Abstract
Background
Women with antiphospholipid syndrome (APS) or antiphospholipid antibodies (aPLs) are at high risk for obstetric complications, including recurrent pregnancy loss (RPL). However, effective treatments for RPL are lacking.
Objective
This study aimed to reveal the function and underlying mechanism of hyperoside (Hyp) in RPL associated with antiphospholipid antibodies (aCLs).
Methods
The pregnant rats (N = 24) were divided randomly into four groups: normal human-IgG (NH-IgG); aCL-pregnancy loss (aCL-PL); aCL-PL + Hyp (40 mg/kg/day); aCL-PL + low molecular weight heparin (LMWH, 525 μg/kg/day). HTR‐8 cells were treated with 80 μg/mL aCL to establish the cell models of miscarriage.
Results
In pregnant rats, aCL-IgG injection raised the abortion rate of embryos, while Hyp treatment inhibited the effects. Additionally, Hyp inhibited the platelet activation and uteroplacental insufficiency caused by aCL. In vivo and in vitro experiments further suggested that Hyp suppressed aCL-induced inflammation and apoptosis by downregulating NOD-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome-related factors and decreasing apoptotic rates. After aCL administration, Hyp therapy downregulated the expression of purinergic ligand-gated ion channel 7 (P2X7), which is reported to induce cytokine release and apoptosis. Furthermore, we found that the treatment of 3′-O-(4-Benzoyl) benzoyl-ATP (BzATP, an agonist of the P2X7 receptor) reversed the inhibitory effects of Hyp on cell function.
Conclusions
Hyp exerts protective effects on aCL-induced pregnancy loss by preventing platelet activation-mediated P2X7/NLRP3 pathway. Therefore, Hyp may provide a feasible pharmaceutical strategy for the treatment of RPL.

Keywords
Introduction
Antiphospholipid syndrome (APS) is an autoimmune disease that can cause thrombosis, pregnancy loss, and preterm delivery. 1 Antiphospholipid antibodies (aPLs), the hallmark of APS, are acquired antibodies against negatively charged phospholipids.2,3 The aPLs include lupus anticoagulant (LA), anticardiolipin antibodies (aCLs), and anti-beta-2 glycoprotein I antibodies (anti-B2GPI). 4 It is well known that aCLs are linked to uteroplacental insufficiency, decidual vasculopathy, and placental thrombosis, ultimately resulting in adverse pregnancy outcomes. 5 The aCL has been revealed to be tightly associated with recurrent pregnancy loss (RPL), 6 and the incidence of aPLs in RPL patients ranges from 15% to 20%. 3 However, the mechanism of aCL-induced RPL has not been fully revealed yet.
It is claimed that a third of patients with APS suffer from thrombocytopenia due to the binding and activation of platelets.7,8 The activated platelets can activate the purinergic receptor, ligand-gated ion channel 7 (P2X7) by releasing adenosine triphosphate (ATP) to induce the release of cytokines and trigger apoptosis. 9 It is well known that P2X7 exists on a variety of cell membranes. Under the action of ATP, P2X7 can activate a range of intracellular signaling pathways and participate in various physiological processes, such as inflammation, neurotransmitter release, and oxidative stress.10-12 In neurodegenerative and neoplastic disorders, inhibiting the P2X7 receptor may lessen the degree of inflammation. 13 Furthermore, it is reported that P2X7 mediates the NOD-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome activation and pyroptosis in human placental Hofbauer cells. 14 The NLRP3 inflammasome is a cytosolic multiprotein complex that responds to cellular perturbations. Subsequently, the assembly of NLRP3 inflammasome causes the activation of caspase-1, which promotes the maturation and release of inflammatory cytokines, including interleukin-1β (IL-1β) and IL-18, as well as inflammatory cell death. 15 These findings suggest that P2X7 acts as a physiological function by activating inflammation in many human diseases. However, it was unknown whether P2X7/NLRP3 inflammasome activation contributed to the pathogenesis of RPL with aCL administration.
Hyperoside (Hyp) is a unique flavonoid in okra (Abelmoschus esculentus) and exerts a vital role in anti-inflammation, anti-depression, and anti-cancer. 16 For example, Hyp suppresses inflammation effectively in mice with acute lung injury, 17 and Hyp protects human umbilical vein endothelial cells against aCL-induced damage by triggering autophagy. 18 In addition, the protective effects of Hyp against RPL have also been described, and Hyp is confirmed to attenuate pregnancy loss by activating autophagy and inhibiting inflammation in a rat model. 19 Also, it is reported that Hyp exerts an inhibitory effect on NLRP3 inflammasome activation in human and mouse macrophages. 20 However, the molecular mechanisms by which Hyp relieves pregnancy loss via the P2X7/NLRP3 inflammasome remain to be further determined.
In this study, we found that the expression of platelet activation factor receptor (PAFR) and platelet-activating factor 4 (PF4) was upregulated in the placental tissues of aCL-triggered rats. The connection between platelet activation and P2X7/NLRP3 pathway was explored in aCL-induced pregnancy loss. Furthermore, the functions and potential mechanisms of Hyp in RPL associated with aCL were explored by in vivo and in vitro experiments.
Materials and methods
Preparation of IgG
In this study, the antibodies of normal human-IgG (NH-IgG) and aCL-IgG were obtained from the serum of healthy humans and APS patients at the First Affiliated Hospital of Henan University of Chinese Medicine. The antibodies were purified as previously described. 19 In brief, with continual stirring, ammonium sulfate (Aladdin, China) was added to the human serum to give 33% saturation. After the centrifugation at 10,000 rpm for 10 min, the supernatant was then kept at 4°C to precipitate the proteins with 50% saturated ammonium sulfate. Subsequently, the proteins precipitated in the serum were centrifuged and resuspended in phosphate buffer saline (PBS, Aladdin, China). After dialysis, 1% BaCl2 (Aladdin, China) was used to check for the removal of ammonium sulfate. Finally, the IgG containing the aCL fraction was concentrated by centrifugation at 4,000 rpm in an ultrafilter tube.
Animal model
Female and male Sprague Dawley (SD) rats were fed ad libitum and water ad libitum. The male rats (N = 16) were allowed to mate with the female ones (N = 32) in 1:2 ratios overnight. The rats were considered pregnant preliminarily if there was milky white or light-yellow jelly (vaginal plug) the next morning,21,22 and the day of vaginal plug-positive was recorded as Gestation Day (GD) 0. In our experiments, female rats (N = 26) were pregnant, and pregnant rats (N = 24) were randomly divided into four groups (NH-IgG, aCL-PL, aCL-PL + Hyp, aCL-PL + LMWH (low molecular weight heparin), N = 6 in each group). On GD eight and GD 12, rats in the three aCL-PL groups were respectively injected with aCL-IgG (75 mg/kg) at multiple subcutaneous sites on the back, and rats in the NH-IgG group were injected with the same amount of NH-IgG at the same site. During GD eight to GD 12, the rats in the aCL-PL + Hyp group were given intragastric administration of Hyp (40 mg/kg/day, Aladdin, China),19,23 and rats in the aCL-PL + LMWH group were given a subcutaneous injection of LMWH (525 μg/kg/day, Meilun Biotech, China). After the last administration for 24 h, the rats were euthanized by 40% carbon dioxide inhalation. Then the placentas and embryos were photographed, and the embryos were weighed and used for recording the absorption rate. During the experiment, the pregnancy status of the rats was confirmed. All the animal experiments were approved by the Institutional Animal Care and the animal ethics committee of the First Affiliated Hospital of Henan University of Chinese Medicine (ethics approval number: YFYDW2019025).
Cell culture and treatment
Human first-trimester trophoblast cells (HTR-8) were purchased from Zhong Qiao Xin Zhou Biotechnology (China), and 1 × 106 cells/well were seeded in 6-well plates and cultured in Roswell Park Memorial Institute-1640 medium (Solarbio, China) containing 10% fetal bovine serum (Tianhang Biotechnology, China). The cells were inoculated overnight in an incubator at 37°C and 5% CO2.
HTR-8 cells were treated with different concentrations of aCL (5, 10, 20, 40, 80, or 160 μg/mL) for 48 h. To evaluate the effects of Hyp on aCL-induced pregnancy loss, HTR-8 cells treated with different concentrations of Hyp (2.5, 5, 10, 20, 40, or 80 μM) for 2 h received the treatment of 80 μg/mL aCL. For the experiments about inflammation and apoptosis, HTR-8 cells were divided into the following groups.
Control: The control cells were maintained in similar conditions without any treatment.
aCL: The cells were treated with 80 μg/mL aCL for 48 h.
aCL + Hyp: The cells were treated with 40 μM Hyp for 2 h before aCL administration.
aCL + Hyp + BzATP: The cells were treated with 40 μM Hyp and 100 μM 3′-O-(4-Benzoyl) benzoyl-ATP (BzATP, GlpBio, USA) for 2 h before aCL administration.
aCL + BBG: The cells were treated with 100 nM brilliant blue G (BBG) for 2 h before aCL administration.
Enzyme-linked immunosorbent assay (ELISA)
The contents of PAFR and PF4 in the plasma of rats were measured using ELISA Kit for PAFR and PF4 (Uscnk, China) respectively. The contents of IL-1β and IL-18 in the supernatant of rat placenta tissues were measured using the Rat IL-1β ELISA Kit (Lianke Biotech, China) and Rat IL-18 ELISA Kit (Fine Biotech, China) respectively. The contents of IL-1β and IL-18 in the supernatant of HTR-8 cells were measured using the Human IL-1β ELISA Kit and Human IL-18 ELISA Kit (Lianke Biotech, China) according to the manufacturer’s instructions. The corresponding concentration was calculated according to the standard curve.
Haematoxylin and Eosin (H&E) staining
Paraffin sections (5 μm thick) from pregnant rats were prepared, and slides were stained with hematoxylin for 5 min and then washed for 5 min, followed by staining with eosin staining for 3 min. Then stained slides were photographed with a conventional microscope (Olympus, Japan). The images were analyzed using Image J-Pro Plus 6.0 software and the maternal blood area of the placentas was carried out as previously mentioned. 24 Each placenta was estimated based on the analysis of six nonconsecutive sections.
Protein extraction and western blot
Cell lysates were collected in the cell lysis buffer (Beyotime, China). Subsequently, the mixture was ground with liquid nitrogen, incubated on ice for 5 min, and centrifuged at 4°C for 5 min at 10,000 g. The protein content was determined according to the instructions of the BCA Protein Assay Kit (Beyotime, China). Equal amounts of protein were separated by electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, USA). Western blot analysis was performed using P2X7 antibody (Abclonal, China), NLRP3 antibody (Affinity, China), apoptosis-associated speck-like protein (ASC) antibody (Abclonal, China), cleaved caspase-1 antibody (Affinity, China), B cell lymphoma 2 (Bcl2) antibody (Affinity, China), BCL2-Associated X (Bax) antibody (Abclonal, China), cleaved-Caspase-3 antibody (Affinity, China), and β-actin (Affinity, China) overnight at 4°C and subsequently incubated in a corresponding horseradish peroxidase-conjugated secondary antibody (Beyotime, China) for 45 min at 37°C. The membranes were visualized using the chemiluminescence (ECL) kit (Beyotime, China).
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining
The assay was performed by the Situ Cell Death Detection Kit (Roche, Switzerland) according to the manufacturer’s procedure. Paraffin-embedded sections of placental tissues were incubated at 60°C for 2 h, followed by dewaxing and rehydrating in xylene and graded ethanol (95%, 85%, and 75%) solutions. The sections of tissues or cells were permeabilized by 0.1% Triton X-100 (Beyotime, China) and then stained with TUNEL fluorescence staining reagents for 60 min at 37°C in the dark. After washing in PBS, the sections were coverslipped with 4′,6-diamidino-2-phenylindole, dihydrochloride (DAPI, Aladdin, China). Fluorescent images of apoptotic cells were captured, and ImageJ software was used to analyze TUNEL staining.
Cell Counting Kit-8 (CCK-8)
Cell viability was determined using the CCK-8 Cell Proliferation and Cytotoxicity Assay Kit (Solarbio, China). Briefly, the treated HTR-8 cells were plated into 96-well plates. Following the addition of the CCK-8 solution (10 µL) to each well, the cells were incubated for 2 h at 37°C. The optical density was measured at 450 nm using a microplate reader (BioTek, USA).
Statistical analysis
Statistical significance was calculated using GraphPad Prism software v.8.0. Group differences were examined by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc analysis and a p value <0.05 was considered significant.
Results
Effect of Hyp on abnormalities of the placentas in aCL-induced rats
To determine whether Hyp played a protective role in aCL-induced pregnancy loss, we first established the rat models by aCL-IgG injection. As shown in Figure 1(a), the volume of the uterus was reduced and the number of embryos was decreased after aCL administration. It was partially reversed after the treatment of Hyp. In addition, Hyp therapy increased embryonic weight and decreased the abortion rate in aCL-induced rats (Figure 1(b)). The results of ELISA indicated that the levels of PAFR and PF4 were decreased in the aCL-PL + Hyp group compared with the aCL-PL group (Figure 1(c)). The results of H&E staining in Figure 1(d) showed that there were few maternal erythrocytes (small, enucleated cells) in the labyrinth zone of placenta tissues after aCL activation, causing a decreased blood space of the placenta and uteroplacental insufficiency. It was relieved by the treatment with Hyp or LMWH (as a positive control). Hyp inhibits platelet activation and abnormalities of the placentas in rats with pregnancy loss. (a) The intact placentas and the isolated embryos were photographed in pregnant rats. (b) Embryo weight and abortion rate in pregnant rats were detected respectively. (c) The levels of platelet activation factor receptor (PAFR) and platelet factor 4 (PF4) in the plasma were quantified by ELISA. (d) Histological analyses of hematoxylin-eosin-stained placenta tissues, and the quantification of maternal erythrocytes in the placentas using an automated method of morphometric analysis. **p < 0.01 versus the NH-IgG group, #p < 0.05, and ##p < 0.01 versus the aCL-PL group. NH-IgG, normal human-IgG; aCL-PL, anticardiolipin antibody-pregnancy loss; Hyp, Hyp; LMWH, low-molecular-weight heparin.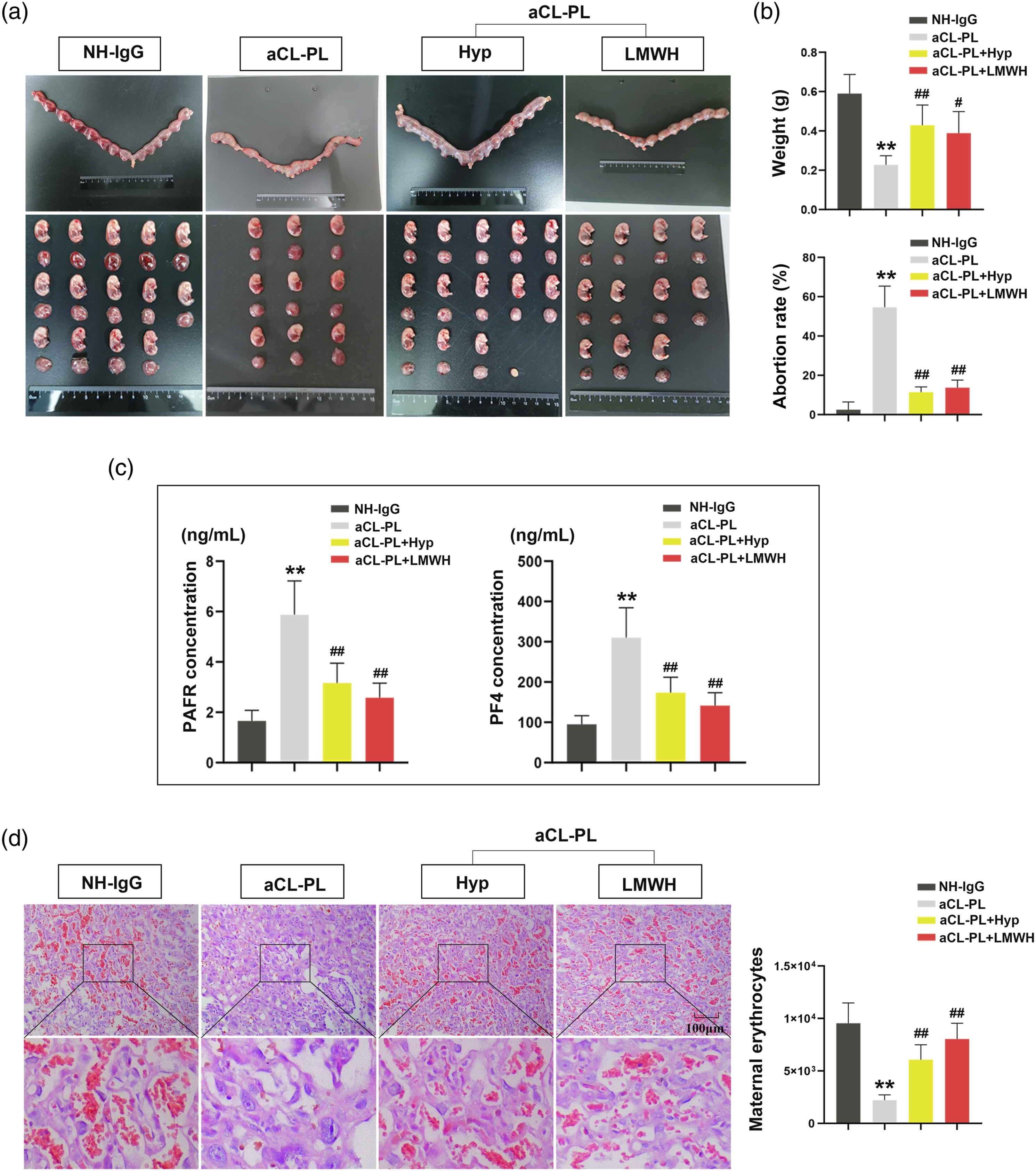
Effect of Hyp on the modulation of inflammation and apoptosis in aCL-induced rats
As a downstream factor of platelet activation, P2X7 was used to investigate the function of Hyp on P2X7-regulated signaling pathways. The results showed that Hyp decreased the protein amounts of P2X7, cleaved-Caspase-1, ASC, and NLRP3 in aCL-induced rats (Figure 2(a)). In addition, the concentration of IL-1β and IL-18 was enhanced after aCL activation, but it was inhibited by Hyp administration (Figure 2(b)). TUNEL staining showed that the apoptotic rate in the placental tissues of rats was accelerated in the aCL-PL group compared with the NH-IgG group, while Hyp treatment presented the opposite effects (Figure 2(c)). Hyp increased the protein amount of Bcl2 and decreased the levels of Bax and cleaved-Caspase-3 after aCL-IgG injection (Figure 2(d)). Hyp attenuates inflammatory response and apoptosis in aCL-induced rats. (a) Western blot analysis for P2X7, cleaved Caspase-1, ASC, and NLRP3. (b) The levels of IL-1β and IL-18 in the placental tissues were determined by ELISA. (c) Assessment of apoptosis using TUNEL analysis in placental tissues, and the quantification of the rate of TUNEL-positive cells/total cells. (d) Western blot analysis for Bcl2, Bax, and cleaved Caspase-3. **p < 0.01 versus the NH-IgG group, ##p < 0.01 versus the aCL-PL group.
Effect of aCL and Hyp at different concentrations on trophoblast cells
HTR-8 cells with the treatment of aCL and Hyp were used to detect cell viability. When treated with 80 and 160 μg/mL aCL, the cells exhibited significantly enhanced cell viability in contrast to the control group (Figure 3(a)). As shown in Figure 3(b), cells treated with 40 μM or 80 μM Hyp showed higher cell viability compared to controls. Hyp attenuates cellular damage in aCL-induced trophoblast cells. (a) CCK-8 assay was used to detect the cell viability in HTR-8 cells treated with different concentrations of aCL (5, 10, 20, 40, 80, or 160 μg/mL) for 48 h. (B) HTR-8 cells were treated with different concentrations of Hyp (2.5, 5, 10, 20, 40, or 80 μM) for 2 h. After the treatment of Hyp, HTR-8 cells were treated with 80 μg/mL aCL for 48 h, and CCK-8 assay was used for the detection of cell viability. **p < 0.01 versus the control group, #p < 0.05 versus the Hyp-0 μM group.
Effect of Hyp on the modulation of inflammation and apoptosis in aCL-induced trophoblast cells
The aCL-induced trophoblast cells were used to verify the protective effects of Hyp on pregnancy loss. Western analysis showed that Hyp downregulated the expression of P2X7, cleaved-Caspase-1, ASC, and NLRP3 after aCL activation, which was restored in the presence of BzATP (an agonist of the P2X7 receptor). Consistent with the effect of Hyp treatment, BBG (an antagonist of the P2X7 receptor) inhibited the expression of NLRP3 inflammasome (Figure 4(a)). In the presence of Hyp, the levels of IL-1β and IL-18 were decreased in aCL-induced trophoblast cells (Figure 4(b)). As shown in Figure 4(c), Hyp administration upregulated the expression of Bcl2 and downregulated the expression of Bax and cleaved-Caspase-3. The TUNEL analysis demonstrated that the apoptotic rate was increased after aCL administration, and the treatment of Hyp reversed the effects (Figure 4(d)). Hyp attenuates inflammatory response and apoptosis in aCL-induced trophoblast cells. (a) Western blot analysis for P2X7, cleaved Caspase-1, ASC, and NLRP3. (b) The levels of IL-1β and IL-18 in the cellular supernatant were determined by ELISA. (c)Western blot analysis for Bcl2, Bax, and cleaved Caspase-3. (d) Assessment of apoptosis using TUNEL analysis in HTR-8 cells. **p < 0.01 versus the control group, ##p < 0.01 versus the aCL group, &p < 0.05 and &&p < 0.01 versus the Hyp + aCL group. BBG, brilliant blue G.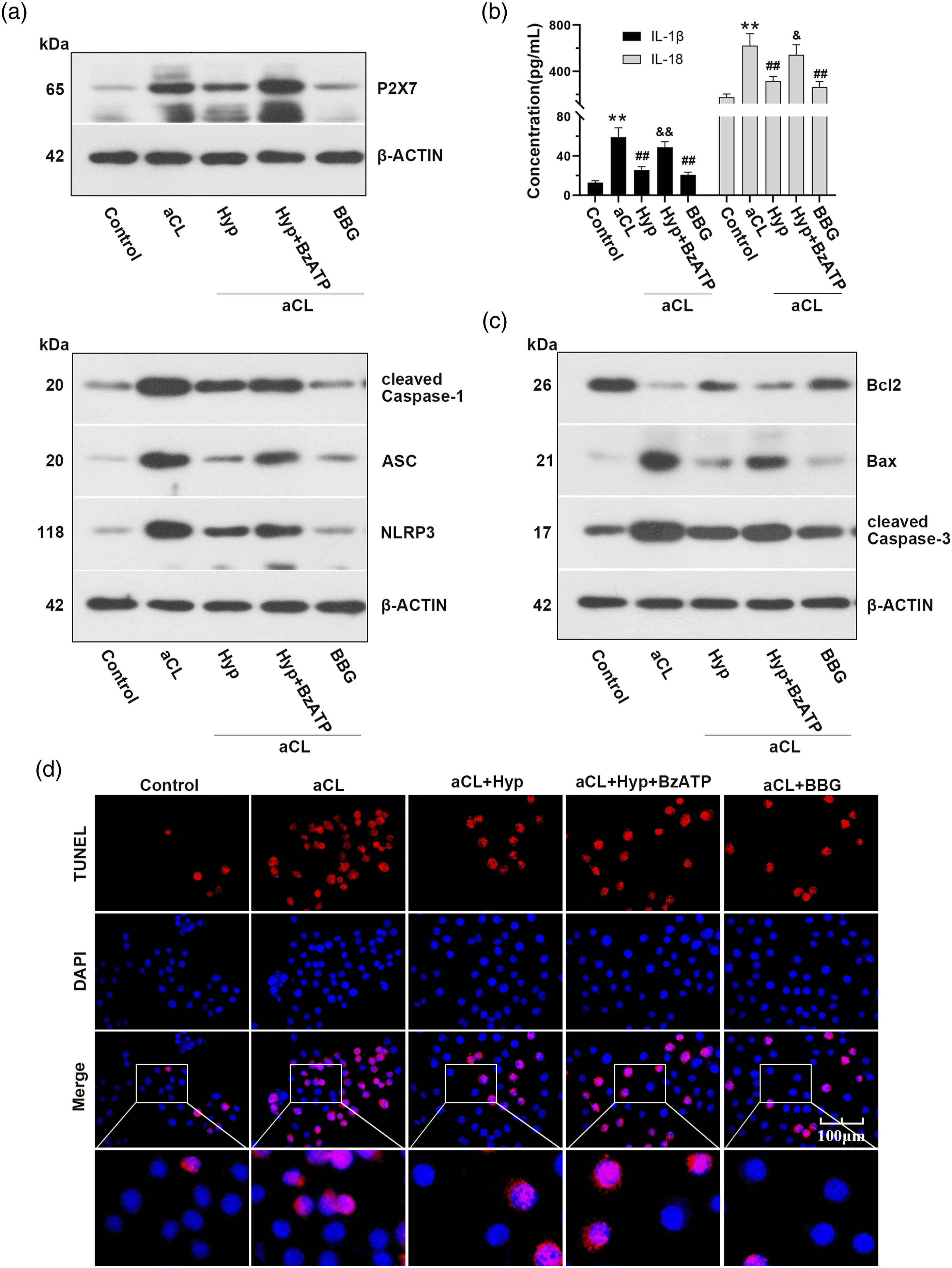
Discussion
APS is a systematic autoimmune disorder, which is defined by thrombosis or obstetric events in patients with persistent aPLs. 25 Obstetric APS is characterized by several features, including fetal loss after 10 weeks of pregnancy, severe preeclampsia, intrauterine growth restriction, or recurrent preterm birth. 25 Miscarriage is the most common complication of pregnancy, with a clinically recognized abortion rate of 15%. 26 Until now, there is no generally accepted treatment for RPL. In Asian societies, Chinese herbal medicine has been widely used for thousands of years and it has become an alternative to western medicine in recent years. 27 In the present study, the promoted platelet activation and pathological changes in aCL-subjected rats were inhibited after Hyp therapy, suggesting that Hyp protected against aCL-induced pregnancy loss. Previous study indicates that Hyp attenuates pregnancy loss by activating autophagy and suppressing inflammation. 19 Consistent with our study, Hyp indeed held protective properties against pregnancy loss.
The evidence available indicates that platelets are versatile cells involved in many other pathophysiological processes. 28 For example, platelets have been recognized to play an important role in immunity,29, 30 and platelets boost the inflammasome capacity in human macrophages and neutrophils. Accumulating evidence suggests that platelet activation is associated with NLRP3 inflammasome, thereby affecting inflammatory response. This is exemplified in the work that platelets are revealed to regulate NLRP3 transcription, leading to the increase of ASC oligomerization, caspase-1 activity, and IL-1β secretion. 31 Beyond that, platelet inhibition can impede NLRP3 inflammasome activation and prevent kidney injury. 32 It is well-established that NLRP3 inflammasome is a multiprotein complex that mediates the activation of the enzyme caspase-1, which further induces the maturation and secretion of the pro-inflammatory factor IL-1β. 33 By investigating the effects of Hyp on the NLRP3 inflammasome, we found that Hyp downregulated the expression of NLRP3 inflammasome-related factors in aCL-induced rats. The increased production of IL-1β and IL-18 caused by aCL activation was also inhibited in the presence of Hyp. In addition, early RPL is thought to be caused by inadequate trophoblast proliferation, shallow invasion, and an excessive rate of trophoblast apoptosis. 34 Here, the anti-inflammatory and anti-apoptotic roles of Hyp were conformed in aCL-induced trophoblasts, which indicated that Hyp might exert protective effects on RPL. In addition, trophoblastic cells with the treatment of BzATP or BBG confirmed that Hyp not only affected the NLRP3 inflammasome but also mediated the P2X7 receptor in response to inflammation.
It is well known that the P2X7 ion channel can connect with the NLRP3 inflammasome complex, leading to the release of cytokines IL-1β and IL-18, and the activation of the P2X7-NLRP3-IL-1β pathway can cause microglial activation (pro-inflammatory) and neuroinflammation. 35 The activation of the P2X7 receptor, together with the involvement of NLRP3 inflammasome and pro-inflammatory cytokines is also well-proven in the liver. 36 After an ischemic stroke in the mouse, the P2X7/NLRP3 pathway may be able to initiate caspase-3-dependent neuronal apoptosis. 37 In addition, the inhibited effects of Hyp on apoptosis have been reported in osteoarthritis, diabetic retinopathy, and acute kidney injury.38-40 On this basis, the apoptotic rate in aCL-activated rats and cells was detected, and we found that Hyp treatment attenuated apoptosis by downregulating the expression of Bax and cleaved-Caspase-3. The results indicated that Hyp indeed exerts a vital role via suppressing inflammasome and apoptosis in pregnancy loss.
Increasing evidence has highlighted the alleviating effects of Hyp in many diseases,38, 41-43 and Hyp functions through different signaling pathways. For example, Hyp attenuates pregnancy loss by regulating mTOR/S6K and TLR4/MyD88/NF-kB signaling pathways. 19 Hyp has been described to involve in the treatment of Parkinson’s disease by inhibiting the activation of the NLRP3 inflammasome. 44 Whether Hyp exerts a protective effect through the P2X7 receptor in RPL is unknown. Hereon, we found that Hyp inhibited the P2X7/NLRP3 pathway to relieve the pathologies of miscarriage caused by aCL. In reviewing the literature, no data was found on the association between Hyp and platelet. Numerous pieces of evidence suggest that LMWH plays an important role in the prevention of recurrent placenta-mediated pregnancy complications.45,46 Moreover, women with aCL and a history of either prior thrombotic events or pregnancy loss may benefit from treatment with LMWH. 47 The most obvious finding in this study was that Hyp or LMWH could inhibit the expression of platelet activation factors and different degrees of pathological pregnancy in aCL-induced abortion.
In conclusion, we found that Hyp inhibited platelet activation and uteroplacental insufficiency in rats with pregnancy loss. In vivo and in vitro experiments indicated that Hyp protected against aCL-induced pregnancy loss by decreasing the secretion of proinflammatory cytokines IL-1β and IL-18 via inhibiting the P2X7/NLRP3 pathway. Furthermore, Hyp attenuated apoptotic rate after aCL activation. Collectively, our study suggested that, for the first time, Hyp exerts the anti-inflammatory and anti-apoptotic roles against aCL-induced pregnancy loss via the regulation of the P2X7/NLRP3 pathway. Therefore, Hyp may be an attractive pharmacological target for the treatment of RPL.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Henan Province Chinese Medicine Scientific Research Special Project (20-21ZY2014) and the Youth Project of National Natural Science Foundation of China (82104911).
