Abstract
Background
Cisplatin (DDP) resistance in ovarian cancer (OC) patients usually leads to treatment failure and increased mortality. Anlotinib has been shown to improve progression-free survival and overall survival in patients with platinum-resistant ovarian cancer, but the mechanism is unclear. This study aims to explore the mechanism by which anlotinib ameliorates platinum resistance in OC cells.
Methods
Cell viability was detected by the 3-4,5-dimethylthiazol-2,5-diphenyltetrazolium bromide (MTT) method, and the apoptosis rate and changes in the cell cycle distribution were evaluated by flow cytometry. Bioinformatics analysis was used to predict the potential gene target of anlotinib in DDP-resistance SKOV3 cells, and its expression was verifies it by RT-qPCR, western blotting and immunofluorescence staining. Finally, ovarian cancer cells overexpressing AURKA were constructed, and the predicted results were verified by animal experiments.
Results
Anlotinib effectively induced apoptosis and G2/M arrest in OC cells and decreased the proportion of EdU-positive cells. AURKA was identified as a possible key target of anlotinib for inhibiting tumorigenic behaviors in SKOV3/DDP cells. Through combined immunofluorescence and western blot analyses, it was demonstrated that anlotinib could effectively inhibit the protein expression of AURKA and upregulate the expression of p53/p21, CDK1, and Bax protein. After overexpression of AURKA in OC cells, the induction of apoptosis and G2/M arrest by anlotinib were significantly inhibited. Anlotinib also effectively inhibited the growth of tumors in nude mice injected with OC cells.
Conclusions
This study demonstrated that anlotinib can induce apoptosis and G2/M arrest in cisplatin-resistant ovarian cancer cells through the AURKA/p53 pathway.
Introduction
Ovarian cancer is a solid malignant tumor in the female reproductive system and ranks behind only cervical cancer and endometrial cancer in incidence. 1 In recent years, exploring the mechanism of drug resistance in ovarian cancer and developing novel drug resistance reversal agents have become hot research emphases.
Anlotinib is a multitargeted tyrosine kinase inhibitor that inhibits tumor progression by targeting angiogenesis-related growth factor receptors, including VEGFR 1/2/3, FGFR 1/2/3, and other tumor-associated kinases, such as c-Kit and Ret. 2 It is approved in China as a single agent for second or later-line treatment of patients with locally advanced or metastatic non-small cell lung cancer (NSCLC). 3 In some clinical studies, anlotinib treatment resulted in modest improvements in progression-free survival and overall survival in patients with platinum-resistant or platinum-refractory ovarian cancer. This suggests that anlotinib may be a new treatment option for patients with platinum-resistant or platinum-refractory ovarian cancer. 4 However, more research is needed to prove whether anlotinib can reverse drug resistance in ovarian cancer and by what mechanism tumor suppression is achieved.
Aurora Kinase A (AURKA) is a serine/threonine kinase essential for controlling mitotic progression, centrosome maturation/separation, and mitotic spindle function. 5 Recent studies have mentioned that AURKA may be involved in the development and progression of cancer and tumorigenesis. AURKA has been shown to be significantly upregulated in most ovarian cancer tissues. 6 Although there are no studies on AURKA in drug-resistant ovarian cancer cells, cisplatin-resistant SKOV3 ovarian cancer cells showed higher levels of AURKA expression. Therefore, it is hypothesized that the increased expression of AURKA may be associated with the cisplatin resistance of SKOV3 cells. The GEO data (GSE109468 and GSE163574) showed that anlotinib significantly inhibited AURKA expression in both synovial sarcoma and pancreatic cancer. Therefore, this study focused on whether anlotinib can reverse the DDP resistance-related properties of ovarian cancer cells through AURKA.
Materials and methods
Reagents and cell culture
The ovarian cancer cell line SKOV3 was obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). The DDP-resistant SKOV3 ovarian cancer cell line (SKOV3/DDP) was purchased from Zhejiang Ruyao Biotechnology Co. Cells were cultured in Dulbecco’s modified Eagle’s medium (11965092, DMEM, Invitrogen, USA) supplemented with 10% fetal bovine serum (FBS, Invitrogen, Carlsbad, CA, USA), 1% streptomycin, and 1% ampicillin. Passage cultures were incubated in a constant temperature incubator at 37°C containing 5% CO2.
Cell proliferation assay
Cells were inoculated into each well of a 96-well plate (6 × 103 cells/well) and incubated for 24 h (37°C in a constant temperature incubator with 5% CO2). Subsequently, the cells were treated with different concentrations (0, 0.5, 1, 5, 10, 20, 40, and 80 μM) of cisplatin or anlotinib for 24 h. After that, the MTT assay was used for detection. 7 The results are expressed as the cell inhibition rate. SKOV3/DDP cells, cells were subgrouped in grouping A: 1. DDP group (5 μM DDP treatment), 2. DDP+AURKA group (5 μM DDP treatment after overexpression of AURKA in SKOV3/DDP cells), 3. DDP+anlotinib (An) group (5 μM DDP and 5 μM anlotinib treatment) and 4. DDP + AURKA + An group (5 μM DDP and 5 μM anlotinib treatment in SKOV3/DDP cells overexpressing AURKA after administration of 5 μM anlotinib treatment). The MTT assay was performed after treating the cells according to the grouping for 24 h. Inhibition rate = 1 - OD of the drug treatment group/OD of the control group.
Flow cytometric analyses of apoptosis and the cell cycle
Cells were subgrouped in grouping B: 1. SKOV3 group; 2. 0 μM group; 3. 0.5 μM group; 4. 1 μM group; 5. 5 μM group. Subgroups 2, 3, 4, and 5 contained SKOV3/DDP cells treated with 0, 0.5, 1, 5 μM anlotinib, respectively, for 24 h. Apoptosis and cell cycle was detected by flow cytometry. 8 Data were obtained by FlowJo V10 (BD Biosciences, USA). For grouping A, cells were processed according to the grouping information, and the cell cycle analysis was performed.
EdU incorporation assay
Cells in groupings A and B were treated according to the subgroups after 24 h. It was sufficient to refer to the instructions of the BeyoClick™ EdU-647 Cell Proliferation Assay Kit (C0081S, Beyotime, China), according to the method of Yang et al. 9
Bioinformatics analysis
The dataset containing differential expression gene related to cisplatin resistance in SKOV3 cells (GSE98559) was first searched through the GEO database (https://www.ncbi.nlm.nih.gov/geo/). The GEO database was then searched for datasets of differential gene expression after anlotinib treatment in synovial sarcoma cells (GSE109468) and pancreatic cancer cells (GSE163574). The data were screened for genes that were downregulated after anlotinib treatment but upregulated in SKOV3 cisplatin-resistant cell lines, and these overlapping enriched genes were identified as potential genes. Canonical SMILES for anlotinib were obtained using the Pubchem database (https://pubchem.ncbi.nlm.nih.gov/), and anlotinib targets (using SMILES notation) were predicted through online sites such as SEA (https://sea.bkslab.org/) and Swisstarget (http://swisstargetprediction.ch/). Finally, the GEO database screening results and anlotinib target prediction results were analyzed, and AURKA was identified as a key target gene through which anlotinib to exert its inhibitory effect on cisplatin-resistant ovarian cancer cells.
The interaction relationship between anlotinib and the AURKA protein was then predicted. Firstly, the 3D structural formula of anlotinib was downloaded from Pubchem. The structure of the AURKA protein (PDB ID: 2BMC) was then obtained from Protein Data Bank (https://www.rcsb.org). Finally, the molecular binding energy between anlotinib and AURKA was calculated by PyMOL, and AutoDock molecular docking software was used to visualize the simulated docking results. The specific method can be found in the study of Daniel et al. 10
RNA isolation and real-time quantitative PCR (RT-qPCR)
Cells from grouping B were collected after 24 h of treatment. Total RNA was isolated using TRIzol reagent. Two micrograms of mRNA were reverse transcribed into cDNA using the first Strand cDNA Synthesis Kit (gDNA Purge) kit (E042, Novoprotein, China). qPCR was performed using a SYBR qPCR SuperMix Plus (E096, Novoprotein) kit, and the following thermal cycling program was run on a 7500 Fast Real-Time PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc.). β-actin was used as the internal reference gene for normalization. The relative expression of target genes was calculated using the 2−ΔΔCt method. 11 The amplification primers were designed through the NCBI online website with the following sequences: AURKA-Forward: 5′-GCA ACC AGT GTA CCT CAT CCT G-3’; AURKA-Reverse: 5′-AAG TCT TCC AAA GCC CAC TGC C-3’; β-actin-Forward: 5′- CAC CAT TGG CAA TGA GCG GTT C-3’; β-actin-Reverse: 5′- AGG TCT TTG CGG ATG TCC ACG T-3’.
Western blotting
Cells from groupings A and B (5 × 106) as well as nude mouse tumor tissues (100 mg) were taken and RIPA lysis buffer (20 μL of phosphatase inhibitor Cocktail per mL of lysis buffer) was added, and the cells and tissues were homogenized with a tissue crusher. Western blot was used to detect the protein expression levels of Bax, Bcl2, CDK1, p53, AURKA, and β-actin according to Yang et al. 9 The ChemiDoc-It Imaging System was used for imaging. The optical density values were quantified by ImageJ software. β-actin was used as the internal reference protein for normalization. All the above antibodies were purchased from the Abcam (UK).
Cellular immunofluorescence
SKOV3 and SKOV3/DDP cells were cultured on coverslips, and immunofluorescence staining was performed after 24 h of treatment of cells according to cell grouping B. Referring to the method of Liu et al. 12 Antibodies are mainly involved: AURKA p53, and Goat Anti-Rabbit IgG-FITC (ab6717, Abcam). Cells were visualized and imaged by fluorescence microscopy (DM500, Leica), and cells in different fields of view were observed and imaged. IPP6.0 was used to statistically quantified the results.
AURKA overexpression plasmid construction
First, the CDS of AURKA was searched in the NCBI database, and primers were designed for amplification of the full-length sequence. A XbaI digestion site was introduced at the upstream 5′ end of the primer sequence, and a BamHI digestion site was introduced at the downstream 3′ end of the primer. The pCDH-CMV-GFP-puro vector was used for plasmid construction, and the PCR product and vector were double digested using the XbaI and BamHI restriction endonucleases (New England Biolabs, Inc., USA) and ligated overnight at 16°C using T4 DNA ligase (Takara, Japan). The recombinant plasmids were transformed into the DH5α competent cells by heat shock at 42°C and spread on solid LB medium for culture. The single clones were selected and sequenced. After the successful construction of the pCDH-AURKA recombinant plasmid was confirmed, plasmid extraction was performed for lentiviral infection. pCDH-AURKA was amplified after introduction of a XbaI digestion site at the 5′ end of the CCNA2 upstream primer and an BamHI digestion site at the 3′ end of the downstream primer. The amplification primer sequences were as follows: AURKA-XbaI-F: 5′-TGC TCT AGA ATG GAC CGA TCT AAA GAA AAC TGC-3′, AURKA BamHI-R: 5′-AGT GGA TCC AG ACT GTT TGC TAG CTG ATT C-3’. The length of the amplification product was 1209 bp.
Lentiviral infection of ovarian cancer cells
Recombinant lentivirus was prepared by cotransfection. The recombinant plasmid (pCDH-AURKA), Δ8.91, and pVSV-G (mass ratio 10:10:1) were transfected into 293T cells by the cationic lipid complex method (X-TremeGENE HP DNA transfection reagent, Roche). Viruses were collected after 72 h. The viral suspension was passed through a 0.45 μm filter and added to infect the target cells at an exact titer for transduction in the presence of polybrene (final concentration 2 μg/mL). After 24 h of transduction and the visualization of a considerable fluorescence signal under the microscope, cell resistance screening was performed by replacing the conventional medium with medium containing 1 μg/mL puromycin and maintaining cell growth for 7–9 days. The medium was then replaced with a complete medium without puromycin. Cells with stable expression were subjected to qPCR and western blotting techniques to verify AURKA overexpression in ovarian cancer cells.
Investigation of the regulation of AURKA by anlotinib
After 24 h of treatment of cells according to grouping A, the following experimental assays were performed: 1. The viability rate in each group of cells after 24 h was detected by an MTT assay; 2. The cell cycle phases were detected by flow cytometry (because cells with overexpression of AURKA expressed green fluorescence, apoptosis detection by flow cytometry is affected); 3. An EdU incorporation assay was performed to quantify cell proliferation within 4 h (24–28 h after anlotinib treatment); 4. Western blotting was performed to evaluate the expression of apoptosis-related proteins (Bax and Bcl2), cell cycle-related proteins (CDK1), and AURKA/p53/p21 proteins. The above study verified that anlotinib affects cell proliferation and apoptosis levels through the AURKA/p53 pathway.
Subcutaneous xenograft experiment in nude mice
Eighteen 4-week-old BalB/C athymic nude mice were used as experimental animals. The nude mice were randomly divided into three groups: 1, control group (n = 6, injected with an equal volume of 0.9% NaCl); 2, DDP group (n = 6, treated with 3 mg/kg cisplatin after tumor formation, i.p); and 3, DDP+anlotinib group (n = 6, treated with 3 mg/kg cisplatin +3 mg/kg anlotinib, i.p). All mice were injected subcutaneously with 100 μL of a suspension of SKOV3/DDP cells (containing 1 × 106 cells) in the dorsal scapular region. Tumor size and volume were measured twice during every 7-day period using a caliper, tumor volume = 1/2 (length × width 2 ). Once the mean tumor volume was 100 mm3, the mice were administered intraperitoneal injections every 3 days for a total of 28 days. The nude mice were sacrificed on Day 28, and tumor tissues were obtained.13,14 Some tumor tissues were used for western blotting to measure the expression levels of apoptosis-, cell cycle-, and AURKA/p53-related proteins. It was approved by the Ethics Committee of the Second Affiliated Hospital of Harbin Medical University (No. SYDW2023-004).
Immunohistochemical (IHC) analysis
IHC staining was performed on 5 μm paraffin sections of tumor tissue as described in the study of Yang et al. 9 AURKA, p53, and Ki67 antibodies were mainly involved. Images were acquired under a light microscope (DM500, Leica). Three sites were randomly selected for each set of micrographs, and the optical density values were quantified and analyzed using IPP6.0.
Statistical analysis
All data in the current study are presented as the mean ± standard deviation values based on triplicate experiments. All results were calculated using GraphPad Prism Software (version Prism 8; GraphPad Software, Inc.). Student’s t-test was used for two-group comparisons. p < 0.05 was considered to indicate a statistically significant difference.
Results
Anlotinib affects the viability and proliferation of SKOV3/DDP cells
This part of the study focused on functional effects assays to demonstrate the inhibitory effect of anlotinib on cisplatin-resistant SKOV3 cells. First, the sensitivity of SKOV3/DDP cells to cisplatin at concentrations ranging from 1-80 μM was significantly lower than that of wild-type SKOV3 cells (Figure 1(a), p < 0.05), as measured by the MTT assay. The IC50 of cisplatin in SKOV3 cells was 4.331 μM. The IC50 of cisplatin in SKOV3/DDP cells was 47.68 μM. In contrast, in the anlotinib cell sensitivity study, the IC50 was 17.34 μM in SKOV3/DDP cells treated with anlotinib only. In cells cotreated with DDP and anlotinib, the IC50 was 4.047 (Figure 1(b)). When the anlotinib concentration was greater than 0.5 μM, the difference in cell viability between the two groups was statistically significant (p < 0.05). The percentages of EdU-positive SKOV3/DDP cells in a proliferative state at 4 h were 20.6 ± 3.2, 14.6 ± 2.2, and 9.5 ± 0.5 after treatment with 0.5, 1, and 5 μM anlotinib treatment, respectively, significantly lower than the percentage of 26.8 ± 4.9 in the 0 μM group (Figure 1(c), p < 0.05). Anlotinib affects the viability and proliferation of SKOV3/DDP cells. (a)-(b). MTT assay of the effect of DDP and anlotinib on the viability of SKOV/DDP cells; (c). EdU incorporation assay of cell proliferation and statistical quantification of the proportion of EdU positive cells by IPP 6.0. *p < 0.05, and **p < 0.01 indicate statistically significant differences compared with the control group; #p < 0.05 and ##p < 0.01, indicate statistically significant differences compared with the 0 μM group.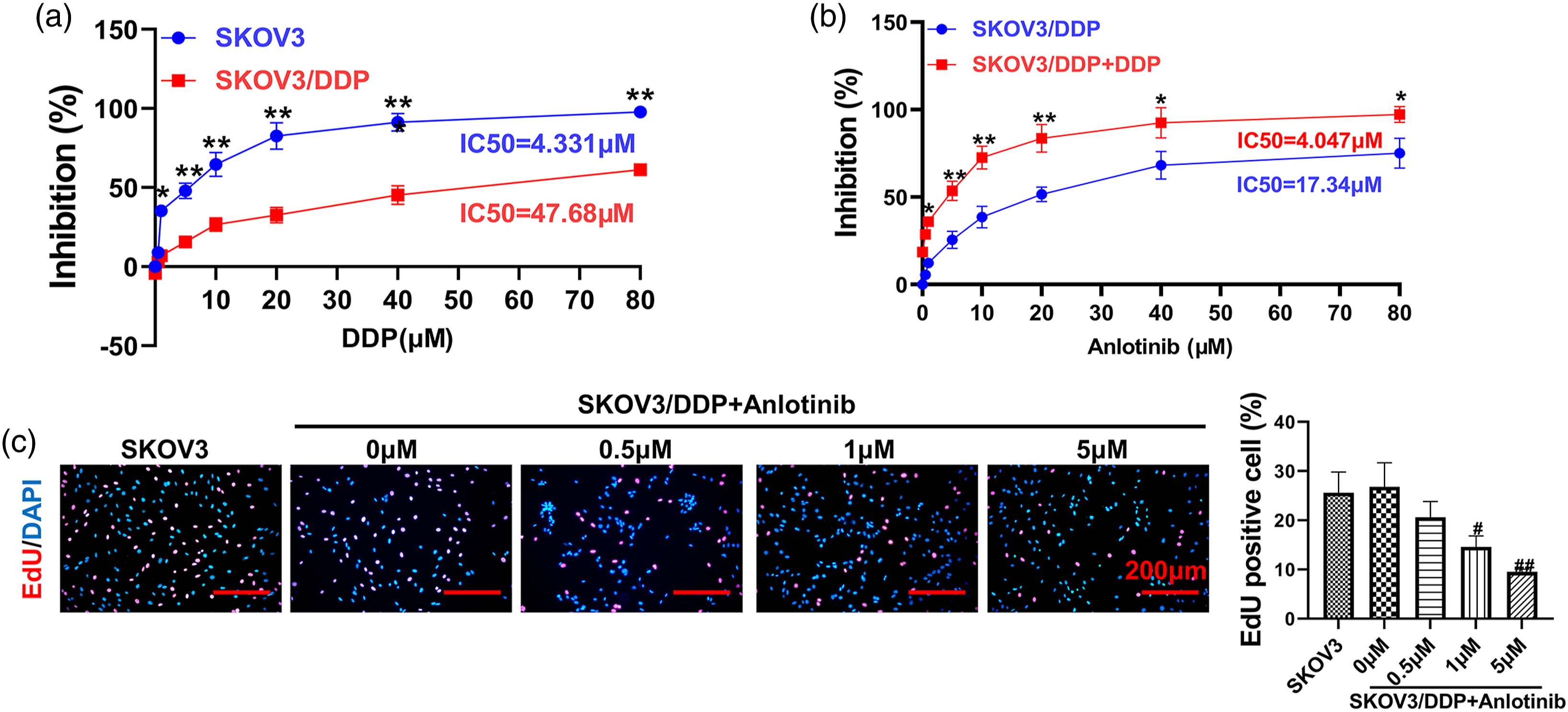
Anlotinib affects the G2/M phase arrest and apoptosis of SKOV3/DDP cells
The results of flow cytometry assays of apoptosis (Figure 2(a)) and the cell cycle (Figure 2(b)) suggested that 0.5, 1, and 5 μM anlotinib enhanced apoptosis levels in SKOV3/DDP cells (p < 0.05 compared with the 0 μM anlotinib group). The percentage of SKOV3/DDP cells in the G2/M phase were 18.0 ± 2.2, 24.0 ± 1.1, and 35.9 ± 1.2 after treatment with 0.5, 1, and 5 μM anlotinib, respectively, significantly higher than the percentage of 11.3 ± 0.8 in the 0 μM group (p < 0.05). Anlotinib affects the G2/M phase arrest and apoptosis of SKOV3/DDP cells. (a). Detection of apoptosis by flow cytometry and statistical quantification; (b). Cell cycle analysis by flow cytometry and statistical quantification by FlowJo V10. *p < 0.05, and **p < 0.01 indicate statistically significant differences compared with the SKOV3 group; #p < 0.05 and ##p < 0.01, indicate statistically significant differences compared with the 0 μM group.
Effect of anlotinib on p53/AURKA pathway in the SKOV3/DDP cells
In this part of the study, data from the GEO, SEA, and SwissTarget databases were combined to predict the therapeutic targets of anlotinib in SKOV3/DDP cells, and AURKA was identified as a possible potential target of anlotinib. The molecular docking simulations of the AURKA protein in complex with anlotinib (Figure 3(a)) showed a covalent bond between anlotinib and the AURKA protein with a binding energy of −8.9 kcal/mol. GEO data analysis showed that the gene expression level of AURKA was significantly higher in SKOV3/DDP cells than in SKOV3 cells (Figure 3(b), p < 0.01). In this experiment, the AURKA gene expression level was significantly higher in SKOV3/DDP cells than in SKOV3 cells (Figure 3(c), p < 0.01). The expression of the AURKA gene was significantly inhibited in SKOV3/DDP cells after 0.5, 1, and 5 μM anlotinib treatment (p < 0.01). The results of the cellular immunofluorescence assay demonstrated that 0.5, 1, and 5 μM anlotinib treatment significantly inhibited AURKA protein expression in SKOV3/DDP cells while upregulating the expression of the downstream pathway oncoprotein p53 (Figure 3(d), p < 0.01). Effect of Anlotinib on p53/AURKA pathway in the SKOV3/DDP cells. (a). Visualization of anlotinib and AURKA protein docking by PyMOL software; (b). Gene expression level of AURKA genes in the GEO database of differentially expressed genes in SKOV3 and SKOV3/DDP cells; (c). Measurement of the cellular AURKA gene expression level by qPCR; (d). Immunofluorescence analysis of AURKA and p53 protein subcellular localization and expression levels, and statistical quantification of the fluorescence intensity by IPP6; *p < 0.05 and **p < 0.01 indicate statistically significant differences compared with the SKOV3 group; #p < 0.05 and ##p < 0.01, indicate statistically significant differences compared with the 0 μM group.
Anlotinib regulates SKOV3/DDP cell cycle and expression of apoptotic proteins through p53/AURKA pathway
Furthermore, western blot analysis (Figure 4(a) to (g)) demonstrated that 0.5, 1, and 5 μM anlotinib treatment significantly inhibited AURKA protein expression in cells, and upregulated the expression of p53/p21. In contrast, the expression of the proapoptotic protein Bax was upregulated, that of the antiapoptotic protein Bcl2 was downregulated, and that of the G2/M checkpoint-phase process-related protein CDK1 was significantly inhibited (p < 0.05). Anlotinib regulates SKOV3/DDP cell cycle and expression of apoptotic proteins through p53/AURKA pathway. (a). Western blot analysis of AURKA/p53/p21/CDK1/Bcl2/Bax protein expression levels; (b)-(g). The optical density values of the protein bands from the western blot were statistically quantified. *p < 0.05 and **p < 0.01 indicate statistically significant differences compared with the SKOV3 group; #p < 0.05 and ##p < 0.01, indicate statistically significant differences compared with the 0 μM group.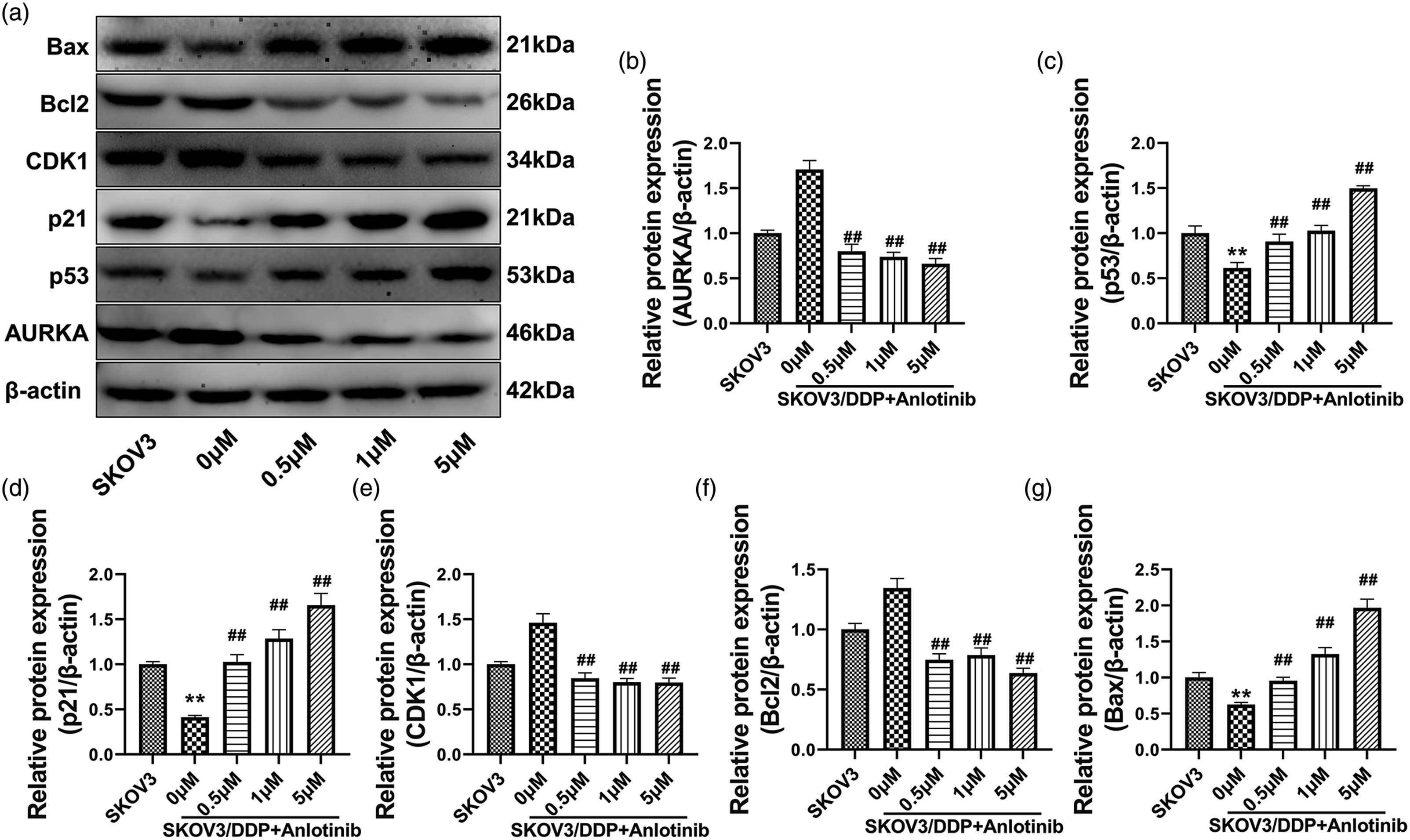
Overexpression of AURKA reversed the effect of anlotinib on the proliferation and cell cycle of SKOV3/DDP cells
To demonstrate that AURKA was responsible for the inhibitory effect of anlotinib on SKOV3/DDP cells, we generated AURKA-overexpressing SKOV3/DDP cells for a functional rescue assay. Successful overexpression of the AURKA gene and protein in SKOV3/DDP cells was demonstrated by qPCR (Figure 5(a)) and western blotting (Figure 5(b) and (c)), respectively. The inhibitory effect of anlotinib on the viability of SKOV3/DDP cells was significantly decreased, as shown by the MTT assay (Figure 5(d)), after overexpression of AURKA (p < 0.01). Overexpression of AURKA reverses the apoptosis-inducing and cell cycle-blocking effects of anlotinib on SKOV3/DDP cells. (a). The level of AURKA gene overexpression in SKOV3/DDP cells was validated by qPCR; (b)-(c). Western blot analysis of AURKA protein expression levels and statistical quantification of band optical density values; (d). MTT assay of cell viability; (e). Cell cycle analysis by flow cytometry; (f). EdU incorporation assay of cell proliferation; *p < 0.05 and **p < 0.01 indicate statistically significant differences compared with the control group; #p < 0.05 and ##p < 0.01 indicate statistically significant differences compared with the DDP+An group.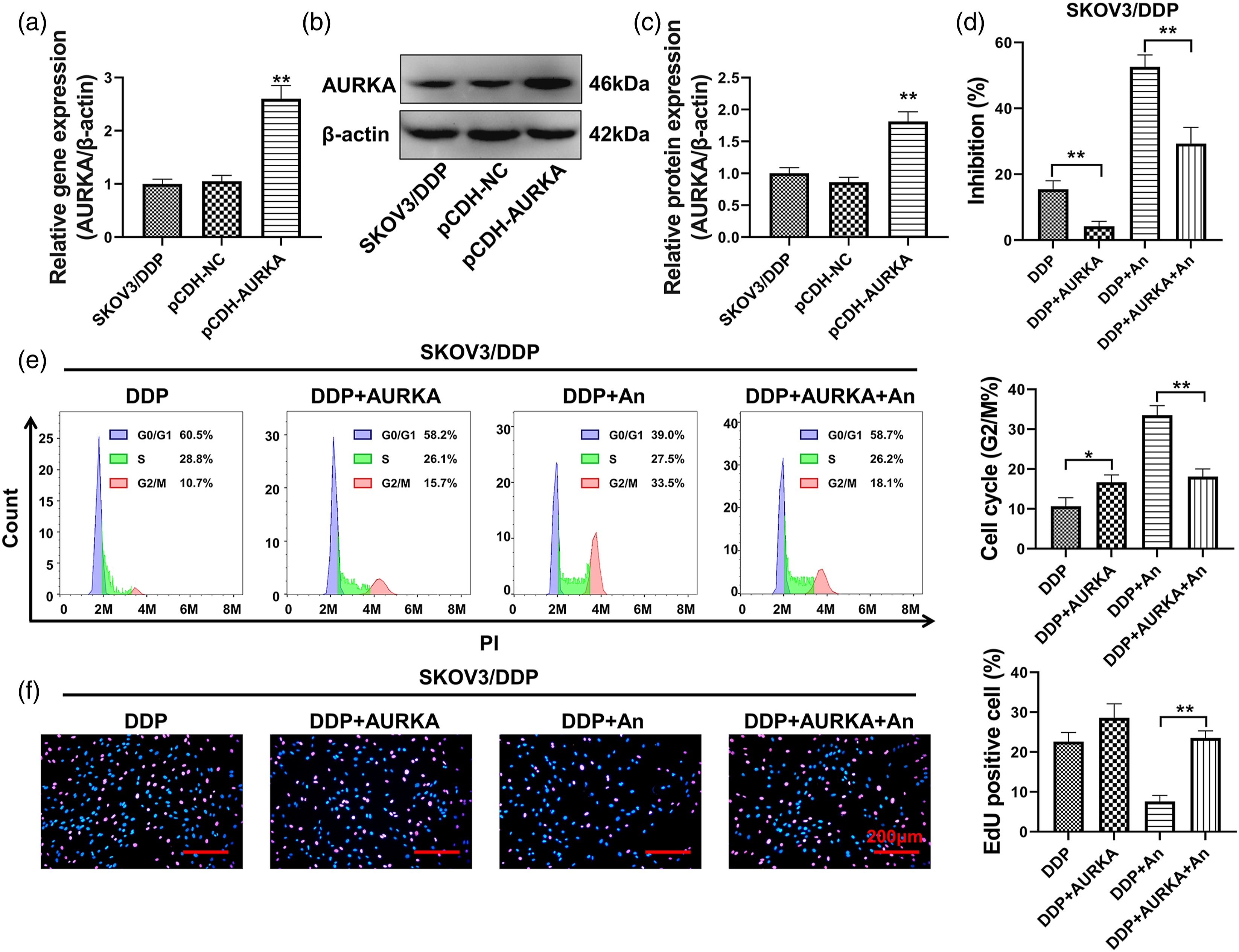
Of interest, the proportion of SKOV3/DDP cells in the G2/M phase was significantly increased after AURKA overexpression (p < 0.05 compared with the DDP group). Overexpression of AURKA significantly reduced the effect of anlotinib on inducing G2/M arrest in SKOV3/DDP cells compared with that in DDP+An group (p < 0.01) (Figure 5(e)). The EdU incorporation assay results showed (Figure 5(f)) that the percentage of SKOV3/DDP cells in a proliferative state was increased after AURKA overexpression. However, no significant difference was observed (p > 0.05 compared with the DDP group). Overexpression of AURKA significantly attenuated the inhibitory effect of anlotinib on the proliferation of SKOV3/DDP cells compared with that in the DDP+An group (p < 0.01).
Overexpression of AURKA reversed the effect of anlotinib on cell cycle and apoptotic proteins in SKOV3/DDP cells
The results of western blotting assay (Figure 6(a) to (g)), showed that overexpression of AURKA reversed the upregulation of Bax and p53/p21 proteins expression and the inhibition of AURKA, CDK1, and Bcl2 proteins expression induced by anlotinib in SKOV3/DDP cells (p < 0.01). Overexpression of AURKA reversed the effect of anlotinib on cell cycle and apoptotic proteins in SKOV3/DDP cells. (a) Western blot analysis of AURKA/p53/p21/CDK1/Bcl2/Bax protein expression levels; (b)-(g). The optical density values of the protein bands from the western blot results were statistically quantified. *p < 0.05 and **p < 0.01 indicate statistically significant differences compared with the control group; #p < 0.05 and ##p < 0.01 indicate statistically significant differences compared with the DDP+An group.
Anlotinib inhibits the growth of tumors derived from SKOV3/DDP cells in tumor-bearing nude mice via the AURKA/p53 pathway
Figure 7(a) shows the tumors stripped from tumor-bearing nude mice at the end of the experiment. Anlotinib was determined to significantly inhibit the growth of SKOV3/DDP tumors in nude mice, with statistically significant differences between these groups and the control group (Figure 7(b), p < 0.05). The IHC analysis assay results showed (Figure 7(c)) that positive staining for the AURKA and Ki67 proteins was decreased after anlotinib treatment, while strong positive p53 protein staining was observed in the DDP+An group. The western blot results further confirmed (Figure 7(d)–(l)) that treatment with anlotinib downregulated AURKA, Bcl2, and CDK1 protein expression and upregulated p53, p21, and Bax protein expression in cisplatin-resistant tumors. Anlotinib inhibits the growth of tumors derived from SKOV3/DDP cells in nude mice. (a).Tumor map of tumor-bearing nude mice; (b). Tumor size calculation results; (c). Immunohistochemical analysis of AURKA, p53 and Ki67 protein expression and distribution in tumor tissues; (d). Relative expression of AURKA and p53; (e). Ki67 protein expression rate; (f)-(l). Quantification of the expression levels and optical density values of AURKA/p53/p21/CDK1/Bcl2/Bax proteins by western blotting. *p < 0.05 and **p < 0.01 indicate the statistical significance of the differences between pairs of connected groups.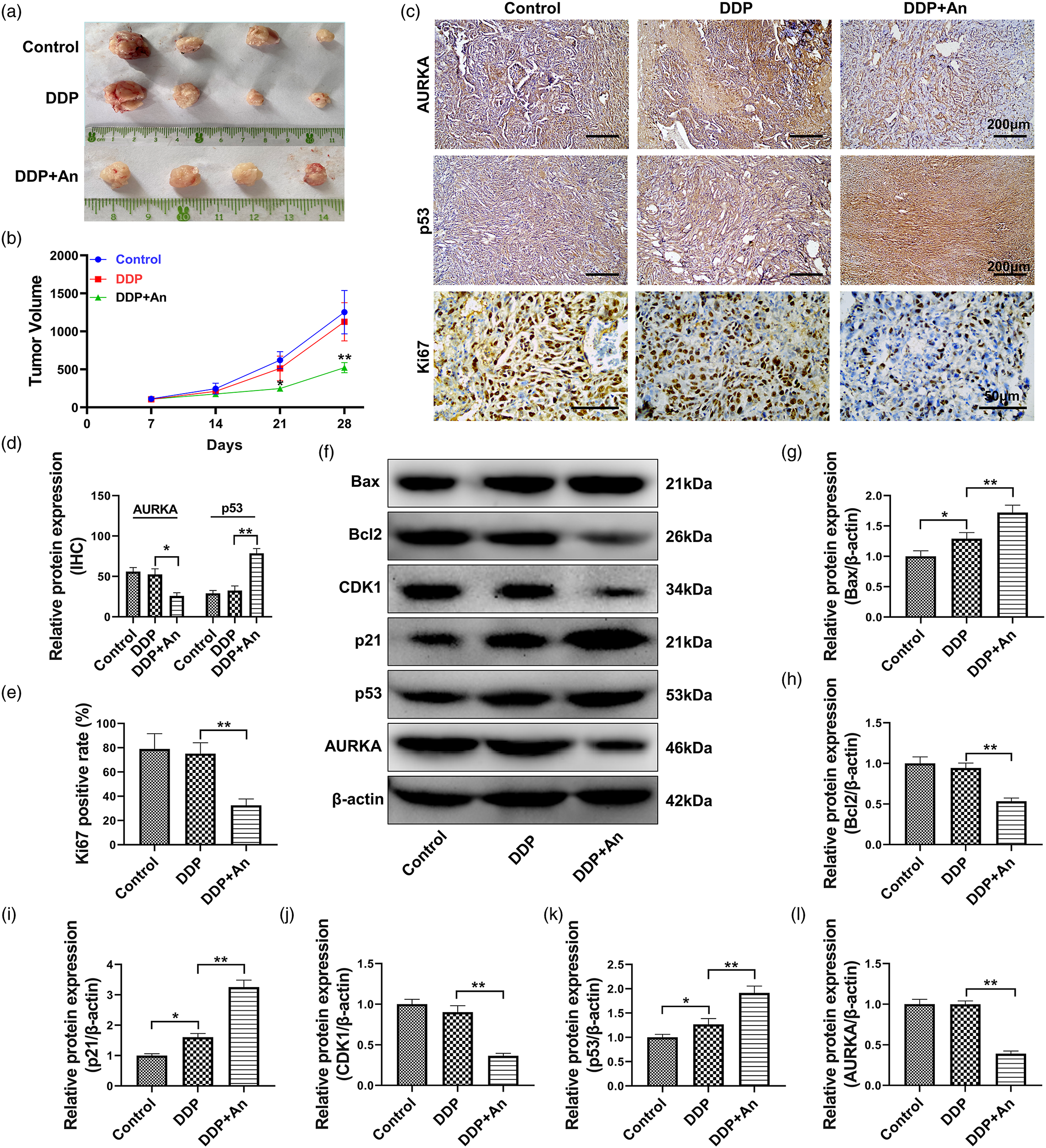
Discussion
Ovarian cancer is a highly malignant disease with the highest mortality rate, which poses a serious threat to women’s life and health. 1 Due to the lack of specific clinical symptoms and difficulty in early diagnosis of ovarian cancer, most ovarian cancer are often at an advanced stage when detected. The main treatments for patients with advanced ovarian cancer are currently extensive cytoreductive surgery and postoperative chemotherapy based on cisplatin (DDP). However, the 5-year survival rate of ovarian cancer patients is still unsatisfactory, only 20%–30%. 15 After treatment with DDP-based chemotherapy regimens, approximately 1/4 of patients develop primary drug resistance, and 80% of them eventually develop secondary drug resistance.16–19 Drug resistance can directly affect chemotherapy outcomes and patient prognosis.
Through a cell model, the present study elucidated a novel molecular mechanism by which anlotinib reverses cisplatin resistance in SKOV3 ovarian cancer cells. In brief, anlotinib was first demonstrated to induce apoptosis and G2/M arrest in SKOV3/DDP cells, thereby inhibiting cell proliferation. This demonstrated that anlotinib was able to reverse SKOV3 cisplatin resistance in SKOV3 cells. Subsequently, the downstream p53 and other downstream proteins of AURKA was examined, and the results showed that anlotinib significantly inhibited the expression while upregulating the oncogene p53 and promoting the effects of p53 on apoptosis and cell cycle progression. In contrast, the findings of Dennis et al. on glioma are similar to those observed in this study, i.e., that inhibition of AURKA leads to G2/M arrest, which blocks an essential process of DNA damage repair and induces apoptosis. 20 Jumpei et al. also demonstrated the effect of AURKA on cell cycle G2/M arrest. 21 The prognosis of patients with high expression is poor. 22 It has been shown that increased expression of AURKA significantly inhibits the expression of the tumor suppressor protein TP53, resulting in cancer cell resistant to chemotherapeutic agents, thus promoting cancer progression.23,24 This suggests that chemotherapeutic drug resistance in numerous cancers may be associated with abnormal upregulation of AURKA.
The tumor suppressor gene p53 is vital in promoting a favorable antitumor response to platinum-based drugs. Normally, DNA damage caused by cisplatin can lead to dissociation of the Mdm2-Mdm4-p53 complex to increase free p53 protein levels, thereby resulting in translocation of p53 to the nucleus where it binds to specific DNA sequences to activate the expression of target genes, such as p21 and Bax.25,26 This transcriptional activity is essential for p53-dependent cellular effects, such as cell cycle arrest and programmed cell death. Thus, inhibition of p53 function is also necessary for tumor cisplatin resistance.
Activation of the p53-p21 pathway has been reported to induce G2/M phase arrest after DNA damage.27,28 In ovarian cancer, p53 has been shown to reduce the phosphorylation level of CDK1 by enhancing the expression of p21, thereby causing G2/M arrest in cancer cells. 29 CDK1 is a cell cycle protein-dependent kinase that normally drives cellular entry into mitosis and is also known as Cdc2. CDK1 is an essential target of the rapid arrest pathway that mediates the G2 checkpoint response to DNA damage. When mammalian cells contain damaged DNA, the p53 tumor suppressor and transcriptional repressor Rb families work in concert to downregulate a large number of genes encoding proteins required for the G2/M transition. 30 In contrast, this study demonstrated that anlotinib treatment upregulated p53/p21, an oncoprotein associated with cisplatin resistance. It significantly inhibited CDK1, a protein related to G2/M phase cell cycle arrest, after downregulation of AURKA. Additionally, Bax, a protein associated with apoptosis, was significantly upregulated. The explanation for these phenomena was based on the premise that the AURKA/p53 pathway was activated.
To further confirm that anlotinib reverses cisplatin-resistance through the AURKA/p53 pathway on cisplatin-resistant ovarian cancer cells, a lentiviral technique was combined with an AURKA gene overexpression cell construct. As expected, the apoptosis-inducing effect of anlotinib on SKOV3/DDP cells mediated by the cycle inhibitor was partially reversed after overexpression of AURKA. Although an increased proportion of the G2/M phase cells were observed in the group of SKOV3/DDP cells without anlotinib treatment, this partial increase could increase the proportion of cells in a proliferative state, as determined from the results of the EdU incorporation assay. The proportion of cells in the DNA replication phase, which favors cell growth, increased during the 4 h assay periodh. In contrast, after 24 h of anlotinib treatment in SKOV3/DDP cells, although the proportion of cells in the G2/M phase was increased, the proportion of EdU-positive cells in turn was dramatically decreased, presumably, within 24 h after anlotinib treatment, most SKOV3/DDP cells had completed the G1-S-G2 transitions, but the G2/M phase transition was blocked, resulting in cells in this interphase cessation of cell growth. As a result, the proportion of cells in which normal DNA replication could continue decreased dramatically within 4 h after the addition of EdU. This resulted in an increased proportion of G2/M-phase cells but a reduced proportion of EdU positivity cells. Due to the low relevance of this phenomenon to the main mechanism investigated in this study, only a theoretical analysis of the phenomenon is presented here.
Does anlotinib have the same oncosuppressive effect on wild-type SKOV3 cells? The answer is yes. Anlotinib significantly inhibited the viability of wild-type SKOV3 cells, as observed in preliminary experiments. However, the starting point for these studies was based on comparing SKOV3 cells with SKOV3/DDP cells. The tumor suppressor pathway that may be triggered in SKOV3/DDP cells is the AURKA/p53 pathway, but whether this is a conventional pathway for targeted ovarian cancer treatment requires additional analysis of data from paracancerous versus cancerous tissue, healthy individuals, or ovarian cancer patients. In the present study, immunofluorescence assays revealed that the AURKA protein was localized to the cytoplasm in nonviable SKOV3 cells, showing strong positive cytoplasmic expression. Usually, AURKA is localized primarily in the nucleus and partially distributed in the cytoplasm and microtubules. Its specific protein localization may trigger different mechanisms related to disease treatment. 31 Therefore, SKOV3 cells after treated with anlotinib were not included in subsequent experimental analyses in this study.
In summary, this study first identified the potential target gene of anlotinib, AURKA, by GEO database and bioinformatics analyses. It was demonstrated that anlotinib could inhibit AURKA and upregulate the p53 pathway to induce apoptosis and G2/M arrest in cisplatin-resistant ovarian cancer cells. The therapeutic effect of anlotinib on cisplatin-resistant ovarian cancer was further confirmed by injecting SKOV3/DDP cells into nude mice. This provides strong evidence for combination treatment with anlotinib in cisplatin-resistant patients in the clinic. Additionally, in a following study, anlotinib will be further investigated to see if it is equally therapeutic for overcoming multidrug resistance in ovarian cancer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
