Abstract
Purpose
Pulmonary fibrosis (PF) is an inescapable problem. Diacerein, a chondro-protective drug, has antioxidant and anti-inflammatory effects. Its effect on PF injury has not yet been fully clarified. Therefore, the current study aimed to detect its protective effect on lung tissue with the explanation of possible underlying mechanisms.
Methods
Adult male albino rats were assigned to four groups: control group, diacerein control group, PF non-treated group, and PF diacerein pretreated group. Lung tissue oxidative stress parameters, inflammatory biomarkers mainly Toll-like receptors-4 (TLR4), and myeloid differentiation factor 88 (MyD88) levels were determined. Histopathological examination of lung tissue and immunohistochemical studies of nuclear factor-kappa B (NF-κB), and transforming growth factor- β (TGF-β) were also done.
Results
Diacerein pretreatment has the ability to restore the PF damaging effect, proved by the reduction of the oxidative stress and lung tissue inflammation via downregulation of TLR4/NF-κB signaling pathway together with the restoration of TGF-β level and improvement of the histopathological and immunohistochemical study findings in the lung tissue.
Conclusion
These results suggested the protective effect of diacerein on PF relies on its antioxidant and anti-inflammatory effects reducing TLR4/NF-κB signaling pathway.
Introduction
Pulmonary fibrosis (PF) is a serious disease with high mortality and morbidity rate. PF is one of the most prevalent interstitial lung diseases, affects approximately 9.3 per 100,000 person per year globally. 1 It describes a condition in which the normal lung anatomy is lost by a process of active lung remodeling with infiltration of inflammatory cells, formation of reactive oxygen species (ROS) and deposition of extracellular matrix protein into the lung parenchyma. 2 The architecture of interstitial gaps changes over time, resulting in decreased gas exchange and possibly death. 3
With good therapeutic success, bleomycin (BLM), a cytotoxic antibiotic, is used in combination with other chemotherapeutic agents to treat squamous cell carcinomas, malignant lymphomas, and testicular cancer.4,5 The most harmful side effect of BLM use is PF since it causes oxidative stress and inflammation in the lung tissue, which finally results in lung fibrosis. 6
It has been demonstrated that multiple factors implicated in the development and progression of PF including nuclear factor‐kappa B (NF‐κB), tumor necrosis factor-α (TNF-α), interleukins-1β (IL-1β), and transforming growth factor- β (TGF-β). 7 Toll-like receptors (TLR) are a major family of transmembrane recognition proteins which include 10 members. Recently, Toll-like receptor 4 (TLR4) proved to play a pivotal role in mediating the induction of the inflammatory response to stress. 8 Activation of TLR4 results in recruitment of its downstream via myeloid differentiation factor 88 (MyD88) which ultimately stimulates the production of proinflammatory cytokines including NF‐κB, TNF-α and IL-1β.8,9
Diacerein (DIA) is a derivative of anthraquinone; that was developed for the elaboration of the osteoarthritis disease. DIA inhibits IL-1 converting enzyme; key limiting enzyme for the secretion of IL-1β cytokine along with suppression of IL-1β binding to its receptor. 10 Earlier researches proved that DIA significantly has a protective effect against inflammatory damage in different animal and human studies,11–14 due to its ability to inhibit the production of inflammatory cytokines (TNF-α and IL-1β) as well as down regulation of the IL-1β receptor.15,16 Also, DIA was proved to downregulate the activation of the TLR4/NF-κB - mediated signaling pathways with subsequent suppression of inflammatory cytokines production. 17
PF is being treated with a variety of anti-inflammatory medications, including corticosteroids and immunosuppressive medicines, but there is still no fully effective cure. 18 Subsequently, the present study was done to investigate the possible protective role of DIA in male rats subjected to BLM-induced PF with the exploration of probable mechanisms mediating its action and the emphasis on the TLR4/MyD88/NF-κB signaling pathway.
Materials and methods
Animals
Thirty–two healthy male albino rats (180–240 g), aged 10 weeks were acquired from the National Research Center (Giza, Egypt). Feeding of rats was done using a standard commercial diet and a tap water. Rats were left to accommodate to the laboratory environment for 1 week before their involvement in the experiment. They were kept at 24 ± 2°C with a 12-h dark/light cycle. The used procedures were following the ARRIVE guidelines and were carried out in accordance with the U.K. Animals Act, 1986. The study was approved by the board of faculty of medicine, Minia university, with approval number (1012021).
Chemicals
DIA was obtained from (Eva Pharma, Egypt), dissolved in 1% carboxymethylcellulose (1% CMC), and freshly prepared before use. 19
Induction of lung fibrosis
Induction of PF using BLM was done using the method previously described by Mohamed et al., 20 rats were anesthetized via ketamine/xylazine (100/10 mg/kg) 21 and then placed in a supine position with the extremities pull caudally to help exposure of the trachea. After that, the trachea was exposed through an anterior neck incision, and a direct puncture with a 24-gauge needle on a 1 mL tuberculin syringe was performed at two to four tracheal rings below the larynx. Saline or BLM single dose of 5 mg/kg was injected into the trachea at day 1 then the tuberculin syringe was removed. Finally, the neck was repaired with sutures. After 2 weeks, animals were anesthetized, sacrificed and lungs of each animal were surgically removed.
Experimental design
Group 1: Control group: received CMC p.o daily for 2 weeks as vehicle along with a single dose saline instilled intratracheally in the first day of the experiment.
Group 2: DIA group: received DIA (25 mg/kg/day) p.o daily for 2 weeks 22 along with a single dose saline instilled intratracheally in the first day of the experiment
Group 3: BLM group: received CMC p.o once daily as vehicle for 2 weeks and a single intratracheal BLM (5 mg/kg) single dose instilled intratracheally dissolved in saline in the first day experiment. 20
Group 4: DIA treated group: received DIA (25 mg/kg/day) p. o suspended in CMC for 2 weeks 22 + BLM (5 mg/kg) single dose at the first day of experiment as previously described.
The dose of DIA was determined following our preliminary experiments and based on other previous studies.
Sample collection
At the end of the experimental period, the animals were anesthetized, weighted then sacrificed. Lung of each rat was weighted for calculation of lung/weight ratio. The lung was washed by cold saline and divided into parts, one part was put in 10% formalin for histopathological examination and the other parts were homogenized using ice-cold phosphate buffer (0.01 M, pH 7.4; 20% w/v). The homogenate was centrifuged for 15 min at 5000 r/min and the supernatant was stored at –80°C till use to measure the biochemical parameters.
Assessment of physical parameters
At the end of experiment, total body weights as well as lung weight from each animal were assessed.
Assessment of lung index
Lung tissue was weighed to determine lung index. Lung index was determined by dividing lung weight (g) by body weight (g) and multiplying by 100.
Assessment of wet/dry (W/D) ratio
The content of lung tissue edema was evaluated by calculating W/D ratio. Rats were sacrificed at 14 days after BLM exposure, subsequently the lungs were removed and “wet” weights were recorded. Thereafter, the lungs were then placed in an incubator at 60°C for 48 h for eliminating moisture and the “dry” weights was observed. Finally, the W/D ratios were calculated.
Biochemical analysis
Assessment of lung tissue oxidative stress/antioxidant biomarkers
According to the previously prescribed method of Marklund and Marklund 23 one unit of super oxide dismutase (SOD) is equal to the amount of enzyme which prevents pyrogallol autoxidation by 50%. A spectrophotometer was used to measure enzyme activity at 420 nm. Lung tissue reduced glutathione (GSH) was determined following Ellman’s way, the yellow color produced by reduction of Ellman’s reagent. The absorbance of the formed chromogen was measured spectrophotometrically at 412 nm. 24 Malondialdehyde (MDA) was quantified by using the thiobarbituric acid method and the sample absorption was measured at 535 nm. 25
Measurements of TLR4 and Myd88 in lung tissue
Lung tissue TLR4 and Myd88 were evaluated by enzyme-linked immunosorbent assay (ELISA) kits (Elabscience Biotechnology Inc., USA and ELISA Genie Co., Dublin, Ireland, respectively), Catalog No: E-EL-R0990, and SKU: RTFI01303, respectively. Those ELISA kits used the Sandwich-ELISA principle. The micro ELISA plate provided in this kit has been pre-coated with an antibody specific to rat TLR4 and Myd88 respectively. Standards or samples are added to the micro ELISA plate wells and combined with the specific antibody. Then a biotinylated detection antibody specific for rat TLR4, and Myd88 respectively and Avidin-Horseradish Peroxidase (HRP) conjugate are added successively to each micro plate well and incubated. Free components are washed away. The substrate solution is added to each well. Only those wells that contain rat TLR4, and Myd88 respectively, biotinylated detection antibody and Avidin-HRP conjugate will appear blue in color. The enzyme-substrate reaction is terminated by the addition of stop solution and the color turns yellow. The optical density (OD) is measured spectrophotometrically at a wave length of 450 nm ± 2 nm. The OD value is proportional to the concentration of rat TLR4 and Myd88 respectively. You can calculate the concentration of rat TLR4 and Myd88 respectively in the samples by comparing the OD of the samples to the standard curve.
Histological examination of lung tissue
Lung tissue sections from all groups fixed in 10% formalin, embedded in paraffin and dehydrated in ascending concentrations of ethyl alcohol (70%–100%). 5 µm tissue sections were stained with hematoxylin and eosin (H&E) and Masson trichrome. 26
Modified passmore histological scoring to evaluate lung damage.

Immunohistochemical study of NF-κB p65 and TGF-β1 in lung tissue
Immunocytochemical staining was performed according the manufacture instructions using NF-κB p65 rabbit polyclonal antibody (PA-16545) used as a marker for inflammation, hypoxia and oxidative stress. TGF-β1 rabbit monoclonal antibody (MA515065) used as a marker for cell growth, healing, fibrosis, inflammation, oncogenesis, erythropoiesis. All were obtained from Thermo fisher Company, USA. 4 μm sections were cut for immune-labeling.
The procedure was performed regarding the manufacture instructions. Briefly, sections were deparaffinized in xylene, rehydrated in descending concentrations of alcohol, and immersed in 0.1% hydrogen peroxide for 15 min to prevent the endogenous peroxidase activity. For antigen retrieval, the slides were boiled in 10 mm citrate buffer solution (PH 6) for 10–20 min followed by cooling at room temperature for 20 min. Sections were washed by phosphate buffer then incubated in the ultravision block at room temperature for 5 min to prevent the non-specific background staining.
Sections were incubated with antibody for 60 min at room temperature. The reaction was seen with the usage of ultra-vision one detection System, HRP Polymer & (diaminiobenzide) DAB Chromogen (Thermo Fisher Company, USA). After fulfillment of the reaction, counterstaining was performed with the usage of Meyer’s hematoxylin and dehydrated by exposure to ascending grades of alcohol then cleared by xylene. Coverslip using permanent mounting media was put.
The changes in the immunohistochemical reaction of the lung tissue sections of the treated animals were observed through the comparison with the corresponding tissue of the control rats. In negative control slides, the same system was performed except the primary antibodies were not added.
Photography
Olympus light microscopy (Olympus, Japan), at the Histology and Cell Biology Department at Faculty of Medicine, Minia University, was used for examination of the slides and capturing images for the tissue sections. Slides were photographed by the usage of an Olympus digital camera. Images were saved as jpg and processed using Adobe Photoshop 7 to standardize contrast, brightness, and background color.
Morphometric analysis
Image J 22 software in the faculty of Dentistry, Minia University was used for measuring the area fraction of NF-κB p65 and TGF-β1 antibody immunopositivity. Area fraction was measured in a standard measuring frame per 10 images in each group using a magnification ×400 by light microscopy was transferred to the monitored screen. Regions containing positively immunostained tissues were used for estimation independent on the intensity of staining.
Statistical analysis
Results were expressed as means (S.E.M.) and analyzed by One-way analysis of variance followed by Tukey’s test. p values < 0.05 were considered significant. Graph Pad Prism software was used (version 5.01 for Windows, GraphPad Software, San Diego California USA, www.graphpad.com).
Results
Physical assessment
Effect on the lung weight/body weight ratio
Administration of BLM caused a significant increase in the lung weight/body weight ratio compared to control group. Administration of DIA in combination with BLM caused a significant decrease in the lung weight/body weight ratio compared to BLM group (Figure 1(a)). Effect of diacerein (DIA) for 14 days on lung weight/body weight ratio and on lung wet/dry weight ratio in bleomycin (BLM) induced lung fibrosis.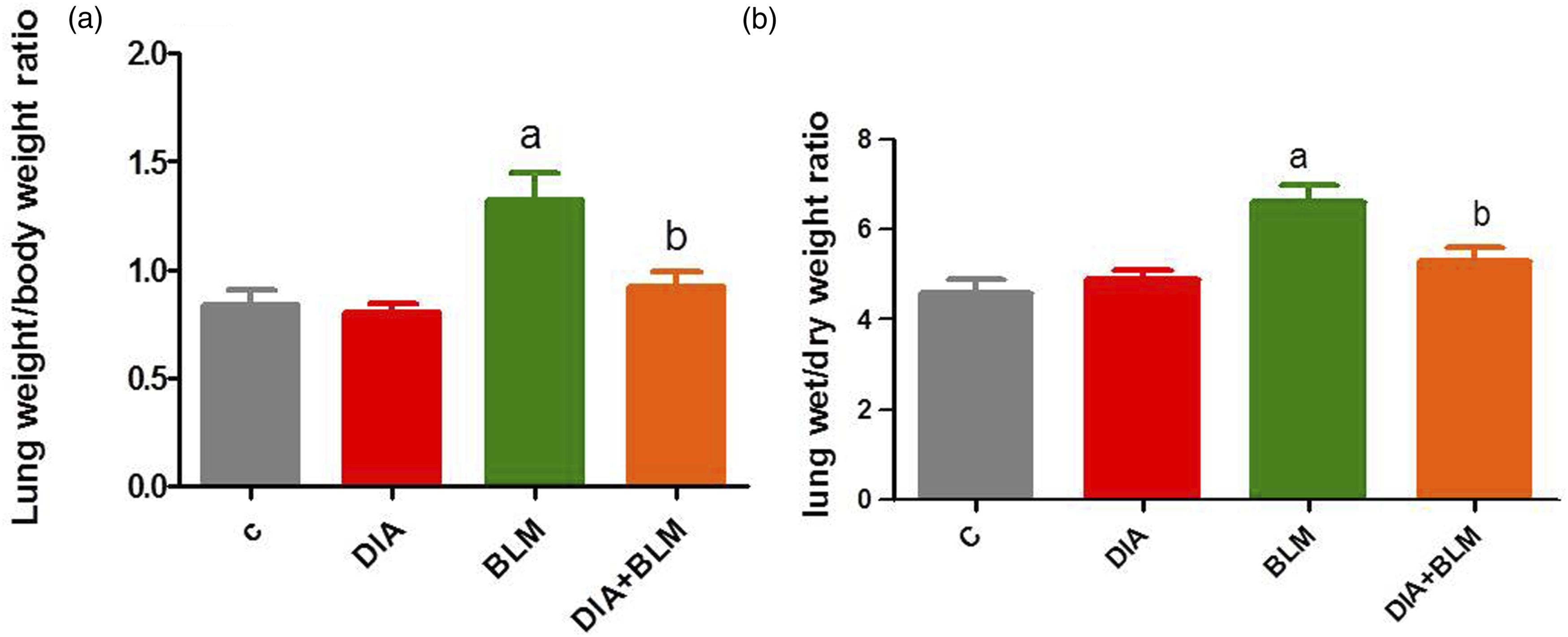
Effect on lung wet/dry weight ratio
Administration of BLM caused a significant increase in the lung wet/dry weight ratio compared to control group. Administration of DIA in combination with BLM caused a significant decrease in the lung wet/dry weight ratio compared to BLM group (Figure 1(b)).
Effect of DIA on lung oxidative stress/antioxidant biomarkers
The lung tissue SOD and GSH levels were significantly decreased in BLM induced PF in rats when compared to the control group. DIA pretreated group displayed a significant elevation in their levels in relation to the BLM induced lung fibrosis group. A significant elevation in lung tissue MDA level in comparison to the control group was evident while DIA pretreatment succeeded in significantly reducing their lung tissue level in comparison to BLM induced lung fibrosis in rats (Figure 2). Effect of Diacerein (DIA) for 14 days on: superoxide dismutase (SOD), reduced glutathione (GSH) and lung malondialdehyde (MDA) in BLM- induced lung fibrosis.
Effect of DIA on lung tissue contents of TLR4 and MyD88
The lung tissue levels of TLR4 and MyD88 were significantly elevated in the BLM induced lung fibrosis group in comparison with the control group and their levels were significantly decreased in DIA pretreated rats (Figure 3). Effect of diacerein (DIA) for 14 days on lung toll- like receptor (TLR4) and lung myeloid differentiation response 88 (MyD88) in BLM- induced lung fibrosis.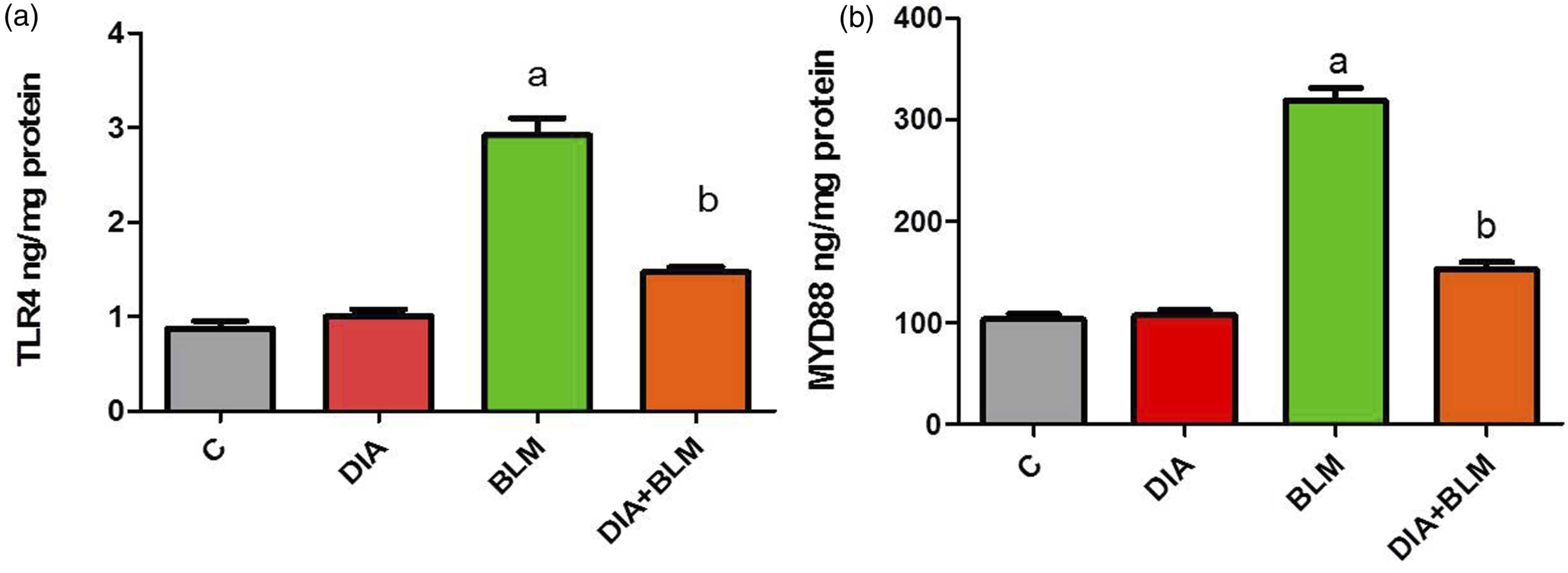
Effect of DIA on lung tissue histological examination
Figure 4(a) and (b) of the control and DIA control groups displayed a normal histological structure of the lung tissue. The alveoli (A) and alveolar sacs (S) were patent and lined with pneumocytes type I (PI) and II (PII). PI was flat and more numerous, and PII-rounded cells that might appear protruded into the alveolar lumen. The interalveolar septa were delicate containing blood capillaries (BC) and macrophages. Effect of DIA on lung tissue histological examination.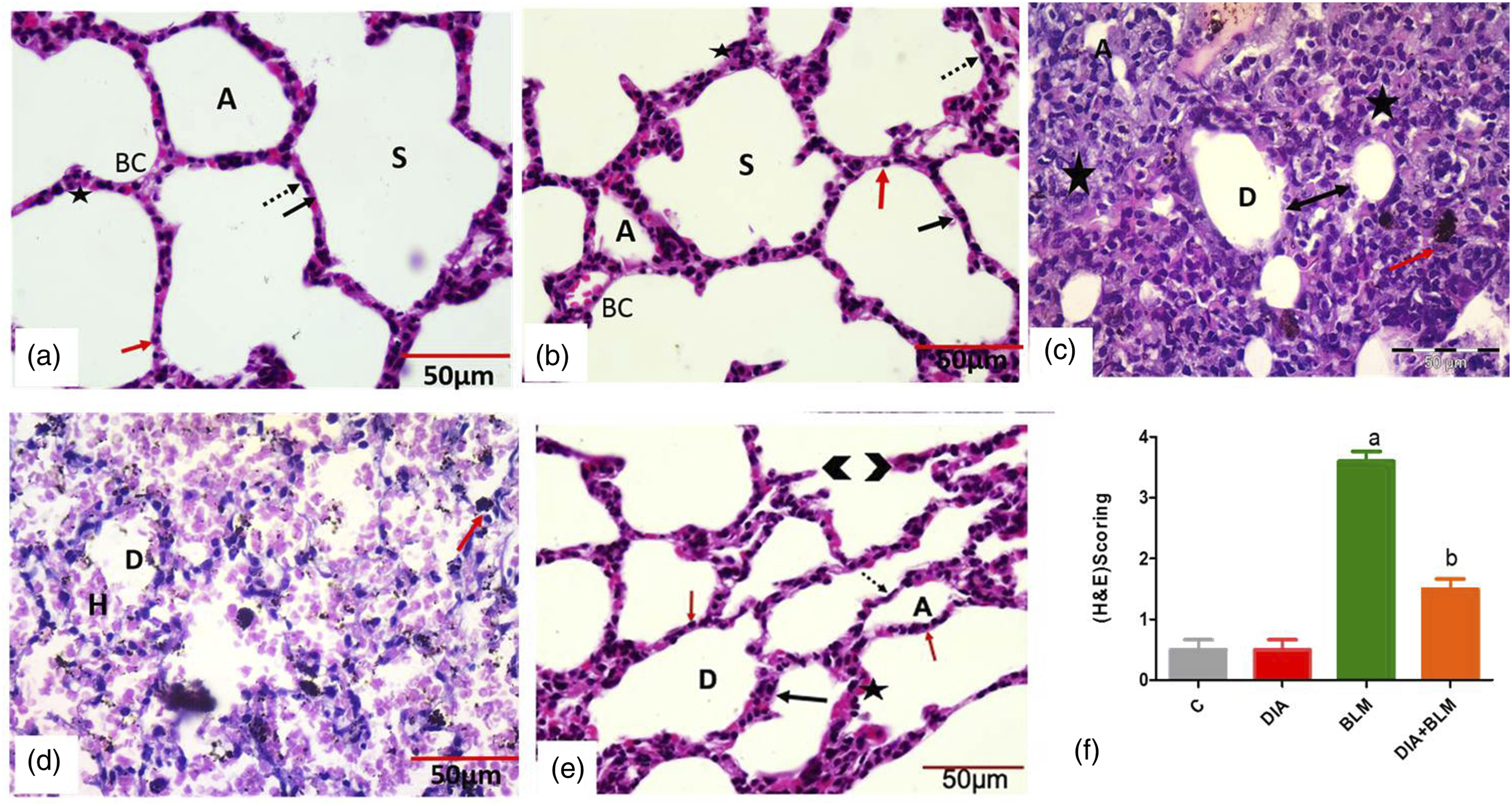
Figure 4(c) and (d) of BLM group showed distorted histological architecture of the lung tissue with collapsed (C) and compensatory dilated (D) alveoli with thick inter-alveolar septa studded with extravasated RBCs and inflammatory cells infiltration. Areas of hemorrhage and extravasated RBCs inside the alveoli. Numerous hemosiderin laden alveolar and interstitial macrophages were obviously noticed. Areas of consolidation and interstitial edema were noticed. While in Figure 4(e) Rats of the DIA pretreated group displayed a significant improvement in the previously mentioned histological findings in comparison to the BLM group. The previously mentioned results matched with the enhancement of the histopathological finding of the modified Passmore score (Figure 4(f)).
As regard staining with Masson trichrome for detection of collagen deposition, the control and DIA groups showed minimal collagen deposition around the blood capillaries and within interalveolar septum (Figure 5(a) and (b)). While In BLM group untreated group showed a marked increase in collagen deposition around the blood vessel and within interalveolar septum (Figure 5(c)). Rats of the DIA pretreated group showed mild collagen deposition around the blood vessel and within interalveolar septum (Figure 5(d)). The previously mentioned results matched with the enhancement of the mean area fraction of collagen fibers (Figure 5(e)). Effect of DIA on lung tissue histopathological collagen fibers deposition score.
Effect of DIA on lung immunohistochemical staining of anti-NF-κB, and anti TGF-β antibodies
In the control and DIA control groups, the mean area fraction of anti-NF-κB, and anti TGF-β antibodies detected in the lung tissue showed low expression (Figures 6(a) and (b) and 7(a) and (b)). In the BLM group, the mean area fraction was significantly elevated in comparison to the control group (Figures 6 and 7(c)), Whereas, anti-NF-κB, and anti TGF-β antibodies area fraction in DIA pretreated rats was significantly decreased in comparison to the BLM group (Figures 6 and 7(d)). Semi-quantitative analysis of anti-NF-κB, and anti TGF-β antibodies area fraction (Figures 6 and 7(e)). Effect of DIA on lung immunohistochemical staining of anti-NF-κB antibody. Effect of DIA on lung immunohistochemical staining of anti TGF-β antibody.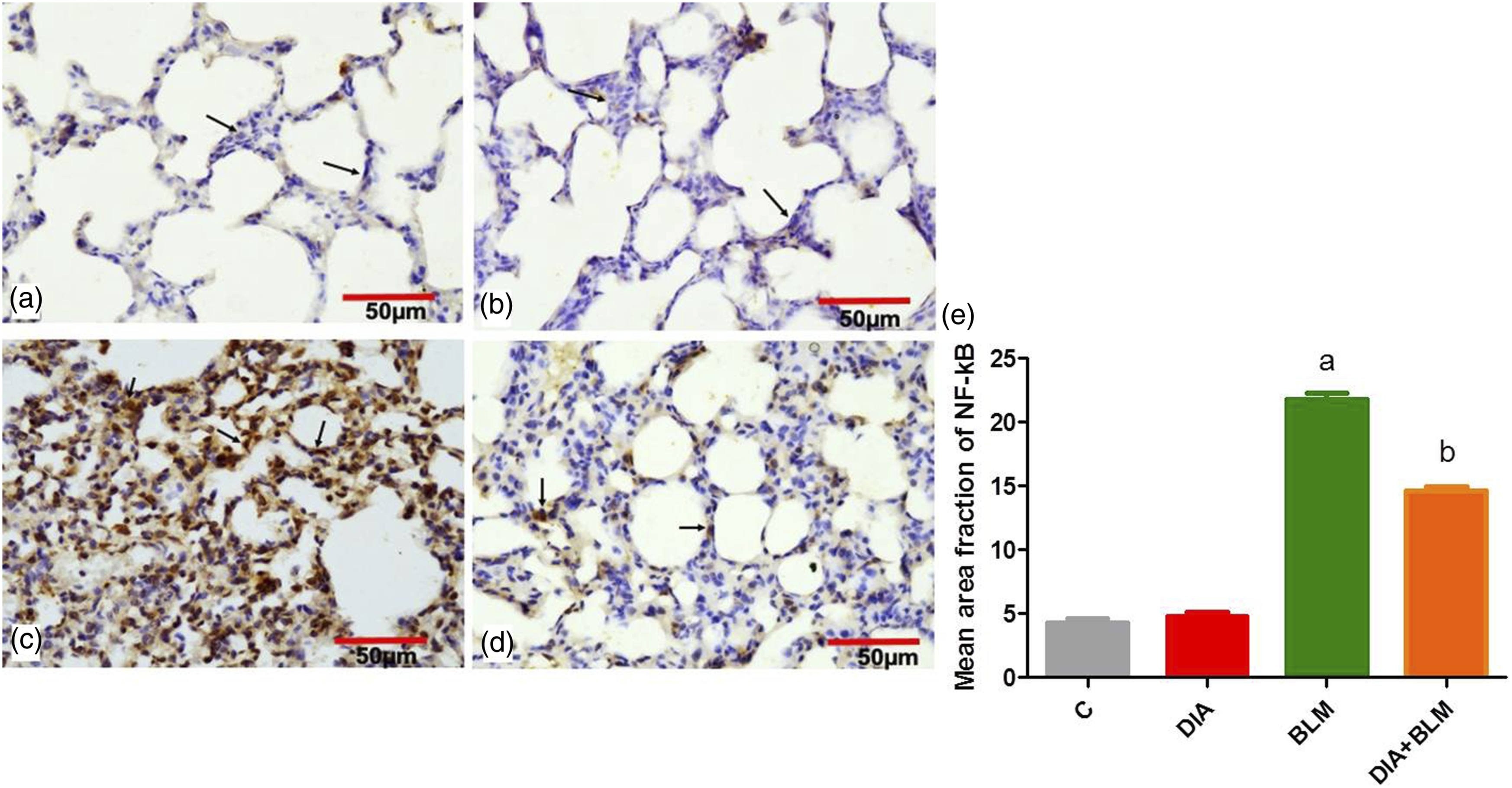

Discussion
A number of diffuse parenchymal lung illnesses progress to pulmonary fibrosis, a potentially fatal condition marked by increasing dyspnea and deteriorating pulmonary function, 28 it is marked by excessive matrix deposition and deterioration of the lung architecture which finally leads to respiratory insufficiency. 29
The pathophysiological basis of PF has been a source of controversy for decades. 30 PF can be triggered by a persistent irritant, such as toxic chemicals, allergens, radiation and environmental particles. 31 Till now, there is no effective treatment for PF. 32 Inflammation, oxidative stress, and accumulation of inflammatory cells in the airways are thought to play key roles in the pathophysiological process of PF.20,33
Numerous animal models have been developed in order to evaluate potential therapeutics and better understand the pathogenesis of PF. 34 BLM is the chosen agent for simulating PF in rodents in general. The most common route of administration of BLM is intratracheal which cause lung toxicity and fibrosis. 35
Seven days after delivery, intratracheal instillation increases alveolar epithelial cell damage and triggers an inflammatory response resembling acute lung injury. The rise in profibrotic markers’ expression is correlated with the rise in proinflammatory cytokines. After the acute phase, the injured sites undergo a subacute phase with fibroblast proliferation and differentiation. 36 The onset of collagen deposition and fibrosis can be observed around day 14.
The current study simultaneously evaluated the effects of IL-1β antagonist: (DIA), on BLM-induced PF in rats. The design of this study allowed us to record the early evidences of lung injury and affection on lung weight/body ratio, wet/dry ratio. In additions to record the evidence of lung inflammation, fibrosis and oxidative stress processes.
The present study revealed that BLM caused a significant increase in the lung weight/body weight ratio as administration of BLM caused significant decrease in the body weight in comparison with the control group, 37 this may indicate BLM-induced systemic toxicity and pulmonary damage associated with pulmonary edema. 38 DIA administration improved lung weight/body weight ratio via its anti-edematous effect via its anti-inflammatory effect. 39 DIA anti-inflammatory due to blocking the synthesis of TNF-α, IL-6, and IL-1β via the inhibition of the activation of the NF-κB pathway. 11
In addition, DIA has been found that it can prevent and treat metabolic diseases such as obesity, hyperlipidemia, 40 as it causes diarrhea leading to body weight loss; 41 the unabsorbed DIA in the gastrointestinal tract (GIT) is totally converted to rhein (producing laxation) before reaching the systemic circulation explains this effect. 42
The lung W/D weight ratio was used to determine pulmonary edema. 43 In our study, BLM administration exhibited a significant increase in W/D ratio compared with control group because of pulmonary edema which is in line with other previous study. 44 In contrast, treatment with DIA ameliorate W/D ratio compared to BLM group, this indicates its ability to decrease pulmonary edema because of its anti-inflammatory effect through inhibition of IL-1β and TNF-α and anti-hypersensitivity properties of DIA which coincide with previous studies of.30,39
Oxidative stress with excessive ROS production and release of inflammatory mediators are considered to be the causative factors of BLM-associated damage in the lung tissue due to DNA oxidation with lipids peroxidation which subsequently increases MDA levels. 40 Imbalance between free radical formation and antioxidant defense mechanism causes lung tissue damage. 45
SOD enzyme and GSH are the main line of defense against oxidative stress. They promote the oxidation of oxygen to be inactive substances. Usually, the harmful sequel of superoxide is inhibited by SOD by converting them into hydrogen peroxide. In PF levels of these enzymes in lung tissues become exhausted as a consequence of oxidative damage caused by free radicals and causes impairment of these defense mechanisms making hydrogen peroxide split into hydrogen and oxygen free radicals and destroy biological molecules, like amino acids, nucleic acids, and membrane transport proteins. 36
MDA is a byproduct of lipid peroxidation occurring in the tissue. 46 In PF, levels of MDA in the lung tissue increases significantly. 47 Lung injury was detected in the current model with significant alterations in oxidative stress markers. The present study found that BLM administration resulted in a decrease in GSH, SOD levels and increase in MDA level in the lung tissue. 48 More else the results of the present study are consistent with previous study of Mansouri et al. 33 which reported the increased lipid peroxidation and decreased anti-oxidant systems in BLM-induced PF during the evaluation of anti-oxidant and anti-inflammatory profile of Zingerone in experimentally-induced PF.
Interestingly, DIA significantly improved oxidative stress in BLM-induced PF in the current study in the form of restoration of normal levels of SOD and GSH, with reduction of MDA levels in the lung tissue due to their potential antioxidant activity. Our data agree with the previously reported study of Abd-Ellatif et al. 13 which assessed the effect DIA against glycerol-induced acute kidney injury by modulating oxidative stress, inflammation, apoptosis, and necroptosis.
In the histopathological findings of the current study, BLM-induced PF group showed distorted histological architecture of the lung tissue with collapsed and compensatory dilated alveoli with thick inter-alveolar septa studded with extravasated red blood cells (RBCs) and inflammatory cells infiltration, areas of hemorrhage and extravasated RBCs inside the alveoli, areas of consolidation and interstitial edema were noticed, to some extent similar findings were reported by Latta et al. 49 Also, administration of DIA ameliorates the histopathological finding in which lung tissue apparently returned to normal whereas revealed well-formed alveoli and alveolar sacs, thick inter-alveolar septa with marked decrease in inflammatory cells infiltration in some areas, marked decrease in vascular congestion due to its anti-inflammatory and anti-oxidant effect.50,51
Oxidative stress activates several intracellular signaling pathways resulting in up-regulation of pro-inflammatory cytokine synthesis. 52 An essential mechanistic link between oxidation and inflammation is related to increased pro-inflammatory cytokines e.g. NF-kB and TNF-α. 53
Given TLR is a key regulator of inflammation which can recognize and bind to specific molecular structures of host apoptosis surfaces. 54 Activation of TLR4 results in the recruitment of its downstream via MyD88 leading to activation of NF-κB by translocation into the nucleus upregulating several pro-inflammatory factors levels e.g., IL-1β and TNF-α It is worth mentioned that TLR4 regulates inflammation and tissue injury in different animal models, for example; ischemia/reperfusion, Alzheimer’s disease and autoimmune disorders. 55
TLR4 is an essential mediator of inflammation that has a role in the pathophysiology of a variety of lung diseases. 55 It has been shown to activate both proinflammatory and profibrotic pathways in PF.56,57 TLRs are thought to have a role in the development of PF through regulating inflammation and wound healing. 58
TLR4-mediated downstream signaling pathways included primary MyD88 dependent and independent pathways. Primary MyD88 dependent pathway mainly regulated the expression of a variety of inflammation related genes, which transmitted intracellular signals via TLR domain of MyD88. This could activate the transcription factor NF-κB, causing the inflammatory factors IL-1, IL-6, and TNF-α to be released. 59 Also, MyD88 is essential in mediating the classic airway inflammatory response to repetitive organic dust extract, 60 it is also a therapeutic target for inflammatory lung diseases. 61
NF-
In the current study, administration of DIA reduced significantly level of TLR4 and MyD88 in the lung tissue and reduce the immunoexpression of NF-
TGF- β1 has been identified to influence a variety of biological processes, including cell proliferation, differentiation, migration, apoptosis, and the production of extracellular matrix. 66 Particularly, it is thought be a critical mediator of fibrosis. Immunoexpression of TGF- β1 levels in the lung tissue of rats with PF increased significantly, indicating that TGF- β1 is closely linked to PF. 67 TGF- β1 may inhibit epithelial cell proliferation, re-program epithelial cells into mesenchymal cells, and stimulate extracellular matrix formation in PF through modulating downstream regulators. 68 In this present study, BLM-induced PF was manifested by elevation of the lung TGF- β1 immunoexpression, our results are in accordance with previous study. 55 Furthermore, the current findings showed that administration of DIA ameliorated the effect of BLM evidently by decreasing level of TGF- β1 immunoexpression in lung tissue. This indicates the beneficial effect of DIA in reducing the fibrosis which agrees with Barakat and his colleges. 69
Conclusion
The present study concluded that ROS and inflammatory pathways may largely participate in the mechanism of pathogenesis of PF. DIA (25 mg/kg orally) can alleviate the inflammatory response mediated by the oxidative stress injury. An evidence was presented by our findings supporting the concept that DIA has antioxidant and anti-inflammatory effects, via its ability to suppress TLR4/MyD88/NF-
Footnotes
Authors’ contributions
AA, EF, MM, and HH suggested the point of research suggestion, performed the experiment, statistically analyzed the data, write, revised, and proofread the final manuscript. AA carried out the histological and immunohistochemical part of the experiment and wrote the corresponding parts of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
