Abstract
Background
Syringaresinol processes anti-inflammatory and antioxidative activity. However, the effects of syringaresinol on cardiorenal fibrosis caused by cardiorenal syndrome type 2 (CRS2) are unclear.
Methods
Molecular docking predicted binding activity of syringaresinol to heat shock protein 90 (HSP90). The toxicity of a 4-weeks treatment with 20 mg/kg of syringaresinol was observed by measuring serum pro-inflammatory cytokines levels and by cardiorenal pathology. A CRS2 rad model was established by myocardial infarction using ligation over an 8 week-period. Rats were divided into five groups, including sham, CRS2, pimitespib, syringaresinol, and HSP90 + syringaresinol. Rats were received a 4-weeks daily treatment with 10 mg/kg pimitespib (a HSP90 inhibitor) or 20 mg/kg syringaresinol. Recombinant adeno-associated virus (rAAV) carrying a periostin (PE) promoter driving the expression of wild-type HSP90 (rAAV9-PE-HSP90, 1 × 1011 μg) was treated intravenously once in CRS2 model rats. Cardiorenal function and pathology were assessed. Expressions of HSP90 and TGF-β1 in the myocardium and kidney were measured by immunohistochemistry and western blotting.
Results
Syringaresinol showed good binding activity with HSP90, and no signs of toxicity in rats following treatment. Pimitespib or syringaresinol significantly improved the cardiorenal function and fibrosis in rats with CRS2. Meanwhile, the rAAV9-PE-HSP90 injection obviously blocked the effects of syringaresinol.
Conclusions
Syringaresinol targets HSP90 to suppress CRS2-induced cardiorenal fibrosis, providing a promising therapeutic drug for CRS2.
Introduction
Cardiorenal syndrome (CRS) is generally defined as a pathophysiological disorder of the heart and kidneys, in which acute or chronic dysfunction of one organ can induce acute or chronic dysfunction in the other. 1 Five subtypes of CRS were effectively classified by the Acute Dialysis Quality Group in 2008. 2 CRS type 2 (CRS2) is characterized by chronic abnormalities in cardiac function leading to kidney injury or dysfunction. The pathogenesis of CRS2 is complex, in which many factors are involved, including neurohormonal activation, endothelial dysfunction, oxidative stress, and inflammation.3–5 Current treatment of CRS2 mainly involves maintaining the stable hemodynamics and reducing the secretion of inflammatory factors that can lead to damage to renal function. Many drugs have been used to treat CRS2, such as angiotensin converting enzyme inhibitors (ACEIs), angiotensin receptor blockers, β-blockers and vasopressin receptor 2 antagonists. 6 However, these drugs target the symptomatic improvement of heart failure in a setting of maintained renal function. Only limited evidence is used to guide the treatment of CRS2.
It is well recognized that renal fibrosis caused by chronic heart failure is an important pathological feature of CRS2, which could develop into end-stage renal disease. 7 Heat shock protein 90 (HSP90) is one of the most abundant molecular chaperone proteins and is involved in cell survival, growth, and differentiation.8,9 HSP90 has been reported to be activated in many fibrotic diseases, for example cardiac fibrosis, 10 renal fibrosis, 9 and pulmonary fibrosis. 11 In these fibrotic diseases, HSP90 regulates transforming growth factor-β (TGF-β)-related pathways to accelerate fibrosis. Inhibition of HSP90 effectively blocks the profibrotic effects of TGF-β-related pathways.8–10,12 Importantly, in CRS2-induced cardiorenal fibrosis, the TGF-β signaling is activated and promotes fibrogenesis through Wnt/β-catenin pathway. 4 These findings indicate that HSP90 could play an important role in the CRS2-induced cardiorenal fibrosis through regulating TGF-β signaling. Nonetheless, the role of HSP90 in CRS2-induced cardiorenal fibrosis is unclear.
Syringaresinol is extracted from various parts of plants, such as the cortex, Amelia seeds and flax seed. 13 Many evidences have shown that syringaresinol processes anti-inflammatory and antioxidative activity.14–16 In addition, syringaresinol could protect against fibrosis of type 1 diabetic cardiomyopathy by inhibiting the TGF-β/Smad pathway. 16 Syringaresinol also could attenuate sepsis-induced cardiac dysfunction by inhibiting inflammation and pyroptosis in mice. 14 However, the effects of syringaresinol on CRS2 are unclear. Is not clear whether syringaresinol could inhibit TGF-β signaling through HSP90 to suppress CRS2-induced cardiorenal fibrosis.
In this study, we found that syringaresinol showed a good binding activity to HSP90. We evaluated whether syringaresinol could suppress CRS2-induced cardiorenal fibrosis through HSP90-mediated TGF-β signaling. An HSP90 inhibitor as a positive control was used to analyze the underlying mechanism of syringaresinol activity in rats with CRS2.
Methods
Molecular docking
The three-dimensional (3D) structure of HSP90 (PDB format) was downloaded from the PDB database (https://www.rcsb.org/), and the 3D structure of syringaresinol (CAS: 21453-69-0, PDB format) was downloaded from the PubChem (https://pubchem.ncbi.nlm.nih.gov/). The mechanical optimization, hydrogenation, and charge of the ligand were determined using UCSF chimera software (https://www.cgl.ucsf.edu/chimera/). 17 Molecular docking was analyzed by the AutoDock Vina tool. A grid box was generated that could cover the entire protein binding site and allowed all ligands to move freely.
Animals
Forty male Sprague Dawley (SD) rats (200 ± 20 g) were purchased (Beijing Vital River Laboratory Animal Technology Co., Ltd, China) and housed in the specific pathogen-free grade laboratory animal room (temperature, 23°C ± 2°C; humidity, 40% ± 5%) with a 12-/12- h light/dark cycle, free water, and food.
All animal experiments were approved by the Institutional Animal Care and Use Committee of Yantai Hospital of Traditional Chinese Medicine, and were performed in accordance with the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals.
CRS2 model
The anesthetized rats were underwent ligation of the left anterior descending coronary artery for 8 weeks.18–20 Before recovery from anesthesia, the mice were remained on a heating blanket. After surgery, the rats were treated with penicillin (8 × 105 U/d) for 3 days. For sham animals, the same procedures were performed without ligation.
Animal treatments
Treatment 1
To analyze the effects of syringaresinol on normal rats, 15 rats (5 rats in each group) were randomly divided into 3 groups: normal group, 1% DMSO group and syringaresinol group. Rats were received daily with 1% DMSO or 20 mg/kg syringaresinol by oral administration for 4 weeks. Syringaresinol (CAS: 21453-69-0, #S880440, Macklin, Shanghai, China) was dissolved in 0.1% DMSO. The dose of syringaresinol referred that used by Li et al. 16
After 4 weeks, rats were anesthetized with pentobarbital sodium (45 mg/kg) and euthanized. Blood samples were obtained from the tail vein. Levels of tumor necrosis factor (TNF)-α, interleukin (IL)-6 and IL-1β in serum were analyzed by enzyme-linked immunosorbent assay (ELISA) kits. Renal and cardiac pathologies were observed by hematoxylin and eosin (HE) staining.
Treatment 2
To confirm our hypothesis that syringaresinol suppresses CRS2-induced cardiorenal fibrosis through HSP90-mediated TGF-β signaling, rats were divided into 5 groups (5 animals in each group): sham, CRS2, pimitespib, syringaresinol, and HSP90 + syringaresinol groups.
In the sham group, rats were housed without ligation for 4 weeks, then treated daily with 1% DMSO for 4 weeks. In the CRS2 group and the pimitespib group, rats were liagated for 4 weeks, and then daily treated with 1% DMSO or 10 mg/kg pimitespib, respectively, for 4 weeks by oral administration. The dose of pimitespib (#S7716, Selleck, Shanghai, China), an HSP 90 inhibitor, referred that used by EI-Kasher et al. 21 Similarly, rats in the syringaresinol group were ligated for 4 weeks, then daily treated with 20 mg/kg syringaresinol for 4 weeks by oral administration. In the HSP90 + syringaresinol group, rats were ligated for 4 weeks, then treated intravenously once with 1 × 1011 μg recombinant adeno-associated virus (rAAV) carrying a periostin (PE) promoter that drives wild-type HSP90 expresssion (rAAV9-PE-HSP90, Kunshan Renyuan Biotechnology Company, Suzhou, China). 10 After that, 20 mg/kg of syringaresinol was treated orally every day for 4 weeks.
The cardiorenal function of rats in each group was assessed, and then the hearts and kidneys were harvested for further analysis. For histological analysis, the hearts and kidneys were fixed in 4% paraformaldehyde. For western blotting analysis, hearts and kidneys were stored at −80°C until use.
ELISA
The levels of TNF-α (#PT516, Beyotime, China), IL-6 (#PI335, Beyotime) and IL-1β (#PI303, Beyotime) in serum were analyzed by ELISA kits according to the manufacturer’s instructions.
Echocardiographic assessment of cardiac function
Rats were anaesthetized with pentobarbital sodium (45 mg/kg) and views of the parasternal long and short axis of the left ventricular (LV) at the level of the tips of the papillary muscle were obtained using the Vevo 2100 Imaging System (VisualSonics Inc). Two-dimensional (2D) targeted M-mode images were obtained from the anterior and posterior walls. The left ventricular (LV) ejection fraction (LVEF%), LV fractional shortening (LVFS%) and LV end diastolic volume (LVEDV, μL) were analyzed in this study.
Evaluation of renal function
After the last treatment, urine and blood samples from the tail vein were collected after 24 h. The concentrations of urinary kidney injury molecule-1 (Kim-1) were analyzed by a rat Kim-1 ELISA kit (#PK683, Beyotime, China). Serum creatinine levels were measured by a rat creatinine serum detection kit (orb865241, Biorbyt). Proteinuria levels were measured by a rat microalbuminuria ELISA kit (#E-EL-R0025c, Elabscience®, Wuhan, China).
HE and Masson staining
In accordance with previous protocols, 22 the fixed kidneys and hearts were dehydrated with 80%, 90%, 95%, and 100% ethanol. They were then embedded in paraffin and cut into 3-μm thick sections. The sections were dewaxed twice with xylene (first for 10 min, then for 5 min) and hydrated using an ethanol gradient (100%, 100%, 95%, 90%, and 85%; 1 min each). After washing with water, the sections were respectively stained with HE (#G1120, Solarbio, China) and Masson (#G1346, Solarbio) solutions according to the instructions. After washing with water for 30 s, the sections were dehydrated with ethanol (85%, 90%, twice at 95% and twice at 100%), then cleared with xylene. Finally, the slices were sealed with neutral gum. The results were visualized under a light microscope.
Immunohistochemistry staining
As reported previously, 23 the kidney and heart sections (3 μm) were dewaxed and hydrated, then placed in 0.01 mol/L sodium citrate buffer (pH = 6.0) and heated to boiling twice (over 10-min interval). After washing with PBS for 3 times (5 min each), the sections were incubated with 3% H2O2 for 10 min, and then incubated with anti-rabbit HSP90 primary antibodies (1:1000, #ab203126, Abcam) overnight at 4°C. After being washing with PBS, the sections were incubated with goat anti-rabbit IgG (HRP) secondary antibody (1:500, #S0001, Affinity) at room temperature for 60 min. After staining with diaminobenzidine (DAB), the sections were dehydrated with ethanol, purified with xylene and sealed with neutral gum. The sections were visualized using a light microscope. The positive expression (positive area/all area) was analyzed using ImageJ software.
Western blotting
Proteins from frozen kidney and heart tissues were collected using RIPA buffer (#R0010, Solarbio). Proteins (30 μg) were separated by 12% SDS‒PAGE (Bio-Rad, China) and transferred to PVDF membranes after being lysed. Before incubation with antibodies, membranes were cut and incubated with primary antibodies overnight at 4°C. Primary antibodies, including anti-rabbit HSP90 (1:500, #ab203126, Abcam), anti-rabbit TGF-β1 (1:500, #ab215715, Abcam) and anti-rabbit GAPDH (1:1000, #ab9487, Abcam), were diluted with 5% bovine serum albumin (BSA). After 3 times washing with TBS-0.01% Tween 20, the membranes were incubated with goat anti-rabbit IgG (HRP) (1:5000, #ab6721, Abcam) for 2 h at room temperature. After washing, the signals were visualized with an enhanced chemiluminescence reagent (#D085075, Bio-Rad). The proteins bands were quantified by ImageJ software and expressed as relative optical density.
Statistical analysis
Quantitative analysis of the results of Masson, immunohistochemistry and western blotting was performed by ImageJ software (National Institutes of Health, USA). Data were showed as mean ± standard deviation by GraphPad Prism 8.0 software (GraphPad Software Inc., CA). Statistical differences among multiple groups were compared by one-way analysis of variance following the Tukey test. Statistical significance was set at a value of p < 0.05.
Results
Effects of syringaresinol on normal rats
In this study, we found syringaresinol showed a good binding activity (score = −6.7 kcal/mol) with HSP90 by molecular docking analysis (Figure 1(a)). Rats were treated with 1% DMSO or 20 mg/kg syringaresinol by oral administration for 4 weeks to analyze the effects of syringaresinol on normal rats. With regard to serum levels of the pro-inflammatory cytokines, TNF-α, IL-6, and IL-1β, there were no significant differences observed among the groups (Figure 1(b) and Supplemental Table 1). The kidney and myocardium pathology of rats was observed by HE staining (Figure 1(c)), and no significant differences were observed among the groups. Effects of syringaresinol (20 mg/kg/d) treatment on normal rats. Rats were treated with 1% DMSO or 20 mg/kg syringaresinol by oral administration for 4 weeks. (a) The binding of syringaresinol to HSP90 was analyzed by molecular docking. (b) The serum levels of TNF-α, IL-6 and IL-1βwere analyzed by ELISA kits. (c) The pathology of kidney and myocardium of rats was observed by HE staining (Scale = 50 μm). NS: no significance.
Syringaresinol treatment improved cardiorenal function through HSP90 in rats with CRS2
The M-mode echocardiograms of each group were obtained (Figure 2(a)), and the LVEF%, LVFS% and LVEDV were analyzed in each group (Figure 2(b) and Supplemental Table 2). Compared with the sham rats, the LVEF% and LVFS% were significantly decreased, while the LVEDV was significantly increased in rats with CRS2 (p < 0.01). After treatment with the HSP90 inhibitor pimitespib (10 mg/kg) for 4 weeks, the cardiac function of rats with CRS2 was clearly improved compared to the CRS2 rats (p < 0.05). Importantly, better effects on improving cardiac function were showed in CRS2 rats with 20 mg/kg of syringaresinol administration for 4 weeks when compared with the CRS2 rats (p < 0.01). However, the effects of syringaresinol were significantly suppressed by the rAAV9-PE-HSP90 treatment (p < 0.05). Syringaresinol improved cardiorenal function through HSP90 in rats with CRS2. Rats with CRS2 were treated daily with 10 mg/kg pimitespib or 20 mg/kg syringaresinol for 4 weeks. For the HSP90 + syringaresinol group, rats with CRS2 were received a single intravenously administration of 1 × 1011 μg AAV9-PE-HSP90, then were daily treated with 20 mg/kg of syringaresinol for 4 weeks. (a) Representative M-mode echocardiograms for each group. (b) Left ventricular (LV) ejection fraction (LVEF%), LV fractional shortening (LVFS%) and LV end diastolic volume (LVEDV, μL). (c) Urinary Kim-1, proteinuria and serum creatinine levels. *p < 0.05, **p < 0.01.
The renal function of rats in each group (Figure 2(c) and Supplemental Table 3), was also evaluated. Urinary Kim-1, proteinuria, and serum creatinine levels were significantly upregulated in rats with CRS2 compared with the sham rats (p < 0.01). After pimitespib treatment, the levels of the above renal factors were clearly downregulated compared with the CRS2 rats (p < 0.05). Furthermore, the levels of the above renal factors were significantly downregulated after treatment with syringaresinol compared with the CRS2 rats (p < 0.01). In contrast, the function of syringaresinol was obviously blocked by the rAAV9-PE-HSP90 treatment (p < 0.01).
Syringaresinol treatment improved cardiorenal fibrosis through HSP90 in CRS2 rats
The cardiorenal pathology of rats in each group was observed by HE staining (Figure 3). In Figure 3(a), it showed that the CRS2-induced myocardial necrosis (black arrows) compared to the sham operated rats. In Figure 3(b), it showed that the CRS2 induced glomerular injury (black arrows) compared to the sham rats. After treatment with pimitespib or syringaresinol, the hypertrophic cardiomyocytes and damaged glomeruli improved. Importantly, the improving effect of syringaresinol was superior to that of pimitespib. However, the rAAV9-PE-HSP90 treatment interfered with the beneficial effects of syringaresinol. Syringaresinol improved cardiorenal pathology through HSP90 in rats with CRS2. The cardiac (a) and renal (b) pathology was observed by HE staining (scale = 50 μm). Black arrows showed the myocardial necrosis and damaged glomeruli.
The cardiorenal fibrosis of each group was observed by Masson staining (Figure 4). It showed that the CRS2-induced cardiac (Figure 4(a) and Supplemental Table 4) and renal (Figure 4(b) and Supplemental Table 4) fibrosis were observed compared to the sham rats (p < 0.01). After treatment with pimitespib or syringaresinol, the cardiorenal fibrosis was significantly suppressed compared with the CRS2 rats (p < 0.01). Importantly, the effects of syringaresinol were obviously blocked by the rAAV9-PE-HSP90 treatment (p < 0.05). Syringaresinol improved the cardiorenal fibrosis through HSP90 in rats with CRS2. The cardiac (a) and renal (b) fibrosis was observed by Masson staining (scale = 100 μm). *p < 0.05, **p < 0.01.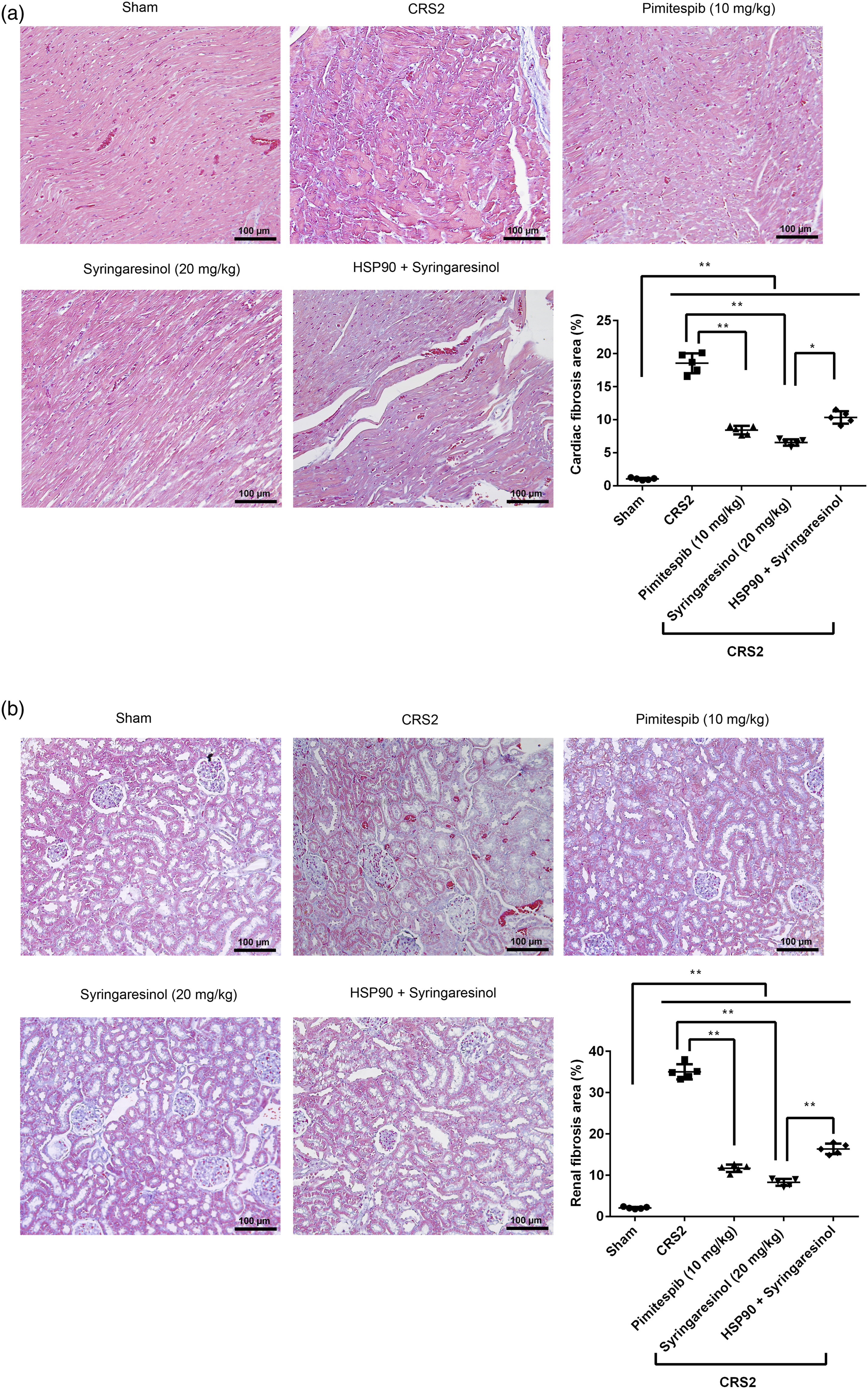
Syringaresinol treatment suppressed HSP90 expression in CRS2 rats
The expression of HSP90 in myocardium and kidney of each group was observed by immunohistochemical staining (Figure 5). The expressions of HSP90 (black arrows) were clearly upregulated in the myocardium (Figure 5(a) and Supplemental Table 5) and kidney (Figure 5(b) and Supplemental Table 5) of CRS2 rats compared with the sham rats (p < 0.01). After treatment with pimitespib or syringaresinol, the expressions of HSP90 were significantly downregulated compared with the CRS2 rats (p < 0.05). Interestingly, the effect of syringaresinol was obviously hampered by the rAAV9-PE-HSP90 treatment (p < 0.05). Expression of HSP90 (black arrows) in the myocardium and kidney of each group was observed by immunohistochemiscal staining (Scale = 50 μm). The positive expression of HSP90 in myocardium (a) and kidney (b) was quantified by ImageJ software. *p < 0.05, **p < 0.01.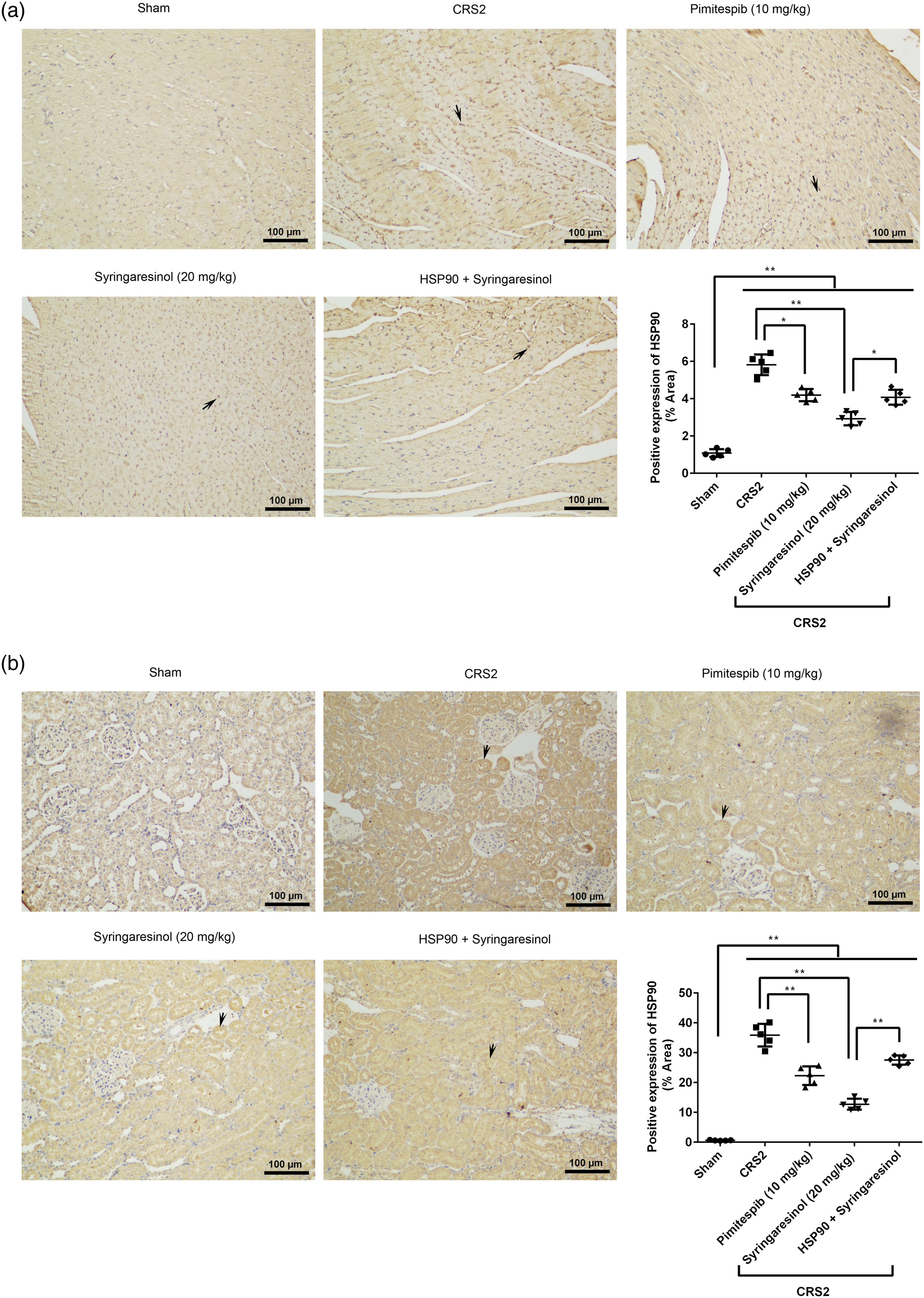
Syringaresinol treatment suppressed the HSP90 and TGF-β1 proteins in CRS2 rats
The expressions of HSP90 and TGF-β1 proteins in myocardium (Figure 6(a)) and kidney (Figure 6(b)) of each group was observed by western blotting. The expressions of HSP90 and TGF-β1 proteins were clearly upregulated in the myocardium (Figure 6(a) and Supplemental Table 6) and kidney (Figure 6(b) and Supplemental Table 7) of CRS2-induced rats compared with the sham rats (p < 0.01). After treatment with pimitespib or syringaresinol, the expressions of HSP90 and TGF-β1 proteins were significantly declined compared with the untreated CRS2 rats (p < 0.05). Furthermore, the effect of syringaresinol was obviously hampered by the rAAV9-PE-HSP90 treatment (p < 0.05). Expressions of HSP90 and TGF-β1 proteins in the myocardium (a) and kidney (b) of each group was observed by western blotting. The relative expression of HSP90 and TGF-β1 proteins was quantified by ImageJ software. *p < 0.05, **p < 0.01.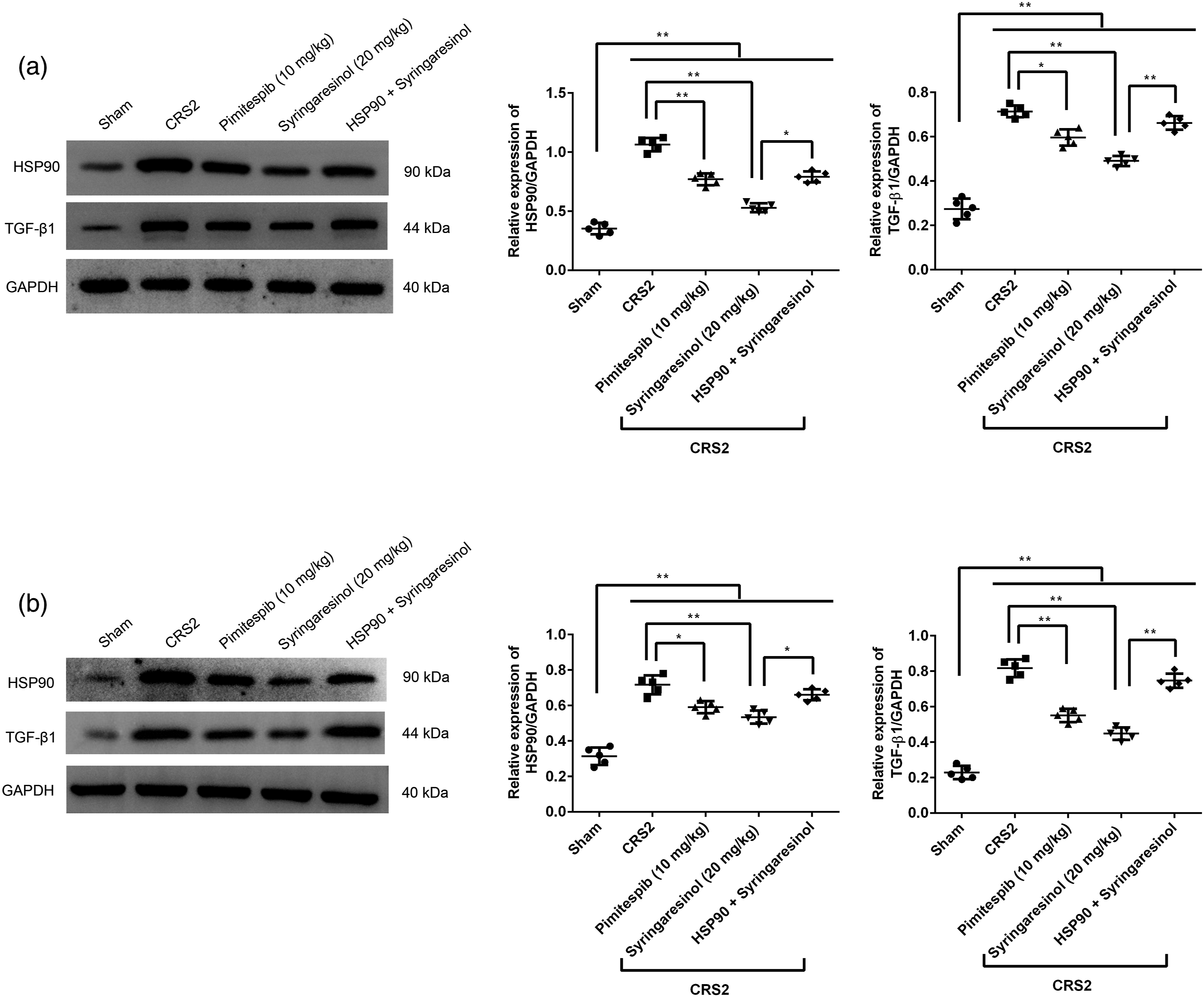
Discussion
The docking score reflects the intermolecular energy (kcal/mol), and represents the stability between a ligand and its receptor. 17 In this study, we found that a good binding activity of syringaresinol with HSP90 and confirmed the safety of the selected dose of syringaresinol through observing changes in the serum levels of proinflammatory cytokines (TNF-α, IL-6 and IL-1β) and the pathology of kidney and myocardium.
In this study, we used pimitespib, a HSP90 inhibitor, as a positive control to analyze the effects of syringaresinol on cardiorenal fibrosis in rats with CRS2. As an oral small molecule inhibitor of HSP90, pimitespib has been approved for treatment of gastrointestinal stromal tumors in Japan. 24 As a treatment for pulmonary fibrosis, pimitespib increases nifuroxazide-induced disruption of the IL-6/STAT3/HIF-1α autocrine loop. 21 HSP90 is activated in many fibrotic diseases.10,12,25 In this work, we also found that HSP90 expressions were upregulated in the myocardia and kidneys of rats with CRS2. Suppression of HSP90 using pimitespib significantly improved the cardiorenal function and fibrosis in CRS2-induced rats, confirming that HSP90 is involved in CRS2-induced cardiorenal fibrosis.
Importantly, syringaresinol treatment in rats with CRS2 improved cardiorenal function and fibrosis achieved better results than the pimitespib treatment, indicating that suppressing HSP90 is one of the underlying mechanisms of syringaresinol. Administration of rAAV9-PE-HSP90 obviously blocked the effects of syringaresinol, further confirming that syringaresinol targets HSP90 in the rats with CRS2. The anti-inflammatory and antioxidative activity of syringaresinol,14–16,26 may also inhibit cardiac fibrosis caused by type 1 diabetic cardiomyopathy. 16 In this study, we also confirmed that syringaresinol could inhibit cardiorenal fibrosis through HSP90 in rats with CRS2. Furthermore, we found that syringaresinol inhibited the TGF-β1 expression in the myocardium and kidney of CRS2 rats. TGF-β1 is the primary factor that drives fibrosis in most, if not all, forms of chronic kidney disease. 27 Our findings suggested that syringaresinol exerts its effects by interfering with HSP90-mediated TGF-β1 signaling in rats with CRS2.
However, there are several limitations to this work that should be considered. The numbers of animals in each group were not enough, which limited the ability to reach statistical significance among groups. In this study, we only investigated one potential mechanism of syringaresinol activity in the treatment of CRS2. Further studies are required to investigate the metabolic activity, bioavailability, and safety profile of syringaresinol to support its application in CRS2 treatment.
Conclusions
Our study determined that syringaresinol targets HSP90 to suppress the cardiorenal fibrosis induced by CRS2. Through downregulation of HSP90-mediated TGF-β1 signaling, syringaresinol administration prevented cardiorenal fibrosis in rats with CRS2. Our findings provide promising findings supporting the application of syringaresinol as a therapeutic agent for CRS2.
Supplemental Material
Supplemental material - Syringaresinol inhibits cardiorenal fibrosis through HSP90 in a cardiorenal syndrome type 2
Supplemental material for Syringaresinol inhibits cardiorenal fibrosis through HSP90 in a cardiorenal syndrome type 2 by Jianjie Wang, Jianqin Zou, Cheng Zhao, Han Yu, Jiajia Teng and Lei Dong in Human & Experimental Toxicology Journal
Footnotes
Acknowledgements
The manuscript was edited by the Charlesworth Author Services.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Yantai Science and Technology (No. 2022YD098).
Data availability statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
