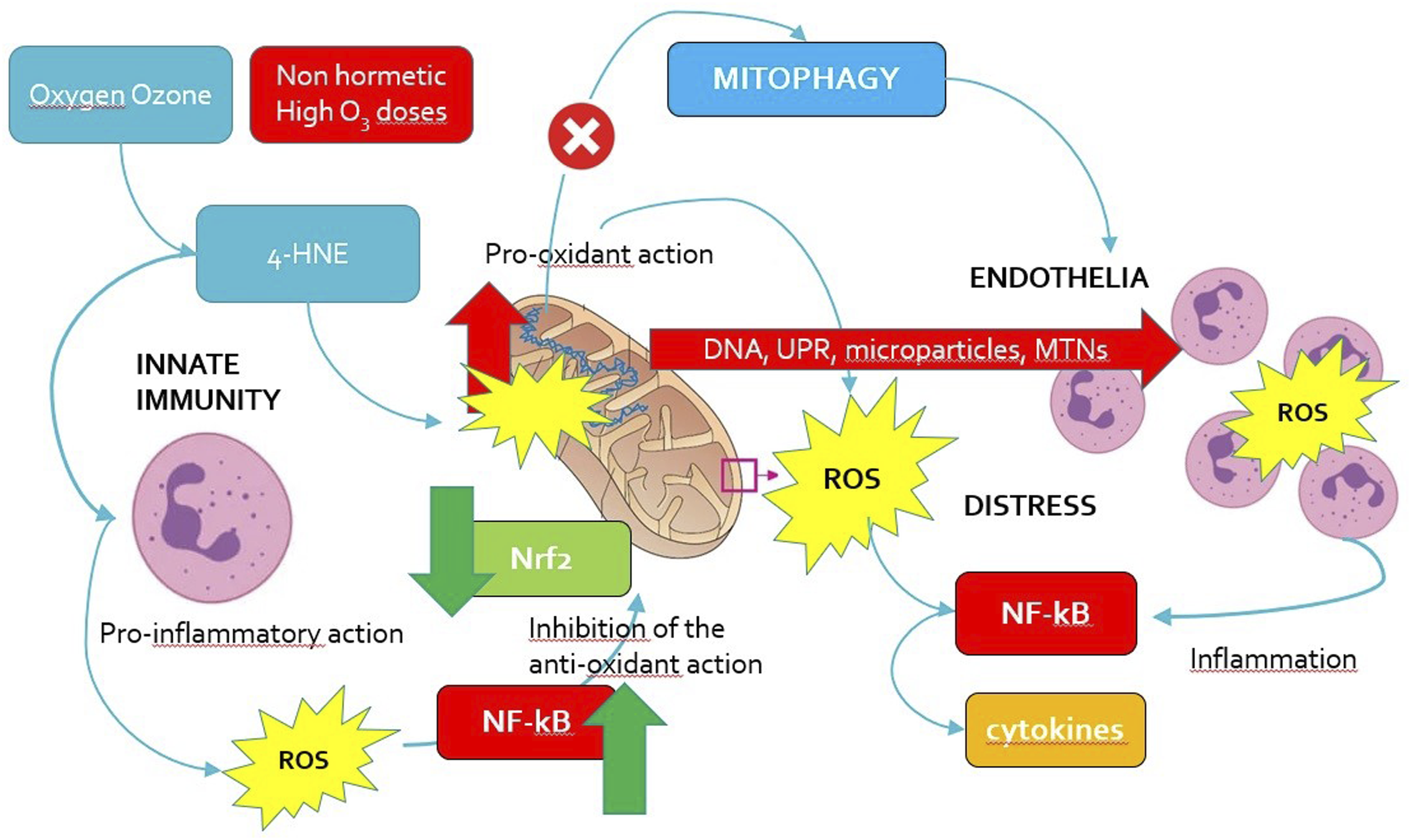
Eustress is a terminology usually associated with a positive and beneficial effect of a stress stimulus on cells, whereas distress is quite the contrary. 63 The leading concept distinguishing eustress from distress, is hormesis and, as regards mitochondria, “mitohormesis”, which does not dismiss the crucial role of ROS. 129 Fundamentally, ROS as signaling molecules, regulate the fine interplay between Nrf2 and NF-κB, which might be considered as major switching tuners of alarming signals for a cell. Briefly speaking, and maybe resulting somehow simplistic, Nrf2 and NF-κB may be represented as the “diplomatic” and the “military” approaches which cells are prompt to settle to address any external or, even internal, insult. In this perspective, inflammation is not merely a circumstance involving simply immunity but a mechanism intrinsically occurring within the “government” of the cell, particularly regarding mitochondria. In this curious and imaginative scenery, any molecule used to elicit an Nrf2-mediated “peace-intended diplomacy”, promotes actually the routinely invested mechanism with which cells stay alive. Using hormesis might simply mean to promote cells in keeping their, yet complex, mechanisms of common survival. So, it is not a treatment, but a cell recalling to the basic mechanisms of survival, i.e., a physiological recovery from an imbalanced condition, in some way.
It is quite difficult to comprehend this remarkable hallmark of living organisms for a certain number of reasonable motivations. First, it is difficult to trust that a toxic substance can trigger a beneficial status within cells, second, cancer cells usually do not respond to hormesis, and third, hormesis is active depending not only on doses but also on different microenvironments.
Fundamentally, any external novel substance (chemical) or physical input are a source of stress for a cell, who strives to maintain its survival machinery and prevent apoptosis. The phenomenological milieu where cells are striving to stand alive and in “good health” is ruled by hormesis. Hormesis is a biological phenomenon characterized by a biphasic dose-response relationship, in which a substance or stressor that is typically harmful at high doses or intensities can have a beneficial effect at low doses or intensities. It is often described as "what doesn't kill you makes you stronger." The hormetic response typically involves a stimulation of protective and repair mechanisms in the body. 130 At low doses or intensities, the stressor induces a beneficial adaptive response that enhances the organism's resilience and improves its overall function. This response can manifest as increased resistance to subsequent stress, improved health, extended lifespan, and enhanced performance.
When a toxic compound is allowed to work “within” the hormetic functional space (usually chaotic in biophysical terms), then this molecule can maintain or even restore the cell survival machinery, so generating benefits. The proper dosage range and administration route for a potential toxicant used in a hormetic way, is highly crucial in medicine, therefore.
This compels physicians using ozone in the medical treatment to comply with standardized and approved protocols, usually via their management and debating in Scientific Societies, such as the Italian Scientific Society of Oxygen Ozone Therapy (SIOOT), based in Bergamo (Northern Italy).
SIOOT organizes High Level Masters, with official Academies (for example the University of Pavia and the University UniCamillus in Rome) about the use of ozone as an adjunct treatment in many immune-inflammatory disorders, involving pain, infection and fatigue. Moreover, an animate and fruitful debate with experts about the chemical biological and clinical role of ozone to reappraise and improve methods and protocols, is continuously promoted in the numerous training courses held by SIOOT.
A leading and sound Scientific Society is paramount to support scientific research on ozone in medicine and promote the widest debate on the topic.
The novelty that ozone can bring to medicine might be simply described as that possibility to adjust and tune the ability of cells to keep its own survival machinery, as ozone is never a pharmaceutical drug able to specifically target and interfere with defined molecular pathways, then inducing a molecular effect. Most probably, ozone may allow cells to rescue their physiological ability in addressing damage and stress.
This is a clear possibility that medicine will gain a “corrective” action, more than an “intervening” role.
Conclusions
Which is the heading message we can bring home with this perspective?
Fundamentally, ozone triggers the Nrf2/Keap1/ARE anti-oxidant pathway but within hormetic ranges and this activation promotes a 4-HNE-mediated anti-inflammatory mechanism. This pro-survival, stress responsive behaviour, is switched on by the involvement of a health mitochondrial function, as mitophagy can be considered the master tuner of pro-survival versus pro-inflammatory responses. The novelty is that ozone may be considered an exogenous regulator of mitochondria turnover and biogenesis. Therefore, the emerging, novel medicine might use mitochondria adjustment to address fundamental immune-inflammatory pathologies, such as autoimmunity and degenerative disorders.
Ozone therapy is a straightforward adjunct therapy able to address immune-based disorders (autoimmunity and inflammation), via the Nrf2-mediated role on NLRP3 and NF-κB, elicited by ozone-derived ozonides, particularly 4-HNE. The anti-oxidative and anti-inflammatory potential, often causes inhibition of nociceptive stimuli, reduction of pain, discomfort and fatigue and lowering of the devasting impact of degenerative diseases, particularly in the musculo-skeletal contexts. Ozone ability in regulating, via HIF-1α and NO, the microvasculature and via MDA the ability of red blood cells to release oxygen, makes ozone a friend, more than a foe, of cardiovascular physiology. However, ozone can have beneficial effects is administered in a low dose range (hormesis) and far from the direct contact with the respiratory system. 27
Footnotes
Acknowledgments
We would like to acknowledge Drssa Francesca Turriceni for her precious aid in preparing the search study and English language corrections.
Author contributions
“Conceptualization, S.C. and L.V.; methodology, M.F., F.V., S.P., U.T., G.R., M.F., L.V.; software, S.P..; validation, L.V., M.F. and U.T.; formal analysis, S.C..; investigation, S.C. L.V.; resources, S.C., M.F.; data curation, S.C.; writing—original draft preparation, S.C.; writing—review and editing, S.C.; visualization, L.V.; supervision, L.V. M. F., G.R., U.T..; project administration, M.F.; funding acquisition, none. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
